As filed with the Securities
and Exchange Commission on July 12, 2024
Registration
No. 333-___________
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM S-3
REGISTRATION
STATEMENT UNDER THE SECURITIES ACT OF 1933
60
DEGREES PHARMACEUTICALS, INC.
(Exact
name of registrant as specified in its charter)
| Delaware |
|
45-2406880 |
(State
or other jurisdiction of
incorporation or organization) |
|
(I.R.S.
Employer
Identification
Number) |
| |
|
|
1025
Connecticut Avenue NW Suite 1000
Washington,
D.C. 20036
(202)
327-5422 |
| (Address,
including zip code, and telephone number, including area code, of registrant’s principal executive offices) |
| |
Geoffrey
Dow
Chief
Executive Officer and President
60
Degrees Pharmaceuticals, Inc.
1025
Connecticut Avenue NW Suite 1000
Washington,
D.C. 20036
(202)
327-5422 |
| (Name,
address, including zip code, and telephone number, including area code, of agent for service) |
| |
Copies
to:
Ross
D. Carmel, Esq.
Philip
Magri, Esq.
Jeffrey Hua, Esq.
Sichenzia
Ross Ference Carmel LLP
1185
Avenue of the Americas, 31st Floor
New
York, New York 10036
Telephone:
(212) 930-9700 |
Approximate
date of commencement of proposed sale to the public: From time to time, after the effective date of this registration statement.
If
the only securities being registered on this Form are being offered pursuant to dividend or interest reinvestment plans, please check
the following box. ☐
If
any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the
Securities Act of 1933, other than securities offered only in connection with dividend or interest reinvestment plans, check the following
box. ☒
If
this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the
following box and list the Securities Act registration statement number of the earlier effective registration statement for the same
offering. ☐
If
this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, please check the following box and list
the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If
this Form is a registration statement pursuant to General Instruction I.D. or a post-effective amendment thereto that shall become effective
upon filing with the Commission pursuant to Rule 462(e) under the Securities Act, check the following box. ☐
If
this Form is a post-effective amendment to a registration statement filed pursuant to General Instruction I.D. filed to register additional
securities or additional classes of securities pursuant to Rule 413(b) under the Securities Act, check the following box. ☐
Indicate
by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting
company or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,”
“smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| |
Large
Accelerated filer |
☐ |
Accelerated
filer |
☐ |
| |
Non-accelerated
filer |
☒ |
Smaller
reporting company |
☒ |
| |
|
|
Emerging
growth company |
☒ |
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided pursuant to Section 7(a)(2)(B) of the Exchange Act. ☐
The
Registrant hereby amends this Registration Statement on such date or dates as may be necessary to delay its effective date until the
Registrant shall file a further amendment that specifically states that this Registration Statement shall thereafter become effective
in accordance with Section 8(a) of the Securities Act of 1933, as amended, or until this Registration Statement shall become effective
on such date as the Commission, acting pursuant to said Section 8(a), may determine.
EXPLANATORY
NOTE
This
registration statement contains two prospectuses:
| |
● |
a
base prospectus, which covers the potential offering, issuance, and
sale by the registrant of up to a maximum aggregate offering price of $15,000,000 of the registrant’s common stock, preferred stock,
debt securities, warrants and units; and |
| |
● |
an
“at the market offering” sales agreement prospectus supplement
covering the offering, issuance and sale by the registrant of up to a maximum aggregate offering price of $1,253,603 of the registrant’s
common stock that may be issued and sold under that certain At the Market Offering Agreement, dated as of July 12, 2024, or the Sales
Agreement, we entered into with WallachBeth Capital LLC as sales agent (the “Sales Agent”). |
The base prospectus immediately follows this explanatory
note. The specific terms of any securities to be offered pursuant to the base prospectus other than the shares under the Sales Agreement
will be specified in a prospectus supplement to the base prospectus. The “at the market offering” prospectus supplement immediately
follows the base prospectus. The common stock that may be offered, issued and sold by the registrant under the “at the market offering”
prospectus supplement is included in the $15,000,000 of securities that may be offered, issued and sold by the registrant under the base
prospectus. Upon termination of the Sales Agreement with the Sales Agent, any portion of the $1,253,603 included in the “at the
market offering” prospectus supplement that is not sold pursuant to the Sales Agreement will be available for sale in other offerings
pursuant to the base prospectus, and if no shares of common stock are sold under the Sales Agreement, the full $15,000,000 of securities
may be sold in other offerings pursuant to the base prospectus and a corresponding prospectus supplement.
The
information in this prospectus is not complete and may be changed. We may not sell these securities or accept an offer to buy these securities
until the Securities and Exchange Commission declares our registration statement effective. This prospectus is not an offer to sell these
securities and is not soliciting an offer to buy these securities in any state where the offer or sale is not permitted.
SUBJECT
TO COMPLETION, DATED JULY 12, 2024
PROSPECTUS
$15,000,000
Common
Stock
Preferred
Stock
Warrants
Debt
Securities
Units

60
Degrees Pharmaceuticals, Inc.
From time
to time, we may offer and sell shares of preferred stock, common stock, debt securities or warrants to purchase preferred stock, common
stock or any combination of these securities, either separately or in units, in one or more offerings in amounts, at prices and on terms
that we will determine at the time of the offering. The debt securities and warrants may be convertible into or exercisable or exchangeable
for preferred stock, common stock or debt securities and the preferred stock may be convertible into or exchangeable for common stock.
The aggregate initial offering price of all securities sold by us under this prospectus will not exceed $15,000,000.
We
may offer securities through underwriting syndicates managed or co-managed by one or more underwriters or dealers, through agents or
directly to purchasers. The prospectus supplement for each offering of securities will describe in detail the plan of distribution for
that offering. For general information about the distribution of securities offered, please see “Plan of Distribution”
in this prospectus. Each time our securities are offered, we will provide a prospectus supplement containing more specific information
about the particular offering and attach it to this prospectus. The prospectus supplements may also add, update or change information
contained in this prospectus. This prospectus may not be used to offer or sell securities without a prospectus supplement that
includes a description of the method and terms of that offering.
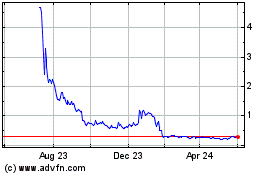
Our common stock and tradeable warrants are quoted
on The Nasdaq Capital Market under the symbols “SXTP” and “SXTPW,” respectively. The last reported sale price
of our common stock and tradeable warrants on The Nasdaq Capital Market on July 11, 2024 was $0.2460 and $0.0683, respectively.
The aggregate market value of our outstanding common stock held by
non-affiliates is $3,761,186, based on 12,206,116 shares of outstanding common stock, of which 10,715,629 shares are held by non-affiliates,
and a share price of $0.351 per share, which was the closing sale price of our common stock as quoted on The Nasdaq Capital Market on
June 7, 2024. Pursuant to General Instruction I.B.6 of Form S-3, in no event will we sell our securities in a public primary offering
with a value exceeding more than one-third of our public float in any 12-month period so long as our public float remains below $75,000,000.
As of the date of this prospectus, we have not offered any securities during the past twelve months pursuant to General Instruction I.B.6
of Form S-3. You are urged to obtain current market quotations of our common stock.
If
we decide to seek a listing of any preferred stock, purchase contracts, warrants, subscriptions rights, depositary shares or units offered
by this prospectus, the related prospectus supplement will disclose the exchange or market on which the securities will be listed, if
any, or where we have made an application for listing, if any.
Other
than our common stock, we have not yet determined whether the other securities that may be offered by this prospectus will be listed
on any exchange, interdealer quotation system or over-the-counter market. If we decide to seek the listing of any such securities upon
issuance, the prospectus supplement relating to those securities will disclose the exchange, quotation system or market on which those
securities will be listed.
We are an “emerging growth company” and a “smaller
reporting company” as defined in the Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”), and have elected
to comply with certain reduced public company reporting requirements. See “Summary - Implications of Being an Emerging Growth
Company and Smaller Reporting Company.”
Investing
in our securities involves a high degree of risk. See “Risk Factors” beginning on page 18 and any risk factors in
our most recent Annual Report on Form 10-K, which is incorporated by reference herein, as well as in any other recently filed
quarterly or current reports and, if any, in the relevant prospectus supplement. We urge you to carefully read this prospectus and
the accompanying prospectus supplement, together with the documents we incorporate by reference, describing the terms of these
securities before investing.
Neither
the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or passed
upon the adequacy or accuracy of this prospectus. Any representation to the contrary is a criminal offense.
The
date of this prospectus is July 12, 2024.
TABLE
OF CONTENTS
ABOUT
THIS PROSPECTUS
This prospectus is part of a registration statement
on Form S-3 that we filed with the Securities and Exchange Commission (the “SEC” or the “Commission”) utilizing
a “shelf” registration process. Under this shelf registration process, we may offer and sell, either individually or in combination,
in one or more offerings, any of the securities described in this prospectus, for total gross proceeds of up to $15,000,000. This prospectus
provides you with a general description of the securities we may offer. Each time we offer securities under this prospectus, we will provide
a prospectus supplement to this prospectus that will contain more specific information about the terms of that offering. We may also authorize
one or more free writing prospectuses to be provided to you that may contain material information relating to these offerings. The prospectus
supplement and any related free writing prospectus that we may authorize to be provided to you may also add, update or change any of the
information contained in this prospectus or in the documents that we have incorporated by reference into this prospectus.
We
urge you to read carefully this prospectus, any applicable prospectus supplement and any free writing prospectuses we have authorized
for use in connection with a specific offering, together with the information incorporated herein by reference as described under the
heading “Incorporation of Documents by Reference,” before investing in any of the securities being offered. You should
rely only on the information contained in, or incorporated by reference into, this prospectus and any applicable prospectus supplement,
along with the information contained in any free writing prospectuses we have authorized for use in connection with a specific offering.
We have not authorized anyone to provide you with different or additional information. This prospectus is an offer to sell only the securities
offered hereby, but only under circumstances and in jurisdictions where it is lawful to do so.
The
information appearing in this prospectus, any applicable prospectus supplement or any related free writing prospectus is accurate only
as of the date on the front of the document and any information we have incorporated by reference is accurate only as of the date of
the document incorporated by reference, regardless of the time of delivery of this prospectus, any applicable prospectus supplement or
any related free writing prospectus, or any sale of a security.
This
prospectus contains summaries of certain provisions contained in some of the documents described herein, but reference is made to the
actual documents for complete information. All of the summaries are qualified in their entirety by the actual documents. Copies of some
of the documents referred to herein have been filed, will be filed or will be incorporated by reference as exhibits to the registration
statement of which this prospectus is a part, and you may obtain copies of those documents as described below under the section entitled
“Where You Can Find Additional Information.”
We
have not authorized any dealer, agent or other person to give any information or to make any representation other than those contained
or incorporated by reference in this prospectus and any accompanying prospectus supplement. You must not rely upon any information or
representation not contained or incorporated by reference in this prospectus or an accompanying prospectus supplement. This prospectus
and the accompanying prospectus supplement, if any, do not constitute an offer to sell or the solicitation of an offer to buy any securities
other than the registered securities to which they relate, nor do this prospectus and the accompanying prospectus supplement, if any,
constitute an offer to sell or the solicitation of an offer to buy securities in any jurisdiction to any person to whom it is unlawful
to make such offer or solicitation in such jurisdiction. You should not assume that the information contained in this prospectus and
the accompanying prospectus supplement, if any, is accurate on any date subsequent to the date set forth on the front of such document
or that any information we have incorporated by reference is correct on any date subsequent to the date of the document incorporated
by reference, even though this prospectus and any accompanying prospectus supplement is delivered or securities are sold on a later date.
References
in this prospectus to the terms “60 Degrees Pharmaceuticals, Inc.,” “60 Degrees Pharmaceuticals,” “60P,”
the “Company,” “we,” “us,” “our” or other similar terms refer to 60 Degrees Pharmaceuticals,
Inc., a Delaware corporation, and our subsidiaries.
CAUTIONARY
NOTE REGARDING FORWARD-LOOKING STATEMENTS
This
prospectus contains “forward-looking statements.” Forward-looking statements reflect the current view about future events.
When used in this prospectus, the words “anticipate,” “believe,” “estimate,” “expect,”
“future,” “intend,” “plan,” or the negative of these terms and similar expressions, as they relate
to us or our management, identify forward-looking statements. Such statements include, but are not limited to, statements contained in
this prospectus relating to our business strategy, our future operating results and liquidity and capital resources outlook. Forward-looking
statements are based on our current expectations and assumptions regarding our business, the economy and other future conditions. Because
forward–looking statements relate to the future, they are subject to inherent uncertainties, risks and changes in circumstances
that are difficult to predict. Our actual results may differ materially from those contemplated by the forward-looking statements. They
are neither statements of historical fact nor guarantees of assurance of future performance. We caution you therefore against relying
on any of these forward-looking statements. Important factors that could cause actual results to differ materially from those in the
forward-looking statements include, without limitation:
| |
● |
Our
ability to effectively operate our business segments; |
| |
|
|
| |
● |
Our
ability to manage our research, development, expansion, growth and operating expenses; |
| |
|
|
| |
● |
Our
ability to evaluate and measure our business, prospects and performance metrics;
|
| |
● |
Our
ability to compete, directly and indirectly, and succeed in a highly competitive and evolving industry; |
| |
|
|
| |
● |
Our
ability to respond and adapt to changes in technology and customer behavior; |
| |
|
|
| |
● |
Our
ability to protect our intellectual property and to develop, maintain and enhance a strong
brand; and
|
| |
● |
other
factors (including the risks contained in the section of this prospectus entitled “Risk Factors”) relating to
our industry, our operations and results of operations. |
Should
one or more of these risks or uncertainties materialize, or should the underlying assumptions prove incorrect, actual results may differ
significantly from those anticipated, believed, estimated, expected, intended or planned.
Factors
or events that could cause our actual results to differ may emerge from time to time, and it is not possible for us to predict all of
them. We cannot guarantee future results, levels of activity, performance or achievements. Except as required by applicable law, including
the securities laws of the United States, we do not intend to update any of the forward-looking statements to conform these statements
to actual results.
MARKET,
INDUSTRY AND OTHER DATA
This
prospectus and any applicable prospectus supplement and the documents incorporated by reference herein and therein contain estimates,
projections, market research and other information concerning, among other things, our industry, our business and markets for our products
and services. Unless otherwise expressly stated, we obtain this information from reports, research surveys, studies and similar data
prepared by market research firms and other third parties, industry, technology and general publications, government data and similar
sources as well as from our own internal estimates and research and from publications, research, surveys and studies conducted by third
parties on our behalf. Information that is based on estimates, projections, market research or similar methodologies is inherently subject
to uncertainties and actual events or circumstances may differ materially from events and circumstances that are reflected in this information.
As a result, you are cautioned not to give undue weight to such information.
TRADEMARKS
Solely
for convenience, our trademarks and tradenames referred to in this prospectus may appear without the ® or ™ symbols,
but such references are not intended to indicate in any way that we will not assert, to the fullest extent under applicable law, our
rights to these trademarks and tradenames. All other trademarks, service marks and trade names included or incorporated by reference
into this prospectus or the accompanying prospectus are the property of their respective owners. We do not intend our use or display
of other companies’ trade names, trademarks or service marks to imply relationships with, or endorsements or sponsorship of us
by, these other companies.
PROSPECTUS
SUMMARY
This
summary provides a brief overview of the key aspects of our business and our securities. The reader should read the entire prospectus
carefully, especially the risks of investing in our securities discussed under “Risk Factors.” Some of the statements contained
in this prospectus, including statements under “Summary” and “Risk Factors” as well as those noted in the documents
incorporated herein by reference, are forward-looking statements and may involve a number of risks and uncertainties. Our actual results
and future events may differ significantly based upon a number of factors. The reader should not put undue reliance on the forward-looking
statements in this document, which speak only as of the date on the cover of this prospectus.
Overview
We are a specialty pharmaceutical company with a goal of using cutting-edge
biological science and applied research to further develop and commercialize new therapies for the prevention and treatment of infectious
diseases. We have successfully achieved regulatory approval of Arakoda, a malaria preventative treatment that has been on the market since
late 2019. Currently, 60P’s pipeline under development covers development programs for vector-borne, fungal, and viral diseases
utilizing three of the Company’s future products: (i) new products that contain the Arakoda regimen of Tafenoquine; (ii) new products
that contain Tafenoquine; and (iii) Celgosivir.
Mission
Our mission is to address the unmet medical need associated with infectious
diseases through the development and commercialization of new small molecule therapeutics, focusing on synthetic drugs (made by chemists
in labs, excluding biologics) with good safety profiles based on prior clinical studies, in order to reduce cost, risk, and capitalize
on existing research. We are seeking to expand Arakoda’s use beyond malaria prevention and to demonstrate clinical benefit for other
disease indications. We are further testing the viability of another product (Celgosivir) to determine whether to advance it into further
clinical development, and may seek to develop and license other molecules in the future. Celgosivir is being considered for development
as an antiviral product for a number of diseases.
Market
Opportunity
Malaria
Prevention
In 2018, the FDA approved Arakoda
for malaria prevention in individuals 18 years and older. Arakoda entered the U.S. supply chain in the third quarter of 2019, just
prior to the COVID-19 pandemic. As the approved indication is for travel medicine, and international travel was substantially
impacted by the pandemic, we did not undertake any active marketing efforts for Arakoda. Following our recent financing the Company
hired a Chief Commercial Officer and commissioned IQVIA market data and a qualitative marketing demand study. That research,
recently completed, suggests that prescribing for malaria prevention therapies has returned to pre-pandemic levels, and that the
total U.S. market represents around 1.1 million prescriptions (one prescription per three weeks of travel). Based on consumer and
HCP demand research, the Company estimates that the accessible market for Arakoda represents about one third of this volume (about
330,000 prescriptions). Barriers to entry include low brand awareness in the prescriber community and the low cost of some of the
generic alternatives. In the second half of 2024 we will conduct a pilot commercialization study to confirm these barriers can be
overcome (see “Strategy”).
Treatment
and Prevention of Tick-Borne Disease (Babesiosis)
We are repositioning the Arakoda regimen of Tafenoquine for several
potential new therapeutic indications that have substantial U.S. caseloads, as further described below:
| |
● |
Treatment of Chronic Tick-Borne Disease (Babesiosis).
Babesia parasites are co-transmitted by the same ticks that transmit Borrelia, the Lyme disease bacterium. Although Lyme
in the acute phase is generally viewed by the medical community as being treatable with antibiotics, individuals who are not treated,
or fail treatment, may go on to develop long term, and potentially debilitating, chronic symptoms such as fatigue, body aches, and cognitive
problems.1 This condition is defined by the Centers for Disease Control and Prevention (“CDC”) as Post-Treatment
Lyme Disease Syndrome (“PTLDS”) or simply as Lyme in the patient community.2 Although there are no published estimates,
key opinion leaders have stated that as many as 50% of Lyme/PTLDS patients are believed to be co-infected with Babesia parasites,
a diagnosis referred to in the Lyme community as “Chronic Babesiosis.” Prescribers in the Lyme disease community utilize a
number of therapeutic modalities to manage the symptoms of Chronic Babesiosis, including FDA-approved pharmaceuticals such as atovaquone
and azithromycin (these are assumed to suppress the growth of Babesia parasites).3
Recent market data shows that Tafenoquine appears
to be increasingly prescribed by Lyme physicians to manage Chronic Babesiosis. This trend may follow the recent publication of several
case reports demonstrating activity in immunosuppressed patients with acute babesiosis, and animal data showing eradication of Babesia
parasites, Tafenoquine (primarily as Arakoda).4 The Company believes the recent increases in sales of Arakoda have been driven
by organic growth of these activities. There are no formal epidemiological publications articulating the incidence or prevalence of Chronic
Babesiosis, so these metrics must be inferred based on data for PTLDS and the rate of coinfection with Babesia parasites. Thus,
the cumulative case load of Chronic Babesiosis may be as high as1.01 million patients in the United States.5 We believe, based
on our market research that at least 37% of this market, or 375,000 cases, may be addressable with Tafenoquine during the remainder of
its market exclusivity window for malaria. We are undertaking additional research to determine how much additional market capture might
be feasible.
Acute infection with many different organisms (e.g. Borrelia, SARS-Cov-2,
Epstein Barr virus) trigger “Long Syndromes” in a minority of cases, characterized by cognitive dysfunction, fatigue and post-exertional
malaise.6 For many years, such conditions have been confusing to the mainstream medical community because there may not be
formal diagnostic criteria or an established theory of disease. This is changing with the advent of Long COVID, and a recent prominent
paper outlined the pathophysiological mechanisms for the first time.7 Although there is not yet supporting evidence in the
medical literature, some key opinion leaders in the Lyme community have postulated, using the veterinary literature as an analog, that
life-long infection by sequestering forms of Babesia (e.g., B. odocoilei) may be a significant driver of chronic fatigue
symptoms.8 If this is true, the addressable market for antibabesial drugs may be substantially larger than stated above, since
the prevalence of chronic fatigue syndrome in the U.S. is at least 3.3 million cases (excluding Long COVID and PTLDS).9
|
| 1 |
See
https://www.cdc.gov/lyme/signs-symptoms/chronic-symptoms-and-lyme-disease.html. |
| 2 |
See
https://www.cdc.gov/lyme/signs-symptoms/chronic-symptoms-and-lyme-disease.html. |
| 3 |
Conclusions from Company-commissioned market research. |
| 4 |
Conclusions from Company-commissioned market research. |
| 5 |
Maximum
prevalence determined by multiplying the rate of Babesia coinfection in PTLDS patients (52%, from Parveen & Bhanot, Pathogens
2019;8(3):117) by the highest estimate of the cumulative prevalence of PTLDS (1,994,189, from Delong et al. BMC Public Health
2019;19(1):352). Maximum new cases determined by multiplying the number of new Lyme cases per year (476,000, from Krugeler et
al (Emerg Infect Dis 2021;27:616-61) by the number of new cases that subsequently become chronic cases (up to 10%, from
Delong et al. BMC Public Health 2019;19(1):352) by the proportion of such patients coinfected with Babesia (52%, from
Parveen & Bhanot, Pathogens 2019;8(3):117). |
| 6 |
See
https://www.cdc.gov/lyme/signs-symptoms/chronic-symptoms-and-lyme-disease.html. |
| 7 |
Walitt
et al Nature Communications 2024;15:907. |
| 8 |
Lindner
HH. 2022. Chronic babesiosis caused by B. odocoilei: Diagnosis, pathophysiology & treatment. Presentation at the 2022
ILADS scientific meeting, Orlando Florida. |
| 9 |
See
https://www.cdc.gov/nchs/data/databriefs/db488.pdf. |
| |
|
Separately from the clinical indication, based on estimates from industry
experts, there may be somewhere between several hundred and several thousand cases of canine babesiosis each year in the United States,
and thousands more globally. Currently, standard of care treatment for babesiosis in dogs is a ten-day course of atovaquone and azithromycin,
which costs about $1,350 out of pocket. A treatment course of Tafenoquine mirroring the human prophylactic dose in dogs might cost <
$300, offering a compelling alternative to standard of care. The additional resources required to generate enabling data for veterinary
uses are much less expensive than human clinical trials and we are already funding a pilot study at North Carolina State University related
to this indication. |
| |
|
|
| |
● |
Treatment of Acute Babesiosis. There are up to 38,000 cases of potentially
treatable acute symptomatic babesiosis (red blood cell infections caused by deer tick bites) in the United States each year.10
Approximately 650 of these cases are hospitalizations, a smaller fraction of which represents immunosuppressed individuals.11 Symptomatic
babesiosis is usually treated with a minimum ten day course of atovaquone and azithromycin which is extended to six weeks in the immunosuppressed,
who may also experience relapses requiring multiple hospitalizations.12 This is much longer than equivalent serious parasitic
diseases such as malaria where the goal is a three-day regimen. In a recently published case series Tafenoquine in combination with standard
of care cured 80% of immunosuppressed patients with relapsing babesiosis and the investigators stated in a press release that “Tafenoquine
is going to make a huge difference, I think, in people who are severely immunocompromised.”
