Axcella Therapeutics Promotes Margaret Koziel, M.D., to Chief Medical Officer
06 December 2021 - 11:50PM
Business Wire
Axcella Therapeutics (Nasdaq: AXLA), a clinical-stage
biotechnology company pioneering a new approach to treat complex
diseases using multi-targeted endogenous metabolic modulator (EMM)
compositions, today announced that Margaret Koziel, M.D., has been
promoted to the role of Chief Medical Officer and added as a member
of the company’s executive team. Dr. Koziel, who previously served
as Vice President, Clinical Development, joined Axcella in 2019,
bringing a wealth of leadership experience within both biopharma
and academia.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20211206005091/en/
Margaret Koziel, M.D., Axcella
Therapeutics Chief Medical Officer (Photo: Business Wire)
“Margaret joined Axcella at a pivotal stage in the company’s
evolution and has been a key contributor and leader since Day 1,
helping to bring multiple clinical studies to a successful
conclusion, contributing to our multi-national regulatory
engagements and rapidly advancing our lead candidates into
later-stage development,” said Bill Hinshaw, President and Chief
Executive Officer of Axcella. “We look forward to benefitting from
her continued wisdom and guidance as we advance AXA1125 and AXA1665
in three Phase 2 trials with the ultimate aim of treating patients
with a range of complex diseases and conditions.”
As Chief Medical Officer, Dr. Koziel will lead Axcella’s
clinical development and regulatory affairs efforts. Before joining
the company, she held positions of increasing responsibility at
Kaleido Biosciences, Vertex Pharmaceuticals, and the Novartis
Institute for Biomedical Sciences, working across the full spectrum
of clinical development, from target selection through Phase 4
trials. Dr. Koziel has also served as a Professor and Assistant
Vice Provost for Clinical Research at the University of
Massachusetts Medical School, staff physician at the Beth Israel
Deaconess Medical Center and Associate Professor of Medicine at
Harvard Medical School. Dr. Koziel has published nearly 90 papers
in journals such as the New England Journal of Medicine and The
Journal of Clinical Investigation. She obtained her B.A. and M.D.
from Dartmouth and her postgraduate medical training at New England
Deaconess Hospital and Massachusetts General Hospital, affiliates
of Harvard Medical School.
“I am honored and excited by the opportunity to continue working
alongside our strong team at Axcella as we seek to bring forward
much-needed treatments for conditions like Long COVID,
non-alcoholic steatohepatitis (NASH) and overt hepatic
encephalopathy (OHE),” said Dr. Koziel. “With compelling data to
date, a therapeutic approach that resonates with physicians, and a
platform that can be rapidly applied to a number of additional
areas, we have a strong foundation in place.”
About Axcella Therapeutics (Nasdaq:
AXLA)
Axcella is a clinical-stage biotechnology company pioneering a
new approach to treat complex diseases using endogenous metabolic
modulator (EMM) compositions. The company’s product candidates are
comprised of EMMs and derivatives that are engineered in distinct
combinations and ratios to restore cellular homeostasis in multiple
key biological pathways and improve cellular energetic efficiency.
Axcella’s pipeline includes lead therapeutic candidates in Phase 2
development for the reduction in risk of overt hepatic
encephalopathy (OHE) recurrence, the treatment of Long COVID, and
the treatment of non-alcoholic steatohepatitis (NASH). The
company’s unique model allows for the evaluation of its EMM
compositions through non-IND clinical studies or IND clinical
trials. For more information, please visit www.axcellatx.com.
Forward-Looking
Statements
This press release contains forward-looking statements within
the meaning of the Private Securities Litigation Reform Act of
1995, as amended, including, without limitation, statements
regarding the company’s ability to apply its platform to additional
areas and treat patients with a range of complex diseases and
conditions. The words “may,” “will,” “could,” “would,” “should,”
“expect,” “plan,” “anticipate,” “intend,” “believe,” “estimate,”
“predict,” “project,” “potential,” “continue,” “target” and similar
expressions are intended to identify forward-looking statements,
although not all forward-looking statements contain these
identifying words. Any forward-looking statements in this press
release are based on management’s current expectations and beliefs
and are subject to a number of risks, uncertainties and important
factors that may cause actual events or results to differ
materially from those expressed or implied by any forward-looking
statements contained in this press release, including, without
limitation, those related to the potential impact of COVID-19 on
the company’s ability to conduct and complete its ongoing or
planned clinical studies and clinical trials in a timely manner or
at all due to patient or principal investigator recruitment or
availability challenges, clinical trial site shutdowns or other
interruptions and potential limitations on the quality,
completeness and interpretability of data the company is able to
collect in its clinical trials of AXA1665 and AXA1125, other
potential impacts on the company’s business and financial results,
including with respect to its ability to raise additional capital
and operational disruptions or delays, changes in law, regulations,
or interpretations and enforcement of regulatory guidance, whether
data readouts support the company’s clinical trial plans and
timing, clinical trial design and target indications for AXA1665
and AXA1125, the clinical development and safety profile of AXA1665
and AXA1125 and their therapeutic potential, whether and when, if
at all, the company’s product candidates will receive approval from
the FDA or other comparable regulatory authorities, potential
competition from other biopharma companies in the company’s target
indications, and other risks identified in the company’s SEC
filings, including Axcella’s Annual Report on Form 10-K, Quarterly
Report on Form 10-Q and subsequent filings with the SEC. The
company cautions you not to place undue reliance on any
forward-looking statements, which speak only as of the date they
are made. Axcella disclaims any obligation to publicly update or
revise any such statements to reflect any change in expectations or
in events, conditions, or circumstances on which any such
statements may be based, or that may affect the likelihood that
actual results will differ from those set forth in the
forward-looking statements. Any forward-looking statements
contained in this press release represent the company’s views only
as of the date hereof and should not be relied upon as representing
its views as of any subsequent date. The company explicitly
disclaims any obligation to update any forward-looking
statements.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20211206005091/en/
Jason Fredette jfredette@axcellatx.com (857) 320-2236
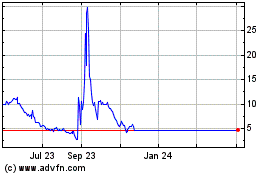
Axcella Health (NASDAQ:AXLA)
Historical Stock Chart
From Mar 2024 to Apr 2024
Axcella Health (NASDAQ:AXLA)
Historical Stock Chart
From Apr 2023 to Apr 2024