Infinity Pharmaceuticals Announces MARIO-275 Phase 2 Data in Patients with Advanced Urothelial Cancer to be Presented at ASCO...
12 January 2021 - 12:05AM
Business Wire
Infinity Pharmaceuticals, Inc. (NASDAQ: INFI) will be presenting
data from MARIO-275 on the first day of the American Society of
Clinical Oncology (ASCO) 2021 Genitourinary Cancers Symposium to be
held virtually, February 11-13, 2021. MARIO-275 is a randomized,
placebo- controlled Phase 2 study evaluating the benefit of adding
eganelisib to nivolumab (Opdivo®) in platinum-refractory, I/O naïve
patients with advanced urothelial cancer over nivolumab
monotherapy, which is approved in this setting.
Presentation Details
Title:
Preliminary analysis of a phase II,
multicenter, randomized, active-control study to evaluate the
efficacy and safety of eganelisib (IPI-549) in combination with
nivolumab compared to nivolumab monotherapy in patients with
advanced urothelial carcinoma.
Presenter:
Piotr Tomczak M.D., Ph.D.
Date:
February 11, 2021 8:00 AM - 6:30
PM EST
Poster Session:
Urothelial Carcinoma
Abstract:
436
About Infinity and Eganelisib
Infinity is an innovative biopharmaceutical company dedicated to
advancing novel medicines for people with cancer. Infinity is
advancing eganelisib, a first-in-class, oral immuno-oncology
development candidate that selectively inhibits PI3K-gamma, in
multiple clinical studies. MARIO-275 is a global, randomized,
placebo-controlled combination study of eganelisib combined with
Opdivo® in I/O naïve urothelial cancer. MARIO-3 is the first
eganelisib combination study in front-line advanced cancer patients
and is evaluating eganelisib in combination with Tecentriq® and
Abraxane® in front-line TNBC and in combination with Tecentriq and
Avastin® in front-line RCC. In collaboration with Arcus
Biosciences, Infinity is evaluating a checkpoint inhibitor-free,
novel combination regimen of eganelisib plus etrumadenant (dual
adenosine receptor antagonist) plus Doxil® in advanced TNBC
patients. With these studies Infinity is evaluating eganelisib in
the anti-PD-1 refractory, I/O-naïve, and front-line settings. For
more information on Infinity, please refer to Infinity's website at
www.infi.com.
Cautionary Note Regarding Forward-Looking Statements
This press release contains forward-looking statements within
the meaning of The Private Securities Litigation Reform Act of 1995
including statements regarding plans to present data from MARIO-275
at the ASCO 2021 Genitourinary Cancers Symposium and the
therapeutic potential of eganelisib. Such statements are subject to
numerous important factors, risks and uncertainties that may cause
actual events or results to differ materially from the Company's
current expectations, including those factors, risks and
uncertainties described under the caption "Risk Factors" included
in Infinity's annual report and quarterly reports filed with the
Securities and Exchange Commission (SEC), and in other filings that
Infinity makes with the SEC, available through the Company’s
website at www.infi.com. Any forward-looking statements contained
in this press release speak only as of the date hereof, and
Infinity does not undertake and expressly disclaims any obligation
to update any forward-looking statements, whether as a result of
new information, future events or otherwise.
Opdivo® is a registered trademark of Bristol Myers Squibb.
Tecentriq® is a registered trademark of Genentech, Inc. Abraxane®
is a registered trademark of Abraxis BioScience, LLC. Avastin® is a
registered trademark of Genentech, Inc. Doxil® is a registered
trademark of Baxter Healthcare Corporation.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210111005366/en/
Ashley Robinson LifeSci Advisors, LLC 617-775-5956
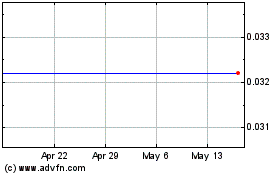
Infinity Pharmaceuticals (NASDAQ:INFI)
Historical Stock Chart
From Apr 2024 to May 2024
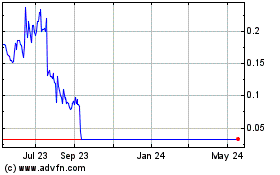
Infinity Pharmaceuticals (NASDAQ:INFI)
Historical Stock Chart
From May 2023 to May 2024