13 |
| |
● |
Prevention of Tick-Borne Diseases. Post-exposure prophylaxis or early
treatment with, respectively, a single dose or several week regimen of doxycycline following a tick-bite is a recognized indication to
prevent the complications of Lyme disease. There may be more than 400,000 such tick bites in the United States requiring medical treatment
each year. This estimate is based on the observation that approximately 50,000 tick bites are treated in U.S. hospital emergency rooms
each year; however, this calculation represents only about 12% of actual treated tick bites based on observations from comparable ex-U.S
health systems.14 Unlike Lyme disease, there is no characteristic rash associated with early infection and no reliable diagnostic
tests. Thus, an individual bitten by a tick cannot know whether they have also been infected with babesiosis. It is likely that a drug
proven to be effective for this indication for babesiosis would also be used in conjunction with Lyme prophylaxis. |
Treatment
and Prevention of Fungal Infections
We are evaluating Tafenoquine for potential utility
in the following fungal diseases:
| |
● |
Treatment
of Candida infections. According to the CDC, there are 50,000 reported cases of candidiasis
(a type of fungal infection) each year in the United States and up to 1,900 clinical cases of C. auris, for which there
are few available treatments.15 Since it has broad-spectrum activity against drug-resistant Candida spp in culture,
Tafenoquine, has the potential to be a market leading therapy for treatment/prevention of C. auris, and to be added to the standard
of care regimens for other Candida infections.16 |
| 10 | This estimate is based on the observations of Krugeler et
al (Emerg Infect Dis 2021;27:616-61) who reported that 476,000 cases of Lyme disease occur in U.S. states where babesiosis
is endemic and Krause et. al. (JAMA 1996;275:1657-16602) who reported that 10% of Lyme disease patients are co-infected with babesiosis
and that according to Krause et al (AJTMH 2003;6:431-436) fact that about 80% of cases are symptomatic (thus 476,000*10%*80% = 38,000
cases of babesiosis per year). |
| 11 | Bloch et al Open Forum Infect Dis 2022;9(11):ofac597. |
| 12 | According to IDSA guidelines. |
| 13 | See Krause et al Clin Infect Dis 2024; doi:10.1093/cid/ciae238
and https://ysph.yale.edu/news-article/antimalarial-drug-is-effective-against-tick-borne-infection-babesiosis/. |
| 14 | Marx et. al., MMWR 2021;70:612-616. |
| 15 | https://www.cdc.gov/fungal/diseases/candidiasis/invasive/statistics.html.;
https://www.cdc.gov/fungal/candida-auris/tracking-c-auris.html. |
|
16 |
Dow and Smith New Microb New Infect 2022;45:100964. |
| |
● |
Prevention of fungal pneumonias. There are up to ~ 91-92,000 new patient cases each year in the United States for which antifungal prophylaxis is recommended, including acute lymphoblastic leukemia (up to 6,540 cases) and large B-cell lymphoma (up to 18,000 cases) patients receiving CAR-T therapy, solid organ transplant patients (up to 42,887 cases), allogeneic (~ 9,000 cases) and autologous (~ 15,000 cases) hematopoietic stem cell transplant patients.17 Despite the availability and use of antifungal prophylaxis, the risk of some patient groups contracting fungal pneumonia exceeds the risk of contracting malaria during travel to West Africa.18 Since it has broad spectrum antifungal effects in cell culture, and activity against Pneumocystis in animal models, Tafenoquine has the potential to be added to existing standard of care regimens for the prevention of fungal pneumonias.19 |
Viral
Diseases
Celgosivir, a potential clinical candidate of 60P’s, has activity
in a number of animal models of important viral diseases such as Dengue and RSV. According to the European CDC, Dengue is associated with
at least 4.1 million cases globally. 20 And, according to the U.S. CDC, RSV is responsible for up to 240,000 hospitalizations
in children less than five years of age and adults greater than 65 years of age in the United States each year.21 As outlined
in the “Strategy” section below, we expect to evaluate Celgosivir in additional non-clinical disease models before
making a decision regarding clinical development.
More
information about our products is provided in the next section, and the status of various development efforts for the above-mentioned
diseases is outlined in Figure A, below.
Figure
A

Products
Arakoda (Tafenoquine) for malaria prevention
We entered into a cooperative research and development
agreement with the United States Army in 2014 to complete development of Arakoda for prevention of malaria.22 With the U.S.
Army, and other private sector entities as partners, we coordinated the execution of two clinical trials, development of a full manufacturing
package, gap-filling non-clinical studies, compilation of a full regulatory dossier, successful defense of our program at an FDA advisory
committee meeting and submitted a new drug application (“NDA”) to the FDA in 2018. The history of that collaboration has
been publicly communicated by the U.S. Army.23
| 17 |
See
statistics for solid organ transplants at the Organ Transplant and Procurement Network at: National data - OPTN (hrsa.gov); See statistics
for hematopoietic stem cell transplant in Dsouza et al Biology of Blood and Bone Marrow Transplantation 202;26: e177-e182; See statistics
for acute lymphoblastic leukemia at: Key Statistics for Acute Lymphocytic Leukemia (ALL) (cancer.org); See statistics for large cell
large B-cell lymphoma at; Diffuse Large B-Cell Lymphoma - Lymphoma Research Foundation; Treatment guidelines recommending antifungal
prophylaxis for these diseases can be reviewed in (i) Fishman et al Clinical Transplantation. 2019;33:e13587, (ii) Hematopoietic
Cell Transplantation (cancernetwork.com), (iii) Cooper et al Journal of the National Comprehensive Cancer Network 2016;14:882-913
and (iv) Los Arcos et al Infection (2021) 49:215–231. |
| 18 |
Aguilar-Guisado et al Clin Transplant 2011;25:E629–38; Mace et al MMWR 202;70:1–35. |
| 19 |
Queener et al JID 1997;165:764-768; Dow and Smith New Microb New Infect 2022;45:100964 |
| 20 |
https://www.ecdc.europa.eu/en/dengue-monthly#:~:text=This%20is%20an%20increase%20of%2032%20653%20cases%
20and%2032,853%20deaths%20have%20been%20reported. |
| 21 |
https://www.cdc.gov/rsv/php/surveillance/index.html#cdc_survey_profile_surveys_used-rsv-burden-estimates. |
| 22 |
In 2014, we signed a cooperative research and development agreement with the United States Army Medical and Materiel Development Activity (Agreement W81XWH-14-0313). Under this agreement, we agreed to submit an NDA for Tafenoquine to the FDA (as Arakoda), while the US Army agreed to finance the bulk of the necessary development activities in support of that goal. |
| 23 |
Zottig et al Military Medicine 2020; 185 (S1): 687. |
The FDA and Australia’s medicinal regulatory
agency, the Therapeutic Goods Administration, subsequently approved Arakoda (brand name in the U.S.) and Kodatef (brand name in Australia),
respectively, for prevention of malaria in travelers in 2018. Prescribing information and guidance for patients can be found at www.arakoda.com.
The features and benefits of Tafenoquine for malaria prophylaxis, some of which have been noted by third-party experts, include: convenient
once weekly dosing following a three day load; the absence of reports of drug resistance during malaria prophylaxis; activity against
liver and blood stages of malaria as well as both the major malaria species (Plasmodium vivax and Plasmodium falciparum);
absence of any black-box safety warnings; good tolerability, including in women and individuals with prior psychiatric medical history;
and a comparable adverse event rate to placebo with up to 12 months continuous dosing.24 Tafenoquine entered the commercial
supply chains in the U.S. and Australia in the third quarter of 2019.
The only limitation of Arakoda is the requirement
for a G6PD test prior to administration.25 The G6PD test must be administered to a prospective patient prior to administration
of Arakoda in order to prevent the potential occurrence of hemolytic anemia in individuals with G6PD deficiency.26 G6PD
is one of the most common enzyme deficiencies and is implicated in hemolysis following administration/ingestion of a variety of oxidant
drugs/food. G6PD must also be ruled out as a possible cause when diagnosing neonatal jaundice. As a consequence, G6PD testing is widely
available in the United States through commercial pathology service providers (e.g., Labcorp, Quest Diagnostics, etc.). Although these
tests have a turn-around time of up to 72 hours, the test needs only to be administered once. Thus, existing U.S. testing infrastructure
is sufficient to support the FDA-approved use of the product (malaria prevention) by members of the armed forces (who automatically have
a G6PD test when they enlist), civilian travelers with a long planning horizon, or repeat travelers.
Tafenoquine for Other (Infectious) Diseases
During the pandemic, we also worked with NIH to
evaluate the utility of Tafenoquine as an antifungal. We, and the NIH, found that Tafenoquine exhibits a Broad Spectrum of Activity in
cell culture against Candida and other yeast strains via a different Mode of Action than traditional antifungals and also exhibits
antifungal activity against some fungal strains at clinically relevant doses in animal models.27 Our work followed Legacy Studies
that show Tafenoquine is effective for treatment and prevention of Pneumocystis pneumonia in animal models.28 We believe
that if added to the standard of care for anti-fungal and yeast infection treatments for general use, Tafenoquine has the potential to
improve patient outcomes in terms of recovery from yeast infections, and prevention of fungal pneumonias in immunosuppressed patients.
There are limited treatment options available for these indications, and Tafenoquine’s novel mechanism of action might also mitigate
problems of resistance. Clinical trial(s) to prove safety and efficacy, and approval by the FDA and other regulators, would be required
before Tafenoquine could be marketed for these indications.
Tafenoquine monotherapy, or use in combination
with other antibabesial medications, clears and eradicates Babesia infections, respectively, in both immunocompetent and immunocompromised
animal models of babesiosis (tick borne red blood cell infections).29 In up to 80% of cases Tafenoquine administered in combination
with antibabesial drugs after prior failure of conventional antibiotics in immunosuppressed babesiosis patients resulted in cures.30
Tafenoquine is also increasingly being utilized by Lyme disease prescribers to manage symptoms of Chronic Babesiosis. Consequently,
we believe that (i) if combined with standard of care products, Tafenoquine has the potential to accelerate parasite clearance and reduce
the duration of illness and treatment with antibiotic therapy in immunosuppressed patients hospitalized with severe illness, (ii) once
appropriate clinical studies have been conducted, it is likely that Tafenoquine would be quickly embraced for post-exposure prophylaxis
of babesiosis in patients with tick bites, and (iii) Tafenoquine could become the leading treatment for Chronic Babesiosis. Clinical trial(s)
to prove safety and efficacy, and approval by FDA and other regulators, would be required before Tafenoquine could be marketed for these
indications.
| 24 |
Tan
and Hwang Journal of Travel Medicine, 2018, 1–2; Baird Journal of Travel Medicine 2018:, 1–13; Schlagenhauf et al Travel
Medicine and Infectious Disease 2022; 46:102268; See Arakoda prescribing information at www.arakoda.com; McCarthy et al CID 2019:69:480-486;
Dow et al. Malar J (2015) 14:473; Dow et al. Malaria Journal 2014, 13:49; Novitt-Moreno et al Travel Med Infect Dis 2022 Jan-Feb;45:102211. |
| 25 |
See
prescribing information at www.arakoda.com. |
| 26 |
See
prescribing information at www.arakoda.com. |
| 27 |
Dow
and Smith, New Microbe and New Infect 2022; 45: 100964. |
| 28 |
Queener
et al Journal of Infectious Diseases 1992;165:764-8). |
| 29 |
Liu
et al. Antimicrobial Agents Chemo 2021;65:e00204-21, Marcos et al. IDCases 2022;27:e01460; Rogers et al. Clin Infect Dis. 2022 Jun
10:ciac473, Prasad and Wormsner. Pathogens 2022;11:1015. |
| 30 |
Krause
et al Clin Infect Dis 2024; doi:10.1093/cid/ciae238. |
Celgosivir
Celgosivir is a host targeted glucosidase inhibitor
that was developed separately by other sponsors for HIV then for hepatitis C.31 The sponsors abandoned Celgosivir after completion
of Phase II clinical trials involving 700+ patients, because other antivirals in development at the time had superior activity. The National
University of Singapore initiated development of Celgosivir independently for Dengue fever. A clinical study, conducted in Singapore,
the results of which were accepted for publication in the peer-reviewed journal Lancet Infectious Diseases, confirmed its safety but the
observed reduction in viral load was lower than what the study was powered to detect.32 Celgosivir (as with other Dengue antivirals)
exhibits greater capacity to cure Dengue infections in animal models when administered prior to symptom onset when compared to administration
post-symptom onset. In animal models, this problem can be addressed by administering the same dose of drug split into four doses per day
rather than two doses per day (as was the case in the Singaporean clinical trial).33 This observation led to the filing and
approval of a patent related to Dengue, which we licensed from the National University of Singapore.
Additional clinical studies would be required
to prove that such a 4x daily dosing regimen would be safe and effective in Dengue patients to regulators’ satisfaction. To that
end, earlier in our history, we, in partnership with the National University of Singapore, and Singapore General Hospital, successfully
secured a grant from the government of Singapore for a follow-on clinical trial. Unfortunately, we were unable at that time to raise matching
private sector funding. We concluded as a result that development of Repositioned Molecules for Dengue, solely and without simultaneous
development for other therapeutic use, despite substantial morbidity and mortality in tropical countries, was an effort best suited for
philanthropic entities. Accordingly, during the pandemic, we undertook an effort (in partnership with NIH’s Division of Microbiology
and Infectious Diseases program and Florida State University) to determine whether Celgosivir might be more broadly useful for respiratory
diseases that have impact in both tropical and temperate countries. Preliminary data suggest that Celgosivir inhibits the replication
of the virus that causes COVID-19 (SARS-CoV-2) in cell culture, and the RSV virus in cell culture and provides benefits in animals. We
have filed and/or licensed patents in relation to Celgosivir for these other viruses as we believe there is potential applications to
fight respiratory diseases that might have more commercial viability than historical development of Celgosivir to combat Dengue fever.
Competitive
Strengths
Our main competitive strength has been our ability
to achieve important clinical milestones inexpensively in therapeutic areas that other entities have found extremely challenging. With
a small virtual management team, we have successfully built productive research partnerships with public and academic entities, and licensed
products with well characterized safety profiles in prior clinical studies, thereby reducing the cost and risk of clinical development.
This business and product model enabled Arakoda to be approved in 2018, with a total operating expense of < $10 million. We plan to
focus in the future on generating proof of concept clinical data sets for the approved Arakoda regimen of Tafenoquine in other therapeutic
areas, all of which is expected to foster and continue our existing tradition of inexpensive product development.
Strategy
Following our initial public offering in July
2023, our initial strategic priority was to conduct a Phase IIB that would have evaluated the potential of the Arakoda regimen of Tafenoquine
to accelerate disease recovery in COVID-19 patients with low risk of disease progression. In October 2023, we made a decision to suspend
this study. This was a consequence of advice previously received from the FDA, which we interpreted to mean that the Agency would not
have granted clearance for the study to proceed unless we redesigned it to (i) enroll a patient population in which receipt of Paxlovid
or Lagevrio would be medically contraindicated, or (ii) compare Tafenoquine to placebo in patients taking a “standard of care”
regimen (defined by the FDA as Lagevrio or Paxlovid). The FDA’s position was somewhat surprising given that neither Paxlovid nor
Lagevrio is indicated for treatment of COVID-19 in low-risk patients. We determined that conducting our study in an alternate population
in the United States would be unfeasible, and that conducting an add-on-to standard of care study might not be Phase III enabling. Accordingly,
the Company made a decision to pivot back to continue commercialization of Arakoda for malaria, and further evaluation of the Arakoda
regimen of Tafenoquine for babesiosis and other diseases. We believe such an approach is both less risky and less expensive.
Moving forward, our general strategy to achieve
profitability and grow shareholder value has three facets: (i) increase sales of Arakoda; (ii) conduct clinical trials to expand the number
of patients who can use Tafenoquine for new indications in the future; and (iii) reposition small molecule therapeutics with good clinical
safety profiles for new indications.
Expansion of U.S. Arakoda Sales
Hiring of Chief Commercial Officer. In
February, 2024, we hired Kristen Landon to lead our commercial efforts to reintroduce Arakoda for malaria prevention and conduct new product
planning initiatives in tick-borne disease for babesiosis. We spent the first quarter analyzing the current landscape in the malaria prevention
market, conducting primary market research among providers and consumers, and assessing agency partners for a virtual/digital marketing
pilot program. Additionally, we kicked off a market assessment on the babesiosis space including desk top research and qualitative interviews
with Key Opinion Leaders in the Infectious Disease and Lyme Community.
| 31 |
Sorbera et al, Drugs of the Future 2005; 30:545-552. |
| 32 |
Low et. al., Lancet ID 2014; 14:706-715. |
| 33 |
Watanabe et al, Antiviral Research 2016; 10:e19. |
P&L Contract Review. We will conduct
a review of all of our supply chain and formulary contracts to determine whether it is possible to increase our margin on Arakoda without
increasing prices, or to compensate for any price adjustments which may be necessary to support repositioning efforts (see below).
Repositioning of Arakoda Relative to Malarone
and Generic Equivalent Atovaquone-Proguanil. A malaria demand study was conducted to assess the attractiveness and acceptability of
the Arakoda product profile and current pricing among health care providers and consumers. The product profile was well received among
both stakeholders; however, price sensitivity on out-of-pocket costs was noted among both groups. Generic atovaquone-proguanil, our primary
competitor is substantially cheaper than Arakoda for the average trip length (three weeks) and has superior formulary positioning (Tier
1 vs. Tier 3). However, generic-atovaquone proguanil does not provide the same level of confidence a traveler may experience from taking
a product with a convenient weekly dosing regimen during travel, that works everywhere in the world against all malaria species and drug
resistant strains, and which requires only a single dose for post-exposure prophylaxis upon return from a malarious area. The value those
advantages confer needs to be communicated with key stakeholders.
Market Segment Definition and Targeting.
We purchased market data to understand the malaria market landscape
over the past decade and identified the current prescribers of Malarone and the generic equivalent atovaquone-proguanil, the main generic
competitor to Arakoda for malaria prophylaxis. Beginning in the third quarter of 2024, we plan to reach out to prescribers covering the
top 80% of atovaquone-proguanil prescribers in order to educate them about the value proposition of Arakoda. We will also compile a list
of the top institutions/organizations that have ex-U.S. deployed workforces and internal occupational health and safety programs, and
target these organizations with messaging regarding the convenience and global effectiveness of Arakoda. We do not initially plan to target
U.S. government agencies as these organizations, such as the Department of Defense, are expected to be extremely price sensitive until
operational considerations justify the use of superior products – for example, the DOD used inexpensive doxycycline for malaria
prevention in the low malaria risk setting of Afghanistan, but chose superior weekly mefloquine, despite safety concerns, for the Ebola
mission to west Africa in 2014, where malaria rates were extremely high.
Digital Revamp and Collateral: We will work with an agency of record to develop a marketing strategy
for the proposed pilot and develop marketing assets that we believe best highlight the features and benefits of Arakoda, namely the convenience
of the travel and post-travel regimen, and global effectiveness. We are currently assessing a co-pay or point of sale offer for travelers
to offset out-of-pocket costs. We launched our Arakoda product website, which went live in April 2024.
Revised Forecast. We have developed
an internal forecast for the malaria and Babesiosis indications and have contracted a third party vendor to validate our
analyses.
Development of the Arakoda Regimen of Tafenoquine
for Babesiosis
In animal models, Tafenoquine monotherapy has
been shown to suppress acute babesiosis infections to the point where the immune system can control them following single or multiple
doses similar to those effective against malaria parasites, and longer regimens alone or in combination with atovaquone leads to complete
radical cure and to the conference of sterile immunity.34 In three case studies in individuals with immunosuppression and/or
refractory parasites, Tafenoquine alone or in combination with various standard of care antimalarials and antibiotics successfully cleared
parasites, leading to three consecutive negative PCR tests, and prevention of further relapses in two of three individuals.35
Our market research has revealed that recent sales growth for Arakoda is primarily attributable to organic growth in prescribing by Lyme
community prescribers for Chronic Babesiosis. Collectively these data suggest Tafenoquine might have utility alone or in combination as
treatment or post-exposure prophylaxis of babesiosis (both acute and chronic).
The Company is planning three clinical trials
to aid further development and commercialization of a Babesiosis indication for Tafenoquine. Trial 1 is a randomized, placebo-controlled,
evaluation of Tafenoquine (200 mg per day for a total of 800 mg) in patients hospitalized with babesiosis who are also taking standard
of care treatment (10 days of atovaquone-azithromycin). The primary endpoint will be time to clinical recovery of 11 common babesiosis
symptoms as reported by patients. The key secondary endpoint will be time to molecular cure as assessed by an FDA-approved Babesia nucleic
acid test that is used for blood donation screening. The study will enroll a minimum of 24 and up to 33 patients before an interim analysis
is conducted, which will include both a test of significance and a sample size re-estimation in case this is required. The study design
was reviewed by the FDA. We have signed a clinical trial agreement with Tufts Medical Group, and are negotiating similar agreement with
two other University Hospitals in the north-eastern United States the study sites). The first patient was randomized on June 25, 2024.
