Eton Gets FDA Approval for Epilepsy Treatment
18 July 2022 - 10:15PM
Dow Jones News
By Dean Seal
Eton Pharmaceuticals Inc. said Monday that regulators have
granted approval for Zonisade, an epilepsy treatment it developed
in partnership with Azurity Pharmaceuticals Inc.
The privately held Azurity said Zonisade is now the first and
only oral liquid formulation of zonisamide approved by the U.S.
Food and Drug Administration. The drug treats partial seizures for
epilepsy patients ages 16 years or older.
Dr. Jim Wheless, chairman of the University of Tennessee Health
Science Center, said Zonisade addresses an important, unmet need
from patients who have difficulty swallowing or are unable to
swallow tablets.
Zonisade was included in Eton's multiproduct oral-solution
partnership with Azurity, which entitles Eton to receive a $5
million payment upon the treatment's launch, as well as royalty on
net sales and up to $15 million in commercial milestones tied to
sales of Zonisade and other products under the companies'
partnership.
Deer Park, Ill.-based Eton said it will incur no market expenses
for the launch, as Azurity will be responsible for commercializing
Zonisade.
Shares of Eton rose 25% to $3.49 in premarket trading after
briefly being halted.
Write to Dean Seal at dean.seal@wsj.com
(END) Dow Jones Newswires
July 18, 2022 08:00 ET (12:00 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
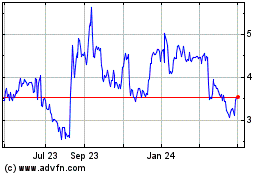
Eton Pharmaceuticals (NASDAQ:ETON)
Historical Stock Chart
From Mar 2024 to Apr 2024
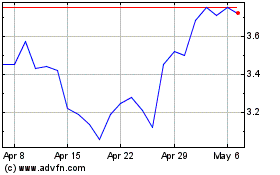
Eton Pharmaceuticals (NASDAQ:ETON)
Historical Stock Chart
From Apr 2023 to Apr 2024