The earliest possible date that date would be available from the interim analysis would be January 31, 2025, assuming a minimum of 24
patients are enrolled prior to September 30, 2024. Further details are available on the clinicaltrials.gov website.36
| 34 |
Liu et al. Antimicrobial Agents Chemo 2021;65:e00204-21. Vydyam et al. J Infect Dis. 2024 Jan 3:jiad315. doi:10.1093/infdis/jiad315. |
| 35 |
Marcos et al. IDCases 2022;27:e01460; Rogers et al. Clin Infect Dis. 2022 Jun 10:ciac473, Prasad and Wormsner. Pathogens 2022;11:1015. |
| 36 |
See: https://classic.clinicaltrials.gov/ct2/show/NCT06207370. |
Trial 2 will be an expanded use study utilizing
commercially available Arakoda. The Company, if approved by an Institutional Review Board (“IRB,” also known as an ethics
committee), plans to offer up to one year of Arakoda at no cost to about 10 patients per year (i.e., immunocompromised patients who have
previously failed standard of care treatment). Informed consent will be obtained from patients to collect a blood sample for PCR testing
at the end of treatment, and patients will be asked to complete a babesiosis symptom questionnaire. The goal of the study is to generate
additional prospective data to confirm the observation by Krause et al in a recent publication that an extended regimen of Tafenoquine
cured 80% of immunocompromised patients with relapsing babesiosis. This study will commence utilizing proceeds from the current offering.
More details about the study can be found on the clinicaltrials.gov website.37
Trial 3 will be an expanded use study utilizing
commercially available Arakoda. The Company, if approved by an IRB, plans to offer an approximately two-month supply of Arakoda at no
cost to patients who have a clinical diagnosis, are willing to submit biological samples for testing, and answer babesiosis and standardized
fatigue inventories before and after treatment. The goal of this study will be to ascertain whether Arakoda treatment improves patient-reported
fatigue symptoms and quality of life in individuals who have a diagnosis of chronic fatigue, symptoms consistent in severity with those
suffered by chronic fatigue patients, and who have molecular evidence of infection with Babesia. This trial will be gated by the outcome
of an epidemiology study we have financed at North Carolina State University (see below) and will require additional funding
In
May 2024, we signed a research and collaboration agreement with North Carolina State University in which the College of Veterinary Medicine
will screen 300 archived blood samples from patients exhibiting symptoms consistent with chronic fatigue symptoms by PCR for the presence
of Babesia spp. In a second phase of the study, positive samples will be sequenced to determine which Babesia spp are present.
The data from this study will help define whether the incidence of Chronic Babesiosis may be more widespread than amongst PTLDS patients,
and also whether it is possible to define a study population for Trial 3 (described) cost effectively.
In March 2024, we initiated, in collaboration
with the North Carolina State University College of Veterinary Medicine, a pilot study of Tafenoquine for treatment of canine babesiosis
in the United States under a sponsored research program. Should this potential collaboration be successful, we believe that the data from
that study may provide supportive data for the clinical babesiosis development program, and could provide proof of concept for an expanded
study to prove utility for veterinary indications.
We believe, if the Company does not become capital-limited,
that the results of the above studies will come to fruition in the first quarter of 2026, potentially facilitating submission of a supplementary
new drug application (or other appropriate regulatory filing) to FDA, with the goal of obtaining marketing approval of Arakoda for treatment
of Babesiosis. If successful, this will allow the Company to actively market Arakoda for Babesiosis.
Parenteral Tafenoquine for Fungal Infections
We plan to support a series of studies in animal
models to determine whether single dose parenteral administration of Tafenoquine exhibits efficacy against Candida spp including
C. auris. These studies are being conducted under a sponsored research agreement with Monash University in Melbourne, Australia.
Combination Partner for Tafenoquine for Malaria
Most new antimalarial treatment products are developed
as drug combinations to proactively combat drug resistance. We believe that Tafenoquine, due to its long half-life and activity against
all parasite species and strains, would be an ideal partner in a drug combination. Recently, Kentucky Technology Inc. (“KTI”),
completed Phase IIA studies in P. vivax malaria, in which they evaluated the safety and efficacy of SJ733, their ATP4 inhibitor
in combination with Tafenoquine as the combination partner drug. It was recently announced that the SJ733 development program would be
partially supported by a grant from the Global Health Innovative Technology Fund (“GHIT”). As part of its shares for services
agreement with KTI, the Company recently received a detailed feasibility assessment and business plan for the project, including an assessment
of potential PRV eligibility, and is considering next steps in relation to potential involvement in this project.
| 37 |
See: https://clinicaltrials.gov/study/NCT06478641. |
Celgosivir for Antiviral Diseases
Reviewing prior studies of Celgosivir for Zika,
Dengue and RSV, it is evident that the drug protects against the pathological effects of viruses through a combination of anti-inflammatory
and antiviral effects. These properties suggest it might have a beneficial effect in several viral diseases. Celgosivir is synthesized
from Castanospermine, which is obtained from botanical sources in low yield, making its inherent cost of goods potentially high. Castanospermine
is also quite water soluble, making it amenable to intravenous formulation. We plan to conduct a proof-of-concept study in a hamster-COVID-19
model to evaluate whether parenterally administered Castanospermine can ameliorate the pathological effects of SARS CoV-2 via modulation
of cytokine response to infection. Following this offering this project will be added to our statement of work for our services agreement
with Florida State University Research Foundation (“FSURF”), and will commence when there are sufficient proceeds from the
sale of FSURF’s 60P shares to support this research. The data generated from the study will allow us to assess whether to move forward
with IND enabling studies of parenteral Castanospermine (or Celgosivir) for viral indications.
Post-Marketing
Requirements
We have an FDA post-marketing requirement
to conduct a malaria prophylaxis study of Arakoda in pediatric and adolescent subjects. We proposed to the FDA, in late 2021, that this
might not be safe to execute given that malaria prevention is administered to asymptomatic individuals and that methemoglobinemia (damage
to the hemoglobin in blood that carries oxygen) occurred in 5% of patients, and exceeded a level of 10% in 3% of individuals in a study
conducted by another sponsor in pediatric subjects with symptomatic vivax malaria.38 The FDA has asked us to propose an alternate
design, for which we submitted a concept protocol in the fourth quarter of 2022, and submitted a full protocol in July, 2024. We estimate
the cost of conducting the study proposed by the FDA, if conducted in the manner suggested by the FDA, would be $2 million, and, due
to the time periods required to secure protocol approvals from the FDA and Ethics Committees, could not be initiated any earlier than
the first quarter of 2026.
Capitalization
and Future Financing
We plan to raise up to $15,000,000 million using
the base prospectus and the prospectus supplement in connection with future sales made pursuant to the Sales Agreement. It is possible
that the funds that are able to be raised using the base prospectus and prospectus supplement may be insufficient to achieve all our
objectives. Therefore, we will be seeking to raise additional funding as non-dilutively as possible, for example in the form of royalty
or debt-based funding or funding from non-profit groups interested in tick-borne diseases. There is no assurance that funds will be available
on acceptable terms, or that additional dilutive funding will not be required.
| 38 |
Velez
et al 2021 - Lancet Child Adolesc Health 2022; 6: 86–95. |
Intellectual Property
We are co-owners, with the U.S. Army, of patents
in the United States and certain foreign jurisdictions directed toward use of Tafenoquine for malaria and have obtained an exclusive worldwide
license from the U.S. Army to practice these inventions. We also have an exclusive worldwide license to use manufacturing information
and non-clinical and clinical data that the U.S. Army possesses relating to use of Tafenoquine for all therapeutic applications and uses
excluding radical cure of symptomatic vivax malaria. We have submitted patent applications in the United States and certain foreign jurisdictions
for use of Tafenoquine for COVID-19, fungal lung infections, tick-borne diseases, and other infectious and non-infectious diseases in
which induction of host cytokines/inflammation is a component of the disease process. The United States Patent and Trademark Office (“USPTO”)
issued our first COVID-19 patent for Tafenoquine in 2023. We have optioned or licensed patents involving Celgosivir for the treatment
and prevention of Dengue (from the National University of Singapore), COVID-19 & Zika (Florida State University), and have pending
patent applications related to Celgosivir for RSV. We have optioned or own manufacturing methods related to Celgosivir. A detailed list
of our intellectual property is as follows:
Patents
| Title |
|
Patent No. |
|
Country |
|
Status |
|
US Patent Date |
|
Application No. |
|
|
Estimated/
Anticipated
Expiration
Date |
| Dosing Regimen For Use Of Celgosivir As An Antiviral Therapeutic For Dengue Virus Infections |
|
2013203400 |
|
Australia |
|
Granted |
|
|
|
|
2013203400+ |
|
|
10-April-2033* |
| Novel Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
2014228035 |
|
Australia |
|
Granted |
|
|
|
|
2014228035 |
|
|
14-Mar-2034* |
| Novel Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
MY-170991-A |
|
Malaysia |
|
Granted |
|
|
|
|
PI2015002372 |
|
|
14-Mar-2034* |
| Novel Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
378015 |
|
Mexico |
|
Granted |
|
|
|
|
MX/a/2015/013115 |
|
|
14-Mar-2034* |
| Novel Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
11201507254V |
|
Singapore |
|
Granted |
|
|
|
|
11201507254V |
|
|
14-Mar-2034* |
| Novel Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
Pending |
|
Singapore |
|
Pending |
|
|
|
|
10201908089V |
|
|
14-Mar-2034* |
| Novel Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
9763921 |
|
US |
|
Issued |
|
9/19/2017 |
|
|
14/772,873 |
|
|
14-Mar-2034^ |
| Novel Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
10517854 |
|
US |
|
Issued |
|
12/31/2019 |
|
|
15/706,845 |
|
|
14-Mar-2034^ |
| Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
11219616 |
|
US |
|
Issued |
|
1/11/2022 |
|
|
16/725,387 |
|
|
14-Mar-2034^ |
| Novel Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naïve Subjects |
|
2015358566 |
|
Australia |
|
Granted |
|
|
|
|
2015358566 |
|
|
02-Dec-2035* |
| Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naïve Subjects |
|
2968694 |
|
Canada |
|
Granted |
|
|
|
|
2968694 |
|
|
02-Dec-2035* |
| Title |
|
Patent No. |
|
Country |
|
Status |
|
US Patent Date |
|
|
Application No. |
|
|
Estimated/
Anticipated
Expiration
Date |
| Novel Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naïve Subjects |
|
10342791 |
|
US |
|
Issued |
|
7/9/2019 |
|
|
15/532,280 |
|
|
02-Dec-2035^ |
| Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naive Subjects |
|
10888558 |
|
US |
|
Issued |
|
1/12/2021 |
|
|
16/504,533 |
|
|
02-Dec-2035^ |
| Novel Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naïve Subjects |
|
Pending |
|
Singapore |
|
Pending |
|
|
|
|
10201904908Q |
|
|
02-Dec-2035* |
| Novel Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naïve Subjects |
|
Pending |
|
EP |
|
Pending |
|
|
|
|
15865264.4 |
|
|
02-Dec-2035* |
| Novel Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naïve Subjects |
|
Pending |
|
Hong Kong |
|
Pending |
|
|
|
|
18103081.4 |
|
|
02-Dec-2035* |
| Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naive Subjects |
|
11,744,828 |
|
US |
|
Issued |
|
9/5/2023 |
|
|
17/145,530 |
|
|
02-Dec-2035^ |
| Novel Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naïve Subjects |
|
Pending |
|
New Zealand |
|
Pending |
|
|
|
|
731813 |
|
|
02-Dec-2035* |
| Regimens of Tafenoquine for Prevention of Malaria in Malaria-Naive Subjects |
|
Pending |
|
US |
|
Pending |
|
|
|
|
18/240,049 |
|
|
02-Dec-2035^ |
| Novel Dosing Regimens Of Celgosivir For The Prevention Of Dengue |
|
2016368580 |
|
Australia |
|
Granted |
|
|
|
|
2016368580 |
|
|
09-Dec-2036* |
| Novel Dosing Regimens Of Celgosivir For The Prevention Of Dengue |
|
Pending |
|
Singapore |
|
Pending |
|
|
|
|
10201912141Y |
|
|
09-Dec-2036* |
| Dosing Regimens Of Celgosivir For The Prevention Of Dengue |
|
11000516 |
|
US |
|
Issued |
|
5/11/2011 |
|
|
16/060,945 |
|
|
09-Dec-2036^ |
| Methods For The Treatment And Prevention Of Lung Infections By Administration Of Tafenoquine |
|
Pending |
|
EP |
|
Pending |
|
|
|
|
21764438.4 |
|
|
02-Mar-2041* |
| Title |
|
Patent No. |
|
Country |
|
Status |
|
US Patent Date |
|
|
Application No. |
|
|
Estimated/
Anticipated
Expiration
Date |
| Methods For The Treatment And Prevention Of Lung Infections By Administration Of Tafenoquine |
|
Pending |
|
China |
|
Pending |
|
|
|
|
202180029643.7 |
|
|
02-Mar-2041* |
| Methods For The Treatment And Prevention Of Lung Infections By Administration Of Tafenoquine |
|
Pending |
|
Australia |
|
Pending |
|
|
|
|
2021231743 |
|
|
02-Mar-2041* |
| Methods For The Treatment And Prevention Of Lung Infections Caused By Gram-Positive Bacteria, Fungus, Or Virus By Administration Of Tafenoquine |
|
Pending |
|
Hong Kong |
|
Pending |
|
|
|
|
62023078645.6 |
|
|
02-Mar-2041* |
| Methods For The Treatment And Prevention Of Lung Infections Caused By Gram-Positive Bacteria, Fungus, Or Virus By Administration Of Tafenoquine |
|
11,633,391 |
|
US |
|
Issued |
|
4/25/2023 |
|
|
17/189,544 |
|
|
05-May-2041^ |
| Methods For The Treatment And Prevention Of Lung Infections Caused By Gram-Positive Bacteria, Fungus, Or Virus By Administration Of Tafenoquine |
|
Pending |
|
US |
|
Pending |
|
|
|
|
18/300,805 |
|
|
02-Mar-2041^ |
| Methods For The Treatment And Prevention Of Lung Infections Caused By Fungus By Administration Of Tafenoquine |
|
Pending |
|
US |
|
Pending |
|
|
|
|
17/683,679 |
|
|
01-Mar-2041^ |
| Methods For The Treatment And Prevention Of Lung Infections Caused By Sars-Cov-2 Virus By Administration Of Tafenoquine |
|
Pending |
|
US |
|
Pending |
|
|
|
|
17/683,718 |
|
|
01-Mar-2041^ |
| Treatment Of Human Coronavirus Infections Using Alpha-Glucosidase Glycoprotein Processing Inhibitors |
|
11369592 |
|
US |
|
Issued |
|
6/28/2022 |
|
|
17/180,140# |
|
|
19-Feb-2041^ |
| Treatment Of Human Coronavirus Infections Using Alpha-Glucosidase Glycoprotein Processing Inhibitors |
|
Pending |
|
US |
|
Pending |
|
|
|
|
17/664,693# |
|
|
19-Feb-2041^ |
| Treatment Of Human Coronavirus Infections Using Alpha-Glucosidase Glycoprotein Processing Inhibitors |
|
Pending |
|
EP |
|
Pending |
|
|
|
|
2021757552# |
|
|
19-Feb-2041* |
| Methods To Treat Respiratory Infection Utilizing Castanospermine Analogs |
|
Pending |
|
PCT |
|
Pending |
|
|
|
|
PCT/US23/26884 |
|
|
05-Jul-2043* |
| Methods To Treat Respiratory Infection Utilizing Castanospermine Analogs |
|
Pending |
|
US |
|
Pending |
|
|
|
|
18/218,202 |
|
|
05-Jul-2043^ |
| Title |
|
Patent No. |
|
Country |
|
Status |
|
US Patent Date |
|
|
Application No. |
|
|
Estimated/
Anticipated
Expiration
Date |
| Methods For The Treatment And Prevention Of Diseases Or Infections With MCP-1 Involvement By Administration Of Tafenoquine |
|
Pending |
|
PCT |
|
Pending |
|
|
|
|
PCT/US23/34169 |
|
|
30-Sep-2043* |
| Methods For The Treatment And Prevention Of Diseases Or Infections With MCP-1 Involvement By Administration Of Tafenoquine |
|
Pending |
|
US |
|
Pending |
|
|
|
|
18/375,070 |
|
|
30-Sep-2043^ |
| Treatment Of Zika Virus Infections Using Alpha Glucosidase Inhibitors |
|
10,328,061+ |
|
US |
|
Issued |
|
|
|
|
15/584,952+ |
|
|
2-May-2037^ |
| Treatment Of Zika Virus Infections Using Alpha Glucosidase Inhibitors |
|
10,561,642+ |
|
US |
|
Issued |
|
|
|
|
15/856,377+ |
|
|
2-May-2037^ |
| Methods For The Treatment And Prevention Of Non-Viral Tick-Borne Diseases And Symptoms Thereof |
|
Pending |
|
PCT |
|
Pending |
|
|
|
|
PCT/US24/25436 |
|
|
19-Apr-2044* |
| Methods For The Treatment And Prevention Of Non-Viral Tick-Borne Diseases And Symptoms Thereof |
|
Pending |
|
PCT |
|
Pending |
|
|
|
|
PCT/US24/25458 |
|
|
19-Apr-2044* |
| Methods For The Treatment And Prevention Of Non-Viral Tick-Borne Diseases And Symptoms Thereof |
|
Pending |
|
PCT |
|
Pending |
|
|
|
|
PCT/US24/25472 |
|
|
19-Apr-2044* |
| Methods For The Treatment And Prevention Of Non-Viral Tick-Borne Diseases And Symptoms Thereof |
|
Pending |
|
US |
|
Pending |
|
|
|
|
18/640,611 |
|
|
19-Apr-2044^ |
| Methods For The Treatment And Prevention Of Non-Viral Tick-Borne Diseases And Symptoms Thereof Of Treating Babesiosis |
|
Pending |
|
US |
|
Pending |
|
|
|
|
18/640,657 |
|
|
19-Apr-2044^ |
| Methods For The Treatment And Prevention Of Non-Viral Tick-Borne Diseases And Symptoms Thereof Of Treating Babesiosis |
|
Pending |
|
US |
|
Pending |
|
|
|
|
18/640,695 |
|
|
19-Apr-2044^ |
| * = |
For foreign patents and applications, the estimated and/or anticipated patent expiration is the date that is twenty years from the PCT filing date. For all issued Australian patents, this estimated date was also confirmed through the Australian patent office web database. |
| ^ = |
For issued U.S. patents, the estimated patent expiration was calculated using information from the front cover of the patent, i.e., 20 years from the date of the nonprovisional filing plus any listed Patent Term Adjustment less any time disclaimed through a Terminal Disclaimer. For pending U.S. applications, the anticipated patent expiration is the date twenty years from the earliest nonprovisional filing date and does not account for possible Patent Term Adjustment (PTA), Patent Term Extension (PTE), or Terminal Disclaimers. |
| & = |
For U.S. provisional applications that are not yet the subject of a nonprovisional or PCT application, the anticipated patent expiration was determined using the assumption that a non-provisional application or PCT will be filed one year after filing the provisional application with a term lasting twenty years from the date of that nonprovisional or PCT filing. This does not account for possible Patent Term Adjustment (PTA), Patent Term Extension (PTE), or Terminal Disclaimers. |
| + = |
60 Degrees Pharmaceuticals, Inc. is not a listed Applicant and Geoffrey S. Dow, Ph.D. is not a listed inventor. |
| # = |
60 Degrees Pharmaceuticals, Inc. is not a listed Applicant, but Geoffrey S. Dow, Ph.D. is a listed inventor. |
All patents not designated with a “+”
list Geoffrey S. Dow, Ph.D. as an inventor.
All patents not designated with a “+”
or a “#” list 60 Degrees Pharmaceuticals, Inc. as an applicant.
All estimated patent expiration dates and anticipated
patent expiration assume payment of any maintenance/annuity fees during the patent term.
Trademarks
| Country |
|
Mark |
|
Status |
|
Application
Number |
|
Date
Filed |
|
Registration
Date |
|
Registration
Number |
|
BIR
Ref Number |
|
Due
Date |
|
Due
Date Description |
| Australia |
|
KODATEF |
|
Registered |
|
1774631 |
|
2-Jun-16 |
|
6/2/2016 |
|
1774631 |
|
0081716-000029 |
|
2-Jun-26 |
|
Renewal
Due |
| Canada |
|
KODATEF |
|
Registered |
|
1785098 |
|
1-Jun-16 |
|
11/26/2019 |
|
TMA1,064,371 |
|
0081716-000028 |
|
26-Nov-29 |
|
Renewal
Due |
| Canada |
|
ARAKODA |
|
Registered |
|
1899317 |
|
15-May-18 |
|
8/20/2020 |
|
TMA1,081,180 |
|
0081716-000053 |
|
20-Aug-30 |
|
Renewal
Due |
| China |
|
KODATEF |
|
Registered |
|
20842242 |
|
2-Aug-16 |
|
9/28/2017 |
|
20842242 |
|
0081716-000035 |
|
27-Sep-27 |
|
Renewal
Due |
| European
Union |
|
KODATEF |
|
Registered |
|
15508872 |
|
3-Jun-16 |
|
9/21/2016 |
|
15508872 |
|
0081716-000034 |
|
3-Jun-26 |
|
Renewal
Due |
| European
Union |
|
ARAKODA |
|
Registered |
|
17900852 |
|
16-May-18 |
|
9/20/2018 |
|
17900852 |
|
0081716-000054 |
|
16-May-28 |
|
Renewal
Due |
| Israel |
|
KODATEF |
|
Registered |
|
285476 |
|
6-Jun-16 |
|
6/6/2016 |
|
285476 |
|
0081716-000033 |
|
6-Jun-26 |
|
Renewal
Due |
| New
Zealand |
|
KODATEF |
|
Registered |
|
1044407 |
|
7-Jun-16 |
|
12/8/2016 |
|
1044407 |
|
0081716-000031 |
|
6-May-26 |
|
Renewal
Due |
| Russian
Federation |
|
KODATEF |
|
Registered |
|
2016720181 |
|
6-Jun-16 |
|
7/10/2017 |
|
623174 |
|
0081716-000032 |
|
6-Jun-26 |
|
Renewal
Due |
| Singapore |
|
KODATEF |
|
Registered |
|
40201707950V |
|
2-May-17 |
|
11/8/2017 |
|
40201707950V |
|
0081716-000040 |
|
2-May-27 |
|
Renewal
Due |
| United
Kingdom |
|
ARAKODA |
|
Registered |
|
17900852 |
|
16-May-18 |
|
9/20/2018 |
|
UK00917900852 |
|
0081716-000054 |
|
16-May-28 |
|
Renewal
Due |
| United
Kingdom |
|
KODATEF |
|
Registered |
|
15508872 |
|
3-Jun-16 |
|
9/21/2016 |
|
UK00915508872 |
|
0081716-000072 |
|
3-Jun-26 |
|
Renewal
Due |
| United
States of America |
|
TQ
100 & TABLET DESIGN |
|
Registered |
|
87608493 |
|
14-Sep-17 |
|
9/11/2018 |
|
5562900 |
|
0081716-000037 |
|
11-Sep-24 |
|
Section
8 & 15 Due |
| United
States of America |
|
ARAKODA |
|
Registered |
|
87688137 |
|
16-Nov-17 |
|
12/31/2019 |
|
5950691 |
|
0081716-000050 |
|
31-Dec-25 |
|
Section
8 & 15 Due |
| United
States of America |
|
KODATEF |
|
Allowed
- 02/16/2021 |
|
90072885 |
|
24-Jul-20 |
|
01/03/2024 |
|
|
|
0081716-000069 |
|
16-Aug-23 |
|
Abandoned |
| United
States of America |
|
KODATEF |
|
Pending |
|
98363219 |
|
24-Jan-18 |
|
01/18/2024 |
|
|
|
0081716-000074 |
|
|
|
|
Key
Relationships & Licenses
On May 30, 2014, we entered into the Exclusive
License Agreement (the “2014 NUS-SHS Agreement”) with National University of Singapore (“NUS”) and Singapore Health
Services Pte Ltd (“SHS”) in which we were granted a license from NUS and SHS with respect to their share of patent rights
regarding “Dosing Regimen for Use of Celgosivir as an Antiviral Therapeutic for Dengue Virus Infection” to develop, market
and sell licensed products. The 2014 NUS-SHS Agreement continues in force until the expiration of the last to expire of any patents under
the patent rights unless terminated earlier in accordance with the 2014 NUS-SHS Agreement. We are obligated to pay royalties at the rate
of 1.5% of gross sales.
On July 15, 2015, we entered into the
Exclusive License Agreement with the U.S. Army Medical Materiel Development Activity (the “U.S. Army”), which was
subsequently amended (the “U.S. Army Agreement”), in which we obtained a license to develop and commercialize the
licensed technology with respect to all therapeutic applications and uses excluding radical cure of symptomatic vivax malaria. This
exclusion does not impact our ability to market Arakoda for the FDA-approved use, which is the prevention of malaria utilizing the
indicated dose in asymptomatic individuals traveling to high-malaria or malaria-prone regions (whereas the license exclusion relates
to its use to treat symptomatic vivax malaria in a patient already presenting with that disease). The term of the U.S. Army
Agreement will continue until the expiration of the last to expire of the patent application or valid claim of the licensed
technology, or 20 years from the start date of the U.S. Army Agreement, unless terminated earlier by the parties. We will be
required to make a minimum annual royalty payment of 3% of net sales (as defined in the U.S. Army Agreement) for net sales < $35
million, and 5% of net sales greater than $35 million, with US government sales excluded from the definition of net sales. In
addition, we must pay fees upon the achievement of certain milestones, including a sales-based milestone fee of $75,000 once cumulative net sales from all sources exceeds $6 million (which milestone was achieved during the year ended December
31, 2023), $100,000 if we are
acquired or merge, and regulatory approval milestone payments once marketing authorizations are achieved in Canada ($5,000) and
Europe ($5,000). Also, we will be required to obtain the U.S. Army Medical Materiel Development Activity’s consent prior to a
change of control of the Company, which consent was obtained on September 2, 2022.
On September 15, 2016, we entered into the Exclusive
License Agreement (the “2016 NUS-SHS Agreement”) with National University of Singapore and Singapore Health Services Pte Ltd
(“SHS”) in which we were granted a license from NUS and SHS with respect to their share of patent rights regarding “Novel
Dosing Regimens of Celgosivir for The Prevention of Dengue” to develop, market and sell licensed products. The 2016 NUS-SHS Agreement
continues in force until the expiration of the last to expire of any patents under the patent rights unless terminated earlier in accordance
with the 2016 NUS-SHS Agreement. We are obligated to pay at the rate of 1.5% of gross sales or minimum annual royalty ($5,000 in 2022
and $15,000 in 2023). In July 2022, we renegotiated the timing of a license fee of $85,000 Singapore Dollars, payable to NUS, such that
payment would be due at the earlier of (i) enrollment of a patient in a Phase II clinical trial involving Celgosivir, (ii) two years from
the agreement date and (iii) an initial public offering.
On February 15, 2021, we entered into the Inter-Institutional
Agreement with FSURF (the “FSURF Agreement”) in which FUSRF granted us the right to manage the licensing of intellectual property
created at FSURF. The term of the FSURF Agreement expires five years from February 15, 2021. After deduction of a 5% administrative fee
by FSURF, capped at $15,000 annually, and reimbursement of patent prosecution expenses, we will receive 20% of license income and FSURF
will receive 80% of license income. Payments of license income shall be paid in U.S. dollars quarterly each year. On February 19, 2021,
we entered into an agreement with FSURF, subsequently amended on February 15, 2023, and again on March 25, 2024, that collectively granted
an option, effective through March 24, 2025, to us to license methods for purifying Castanospermine and its use for the treatment of COVID-19.
On August 19, 2021, we entered into an agreement with FSURF, subsequently amended on February 15, 2023, and again on March 25, 2024, that
collectively granted an option, effective through March 24, 2025, to us to license a patent relating to the use of alpha glucosidase inhibitors
(including Castanospermine and Celgosivir) for treatment of Zika infections.
Ending upon July 12, 2033 or the conversion or
redemption in full of all of the shares of Series A Preferred Stock owned by Knight, we will pay Knight a royalty equal to 3.5% of our
net sales, where “net sales” has the same meaning as in the U.S. Army Agreement. Upon
succeeding with the qualified IPO, at the end of the quarter and each thereafter the royalty will be calculated, and payment will be
made within fifteen days.
On February 13, 2024, our majority-owned Australian
subsidiary, 60P Australia Pty Ltd, and Monash University entered into the Research Services Agreement (the “Agreement”) in
which Monash University agreed to provide research services, including among other things, testing the efficacy of Tafenoquine against
candidemia, confirming suitable fungal infection dosage and determining the pharmacokinetics of Tafenoquine following intraperitoneal
drug administration (collectively, the “Services”). The commencement date of the Agreement was effective as of February 5,
2024, and the commencement of experiments was May 2024 and the anticipated completion date is on November 30, 2024. The Company agreed
to pay Monash University $90,167 AUD on April 1, 2024 and $90,167 AUD upon the completion of the Services.
On March 20, 2024, we signed a sponsored research
agreement with North Carolina State University to conduct a pilot study to evaluate the efficacy of Tafenoquine in canine babesiosis.
The research is expected to be completed by March 30, 2026. The Company will retain ownership of all data and inventions related to the
study, subject to retained right of North Carolina State University to utilize study data or research use and publications. For a six-month
period following notification by the University, the Company retains first right of refusal to negotiate a license to utilize any inventions
or data generated by the University relevant to Tafenoquine but not occurring as a direct result of performing the planned studies. The
Company agreed to pay North Carolina State University $12,000 upon contract execution, $8,000 around October 1st, 2024, then
$3,869 around April 1st, 2025 when work is expected to be completed.
On May 10, 2024, we entered into a sponsored research
agreement with North Carolina State University to conduct a study to evaluate the incidence of Babesia infection amongst archived blood
samples from patients with chronic fatigue and neurocognitive problems. The research will be completed by May 31, 2025. The Company retains
the right to use all study data, joint and University-owned inventions for non-commercial purposes and first right of refusal to negotiate
a royalty bearing license for commercial purposes for any university own intellectual property or ownership interest in jointly-owned
intellectual property. The Company agreed to pay North Carolina State University $37,620 upon contract execution, $22,572 after six months,
then $15,048 upon completion of the contract at twelve months.
On May 29, 2024, we signed a clinical
trial agreement with Tufts Medicine, Inc, which specifies the terms on which Tufts will act as a clinical trial site for our Tafenoquine-Babesiosis
study. The Company retains the first right to negotiate an exclusive license to any intellectual property owned by Tufts Medicine, Inc,
arising from the study.
Corporate
Structure
60
Degrees Pharmaceuticals, Inc. is a Delaware corporation that was incorporated on June 1, 2022.
On
June 1, 2022, 60 Degrees Pharmaceuticals, LLC, a District of Columbia limited liability company (“60P LLC”), entered into
the Agreement and Plan of Merger with 60 Degrees Pharmaceuticals, Inc., pursuant to which 60P LLC merged into 60 Degrees Pharmaceuticals,
Inc. The value of each outstanding member’s membership interest in 60P LLC was correspondingly converted into common stock of 60
Degrees Pharmaceuticals, Inc., par value $0.0001 per share, with a cost-basis equal to $5.00 per share.
Our
majority-owned subsidiary, 60P Australia Pty Ltd, an Australian proprietary company limited by shares (“60P Australia”),
was formed and registered in Queensland on December 3, 2013, and conducts operations in Australia.
60P
Australia previously solely owned a Singaporean subsidiary company, 60P Singapore Pte. Ltd., which dissolved at our election in the second
quarter of 2022.
Going
Concern
Our
independent auditors have issued a report raising substantial doubt of our ability to continue as a going concern. We anticipate that
we will require additional capital to continue as a going concern and expand our operations in accordance with our current business plan.
Suppliers
We
have quality and contract manufacturing agreements relating to Arakoda in place with Piramal Enterprises Limited (API, tablets) and PCI
Pharma Services (secondary packaging) (“PCI”) and supply/quality/pharmacovigilance agreements in place with Biocelect Pty
Ltd, Scandinavian Biopharma, and Knight Therapeutics Inc. (to allow supply of Arakoda/Kodatef to Australia, Europe and Canada/Israel/Latin
America and Russia, respectively). As of the date of this prospectus, we have not supplied any of our products to Russia nor do we anticipate
supplying any of our products to Russia in the near future.
Information
Regarding our Capitalization
As of July 12, 2024, we had 12,206,116
shares of common stock issued and outstanding. Additional information regarding our issued and outstanding securities may be found under
“Market for Common Equity and Related Stockholder Matters” and “Description of Securities.”
Unless
otherwise specifically stated, information throughout this prospectus does not assume the exercise of outstanding options or warrants
to purchase shares of our common stock.
Corporate
Information
Our
principal executive offices are located at 1025 Connecticut Avenue NW Suite 1000, Washington, D.C. 20036. Our corporate website address
is 60degreespharma.com. Our telephone number is (202) 327-5422. The information included on our website is not part of this prospectus.
Implications
of Being an Emerging Growth Company and a Smaller Reporting Company
We
are an “emerging growth company,” as defined in the JOBS Act. We will remain an emerging growth company until the earlier
of (i) the last day of the fiscal year following the fifth anniversary of the date of the first sale of our common stock pursuant to
an effective registration statement under the Securities Act; (ii) the last day of the fiscal year in which we have total annual gross
revenues of $1.235 billion or more; (iii) the date on which we have issued more than $1 billion in nonconvertible debt during the previous
three years; or (iv) the date on which we are deemed to be a large accelerated filer under applicable SEC rules. We expect that we will
remain an emerging growth company for the foreseeable future, but cannot retain our emerging growth company status indefinitely and will
no longer qualify as an emerging growth company on or before the last day of the fiscal year following the fifth anniversary of the date
of the first sale of our common stock pursuant to an effective registration statement under the Securities Act. For so long as we remain
an emerging growth company, we are permitted and intend to rely on exemptions from specified disclosure requirements that are applicable
to other public companies that are not emerging growth companies.
These
exemptions include:
| |
● |
being
permitted to provide only two years of audited financial statements, in addition to any required unaudited interim financial statements,
with correspondingly reduced “Management’s Discussion and Analysis of Financial Condition and Results of Operations”
disclosure; |
| |
● |
not
being required to comply with the requirement of auditor attestation of our internal controls over financial reporting; |
| |
● |
not
being required to comply with any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory
audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial
statements; |
| |
● |
reduced
disclosure obligations regarding executive compensation; and |
| |
● |
not
being required to hold a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments
not previously approved. |
An
emerging growth company can take advantage of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act to
comply with new or revised accounting standards. This allows an emerging growth company to delay the adoption of certain accounting standards
until those standards would otherwise apply to private companies. We have elected to avail ourselves of this extended transition period
and, as a result, we will not be required to adopt new or revised accounting standards on the dates on which adoption of such standards
is required for other public reporting companies.
We
are also a “smaller reporting company” as defined in Rule 12b-2 of the Securities Exchange Act of 1934, as amended (the “Exchange
Act”), and have elected to take advantage of certain of the scaled disclosure available for smaller reporting companies. We will
remain a smaller reporting company until the end of the fiscal year in which (1) we have a public common equity float of more than $250 million,
or (2) we have annual revenues for the most recently completed fiscal year of more than $100 million and a public common equity
float or public float of more than $700 million. We also would not be eligible for status as a smaller reporting company if we become
an investment company, an asset-backed issuer or a majority-owned subsidiary of a parent company that is not a smaller reporting company.
We
have elected to take advantage of certain of the reduced disclosure obligations in the registration statement of which this prospectus
is a part and may elect to take advantage of other reduced reporting requirements in future filings. As a result, the information that
we provide to our stockholders may be different from what you might receive from other public reporting companies in which you hold equity
interests.
RISK
FACTORS
Investing
in our securities involves a high degree of risk. Before deciding whether to invest in our securities, you should consider carefully
the risks and uncertainties described under this section and the section titled “Risk Factors” contained in the applicable
prospectus supplement, and discussed under the section titled “Risk Factors” contained in our most recent Annual Report
on Form 10-K and in our most recent Quarterly Report on Form 10-Q, as well as any amendments thereto reflected in subsequent filings
with the SEC, which are incorporated by reference into this prospectus in their entirety, together with other information in this prospectus,
and the documents incorporated by reference that we may authorize for use in connection with a specific offering. The risks described
in this section and in these documents are not the only ones we face, but those that we consider being material. There may be other unknown
or unpredictable economic, business, competitive, regulatory or other factors that could have material adverse effects on our future
results. Past financial performance may not be a reliable indicator of future performance, and historical trends should not be used to
anticipate results or trends in future periods. If any of these risks actually occur, our business, financial condition, results of operations
or cash flow could be harmed. This could cause the trading price of our securities to decline, resulting in a loss of all or part of
your investment. Please also read carefully the section above titled “Cautionary Note Regarding Forward-Looking Statements.”
USE
OF PROCEEDS
We
intend to use the net proceeds from the sale of the securities as set forth in the applicable prospectus supplement.
DIVIDEND
POLICY
We
have never declared or paid any cash dividends on our capital stock. We intend to retain future earnings, if any, to finance the operation
of our business and do not anticipate paying any cash dividends in the foreseeable future. Any future determination related to our dividend
policy will be made at the discretion of our Board after considering our financial condition, results of operations, capital requirements,
business prospects and other factors our Board deems relevant, and subject to the restrictions contained in any future financing instruments.
THE
SECURITIES WE MAY OFFER
We
may offer and sell, at any time and from time to time:
| |
● |
shares
of our common stock; |
| |
● |
shares
of our preferred stock; |
| |
● |
warrants
to purchase shares of our common stock, preferred stock and/or debt securities; |
| |
● |
debt
securities consisting of debentures, notes or other evidences of indebtedness; |
| |
● |
units
consisting of a combination of the foregoing securities; or |
| |
● |
any
combination of these securities. |
The
terms of any securities we offer will be determined at the time of sale. We may issue debt securities that are exchangeable for and/or
convertible into common stock or any of the other securities that may be sold under this prospectus. When particular securities are offered
by us, a supplement to this prospectus will be filed with the SEC, which will describe the terms of the offering and sale of the offered
securities.
We may offer up to $15,000,000 of securities
under this prospectus. If securities are offered as units, we will describe the terms of the units in a prospectus supplement.
DESCRIPTION
OF CAPITAL STOCK
General
The
following description summarizes some of the terms of our capital stock. Because it is only a summary, it does not contain all the information
that may be important to you and is subject to and qualified in its entirety by reference to our certificate of incorporation, as corrected
(“Certificate of Incorporation”) and amended and restated bylaws (“Bylaws”), which are filed as exhibits to our
most recent Annual Report on Form 10-K and are incorporated by reference herein. We encourage you to read our Certificate of Incorporation
and Bylaws for additional information.
As of July 12, 2024, our authorized capital stock
presently consists of 150,000,000 shares of common stock, par value $0.00001 per share, and 1,000,000 shares of “blank check”
preferred stock, par value $0.00001 per share, of which 80,965 shares of preferred stock have been designated as “Series A Non-Voting
Convertible Preferred Stock” (“Series A Preferred Stock”).
Common
Stock
The
holders of our common stock are entitled to the following rights:
Voting
Rights. Each share of our common stock entitles its holder to one vote per share on all matters to be voted or consented upon
by the stockholders.
Dividend
Rights. Subject to limitations under Delaware law, holders of our common stock are entitled to receive ratably such dividends
or other distributions, if any, as may be declared by our Board out of funds legally available therefor.
Liquidation
Rights. In the event of liquidation, dissolution or winding up of our business, the holders of our common stock are entitled
to share ratably in the assets available for distribution after the payment of all of our debts and other liabilities.
Other
Matters. The holders of our common stock have no subscription, redemption or conversion privileges; in addition, such common
stock does not entitle its holders to pre-emptive rights. All of the outstanding shares of our common stock are fully paid and non-assessable.
Preferred
Stock
Our Certificate of Incorporation authorizes 1,000,000
shares of “blank check” preferred stock, par value $0.0001 per share. We have currently authorized 80,965 shares of Series
A Preferred Stock with the following terms and rights: (i) 6% dividend, (ii) non-voting; (iii) not redeemable; and (iv) convertible into
shares of common stock, solely at the Company’s discretion, determined by (A) multiplying the number of shares of Series A Preferred
Stock to be converted by $100, (B) adding to the result all accrued and accumulated and unpaid dividends on such shares to be converted,
if any, and then (C) dividing the result by a price equal to the lower of (1) $100, (2) the price paid for the shares of common stock
in the IPO and (3) the 10-day volume weighted average share price immediately preceding our election to convert the shares of Series
A Preferred Stock; provided that the conversion of the shares of Series A Preferred Stock does not result in the holder’s ownership
of common stock exceeding 19.9%. As of July 12, 2024, there are 78,803 shares of Series A Preferred Stock issued and outstanding.
The
Board may provide for the issue of any or all of the unissued and undesignated shares of the preferred stock in one or more series, and
to fix the number of shares and to determine or alter for each such series, such voting powers, full or limited, or no voting powers,
and such designation, preferences, and relative, participating, optional, or other rights and such qualifications, limitations, or restrictions
thereof, as shall be stated and expressed in the resolution or resolutions adopted by the Board providing for the issuance of such shares
and as may be permitted by law, without stockholder approval. Our Board is able to determine, with respect to any series of preferred
stock, the terms and rights of that series, including:
| |
● |
the
designation of the series; |
| |
● |
the
number of shares of the series; |
| |
● |
whether
dividends, if any, will be cumulative or non-cumulative and the dividend rate, if any, of the series; |
| |
● |
the
dates at which dividends, if any, will be payable; |
| |
● |
the
redemption rights and price or prices, if any, for shares of the series; |
| |
● |
the
terms and amounts of any sinking fund provided for the purchase or redemption of shares of the series; |
| |
● |
the
amounts payable on shares of the series in the event of any voluntary or involuntary liquidation, dissolution or winding-up of the
affairs of our Company; |
| |
● |
whether
the shares of the series will be convertible into shares of any other class or series, or any other security, of our Company or any
other entity, and, if so, the specification of the other class or series or other security, the conversion price or prices or rate
or rates and provisions for any adjustments to such prices or rates, the date or dates as of which the shares will be convertible,
and all other terms and conditions upon which the conversion may be made; |
| |
● |
the
ranking of such series with respect to dividends and amounts payable on our liquidation, dissolution or winding-up, which may include
provisions that such series will rank senior to our common stock with respect to dividends and those distributions; |
| |
● |
restrictions
on the issuance of shares of the same series or any other class or series; or |
| |
● |
voting
rights, if any, of the holders of the series. |
The
issuance of preferred stock could adversely affect, among other things, the voting power of holders of common stock and the likelihood
that stockholders will receive dividend payments and payments upon our liquidation, dissolution or winding up. The issuance of preferred
stock could also have the effect of delaying, deferring or preventing a change in control of us.
Section
203 of the Delaware General Corporation Law
We
are subject to the provisions of Section 203 of the DGCL regulating corporate takeovers. This statute prevents certain Delaware corporations,
under certain circumstances, from engaging in a “business combination” with:
| |
● |
a
stockholder who owns 15% or more of our outstanding voting stock (otherwise known as an “interested stockholder”); |
| |
● |
an
affiliate of an interested stockholder; or |
| |
● |
an
associate of an interested stockholder, for three years following the date that the stockholder became an interested stockholder. |
A
“business combination” includes a merger or sale of more than 10% of our assets. However, the above provisions of Section
203 do not apply if:
| |
● |
our
Board approves the transaction that made the stockholder an “interested stockholder,” prior to the date of the transaction;
or |
| |
● |
after
the completion of the transaction that resulted in the stockholder becoming an interested stockholder, that stockholder owned at
least 85% of our voting stock outstanding at the time the transaction commenced, other than statutorily excluded shares of common
stock. |
Potential
Effects of Authorized but Unissued Stock
Our
shares of common and preferred stock are available for future issuance without stockholder approval. We may utilize these additional
shares for a variety of corporate purposes, including future public offerings to raise additional capital, to facilitate corporate acquisitions,
payment as a dividend on the capital stock or as equity compensation to our service providers under our equity compensation plans.
The
existence of unissued and unreserved common stock and preferred stock may enable our Board to issue shares to persons friendly to current
management or to issue preferred stock with terms that could render more difficult or discourage a third-party attempt to obtain control
by means of a merger, tender offer, proxy contest or otherwise, thereby protecting the continuity of our management. In addition, our
Board has the discretion to determine designations, rights, preferences, privileges and restrictions, including voting rights, dividend
rights, conversion rights, redemption privileges and liquidation preferences of each series of preferred stock, all to the fullest extent
permissible under the Delaware General Corporation Law and subject to any limitations set forth in our Certificate of Incorporation.
The purpose of authorizing the Board to issue preferred stock and to determine the rights and preferences applicable to such preferred
stock is to eliminate delays associated with a stockholder vote on specific issuances. The issuance of preferred stock, while providing
desirable flexibility in connection with possible financings, acquisitions and other corporate purposes, could have the effect of making
it more difficult for a third-party to acquire, or could discourage a third-party from acquiring, a majority of our outstanding voting
stock.
Also,
if we issue additional shares of our authorized, but unissued, common stock, these issuances will dilute the voting power and distribution
rights of our existing common stockholders.
Transfer
Agent and Registrar
The
transfer agent and registrar for our common stock will be Equity Stock Transfer, LLC (“Equity Stock Transfer”), located at
237 West 37th Street, Suite 602, New York, NY 10018. The phone number and facsimile number for Equity Stock Transfer are (212) 575-5757
and (347) 584-3644, respectively. Additional information about Equity Stock Transfer can be found on its website at www.equitystock.com.
Listing
Our
common stock and Tradeable Warrants have been approved for listing on The Nasdaq Capital Market under the symbols “SXTP”
and “SXTPW,” respectively.
DESCRIPTION
OF WARRANTS
We
may issue warrants for the purchase of shares of our common stock or preferred stock or of debt securities. We may issue warrants independently
or together with other securities, and the warrants may be attached to or separate from any offered securities. Each series of warrants
will be issued under a separate warrant agreement to be entered into between us and the investors or a warrant agent. The following summary
of material provisions of the warrants and warrant agreements is subject to, and qualified in its entirety by reference to, all the provisions
of the warrant agreement and warrant certificate applicable to a particular series of warrants. The terms of any warrants offered under
a prospectus supplement may differ from the terms described below. We urge you to read the applicable prospectus supplement and any related
free writing prospectus, as well as the complete warrant agreements and warrant certificates that contain the terms of the warrants.
The
particular terms of any issue of warrants will be described in the prospectus supplement relating to the issue. Those terms may include:
| |
● |
the
number of shares of common stock or preferred stock purchasable upon the exercise of warrants to purchase such shares and the price
at which such number of shares may be purchased upon such exercise; |
| |
● |
the
designation, stated value and terms (including, without limitation, liquidation, dividend, conversion and voting rights) of the series
of preferred stock purchasable upon exercise of warrants to purchase preferred stock; |
| |
● |
the
principal amount of debt securities that may be purchased upon exercise of a debt warrant and the exercise price for the warrants,
which may be payable in cash, securities or other property; |
| |
● |
the
date, if any, on and after which the warrants and the related debt securities, preferred stock or common stock will be separately
transferable; |
| |
● |
the
terms of any rights to redeem or call the warrants; |
| |
● |
the
date on which the right to exercise the warrants will commence and the date on which the right will expire; |
| |
● |
United
States federal income tax consequences applicable to the warrants; and |
| |
● |
any
additional terms of the warrants, including terms, procedures and limitations relating to the exchange, exercise and settlement of
the warrants. |
Holders
of equity warrants will not be entitled to:
| |
● |
vote,
consent or receive dividends; |
| |
● |
receive
notice as stockholders with respect to any meeting of stockholders for the election of our directors or any other matter; or |
| |
● |
exercise
any rights as stockholders of 60 Degrees Pharmaceuticals. |
Each
warrant will entitle its holder to purchase the principal amount of debt securities or the number of shares of preferred stock or common
stock at the exercise price set forth in, or calculable as set forth in, the applicable prospectus supplement. Unless we otherwise specify
in the applicable prospectus supplement, holders of the warrants may exercise the warrants at any time up to the specified time on the
expiration date that we set forth in the applicable prospectus supplement. After the close of business on the expiration date, unexercised
warrants will become void.
A
holder of warrant certificates may exchange them for new warrant certificates of different denominations, present them for registration
of transfer and exercise them at the corporate trust office of the warrant agent or any other office indicated in the applicable prospectus
supplement. Until any warrants to purchase debt securities are exercised, the holder of the warrants will not have any rights of holders
of the debt securities that can be purchased upon exercise, including any rights to receive payments of principal, premium or interest
on the underlying debt securities or to enforce covenants in the applicable indenture. Until any warrants to purchase common stock or
preferred stock are exercised, the holders of the warrants will not have any rights of holders of the underlying common stock or preferred
stock, including any rights to receive dividends or payments upon any liquidation, dissolution or winding up on the common stock or preferred
stock, if any.
DESCRIPTION
OF DEBT SECURITIES
The
following description, together with the additional information we include in any applicable prospectus supplement or free writing prospectus,
summarizes certain general terms and provisions of the debt securities that we may offer under this prospectus. When we offer to sell
a particular series of debt securities, we will describe the specific terms of the series in a supplement to this prospectus. We will
also indicate in the supplement to what extent the general terms and provisions described in this prospectus apply to a particular series
of debt securities.
We
may issue debt securities either separately, or together with, or upon the conversion or exercise of or in exchange for, other securities
described in this prospectus. Debt securities may be our senior, senior subordinated or subordinated obligations and, unless otherwise
specified in a supplement to this prospectus, the debt securities will be our direct, unsecured obligations and may be issued in one
or more series.
The
debt securities will be issued under an indenture between us and a trustee named in the prospectus supplement. We have summarized select
portions of the indenture below. The summary is not complete. The form of the indenture has been filed as an exhibit to the registration
statement and you should read the indenture for provisions that may be important to you. In the summary below, we have included references
to the section numbers of the indenture so that you can easily locate these provisions. Capitalized terms used in the summary and not
defined herein have the meanings specified in the indenture.
General
The
indenture does not limit the amount of debt securities that we may issue. It provides that we may issue debt securities up to the principal
amount that we may authorize and may be in any currency or currency unit that we may designate. Except for the limitations on consolidation,
merger and sale of all or substantially all of our assets contained in the indenture, the terms of the indenture do not contain any covenants
or other provisions designed to give holders of any debt securities protection against changes in our operations, financial condition
or transactions involving us.
We
may issue the debt securities issued under the indenture as “discount securities,” which means they may be sold at a discount
below their stated principal amount. These debt securities, as well as other debt securities that are not issued at a discount, may be
issued with “original issue discount,” or OID, for U.S. federal income tax purposes because of interest payment and other
characteristics or terms of the debt securities. Material U.S. federal income tax considerations applicable to debt securities issued
with OID will be described in more detail in any applicable prospectus supplement.
We
will describe in the applicable prospectus supplement the terms of the series of debt securities being offered, including:
| |
● |
the
title of the series of debt securities; |
| |
|
|
| |
● |
any
limit upon the aggregate principal amount that may be issued; |
| |
|
|
| |
● |
the
maturity date or dates; |
| |
|
|
| |
● |
the
form of the debt securities of the series; |
| |
|
|
| |
● |
the
applicability of any guarantees; |
| |
|
|
| |
● |
whether
or not the debt securities will be secured or unsecured, and the terms of any secured debt; |
| |
|
|
| |
● |
whether
the debt securities rank as senior debt, senior subordinated debt, subordinated debt or any combination thereof, and the terms of
any subordination; |
| |
● |
if
the price (expressed as a percentage of the aggregate principal amount thereof) at which such debt securities will be issued
is a price other than the principal amount thereof, the portion of the principal amount thereof payable upon declaration of acceleration
of the maturity thereof, or if applicable, the portion of the principal amount of such debt securities that is convertible into another
security or the method by which any such portion shall be determined; |
| |
|
|
| |
● |
the
interest rate or rates, which may be fixed or variable, or the method for determining the rate and the date interest will begin to
accrue, the dates interest will be payable and the regular record dates for interest payment dates or the method for determining
such dates; |
| |
|
|
| |
● |
our
right, if any, to defer payment of interest and the maximum length of any such deferral period; |
| |
|
|
| |
● |
if
applicable, the date or dates after which, or the period or periods during which, and the price or prices at which, we may, at our
option, redeem the series of debt securities pursuant to any optional or provisional redemption provisions and the terms of those
redemption provisions; |
| |
|
|
| |
● |
the
date or dates, if any, on which, and the price or prices at which we are obligated, pursuant to any mandatory sinking fund or analogous
fund provisions or otherwise, to redeem, or at the holder’s option to purchase, the series of debt securities and the currency
or currency unit in which the debt securities are payable; |
| |
|
|
| |
● |
the
denominations in which we will issue the series of debt securities, if other than denominations of $1,000 and any integral multiple
thereof; |
| |
|
|
| |
● |
any
and all terms, if applicable, relating to any auction or remarketing of the debt securities of that series and any security for our
obligations with respect to such debt securities and any other terms which may be advisable in connection with the marketing of debt
securities of that series; |
| |
|
|
| |
● |
whether
the debt securities of the series shall be issued in whole or in part in the form of a global security or securities; the terms and
conditions, if any, upon which such global security or securities may be exchanged in whole or in part for other individual securities;
and the depositary for such global security or securities; |
| |
|
|
| |
● |
if
applicable, the provisions relating to conversion or exchange of any debt securities of the series and the terms and conditions upon
which such debt securities will be so convertible or exchangeable, including the conversion or exchange price, as applicable, or
how it will be calculated and may be adjusted, any mandatory or optional (at our option or the holders’ option) conversion
or exchange features, the applicable conversion or exchange period and the manner of settlement for any conversion or exchange; |
| |
● |
if
other than the full principal amount thereof, the portion of the principal amount of debt securities of the series which shall be
payable upon declaration of acceleration of the maturity thereof; |
| |
|
|
| |
● |
additions
to or changes in the covenants applicable to the particular debt securities being issued, including, among others, the consolidation,
merger or sale covenant; |
| |
|
|
| |
● |
additions
to or changes in the events of default with respect to the securities and any change in the right of the trustee or the holders to
declare the principal, premium, if any, and interest, if any, with respect to such securities to be due and payable; |
| |
|
|
| |
● |
additions
to or changes in or deletions of the provisions relating to covenant defeasance and legal defeasance; |
| |
|
|
| |
● |
additions
to or changes in the provisions relating to satisfaction and discharge of the indenture; |
| |
|
|
| |
● |
additions
to or changes in the provisions relating to the modification of the indenture both with and without the consent of holders of debt
securities issued under the indenture; |
| |
● |
the currency of payment of debt securities if other than U.S. dollars and the manner of determining the equivalent amount in U.S. dollars; |
| |
|
|
| |
● |
whether interest will be payable in cash or additional debt securities at our or the holders’ option and the terms and conditions upon which the election may be made; |
| |
|
|
| |
● |
any restrictions on transfer, sale or assignment of the debt securities of the series; and |
| |
|
|
| |
● |
any other specific terms, preferences, rights or limitations of, or restrictions on, the debt securities, any other additions or changes in the provisions of the indenture, and any terms that may be required by us or advisable under applicable laws or regulations. |
Conversion or Exchange Rights
We will set forth in the applicable prospectus
supplement the terms on which a series of debt securities may be convertible into or exchangeable for our common stock or our other securities.
We will include provisions as to settlement upon conversion or exchange and whether conversion or exchange is mandatory, at the option
of the holder or at our option. We may include provisions pursuant to which the number of shares of our common stock or our other securities
that the holders of the series of debt securities receive would be subject to adjustment.
Consolidation, Merger or Sale
Unless we provide otherwise in the prospectus
supplement applicable to a particular series of debt securities, the indenture will not contain any covenant that restricts our ability
to merge or consolidate, or sell, convey, transfer or otherwise dispose of our assets as an entirety or substantially as an entirety.
However, any successor to or acquirer of such assets (other than a subsidiary of ours) must assume all of our obligations under the indenture
or the debt securities, as appropriate.
Events of Default under the Indenture
Unless we provide otherwise in the prospectus
supplement applicable to a particular series of debt securities, the following are events of default under the indenture with respect
to any series of debt securities that we may issue:
| |
● |
if we fail to pay any installment of interest on any series of debt securities, as and when the same shall become due and payable, and such default continues for a period of 90 days; provided, however, that a valid extension of an interest payment period by us in accordance with the terms of any indenture supplemental thereto shall not constitute a default in the payment of interest for this purpose; |
| |
● |
if we fail to pay the principal of, or premium, if any, on any series of debt securities as and when the same shall become due and payable whether at maturity, upon redemption, by declaration or otherwise or in any payment required by any sinking or analogous fund established with respect to such series; provided, however, that a valid extension of the maturity of such debt securities in accordance with the terms of any indenture supplemental thereto shall not constitute a default in the payment of principal or premium, if any; |
| |
● |
if we fail to observe or perform any other covenant or agreement contained in the debt securities or the indenture, other than a covenant specifically relating to another series of debt securities, and our failure continues for 90 days after we receive written notice of such failure, requiring the same to be remedied and stating that such is a notice of default thereunder, from the trustee or holders of at least 25% in aggregate principal amount of the outstanding debt securities of the applicable series; and |
| |
● |
if specified events of bankruptcy, insolvency or reorganization occur. |
If an event of default with respect to debt securities
of any series occurs and is continuing, other than an event of default specified in the last bullet point above, the trustee or the holders
of at least 25% in aggregate principal amount of the outstanding debt securities of that series, by notice to us in writing, and to the
trustee if notice is given by such holders, may declare the unpaid principal of, premium, if any, and accrued interest, if any, due and
payable immediately. If an event of default specified in the last bullet point above occurs with respect to us, the principal amount
of and accrued interest, if any, of each issue of debt securities then outstanding shall be due and payable without any notice or other
action on the part of the trustee or any holder.
The holders of a majority in principal amount
of the outstanding debt securities of an affected series may waive any default or event of default with respect to the series and its
consequences, except defaults or events of default regarding payment of principal, premium, if any, or interest, unless we have cured
the default or event of default in accordance with the indenture. Any waiver shall cure the default or event of default.
Subject to the terms of the indenture, if an event
of default under an indenture shall occur and be continuing, the trustee will be under no obligation to exercise any of its rights or
powers under such indenture at the request or direction of any of the holders of the applicable series of debt securities, unless such
holders have offered the trustee reasonable indemnity. The holders of a majority in principal amount of the outstanding debt securities
of any series will have the right to direct the time, method and place of conducting any proceeding for any remedy available to the trustee,
or exercising any trust or power conferred on the trustee, with respect to the debt securities of that series, provided that:
| |
● |
the direction so given by the holder is not in conflict with any law or the applicable indenture; and |
| |
● |
subject to its duties under the Trust Indenture Act of 1939 (“Trust Indenture Act”), the trustee need not take any action that might involve it in personal liability or might be unduly prejudicial to the holders not involved in the proceeding. |
A holder of the debt securities of any series
will have the right to institute a proceeding under the indenture or to appoint a receiver or trustee, or to seek other remedies only
if:
| |
● |
the holder has given written notice to the trustee of a continuing event of default with respect to that series; |
| |
● |
the holders of at least 25% in aggregate principal amount of the outstanding debt securities of that series have made written request; |
| |
● |
such holders have offered to the trustee indemnity satisfactory to it against the costs, expenses and liabilities to be incurred by the trustee in compliance with the request; and |
| |
● |
the trustee does not institute the proceeding, and does not receive from the holders of a majority in aggregate principal amount of the outstanding debt securities of that series other conflicting directions within 90 days after the notice, request and offer. |
These limitations do not apply to a suit instituted
by a holder of debt securities if we default in the payment of the principal, premium, if any, or interest on, the debt securities.
We will periodically file statements with the
trustee regarding our compliance with specified covenants in the indenture.
Modification of Indenture; Waiver
We and the trustee may change an indenture without
the consent of any holders with respect to specific matters:
| |
● |
to cure any ambiguity, defect or inconsistency in the indenture or in the debt securities of any series; |
| |
● |
to comply with the provisions described above under “Description of Debt Securities—Consolidation, Merger or Sale”; |
| |
● |
to provide for uncertificated debt securities in addition to or in place of certificated debt securities; |
| |
● |
to add to our covenants, restrictions, conditions or provisions such new covenants, restrictions, conditions or provisions for the benefit of the holders of all or any series of debt securities, to make the occurrence, or the occurrence and the continuance, of a default in any such additional covenants, restrictions, conditions or provisions an event of default or to surrender any right or power conferred upon us in the indenture; |
| |
● |
to add to, delete from or revise the conditions, limitations, and restrictions on the authorized amount, terms, or purposes of issue, authentication and delivery of debt securities, as set forth in the indenture; |
| |
● |
to make any change that does not adversely affect the interests of any holder of debt securities of any series in any material respect; |
| |
● |
to provide for the issuance of and establish the form and terms and conditions of the debt securities of any series as provided above under “Description of Debt Securities—General” to establish the form of any certifications required to be furnished pursuant to the terms of the indenture or any series of debt securities, or to add to the rights of the holders of any series of debt securities; |
| |
● |
to evidence and provide for the acceptance of appointment under any indenture by a successor trustee; or |
| |
● |
to comply with any requirements of the SEC in connection with the qualification of any indenture under the Trust Indenture Act. |
In addition, under the indenture, the rights of
holders of a series of debt securities may be changed by us and the trustee with the written consent of the holders of at least a majority
in aggregate principal amount of the outstanding debt securities of each series that is affected. However, unless we provide otherwise
in the prospectus supplement applicable to a particular series of debt securities, we and the trustee may make the following changes only
with the consent of each holder of any outstanding debt securities affected:
| |
● |
extending the fixed maturity of any debt securities of any series; |
| |
● |
reducing the principal amount, reducing the rate of or extending the time of payment of interest, or reducing any premium payable upon the redemption of any series of any debt securities; or |
| |
● |
reducing the percentage of debt securities, the holders of which are required to consent to any amendment, supplement, modification or waiver. |
Discharge
Each indenture provides that we can elect to be
discharged from our obligations with respect to one or more series of debt securities, except for specified obligations, including obligations
to:
| |
● |
register the transfer or exchange of debt securities of the series; |
| |
● |
replace stolen, lost or mutilated debt securities of the series; |
| |
● |
pay principal of and premium and interest on any debt securities of the series; |
| |
● |
maintain paying agencies; |
| |
● |
hold monies for payment in trust; |
| |
● |
recover excess money held by the trustee; |
| |
● |
compensate and indemnify the trustee; and |
| |
● |
appoint any successor trustee. |
In order to exercise our rights to be discharged,
we must deposit with the trustee money or government obligations sufficient to pay all the principal of, any premium, if any, and interest
on, the debt securities of the series on the dates payments are due.
Form, Exchange and Transfer
We will issue the debt securities of each series
only in fully registered form without coupons and, unless we provide otherwise in the applicable prospectus supplement, in denominations
of $1,000 and any integral multiple thereof. The indenture provides that we may issue debt securities of a series in temporary or permanent
global form and as book-entry securities that will be deposited with, or on behalf of, The Depository Trust Company, or DTC, or another
depositary named by us and identified in the applicable prospectus supplement with respect to that series. To the extent the debt securities
of a series are issued in global form and as book-entry, a description of terms relating to any book-entry securities will be set forth
in the applicable prospectus supplement.
At the option of the holder, subject to the terms
of the indenture and the limitations applicable to global securities described in the applicable prospectus supplement, the holder of
the debt securities of any series can exchange the debt securities for other debt securities of the same series, in any authorized denomination
and of like tenor and aggregate principal amount.
Subject to the terms of the indenture and the
limitations applicable to global securities set forth in the applicable prospectus supplement, holders of the debt securities may present
the debt securities for exchange or for registration of transfer, duly endorsed or with the form of transfer endorsed thereon duly executed
if so required by us or the security registrar, at the office of the security registrar or at the office of any transfer agent designated
by us for this purpose. Unless otherwise provided in the debt securities that the holder presents for transfer or exchange, we will impose
no service charge for any registration of transfer or exchange, but we may require payment of any taxes or other governmental charges.
We will name in the applicable prospectus supplement
the security registrar, and any transfer agent in addition to the security registrar, that we initially designate for any debt securities.
We may at any time designate additional transfer agents or rescind the designation of any transfer agent or approve a change in the office
through which any transfer agent acts, except that we will be required to maintain a transfer agent in each place of payment for the debt
securities of each series.
If we elect to redeem the debt securities of any
series, we will not be required to:
| |
● |
issue, register the transfer of, or exchange any debt securities of that series during a period beginning at the opening of business 15 days before the day of mailing of a notice of redemption of any debt securities that may be selected for redemption and ending at the close of business on the day of the mailing; or |
| |
● |
register the transfer of or exchange any debt securities so selected for redemption, in whole or in part, except the unredeemed portion of any debt securities we are redeeming in part. |
Information Concerning the Trustee
The trustee, other than during the occurrence
and continuance of an event of default under an indenture, undertakes to perform only those duties as are specifically set forth in the
applicable indenture. Upon an event of default under an indenture, the trustee must use the same degree of care as a prudent person would
exercise or use in the conduct of his or her own affairs. Subject to this provision, the trustee is under no obligation to exercise any
of the powers given it by the indenture at the request of any holder of debt securities unless it is offered reasonable security and indemnity
against the costs, expenses and liabilities that it might incur.
Payment and Paying Agents
Unless we otherwise indicate in the applicable
prospectus supplement, we will make payment of the interest on any debt securities on any interest payment date to the person in whose
name the debt securities, or one or more predecessor securities, are registered at the close of business on the regular record date for
the interest.
We will pay principal of and any premium and interest
on the debt securities of a particular series at the office of the paying agents designated by us, except that unless we otherwise indicate
in the applicable prospectus supplement, we will make interest payments by check that we will mail to the holder or by wire transfer to
certain holders. Unless we otherwise indicate in the applicable prospectus supplement, we will designate the corporate trust office of
the trustee as our sole paying agent for payments with respect to debt securities of each series. We will name in the applicable prospectus
supplement any other paying agents that we initially designate for the debt securities of a particular series. We will maintain a paying
agent in each place of payment for the debt securities of a particular series.
All money we pay to a paying agent or the trustee
for the payment of the principal of or any premium or interest on any debt securities that remains unclaimed at the end of two years
after such principal, premium or interest has become due and payable will be repaid to us, and the holder of the debt security thereafter
may look only to us for payment thereof.
Governing Law
The indenture and the debt securities will be
governed by and construed in accordance with the internal laws of the State of New York, except to the extent that the Trust Indenture
Act is applicable.
DESCRIPTION OF UNITS
The following description, together with the additional
information we include in any applicable prospectus supplement, summarizes the material terms and provisions of the units that we may
offer under this prospectus. Units may be offered independently or together with common stock, preferred stock, debt securities and/or
warrants offered by any prospectus supplement, and may be attached to or separate from those securities. While the terms we have summarized
below will generally apply to any future units that we may offer under this prospectus, we will describe the particular terms of any series
of units that we may offer in more detail in the applicable prospectus supplement. The terms of any units offered under a prospectus supplement
may differ from the terms described below.
We will incorporate by reference into the registration
statement of which this prospectus forms a part the form of unit agreement, including a form of unit certificate, if any, that describes
the terms of the series of units we are offering before the issuance of the related series of units. The following summaries of material
provisions of the units, and the unit agreements, are subject to, and qualified in their entirety by reference to, all the provisions
of the unit agreement applicable to a particular series of units. We urge you to read the applicable prospectus supplements related to
the units that we sell under this prospectus, as well as the complete unit agreements that contain the terms of the units.
General
We may issue units comprised of one or more shares
of our common stock or preferred stock, debt securities and warrants in any combination. Each unit will be issued so that the holder of
the unit is also the holder of each security included in the unit. Thus, the holder of a unit will have the rights and obligations of
a holder of each included security. The unit agreement under which a unit is issued may provide that the securities included in the unit
may not be held or transferred separately, at any time or at any time before a specified date.
We will describe in the applicable prospectus
supplement the terms of the series of units, including:
| |
● |
the designation and terms of the units and of the securities comprising the units, including whether, and under what circumstances, those securities may be held or transferred separately; |
| |
● |
the rights and obligations of the unit agent, if any; |
| |
● |
any provisions of the governing unit agreement that differ from those described below; and |
| |
● |
any provisions for the issuance, payment, settlement, transfer or exchange of the units or of the securities comprising the units. |
The provisions described in this section, as well
as those described under “Description of Capital Stock,” “Description of Our Common Stock,” “Description
of Debt Securities” and “Description of Warrants,” will apply to each unit and to any common stock, preferred
stock, debt securities or warrants included in each unit, respectively.
Issuance in Series
We may issue units in such amounts and in numerous
distinct series as we determine.
LEGAL OWNERSHIP OF SECURITIES
We may issue securities in registered form or
in the form of one or more global securities. We describe global securities in greater detail below. We refer to those persons who have
securities registered in their own names on the books that we or any applicable trustee, depositary or warrant agent maintain for this
purpose as the “holders” of those securities. These persons are the legal holders of the securities. We refer to those persons
who, indirectly through others, own beneficial interests in securities that are not registered in their own names, as “indirect
holders” of those securities. As we discuss below, indirect holders are not legal holders, and investors in securities issued in
book-entry form or in street name will be indirect holders.
Book-Entry Holders
We may issue securities in book-entry form only,
as we will specify in the applicable prospectus supplement. This means securities may be represented by one or more global securities
registered in the name of a financial institution that holds them as depositary on behalf of other financial institutions that participate
in the depositary’s book-entry system. These participating institutions, which are referred to as participants, in turn, hold beneficial
interests in the securities on behalf of themselves or their customers.
Only the person in whose name a security is registered
is recognized as the holder of that security. Securities issued in global form will be registered in the name of the depositary or its
participants. Consequently, for securities issued in global form, we will recognize only the depositary as the holder of the securities,
and we will make all payments on the securities to the depositary. The depositary passes along the payments it receives to its participants,
which in turn pass the payments along to their customers who are the beneficial owners. The depositary and its participants do so under
agreements they have made with one another or with their customers; they are not obligated to do so under the terms of the securities.
As a result, investors in a global security will
not own securities directly. Instead, they will own beneficial interests in a global security, through a bank, broker or other financial
institution that participates in the depositary’s book-entry system or holds an interest through a participant. As long as the securities
are issued in global form, investors will be indirect holders, and not legal holders, of the securities.
Street Name Holders
We may terminate a global security or issue securities
in non-global form. In these cases, investors may choose to hold their securities in their own names or in “street name.”
Securities held by an investor in street name would be registered in the name of a bank, broker or other financial institution that the
investor chooses, and the investor would hold only a beneficial interest in those securities through an account he or she maintains at
that institution.
For securities held in street name, we or any
applicable trustee or depositary will recognize only the intermediary banks, brokers and other financial institutions in whose names the
securities are registered as the holders of those securities, and we or any applicable trustee or depositary will make all payments on
those securities to them. These institutions pass along the payments they receive to their customers who are the beneficial owners, but
only because they agree to do so in their customer agreements or because they are legally required to do so. Investors who hold securities
in street name will be indirect holders, not holders, of those securities.
Legal Holders
Our obligations, as well as the obligations of
any applicable trustee and of any third parties employed by us or a trustee, run only to the legal holders of the securities. We do not
have obligations to investors who hold beneficial interests in global securities, in street name or by any other indirect means. This
will be the case whether an investor chooses to be an indirect holder of a security or has no choice because we are issuing the securities
only in global form.
For example, once we make a payment or give a
notice to the holder, we have no further responsibility for the payment or notice even if that holder is required, under agreements with
depositary participants or customers or by law, to pass it along to the indirect holders but does not do so. Similarly, we may want to
obtain the approval of the holders to amend an indenture, to relieve us of the consequences of a default or of our obligation to comply
with a particular provision of the indenture or for other purposes. In such an event, we would seek approval only from the holders, and
not the indirect holders, of the securities. Whether and how the holders contact the indirect holders is up to the holders.
Special Considerations for Indirect Holders
If you hold securities through a bank, broker,
or other financial institution, either in book-entry form because the securities are represented by one or more global securities or in
street name, you should check with your own institution to find out:
| |
● |
the performance of third-party service providers; |
| |
● |
how it handles securities payments and notices; |
| |
● |
whether it imposes fees or charges; |
| |
● |
how it would handle a request for the holders’ consent, if ever required; |
| |
● |
whether and how you can instruct it to send you securities registered in your own name so you can be a holder, if that is permitted in the future; |
| |
● |
how it would exercise rights under the securities if there were a default or other event triggering the need for holders to act to protect their interests; and |
| |
● |
if the securities are in book-entry form, how the depositary’s rules and procedures will affect these matters. |
Global Securities
A global security is a security that represents
one or any other number of individual securities held by a depositary. Generally, all securities represented by the same global securities
will have the same terms.
Each security issued in book-entry form will be
represented by a global security that we deposit with and register in the name of a financial institution or its nominee that we select.
The financial institution that we select for this purpose is called the depositary. Unless we specify otherwise in the applicable prospectus
supplement, DTC will be the depositary for all securities issued in book-entry form.
A global security may not be transferred to or
registered in the name of anyone other than the depositary, its nominee or a successor depositary, unless special termination situations
arise. We describe those situations below under “Special Situations When a Global Security Will Be Terminated.” As
a result of these arrangements, the depositary, or its nominee, will be the sole registered owner and holder of all securities represented
by a global security, and investors will be permitted to own only beneficial interests in a global security. Beneficial interests must
be held by means of an account with a broker, bank or other financial institution that in turn has an account with the depositary or with
another institution that does. Thus, an investor whose security is represented by a global security will not be a holder of the security,
but only an indirect holder of a beneficial interest in the global security.
If the prospectus supplement for a particular
security indicates that the security will be issued in global form only, then the security will be represented by a global security at
all times unless and until the global security is terminated. If termination occurs, we may issue the securities through another book-entry
clearing system or decide that the securities may no longer be held through any book-entry clearing system.
Special Considerations for Global Securities
The rights of an indirect holder relating to a
global security will be governed by the account rules of the investor’s financial institution and of the depositary, as well as
general laws relating to securities transfers. We do not recognize an indirect holder as a holder of securities and instead deal only
with the depositary that holds the global security.
If securities are issued only in the form of a
global security, an investor should be aware of the following:
| |
● |
an investor cannot cause the securities to be registered in his or her name, and cannot obtain non-global certificates for his or her interest in the securities, except in the special situations we describe below; |
| |
● |
an investor will be an indirect holder and must look to his or her own bank or broker for payments on the securities and protection of his or her legal rights relating to the securities, as we describe above; |
| |
● |
an investor may not be able to sell interests in the securities to some insurance companies and to other institutions that are required by law to own their securities in non-book-entry form; |
| |
● |
an investor may not be able to pledge his or her interest in a global security in circumstances where certificates representing the securities must be delivered to the lender or other beneficiary of the pledge in order for the pledge to be effective; |
| |
● |
the depositary’s policies, which may change from time to time, will govern payments, transfers, exchanges and other matters relating to an investor’s interest in a global security; |
| |
● |
we and any applicable trustee have no responsibility for any aspect of the depositary’s actions or for its records of ownership interests in a global security, nor do we or any applicable trustee supervise the depositary in any way; |
| |
● |
the depositary may, and we understand that DTC will, require that those who purchase and sell interests in a global security within its book-entry system use immediately available funds, and your broker or bank may require you to do so as well; and |
| |
● |
financial institutions that participate in the depositary’s book-entry system, and through which an investor holds its interest in a global security, may also have their own policies affecting payments, notices and other matters relating to the securities. |
There may be more than one financial intermediary
in the chain of ownership for an investor. We do not monitor and are not responsible for the actions of any of those intermediaries.
Special Situations When a Global Security Will
Be Terminated
In a few special situations described below, the
global security will terminate and interests in it will be exchanged for physical certificates representing those interests. After that
exchange, the choice of whether to hold securities directly or in street name will be up to the investor. Investors must consult their
own banks or brokers to find out how to have their interests in securities transferred to their own name, so that they will be direct
holders. We have described the rights of holders and street name investors above.
Unless we provide otherwise in the applicable
prospectus supplement, the global security will terminate when the following special situations occur:
| |
● |
if the depositary notifies us that it is unwilling, unable or no longer qualified to continue as depositary for that global security and we do not appoint another institution to act as depositary within 90 days; |
| |
● |
if we notify any applicable trustee that we wish to terminate that global security; or |
| |
● |
if an event of default has occurred with regard to securities represented by that global security and has not been cured or waived. |
The applicable prospectus supplement may also
list additional situations for terminating a global security that would apply only to the particular series of securities covered by the
applicable prospectus supplement. When a global security terminates, the depositary, and not we or any applicable trustee, is responsible
for deciding the names of the institutions that will be the initial direct holders.
PLAN OF DISTRIBUTION
We may sell the securities from time to time pursuant
to underwritten public offerings, direct sales to the public, negotiated transactions, block trades or a combination of these methods.
We may sell the securities to or through underwriters or dealers, through agents, or directly to one or more purchasers. We may distribute
securities from time to time in one or more transactions:
| |
● |
at a fixed price or prices, which may be changed; |
| |
● |
at market prices prevailing at the time of sale; |
| |
● |
at prices related to such prevailing market prices; or |
A prospectus supplement or supplements (and any
related free writing prospectus that we may authorize to be provided to you) will describe the terms of the offering of the securities,
including, to the extent applicable:
| |
● |
the name or names of the underwriters, if any; |
| |
● |
the purchase price of the securities or other consideration therefor, and the proceeds, if any, we will receive from the sale; |
| |
● |
any options under which underwriters may purchase additional securities from us; |
| |
● |
any agency fees or underwriting discounts and other items constituting agents’ or underwriters’ compensation; |
| |
● |
any public offering price; |
| |
● |
any discounts or concessions allowed or reallowed or paid to dealers; and |
| |
● |
any securities exchange or market on which the securities may be listed. |
Only underwriters named in the prospectus supplement
will be underwriters of the securities offered by the prospectus supplement.
If underwriters are used in the sale, they will
acquire the securities for their own account and may resell the securities from time to time in one or more transactions at a fixed public
offering price or at varying prices determined at the time of sale. The obligations of the underwriters to purchase the securities will
be subject to the conditions set forth in the applicable underwriting agreement. We may offer the securities to the public through underwriting
syndicates represented by managing underwriters or by underwriters without a syndicate. Subject to certain conditions, the underwriters
will be obligated to purchase all of the securities offered by the prospectus supplement, other than securities covered by any option
to purchase additional securities from us. Any public offering price and any discounts or concessions allowed or reallowed or paid to
dealers may change from time to time. We may use underwriters with whom we have a material relationship. We will describe in the prospectus
supplement, naming the underwriter, the nature of any such relationship.
We may sell securities directly or through agents
we designate from time to time in an “at the market offering” or other similar offering. We will name any agent involved in
the offering and sale of securities and we will describe any commissions we will pay the agent in the prospectus supplement. Unless the
prospectus supplement states otherwise, our agent will act on a best-efforts basis for the period of its appointment.
We may authorize agents or underwriters to solicit
offers by certain types of institutional investors to purchase securities from us at the public offering price set forth in the prospectus
supplement pursuant to delayed delivery contracts providing for payment and delivery on a specified date in the future. We will describe
the conditions to these contracts and the commissions we must pay for solicitation of these contracts in the prospectus supplement.
We may provide agents and underwriters with indemnification
against civil liabilities, including liabilities under the Securities Act, or contribution with respect to payments that the agents or
underwriters may make with respect to these liabilities. Agents and underwriters may engage in transactions with, or perform services
for, us in the ordinary course of business.
All securities we may offer, other than common
stock, will be new issues of securities with no established trading market. Any underwriters may make a market in these securities, but
will not be obligated to do so and may discontinue any market making at any time without notice. We cannot guarantee the liquidity of
the trading markets for any securities.
LEGAL MATTERS
Unless otherwise indicated in the applicable prospectus
supplement, the validity of the issuance of the securities offered hereby will be passed upon for us by Sichenzia Ross Ference Carmel
LLP located in New York, New York. Additional legal matters may be passed upon for us or any underwriters, dealers or agents, by counsel
that we will name in the applicable prospectus supplement.
EXPERTS
RBSM LLP, an independent registered public accounting
firm, audited our financial statements for the years ended December 31, 2023 and 2022, respectively. We have included our financial statements
in this prospectus and elsewhere in the registration statement in reliance on the reports of RBSM LLP, given their authority as experts
in accounting and auditing.
WHERE YOU CAN FIND MORE INFORMATION
This prospectus is part of the registration statement
on Form S-3 that we filed with the Commission under the Securities Act and does not contain all of the information set forth in the
registration statement. Whenever a reference is made in this prospectus to any of our contracts, agreements or other documents, the reference
may not be complete and you should refer to the exhibits that are part of the registration statement or the exhibits to the reports or
other document incorporated into this prospectus for a copy of such contract agreement or other document. Because we are subject to the
information and reporting requirements under the Exchange Act, we file annual, quarterly and current reports, proxy statements and other
information with the Commission. Our filings with the Commission are available to the public over the Commission’s website at www.sec.gov.
Our Annual Report on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K, including any amendments to those reports,
and other information that we file with or furnish to the Commission pursuant to Section 13(a) or 15(d) of the Exchange Act can also be
accessed free of charge on our website. You may also read and copy any document we file with the SEC at its public reference facility
at 100 F Street, N.E., Washington, D.C., 20549, on official business days during the hours of 10 a.m. to 3 p.m. You may also obtain copies
of the documents at prescribed rates by writing to the Public Reference Section of the SEC at 100 F Street, N.E., Washington, D.C., 20549.
Please call the SEC at 1-800-SEC-0330 for further information on the operation of the public reference facility. In addition, you can
find more information about us on our website at https://60degreespharma.com. Information contained on or accessible through our
website is not a part of this prospectus and is not incorporated by reference herein, and the inclusion of our website address in this
prospectus is an inactive textual reference only.
INCORPORATION OF CERTAIN INFORMATION BY REFERENCE
The SEC allows us to “incorporate by reference”
information that we file with it into this prospectus, which means that we can disclose important information to you by referring you
to those documents. The information incorporated by reference is an important part of this prospectus. The information incorporated by
reference into this prospectus is deemed to be part of this prospectus, and any information filed with the SEC after the date of this
prospectus will automatically be deemed to update and supersede information contained in this prospectus and any accompanying prospectus
supplement.
The following documents previously filed with
the SEC are incorporated by reference in this prospectus:
| |
● |
The Registrant’s Annual Report on Form 10-K for the fiscal year ended December 31, 2023, filed with the SEC on April 1, 2024; |
| |
● |
The Registrant’s Quarterly Report on Form 10-Q for the fiscal quarter ended March 31, 2024, filed with the SEC on May 15, 2024; |
| |
● |
The Registrant’s Preliminary Schedule 14A, filed with the SEC on May 3, 2024, and the Registrant’s Definitive Schedule 14A, filed with the SEC on May 30, 2024; |
| |
● |
The description of the Registrant’s common stock, which is contained in a registration statement on Form 8-A12B filed with the SEC on June 27, 2023, under the Exchange Act, including any amendment or report filed for the purpose of updating such description. |
All filings filed by us pursuant to the Exchange
Act after the date of the initial filing of the registration statement of which this prospectus is a part and prior to effectiveness of
the registration statement shall be deemed to be incorporated by reference into this prospectus.
We also incorporate by reference all additional
documents that we file with the Securities and Exchange Commission under the terms of Sections 13(a), 13(c), 14 or 15(d) of the Exchange
Act that are made after the date of the initial registration statement but prior to effectiveness of the registration statement and after
the date of this prospectus but prior to the termination of the offering of the securities covered by this prospectus. We are not, however,
incorporating, in each case, any documents or information that we are deemed to furnish and not file in accordance with Securities and
Exchange Commission rules.
You should rely only on the information contained
or incorporated by reference in this prospectus. We have not authorized any other person to provide you with different information. If
anyone provides you with different or inconsistent information, you should not rely on it. You should assume that the information appearing
in this prospectus is accurate only as of the date of this prospectus. Our business, financial condition, results of operations and prospects
may have changed since that date.
Any statement contained in a document incorporated
or deemed to be incorporated by reference into this prospectus will be deemed to be modified or superseded for the purposes of this prospectus
to the extent that a statement contained herein, or in any other subsequently filed document which also is or is deemed to be incorporated
by reference herein, modifies or supersedes that statement. The modifying or superseding statement need not state it has modified or superseded
a prior statement or include any other information set forth in the document that it modifies or supersedes. The making of a modifying
or superseding statement is not an admission for any purposes that the modified or superseded statement, when made, constituted a misrepresentation,
an untrue statement of a material fact or an omission to state a material fact that is required to be stated or that is necessary to make
a statement not misleading in light of the circumstances in which it was made. Any statement so modified or superseded will not be deemed,
except as so modified or superseded, to constitute a part of this prospectus.
You may request, and we will provide you with,
a copy of these filings, at no cost, by calling us at (202) 327-5422 or by writing to us at the following address:
60 Degrees Pharmaceuticals, Inc.
1025 Connecticut Avenue NW Suite 1000
Washington, D.C. 20036
Attn: Geoffrey Dow, Chief Executive Officer and
President
$15,000,000
Common Stock
Preferred Stock
Warrants
Debt Securities
Units

60
Degrees Pharmaceuticals, Inc.
PROSPECTUS
July 12, 2024
The information in
this prospectus supplement is not complete and may be changed. We may not sell these securities until the registration statement filed
with the Securities and Exchange Commission is effective. This prospectus is not an offer to sell these securities and is not soliciting
an offer to buy these securities in any jurisdiction where the offer or sale is not permitted.
SUBJECT TO COMPLETION,
DATED JULY 12, 2024
PROSPECTUS SUPPLEMENT
Up to $1,253,603
Common Stock

60
Degrees Pharmaceuticals, Inc.
We have entered into an At the Market Offering Agreement dated as of
July 12, 2024, or the Sales Agreement, with WallachBeth Capital LLC, or the “Sales Agent,” relating to shares of our common
stock, par value $0.0001 per share, offered by this prospectus supplement and the accompanying prospectus. In accordance with the terms
of the Sales Agreement, we may offer and sell shares of our common stock having an aggregate offering price of up to $1,253,603 from time
to time through the Sales Agent, acting as sales agent or principal.
Our common stock is listed on The Nasdaq Capital
Market under the symbol “SXTP.” On July 11, 2024, the last reported sale price of our common stock on The Nasdaq Capital Market
was $0.2460 per share.
Sales of our common stock, if any, under this
prospectus supplement and the accompanying prospectus will be made in sales deemed to be an “at the market offering” as defined
in Rule 415(a)(4) promulgated under the Securities Act of 1933, as amended, or the Securities Act, including sales made directly on or
through The Nasdaq Capital Market or any other existing trading market for our common stock in the United States or to or through a market
maker. The Sales Agent is not required to sell any specific amount of shares of common stock, but will act as our sales agent using commercially
reasonable efforts consistent with its normal trading and sales practices, on mutually agreed terms between the Sales Agent and us. There
is no arrangement for funds to be received in any escrow, trust or similar arrangement.
The compensation to the Sales Agent for sales
of common stock sold pursuant to the Sales Agreement will be an amount equal to 4.0% of the gross proceeds of any shares of common stock
sold under the Sales Agreement. In connection with the sale of the common stock on our behalf, the Sales Agent may be deemed to be an
“underwriter” within the meaning of the Securities Act and the compensation of the Sales Agent may be deemed to be underwriting
commissions or discounts. We have also agreed to provide indemnification and contribution to the Sales Agent with respect to certain liabilities,
including liabilities under the Securities Act or the Securities Exchange Act of 1934, as amended, or the Exchange Act.
The aggregate market value of our
outstanding common stock held by non-affiliates is $3,761,186, based on 12,206,116 shares of outstanding common stock, of which
10,715,629 shares are held by non-affiliates, and a share price of $0.351 per share, which was the closing sale price of our common
stock as quoted on The Nasdaq Capital Market on June 7, 2024. Pursuant to General Instruction I.B.6 of Form S-3, in no event
will we sell our securities in a public primary offering with a value exceeding more than one-third of our public float in any
12-month period so long as our public float remains below $75,000,000. As of the date of this prospectus, we have not offered any
securities during the past twelve months pursuant to General Instruction I.B.6 of Form S-3. You are urged to obtain current
market quotations of our common stock.
Investing in our securities involves a high
degree of risk. See “Risk Factors” beginning on page S-22 of this prospectus supplement and elsewhere in this prospectus
supplement, the accompanying base prospectus and the other documents that are incorporated by reference in this prospectus supplement
and the accompanying prospectus.
Neither the Securities and Exchange Commission
nor any state securities commission has approved or disapproved of these securities or determined if this prospectus supplement or the
accompanying prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
WallachBeth
Capital LLC
The date of this prospectus supplement is July 12, 2024.
TABLE OF CONTENTS
PROSPECTUS SUPPLEMENT
ABOUT THIS PROSPECTUS SUPPLEMENT
This prospectus supplement and the accompanying prospectus are part
of a “shelf” registration statement on Form S-3 that we filed with the Securities and Exchange Commission, or the SEC
or Commission, on July 12, 2024. This document is in two parts. The first part is this prospectus supplement, which describes the terms
of this offering of our common stock and adds to and updates the information contained in the accompanying prospectus. The second part,
the accompanying prospectus, provides more general information, some of which may not apply to this offering. Generally, when we refer
to this prospectus, we are referring to both parts of this document combined. To the extent there is a conflict between the information
contained in this prospectus supplement and the information contained in the accompanying prospectus, you should rely on the information
in this prospectus supplement.
This prospectus supplement and the accompanying
prospectus relate to the offering of shares of our common stock. Before buying any of the shares of common stock offered hereby, we urge
you to read carefully this prospectus supplement and the accompanying prospectus, together with the information incorporated herein by
reference as described below under the heading “Incorporation of Certain Information by Reference.” This prospectus
supplement contains information about the common stock offered hereby and may add to, update or change information in the accompanying
prospectus.
You should rely only on the information contained
in, or incorporated by reference into, this prospectus supplement and the accompanying prospectus. We have not, and the Sales Agent has
not, authorized anyone to provide you with different or additional information.
We are not making offers to sell or solicitations
to buy our common stock in any jurisdiction in which an offer or solicitation is not authorized or in which the person making that offer
or solicitation is not qualified to do so or to anyone to whom it is unlawful to make an offer or solicitation. You should assume that
the information in this prospectus supplement and the accompanying prospectus is accurate only as of the date on the front of the respective
document and that any information that we have incorporated by reference is accurate only as of the date of the document incorporated
by reference, regardless of the time of delivery of this prospectus supplement or the accompanying prospectus or the time of any sale
of our common stock.
This prospectus supplement and the accompanying
prospectus contain summaries of certain provisions contained in some of the documents described herein, but reference is made to the actual
documents for complete information. All of the summaries are qualified in their entirety by the actual documents. Copies of some of the
documents referred to herein have been filed, will be filed or will be incorporated herein by reference as exhibits to the registration
statement, and you may obtain copies of those documents as described below under the section entitled “Where You Can Find More
Information.”
We further note that the representations, warranties
and covenants made by us in any agreement that is filed as an exhibit to any document that is incorporated by reference herein were made
solely for the benefit of the parties to such agreement, including, in some cases, for the purpose of allocating risk among the parties
to such agreements, and should not be deemed to be a representation, warranty or covenant to you. Moreover, such representations, warranties
or covenants were accurate only as of the date when made. Accordingly, such representations, warranties and covenants should not be relied
on as accurately representing the current state of our affairs.
This prospectus supplement and the accompanying
prospectus contain and incorporate by reference market data and industry statistics and forecasts that are based on independent industry
publications and other publicly available information. Although we believe these sources are reliable, we do not guarantee the accuracy
or completeness of this information and we have not independently verified this information. Although we are not aware of any misstatements
regarding the market and industry data presented in this prospectus supplement, the accompanying prospectus or the documents incorporated
herein by reference, these estimates involve risks and uncertainties and are subject to change based on various factors, including those
discussed under the headings “Risk Factors” in this prospectus supplement and the accompanying prospectus, and under
similar headings in the other documents that are incorporated herein by reference. Accordingly, investors should not place undue reliance
on this information.
References in this prospectus to the terms “60
Degrees Pharmaceuticals, Inc.,” “60 Degrees Pharmaceuticals,” “60P,” the “Company,” “we,”
“us,” “our” or other similar terms refer to 60 Degrees Pharmaceuticals, Inc., a Delaware corporation, and our
subsidiaries.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus contains “forward-looking
statements.” Forward-looking statements reflect the current view about future events. When used in this prospectus, the words “anticipate,”
“believe,” “estimate,” “expect,” “future,” “intend,” “plan,” or
the negative of these terms and similar expressions, as they relate to us or our management, identify forward-looking statements. Such
statements include, but are not limited to, statements contained in this prospectus relating to our business strategy, our future operating
results and liquidity and capital resources outlook. Forward-looking statements are based on our current expectations and assumptions
regarding our business, the economy and other future conditions. Because forward–looking statements relate to the future, they are
subject to inherent uncertainties, risks and changes in circumstances that are difficult to predict. Our actual results may differ materially
from those contemplated by the forward-looking statements. They are neither statements of historical fact nor guarantees of assurance
of future performance. We caution you therefore against relying on any of these forward-looking statements. Important factors that could
cause actual results to differ materially from those in the forward-looking statements include, without limitation:
| |
● |
Our ability to effectively operate our business
segments; |
| |
|
|
| |
● |
Our ability to manage our research, development, expansion, growth and operating expenses; |
| |
|
|
| |
● |
Our ability to evaluate and measure our business,
prospects and performance metrics;
|
| |
● |
Our ability to compete, directly and indirectly, and succeed in a highly competitive and evolving industry; |
| |
|
|
| |
● |
Our ability to respond and adapt to changes in technology and customer behavior; |
| |
|
|
| |
● |
Our ability to protect our intellectual property
and to develop, maintain and enhance a strong brand; and |
| |
|
|
| |
● |
other factors (including the risks contained in the section of this prospectus entitled “Risk Factors”) relating to our industry, our operations and results of operations. |
Should one or more of these risks or uncertainties
materialize, or should the underlying assumptions prove incorrect, actual results may differ significantly from those anticipated, believed,
estimated, expected, intended or planned.
Factors or events that could cause our actual
results to differ may emerge from time to time, and it is not possible for us to predict all of them. We cannot guarantee future results,
levels of activity, performance or achievements. Except as required by applicable law, including the securities laws of the United States,
we do not intend to update any of the forward-looking statements to conform these statements to actual results.
MARKET, INDUSTRY AND OTHER DATA
This prospectus and any applicable prospectus
supplement and the documents incorporated by reference herein and therein contain estimates, projections, market research and other information
concerning, among other things, our industry, our business and markets for our products and services. Unless otherwise expressly stated,
we obtain this information from reports, research surveys, studies and similar data prepared by market research firms and other third
parties, industry, medical and general publications, government data and similar sources as well as from our own internal estimates and
research and from publications, research, surveys and studies conducted by third parties on our behalf. Information that is based on estimates,
projections, market research or similar methodologies is inherently subject to uncertainties and actual events or circumstances may differ
materially from events and circumstances that are reflected in this information. As a result, you are cautioned not to give undue weight
to such information.
TRADEMARKS
Solely for convenience, our trademarks and tradenames
referred to in this prospectus may appear without the ® or ™ symbols, but such references are not intended to indicate
in any way that we will not assert, to the fullest extent under applicable law, our rights to these trademarks and tradenames. All other
trademarks, service marks and trade names included or incorporated by reference into this prospectus or the accompanying prospectus are
the property of their respective owners. We do not intend our use or display of other companies’ trade names, trademarks or service
marks to imply relationships with, or endorsements or sponsorship of us by, these other companies.
PROSPECTUS
SUPPLEMENT SUMMARY
This summary provides a brief overview of the
key aspects of our business and our securities. The reader should read the entire prospectus carefully, especially the risks of investing
in our securities discussed under “Risk Factors.” Some of the statements contained in this prospectus, including statements
under “Summary” and “Risk Factors” as well as those noted in the documents incorporated herein by reference, are
forward-looking statements and may involve a number of risks and uncertainties. Our actual results and future events may differ significantly
based upon a number of factors. The reader should not put undue reliance on the forward-looking statements in this document, which speak
only as of the date on the cover of this prospectus.
Overview
We are a specialty pharmaceutical company with a goal of using cutting-edge
biological science and applied research to further develop and commercialize new therapies for the prevention and treatment of infectious
diseases. We have successfully achieved regulatory approval of Arakoda, a malaria preventative treatment that has been on the market since
late 2019. Currently, 60P’s pipeline under development covers development programs for vector-borne, fungal, and viral diseases
utilizing three of the Company’s future products: (i) new products that contain the Arakoda regimen of Tafenoquine; (ii) new products
that contain Tafenoquine; and (iii) Celgosivir.
Mission
Our mission is to address the unmet medical need associated with infectious
diseases through the development and commercialization of new small molecule therapeutics, focusing on synthetic drugs (made by chemists
in labs, excluding biologics) with good safety profiles based on prior clinical studies, in order to reduce cost, risk, and capitalize
on existing research. We are seeking to expand Arakoda’s use beyond malaria prevention and to demonstrate clinical benefit for other
disease indications. We are further testing the viability of another product (Celgosivir) to determine whether to advance it into further
clinical development, and may seek to develop and license other molecules in the future. Celgosivir is being considered for development
as an antiviral product for a number of diseases.
Market Opportunity
Malaria Prevention
In 2018, the FDA approved Arakoda for malaria prevention in individuals
18 years and older. Arakoda entered the U.S. supply chain in the third quarter of 2019, just prior to the COVID-19 pandemic. As the approved
indication is for travel medicine, and international travel was substantially impacted by the pandemic, we did not undertake any active
marketing efforts for Arakoda. travel was substantially impacted by the pandemic, we did not undertake any active marketing efforts for
Arakoda. Following our recent financing the Company hired a Chief Commercial Officer and commissioned IQVIA market data and a qualitative
marketing demand study. That research, recently completed, suggests that prescribing of antimalarial medications has returned to pre-pandemic
levels, and that the total U.S. market represents around 1.1 million prescriptions (one prescription per three weeks of travel). Based
on consumer and HCP demand research, the Company estimates that the accessible market for Arakoda represents about one third of this volume
(about 330,000 prescriptions). Barriers to entry include low brand awareness in the prescriber community and the low cost of some of the
generic alternatives. In the second half of 2024 we will conduct a pilot commercialization study to confirm these barriers can be overcome
(see “Strategy”).
Treatment and Prevention of Tick-Borne
Disease (Babesiosis)
We are repositioning the Arakoda regimen of Tafenoquine to potentially
address several new therapeutic indications that have substantial U.S. caseloads, as further described below:
| |
● |
Treatment of Chronic Tick-Borne Disease
(Babesiosis). Babesia parasites are co-transmitted by the same ticks that transmit Borrelia, the Lyme disease
bacterium. Although Lyme in the acute phase is generally viewed by the medical community as being treatable with antibiotics,
individuals who are not treated, or fail treatment, may go onto to develop long term, and potentially debilitating, chronic symptoms
such as fatigue, body aches and cognitive problems.1 This condition is defined by the CDC as Post-Treatment Lyme Disease
Syndrome (“PTLDS”) or simply as Lyme in the patient community.2 Although there are no published estimates, key opinion
leaders represent that as many as 50% of Lyme/PTLDS patients are believed to be co-infected with Babesia parasites, a
diagnosis referred to in the Lyme community as “Chronic Babesiosis.” Prescribers in the Lyme disease community utilize
a number of therapeutic modalities to manage the symptoms of Chronic Babesiosis, including FDA-approved pharmaceuticals such
as atovaquone and azithromycin (these are assumed to suppress the growth of Babesia parasites).3
Following the publication of several case reports demonstrating activity
in immunosuppressed patients with acute babesiosis, and animal data showing eradication of Babesia parasites, Tafenoquine (primarily
as Arakoda) appears to have begun to be increasingly prescribed by Lyme physicians to manage Chronic Babesiosis.4 The Company
believes the recent increases in sales of Arakoda have been driven by organic growth of these activities. There are no formal epidemiological
publications articulating the incidence or prevalence of Chronic Babesiosis, so these metrics must be inferred based on data for PTLDS
and the rate of coinfection with Babesia parasites. Thus, the cumulative case load of Chronic Babesiosis may be as high as1.01
million patients in the United States.5 We believe, based on our market research that at least 37% of this market, or 375,000
cases, may be addressable with Tafenoquine during the remainder of its market exclusivity window for malaria. We are undertaking additional
research to determine how much additional market capture might be feasible
Acute infection with many different
organisms (e.g. Borrelia, SARS-Cov-2, Epstein Barr virus) trigger “Long Syndromes” in a minority of cases, characterized
by cognitive dysfunction, fatigue and post-exertional malaise.6 For many years, such conditions have been confusing to
the mainstream medical community because there may not be formal diagnostic criteria or an established theory of disease. This is
changing with the advent of Long COVID, and a recent prominent paper outlined the pathophysiological mechanisms for the first
time.7 Although there is not yet supporting evidence in the medical literature, some key opinion leaders in the Lyme
community have postulated, using the veterinary literature as an analog, that life-long infection by sequestering forms of Babesia
(e.g., B. odocoilei) may be significant driver of chronic fatigue symptoms.8 If this is true, the addressable
market for antibabesial drugs may be substantially larger than stated above, since the prevalence of chronic fatigue syndrome in the
U.S. is at least 3.3 million cases (excluding Long COVID and PTLDS).9
Separately from the clinical indication, based on estimates from industry
experts, there may be somewhere between several hundred and several thousand cases of canine babesiosis each year in the United States,
and thousands more globally. Currently, standard of care treatment for babesiosis in dogs is a ten-day course of atovaquone and azithromycin,
which costs about $1,350 out of pocket. A treatment course of Tafenoquine mirroring the human prophylactic dose in dogs might cost <
$300, offering a compelling alternative to standard of care. The additional resources required to generate enabling data for veterinary
uses are much less expensive than human clinical trials and we are already funding a pilot study at North Carolina State University related
to this indication. |
| 1 |
See https://www.cdc.gov/lyme/signs-symptoms/chronic-symptoms-and-lyme-disease.html. |
| 2 |
See https://www.cdc.gov/lyme/signs-symptoms/chronic-symptoms-and-lyme-disease.html. |
| 3 |
Conclusions from Company-commissioned market research. |
| 4 |
Conclusions from Company-commissioned market research. |
| 5 |
Maximum prevalence determined by multiplying the rate of Babesia coinfection in PTLDS patients (52%, from Parveen & Bhanot, Pathogens 2019;8(3):117) by the highest estimate of the cumulative prevalence of PTLDS (1,994,189, from Delong et al. BMC Public Health 2019;19(1):352). Maximum new cases determined by multiplying the number of new Lyme cases per year (476,000, from Krugeler et al (Emerg Infect Dis 2021;27:616-61) by the number of new cases that subsequently become chronic cases (up to 10%, from Delong et al. BMC Public Health 2019;19(1):352) by the proportion of such patients coinfected with Babesia (52%, from Parveen & Bhanot, Pathogens 2019;8(3):117). |
| 6 |
See https://www.cdc.gov/lyme/signs-symptoms/chronic-symptoms-and-lyme-disease.html. |
| 7 |
Walitt et al Nature Communications 2024;15:907. |
| 8 |
Lindner HH. 2022. Chronic
babesiosis caused by B. odocoilei: Diagnosis, pathophysiology & treatment. Presentation at the 2022 ILADS scientific meeting,
Orlando Florida. |
| 9 |
See https://www.cdc.gov/nchs/data/databriefs/db488.pdf. |
| |
● |
Treatment
of Acute Babesiosis. There are up to 38,000 cases of potentially treatable acute symptomatic babesiosis (red blood cell infections
caused by deer tick bites) in the United States each year.10 Approximately 650 of these cases are hospitalizations, a smaller
fraction of which represents immunosuppressed individuals.11 Symptomatic babesiosis is usually treated with a minimum ten
day course of atovaquone and azithromycin which is extended to six weeks in the immunosuppressed, who may also experience relapses requiring
multiple hospitalizations.12 This is much longer than equivalent serious parasitic diseases such as malaria where the goal
is a three-day regimen. In a recently published case series Tafenoquine in combination with standard of care cured 80% of immunosuppressed
patients with relapsing babesiosis and the investigators stated in a press release that “Tafenoquine is
going to make a huge difference, I think, in people who are severely immunocompromised.”13 |
| |
|
|
| |
● |
Prevention
of Tick-Borne Diseases. Post-exposure prophylaxis or early treatment with, respectively,
a single dose or several week regimen of doxycycline following a tick-bite is a recognized
indication to prevent the complications of Lyme disease. There may be more than 400,000 such
tick bites in the United States requiring medical treatment each year. This estimate is based
on the observation that approximately 50,000 tick bites are treated in U.S. hospital emergency
rooms each year but this calculation represents only about 12% of actual treated tick bites
based on observations from comparable ex-U.S health systems.14 Unlike Lyme disease,
there is no characteristic rash associated with early infection and no reliable diagnostic
tests. Thus, an individual bitten by a tick cannot know whether they have also been infected
with babesiosis. It is likely that a drug proven to be effective for this indication for
babesiosis would also be used in conjunction with Lyme prophylaxis. |
Treatment and Prevention of Fungal Infections
We are evaluating Tafenoquine for potential utility in the following
fungal diseases:
| |
● |
Treatment of Candida infections. According to the CDC, there are 50,000 cases
of candidiasis (a type of fungal infection) each year in the United States and up to 1,900 clinical cases of C. auris,
for which there are few available treatments, have been reported to date.15 Since it has broad-spectrum activity against
drug-resistant Candida spp in culture, Tafenoquine, has the potential to be a market leading therapy for treatment/prevention
of C. auris, and to be added to the standard of care regimens for other Candida infections.16 |
| |
● |
Prevention of fungal pneumonias.
There are up to ~ 91-92,000 new patient cases each year in the United States including acute lymphoblastic
leukemia (up to 6,540 cases) and large B-cell lymphoma (up to 18,000 cases) patients receiving CAR-T
therapy, solid organ transplant patients (up to 42,887 cases), allogeneic (~ 9,000 cases) and autologous
(~ 15,000 cases) hematopoietic stem cell transplant patients for whom the use of antifungal prophylaxis
is recommended.17 Despite the availability and use of antifungal prophylaxis, the risk
of some patient groups contracting fungal pneumonia exceeds the risk of contracting malaria during
travel to West Africa.18 Since it has broad spectrum antifungal effects in cell culture,
and activity against Pneumocystis in animal models, Tafenoquine has the potential to be added
to existing standard of care regimens for the prevention of fungal pneumonias.19. |
| 10 |
This estimate is based on the observations of Krugeler et al (Emerg Infect Dis 2021;27:616-61) who reported that 476,000 cases of Lyme disease occur in U.S. states where babesiosis is endemic and Krause et. al. (JAMA 1996;275:1657-16602) who reported that 10% of Lyme disease patients are co-infected with babesiosis and that according to Krause et al (AJTMH 2003;6:431-436) fact that about 80% of cases are symptomatic (thus 476,000*10%*80% = 38,000 cases of babesiosis per year). |
| 11 |
Bloch et al Open Forum Infect Dis 2022;9(11):ofac597. |
| 12 |
According to IDSA guidelines. |
| 13 |
See Krause et al Clin Infect Dis 2024; doi:10.1093/cid/ciae238 and https://ysph.yale.edu/news-article/antimalarial-drug-is-effective-against-tick-borne-infection-babesiosis/. |
| 14 |
Marx et. al., MMWR 2021;70:612-616. |
| 15 |
https://www.cdc.gov/fungal/diseases/candidiasis/invasive/statistics.html.; https://www.cdc.gov/fungal/candida-auris/tracking-c-auris.html. |
| 16 |
Dow and Smith New Microb New Infect 2022;45:100964. |
| 17 |
See statistics for solid organ transplants at the Organ Transplant and Procurement Network at: National data - OPTN (hrsa.gov); See statistics for hematopoietic stem cell transplant in Dsouza et al Biology of Blood and Bone Marrow Transplantation 202;26: e177-e182; See statistics for acute lymphoblastic leukemia at: Key Statistics for Acute Lymphocytic Leukemia (ALL) (cancer.org); See statistics for large cell large B-cell lymphoma at; Diffuse Large B-Cell Lymphoma - Lymphoma Research Foundation; Treatment guidelines recommending antifungal prophylaxis for these diseases can be reviewed in (i) Fishman et al Clinical Transplantation. 2019;33:e13587, (ii) Hematopoietic Cell Transplantation (cancernetwork.com), (iii) Cooper et al Journal of the National Comprehensive Cancer Network 2016;14:882-913 and (iv) Los Arcos et al Infection (2021) 49:215–231. |
| 18 |
Aguilar-Guisado et al Clin Transplant 2011;25:E629–38; Mace et al MMWR 202;70:1–35. |
| 19 |
Queener et al JID 1997;165:764-768; Dow and Smith New Microb New Infect 2022;45:100964 |
Viral Diseases
Celgosivir, a potential clinical candidate of 60P’s, has activity
in a number of animal models of important viral diseases such as Dengue and RSV. According to the European CDC, Dengue is associated with
at least 4.1 million cases globally.20 According to the U.S. CDC, RSV is responsible for up to 240,000 hospitalizations in
children less than five years of age and adults greater than 65 years of age in the United States each year according to the CDC.21
As outlined in the “Strategy” section below, we expect to evaluate Celgosivir in additional non-clinical disease models
before making a decision regarding clinical development.
More information about our products is provided
in the next section, and the status of various development efforts for the above-mentioned diseases is outlined in Figure A, below.
Figure A

Products
Arakoda (Tafenoquine) for malaria prevention
We entered into a cooperative research and development agreement with
the United States Army in 2014 to complete development of Arakoda for prevention of malaria.22 With the U.S. Army, and other
private sector entities as partners, we coordinated the execution of two clinical trials, development of a full manufacturing package,
gap-filling non-clinical studies, compilation of a full regulatory dossier, successful defense of our program at an FDA advisory committee
meeting and submitted a new drug application (“NDA”) to the FDA in 2018. The history of that collaboration has been publicly
communicated by the U.S. Army.23
| 20 |
https://www.ecdc.europa.eu/en/dengue-monthly#:~:text=This%20is%20an%20increase%20of%2032%20653%20cases%
20and%2032,853%20deaths%20have%20been%20reported. |
| 21 |
https://www.cdc.gov/rsv/php/surveillance/index.html#cdc_survey_profile_surveys_used-rsv-burden-estimates. |
| 22 |
In 2014, we signed a cooperative research and development agreement with the United States
Army Medical and Materiel Development Activity (Agreement W81XWH-14-0313). Under this agreement, we agreed to submit an NDA for Tafenoquine
to the FDA (as Arakoda), while the US Army agreed to finance the bulk of the necessary development activities in support of that goal. |
| 23 | Zottig et al Military Medicine
2020; 185 (S1): 687. |
The FDA and Australia’s medicinal regulatory
agency, Therapeutic Goods Administration, subsequently approved Arakoda (brand name in the U.S.) and Kodatef (brand name in Australia),
respectively, for prevention of malaria in travelers in 2018. Prescribing information and guidance for patients can be found at www.arakoda.com.
The features and benefits of Tafenoquine for malaria prophylaxis, some of which have been noted by third-party experts, include: convenient
once weekly dosing following a three day load; the absence of reports of drug resistance during malaria prophylaxis; activity against
liver and blood stages of malaria as well as both the major malaria species (Plasmodium vivax and Plasmodium falciparum);
absence of any black-box safety warnings; good tolerability including in women and individuals with prior psychiatric medical history,
and a comparable adverse event rate to placebo with up to 12 months continuous dosing.24 Tafenoquine entered the commercial
supply chains in the U.S. and Australia in the third quarter of 2019.
The only limitation of Arakoda is the requirement for a G6PD test prior
to administration.25 The G6PD test must be administered to a prospective patient prior to administration of Arakoda
in order to prevent the potential occurrence of hemolytic anemia in individuals with G6PD deficiency.26 G6PD is one of
the most common enzyme deficiencies and is implicated in hemolysis following administration/ingestion of a variety of oxidant drugs/food.
G6PD must also be ruled out as a possible cause when diagnosing neonatal jaundice. As a consequence, G6PD testing is widely available
in the United States through commercial pathology service providers (e.g., Labcorp, Quest Diagnostics, etc.). Although these tests have
a turn-around time of up to 72 hours, the test needs only to be administered once. Thus, existing U.S. testing infrastructure is sufficient
to support the FDA-approved use of the product (malaria prevention) by members of the armed forces (who automatically have a G6PD test
when they enlist), civilian travelers with a long planning horizon or repeat travelers.
Tafenoquine for Other (Infectious) Diseases
During the pandemic, we also worked with NIH to evaluate the utility
of Tafenoquine as an antifungal. We, and the NIH, found that Tafenoquine exhibits a Broad Spectrum of Activity in cell culture against
Candida and other yeast strains via a different Mode of Action than traditional antifungals and also exhibits antifungal activity
against some fungal strains at clinically relevant doses in animal models.27 Our work followed Legacy Studies that show Tafenoquine
is effective for treatment and prevention of Pneumocystis pneumonia in animal models.28 We believe that if added to
the standard of care for anti-fungal and yeast infection treatments for general use, Tafenoquine has the potential to improve patient
outcomes in terms of recovery from yeast infections, and prevention of fungal pneumonias in immunosuppressed patients. There are limited
treatment options available for these indications, and Tafenoquine’s novel mechanism of action might also mitigate problems of resistance.
Clinical trial(s) to prove safety and efficacy, and approval by the FDA and other regulators, would be required before Tafenoquine could
be marketed for these indications.
Tafenoquine monotherapy, or use in combination with other antibabesial
medications, clears and eradicates Babesia infections, respectively, in both immunocompetent and immunocompromised animal models
of babesiosis (tick borne red blood cell infections).29 In up to 80% of cases Tafenoquine administered in combination with
antibabesial drugs after prior failure of conventional antibiotics in immunosuppressed babesiosis patients resulted in cures.30 Tafenoquine
is also increasingly being utilized by Lyme disease prescribers to manage symptoms of Chronic Babesiosis, on the assumption that it eradicates
residual populations of Babesia. Consequently, we believe that (i) if combined with standard of care products, Tafenoquine has
the potential to accelerate parasite clearance and reduce the duration of illness and treatment with antibiotic therapy in immunosuppressed
patients those hospitalized with severe illness and (ii) that once appropriate clinical studies have been conducted, it is likely that
Tafenoquine would be quickly embraced for post-exposure prophylaxis of babesiosis in patients with tick bites and (iii) could become the
leading treatment for Chronic Babesiosis. Clinical trial(s) to prove safety and efficacy, and approval by FDA and other regulators, would
be required before Tafenoquine could be marketed for these indications.
| 24 |
Tan and Hwang Journal of
Travel Medicine, 2018, 1–2; Baird Journal of Travel Medicine 2018:, 1–13; Schlagenhauf et al Travel Medicine and Infectious
Disease 2022; 46:102268; See Arakoda prescribing information at www.arakoda.com; McCarthy et al CID 2019:69:480-486; Dow et al. Malar
J (2015) 14:473; Dow et al. Malaria Journal 2014, 13:49; Novitt-Moreno et al Travel Med Infect Dis 2022 Jan-Feb;45:102211. |
| 25 |
See prescribing information
at www.arakoda.com. |
| 26 |
See prescribing information
at www.arakoda.com. |
| 27 |
Dow and Smith, New Microbe
and New Infect 2022; 45: 100964. |
| 28 |
Queener et al Journal of
Infectious Diseases 1992;165:764-8). |
| 29 |
Liu et al. Antimicrobial
Agents Chemo 2021;65:e00204-21, Marcos et al. IDCases 2022;27:e01460; Rogers et al. Clin Infect Dis. 2022 Jun 10:ciac473, Prasad
and Wormsner. Pathogens 2022;11:1015. |
| 30 |
Krause et al Clin Infect
Dis 2024; doi:10.1093/cid/ciae238. |
Celgosivir
Celgosivir is a host targeted glucosidase inhibitor that was developed
separately by other sponsors for HIV then for hepatitis C.31 The sponsors abandoned Celgosivir after completion of Phase II
clinical trials involving 700+ patients, because other antivirals in development at the time had superior activity. The National University
of Singapore initiated development of Celgosivir independently for Dengue fever. A clinical study, conducted in Singapore, the results
of which were accepted for publication in the peer-reviewed journal Lancet Infectious Diseases, confirmed its safety but the observed
reduction in viral load was lower than what the study was powered to detect.32 Celgosivir (as with other Dengue antivirals)
exhibits greater capacity to cure Dengue infections in animal models when administered prior to symptom onset when compared to administration
post-symptom onset. In animal models, this problem can be addressed by administering the same dose of drug split into four doses per day
rather than two doses per day (as was the case in the Singaporean clinical trial).33 This observation led to the filing and
approval of a patent related to Dengue, which we licensed from the National University of Singapore.
Additional clinical studies would be required to prove that such a
4x daily dosing regimen would be safe and effective in Dengue patients to regulators’ satisfaction. To that end, earlier in our
history, we, in partnership with the National University of Singapore, and Singapore General Hospital, successfully secured a grant from
the government of Singapore for a follow-on clinical trial. Unfortunately, we were unable at that time to raise matching private sector
funding. We concluded as a result that development of Repositioned Molecules for Dengue, solely and without simultaneous development for
other therapeutic use, despite substantial morbidity and mortality in tropical countries, was an effort best suited for philanthropic
entities. Accordingly, during the pandemic, we undertook an effort (in partnership with NIH’s Division of Microbiology and Infectious
Diseases program and Florida State University) to determine whether Celgosivir might be more broadly useful for respiratory diseases that
have impact in both tropical and temperate countries. Preliminary data suggest Celgosivir inhibits the replication of the virus that causes
COVID-19 (SARS-CoV-2) in cell culture, and the RSV virus in cell culture and provides benefits in animals. We have filed and/or licensed
patents in relation to Celgosivir for these other viruses as we believe there is potential applications to fight respiratory diseases
that might have more commercial viability than historical development of Celgosivir to combat Dengue fever.
Competitive Strengths
Our main competitive strength has been our ability to achieve important
clinical milestones inexpensively in therapeutic areas that other entities have found extremely challenging. With a small virtual management
team, we have successfully built productive research partnerships with public and academic entities, and licensed products with well characterized
safety profiles in prior clinical studies, thereby reducing the cost and risk of clinical development. This business and product model
enabled Arakoda to be approved in 2018, with a total operating expense of < $10 million. We plan to focus in the future on generating
proof of concept clinical data sets for the approved Arakoda regimen of Tafenoquine in other therapeutic areas, all of which is expected
to foster and continue our existing tradition of inexpensive product development.
Strategy
Following our initial public offering in July 2023, our initial strategic
priority was to conduct a Phase IIB that would have evaluated the potential of the Arakoda regimen of Tafenoquine to accelerate disease
recovery in COVID-19 patients with low risk of disease progression. In October 2023, we made a decision to suspend this study. This was
a consequence of advice previously received from the FDA, which we interpreted to mean that the Agency would not have granted clearance
for the study to proceed unless we redesigned it to (i) enroll a patient population in which receipt of Paxlovid or Lagevrio would be
medically contraindicated or (ii) compare Tafenoquine to placebo in patients taking a “standard of care” regimen (defined
by the FDA as Lagevrio or Paxlovid). The FDA’s position was somewhat surprising given that neither Paxlovid nor Lagevrio is indicated
for treatment of COVID-19 in low-risk patients. We determined that conducting our study in an alternate population in the United States
would be unfeasible, and that conducting an add-on-to standard of care study might not be Phase III enabling. Accordingly, the Company
made a decision to pivot back to continue commercialization of Arakoda for malaria, and further evaluation of the Arakoda regimen of Tafenoquine
for babesiosis and other diseases. We believe such an approach is both less risky and less expensive.
Moving forward, our general strategy to achieve profitability and grow
shareholder value has three facets: (i) increase sales of Arakoda; (ii) conduct clinical trials to expand the number of patients who can
use Tafenoquine for new indications in the future; and (iii) reposition small molecule therapeutics with good clinical safety profiles
for new indications.
| 31 |
Sorbera et al, Drugs of
the Future 2005; 30:545-552. |
| 32 |
Low et. al., Lancet ID
2014; 14:706-715. |
| 33 |
Watanabe et al, Antiviral
Research 2016; 10:e19. |
Expansion of U.S. Arakoda Sales
Hiring of Chief Commercial Officer. In
February, 2024, we hired Kristen Landon to lead our commercial efforts to reintroduce Arakoda for malaria prevention and conduct new product
planning initiatives in tick-borne disease for babesiosis. We spent the first quarter analyzing the current landscape in the malaria prevention
market, conducting primary market research among providers and consumers, and assessing agency partners for a virtual/digital marketing
pilot program. Additionally, we kicked off a market assessment on the babesiosis space including desk top research and qualitative interviews
with Key Opinion Leaders in the Infectious Disease and Lyme Community.
P&L Contract Review. We will conduct
a review of all of our supply chain and formulary contracts to determine whether it is possible to increase our margin on Arakoda without
increasing prices, or to compensate for any price adjustments which may be necessary to support repositioning efforts (see below).
Repositioning of Arakoda Relative to Malarone
and Generic Equivalent Atovaquone-Proguanil. A malaria demand study was conducted to assess the attractiveness and acceptability of
the Arakoda product profile and current pricing among health care providers and consumers. The product profile was well received among
both stakeholders, however, price sensitivity on out-of-pocket costs was noted among both groups. Generic atovaquone-proguanil, our primary
competitor is substantially cheaper than Arakoda for the average trip length (three weeks) and has superior formulary positioning (Tier
1 vs. Tier 3). However, generic-atovaquone proguanil does not provide the same level of confidence a traveler may experience from taking
a product with a convenient weekly dosing regimen during travel, that works everywhere in the world against all malaria species and drug
resistant strains, and which requires only a single dose for post-exposure prophylaxis upon return from a malarious area. The value those
advantages confer needs to be communicated with key stakeholders.
Market Segment Definition and Targeting.
We purchased market data to understand the malaria market landscape over the past decade and identified the current prescribers of Malarone
and the generic equivalent atovaquone-proguanil, the main generic competitor to Arakoda for malaria prophylaxis. Beginning in the third
quarter of 2024, we plan to reach out to prescribers covering the top 80% of atovaquone-proguanil prescribers in order to educate them
about the value proposition of Arakoda. We will also compile a list of the top institutions/organizations that have ex-U.S. deployed
workforces and internal occupational health and safety programs, and target these organizations with messaging regarding the convenience
and global effectiveness of Arakoda. We do not initially plan to target U.S. government agencies as these organizations, such as the
Department of Defense, are expected to be extremely price sensitive until operational considerations justify the use of superior products
– for example, the DOD used inexpensive doxycycline for malaria prevention in the low malaria risk setting of Afghanistan, but
chose superior weekly mefloquine, despite safety concerns, for the Ebola mission to west Africa in 2014, where malaria rates were extremely
high.
Digital Revamp and Collateral: We will
work with an agency of record to develop a marketing strategy for the proposed pilot and develop marketing assets that we believe best
highlight the features and benefits of Arakoda, namely the convenience of the travel and post-travel regimen and global effectiveness.
We are currently assessing a co-pay or point of sale offer for travelers to offset out-of-pocket costs. We launched our Arakoda product
website which went live in April 2024.
Revised Forecast. We have developed an internal forecast for
the malaria and Babesiosis indications and have contracted a third party vendor to validate our analyses.
Development
of Arakoda Regimen of Tafenoquine for Babesiosis
In animal models, Tafenoquine monotherapy has
been shown to suppress acute babesiosis infections to the point where the immune system can control them following single or multiple
doses similar to those effective against malaria parasites, and longer regimens alone or in combination with atovaquone leads to complete
radical cure and to the conference of sterile immunity.34 In three case studies in individuals with immunosuppression and/or
refractory parasites, Tafenoquine alone or combination with various standard of care antimalarials and antibiotics successfully cleared
parasites leading to three consecutive negative PCR tests, and prevention of further relapses in two of three individuals.35
Our market research has revealed that recent sales growth in Arakoda is primarily attributable to organic growth in prescribing by Lyme
community prescribes for Chronic Babesiosis. Collectively these data suggest Tafenoquine might have utility alone or in combination
as treatment or post-exposure prophylaxis of babesiosis (both acute and chronic).
| 34 |
Liu et al. Antimicrobial
Agents Chemo 2021;65:e00204-21. Vydyam et al. J Infect Dis. 2024 Jan 3:jiad315. doi:10.1093/infdis/jiad315. |
| 35 |
Marcos et al. IDCases 2022;27:e01460;
Rogers et al. Clin Infect Dis. 2022 Jun 10:ciac473, Prasad and Wormsner. Pathogens 2022;11:1015. |
The Company is planning three clinical trials to aid further development
and commercialization of a Babesiosis indication for Tafenoquine. Trial 1 is a randomized, placebo-controlled, evaluation of Tafenoquine
(200 mg per day for a total of 800 mg) in patients hospitalized with babesiosis who are also taking standard of care treatment (10 days
of atovaquone-azithromycin). The primary endpoint will be time to clinical recovery of 11 common babesiosis symptoms as reported by patients.
The key secondary endpoint will be time to molecular cure as assessed by an FDA-approved Babesia nucleic acid test that is used for blood
donation screening. The study will enroll a minimum of 24 and up to 33 patients before an interim analysis is conducted, which will include
both a test of significance and a sample size re-estimation in case this is required. The study design was reviewed by the FDA. We have
signed a clinical trial agreement with Tufts Medical Group, and are negotiating similar agreement with two other University Hospitals
in the north-eastern United States the study sites). The first patient was randomized on June 25, 2024. The earliest possible date that
date would be available from the interim analysis would be January 31, 2025, assuming a minimum of 24 patients are enrolled prior to September
30, 2024. Further details are available on the clinicaltrials.gov website.36
Trial 2 will be an expanded use study utilizing
commercially available Arakoda. The Company, if approved by an Institutional Review Board (“IRB,” also known as an ethics
committee), plans to offer up to one year of Arakoda at no cost to about 10 patients per year (i.e., immunocompromised patients who have
previously failed standard of care treatment). Informed consent will be obtained from patients to collect a blood sample for PCR testing
at the end of treatment, and patients will be asked to complete a babesiosis symptom questionnaire. The goal of the study is to generate
additional prospective data to confirm the observation by Krause et al in a recent publication that an extended regimen of Tafenoquine
cured 80% of immunocompromised patients with relapsing babesiosis. This study will commence utilizing proceeds from the current offering.
More details about the study can be found on the clinicaltrials.gov website.37
Trial 3 will be an expanded use study utilizing commercially available
Arakoda. The Company, if approved by an IRB, plans to offer an approximately two-month supply of Arakoda at no cost to patients who have
a clinical diagnosis, are willing to submit biological samples for testing, and answer babesiosis and standardized fatigue inventories
before and after treatment. The goal of this study will be to ascertain whether Arakoda treatment improves patient-reported fatigue symptoms
and quality of life in individuals who have a diagnosis of chronic fatigue, symptoms consistent in severity with those suffered by chronic
fatigue patients, and who have molecular evidence of infection with Babesia. This trial will be gated by the outcome of an epidemiology
study we have financed at North Carolina State University (see below) and will require additional funding
In May 2024, we signed a research and collaboration
agreement with North Carolina State University in which the College of Veterinary Medicine will screen 300 archived blood samples from
patients exhibiting symptoms consistent with chronic fatigue symptoms by PCR for the presence of Babesia spp. In a second phase of the
study, positive samples will be sequenced to determine which Babesia spp are present. The data from this study will help define
whether the incidence of Chronic Babesiosis may be more widespread than amongst PTLDS patients, and also whether it is possible to define
a study population for Trial 3 (described) cost effectively.
In March 2024, we initiated, in collaboration
with the North Carolina State University College of Veterinary Medicine, a pilot study of Tafenoquine for treatment of canine babesiosis
in the United States under a sponsored research program. Should this potential collaboration be successful, we believe that the data from
that study may provide supportive data for the clinical babesiosis development program, and could provide proof of concept for an expanded
study to prove utility for veterinary indications.
We believe, if the Company does not become capital-limited,
that the results of the above studies will come to fruition in the first quarter of 2026, potentially facilitating submission of a supplementary
new drug application (or other appropriate regulatory filing) to FDA, with the goal of obtaining marketing approval of Arakoda for treatment
of Babesiosis. If successful, this will allow the Company to actively market Arakoda for Babesiosis.
| 36 |
See: https://classic.clinicaltrials.gov/ct2/show/NCT06207370. |
| 37 |
See: https://clinicaltrials.gov/study/NCT06478641. |
Parenteral Tafenoquine for Fungal Infections
We plan to support a series of studies in animal
models to determine whether single dose parenteral administration of Tafenoquine exhibits efficacy against Candida spp including
C. auris. These studies are being conducted under a sponsored research agreement with Monash University in Melbourne, Australia.
Combination Partner for Tafenoquine for Malaria
Most new antimalarial treatment products are developed
as drug combinations to proactively combat drug resistance. We believe that Tafenoquine, due to its long half-life and activity against
all parasite species and strains, would be an ideal partner in a drug combination. Recently, Kentucky Technology Inc. (“KTI”),
completed Phase IIA studies in P. vivax malaria, in which they evaluated the safety and efficacy of SJ733, their ATP4 inhibitor
in combination with Tafenoquine as the combination partner drug. It was recently announced that the SJ733 development program would be
partially supported by a grant from the Global Health Innovative Technology Fund (“GHIT”). As part of its shares for services
agreement with KTI, the Company recently received a detailed feasibility assessment and business plan for the project, including an assessment
of potential PRV eligibility, and is considering next steps in relation to potential involvement in this project.
Celgosivir for Antiviral Diseases
Reviewing prior studies of Celgosivir for Zika, Dengue, and RSV, it
is evident that the drug protects against the pathological effects of viruses through a combination of anti-inflammatory and antiviral
effects. These properties suggest it might have a beneficial effect in several viral diseases. Celgosivir is synthesized from Castanospermine,
which is obtained from botanical sources in low yield, making its inherent cost of goods potentially high. Castanospermine is also quite
water soluble making it amenable to intravenous formulation. We plan to conduct a proof-of-concept study in a hamster-COVID-19 model to
evaluate whether parenterally administered Castanospermine can ameliorate the pathological effects of SARS CoV-2 via modulation of cytokine
response to infection. Following this offering this project will be added to our statement of work for our services agreement with Florida
State University Research Foundation (“FSURF”), and will commence when there are sufficient proceeds from the sale of FSURF’s
60P shares to support this research. The data generated from the study will allow us to assess whether to move forward with IND enabling
studies of parenteral Castanospermine (or Celgosivir) for viral indications.
Post-Marketing Requirements
We have an FDA post-marketing requirement to conduct a malaria prophylaxis
study of Arakoda in pediatric and adolescent subjects. We proposed to the FDA, in late 2021, that this might not be safe to execute given
that malaria prevention is administered to asymptomatic individuals and that methemoglobinemia (damage to the hemoglobin in blood that
carries oxygen) occurred in 5% of patients, and exceeded a level of 10% in 3% of individuals in a study conducted by another sponsor in
pediatric subjects with symptomatic vivax malaria.38 The FDA has asked us to propose an alternate design, for which we submitted
a concept protocol in the fourth quarter of 2022, and submitted a full protocol in July, 2024. We estimate the cost of conducting the
study proposed by the FDA, if conducted in the manner suggested by the FDA, would be $2 million, and, due to the time periods required
to secure protocol approvals from the FDA and Ethics Committees, could not be initiated any earlier than the first quarter of 2026.
Capitalization and Future Financing
We plan to raise up to $15,000,000 million using the base prospectus
and this prospectus supplement in connection with future sales made pursuant to the Sales Agreement. It is possible that the funds that
are able to be raised using the base prospectus and prospectus supplement may be insufficient to achieve all our objectives. Therefore,
we will be seeking to raise additional funding as non-dilutively as possible, for example in the form of royalty or debt-based funding
of from non-profit groups interested in tick-borne diseases. There is no assurance that funds will be available on acceptable terms, or
that additional dilutive funding will not be required.
| 38 |
Velez et al 2021 - Lancet Child Adolesc Health 2022; 6: 86–95. |
Intellectual Property
We are co-owners, with the U.S. Army, of patents
in the United States and certain foreign jurisdictions directed toward use of Tafenoquine for malaria and have obtained an exclusive worldwide
license from the U.S. Army to practice these inventions. We also have an exclusive worldwide license to use manufacturing information
and non-clinical and clinical data that the U.S. Army possesses relating to use of Tafenoquine for all therapeutic applications and uses
excluding radical cure of symptomatic vivax malaria. We have submitted patent applications in the United States and certain foreign jurisdictions
for use of Tafenoquine for COVID-19, fungal lung infections, tick-borne diseases, and other infectious and non-infectious diseases in
which induction of host cytokines/inflammation is a component of the disease process. The United States Patent and Trademark Office (“USPTO”)
issued our first COVID-19 patent for Tafenoquine in 2023. We have optioned or licensed patents involving Celgosivir for the treatment
and prevention of Dengue (from the National University of Singapore), COVID-19 & Zika (Florida State University), and have pending
patent applications related to Celgosivir for RSV. We have optioned or own manufacturing methods related to Celgosivir. A detailed list
of our intellectual property is as follows:
Patents
| Title |
|
Patent No. |
|
Country |
|
Status |
|
US Patent Date |
|
Application No. |
|
|
Estimated/
Anticipated
Expiration
Date |
| Dosing Regimen For Use Of Celgosivir As An Antiviral Therapeutic For Dengue Virus Infections |
|
2013203400 |
|
Australia |
|
Granted |
|
|
|
|
2013203400+ |
|
|
10-April-2033* |
| Novel Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
2014228035 |
|
Australia |
|
Granted |
|
|
|
|
2014228035 |
|
|
14-Mar-2034* |
| Novel Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
MY-170991-A |
|
Malaysia |
|
Granted |
|
|
|
|
PI2015002372 |
|
|
14-Mar-2034* |
| Novel Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
378015 |
|
Mexico |
|
Granted |
|
|
|
|
MX/a/2015/013115 |
|
|
14-Mar-2034* |
| Novel Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
11201507254V |
|
Singapore |
|
Granted |
|
|
|
|
11201507254V |
|
|
14-Mar-2034* |
| Novel Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
Pending |
|
Singapore |
|
Pending |
|
|
|
|
10201908089V |
|
|
14-Mar-2034* |
| Novel Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
9763921 |
|
US |
|
Issued |
|
9/19/2017 |
|
|
14/772,873 |
|
|
14-Mar-2034^ |
| Novel Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
10517854 |
|
US |
|
Issued |
|
12/31/2019 |
|
|
15/706,845 |
|
|
14-Mar-2034^ |
| Dosing Regimens Of Celgosivir For The Treatment Of Dengue |
|
11219616 |
|
US |
|
Issued |
|
1/11/2022 |
|
|
16/725,387 |
|
|
14-Mar-2034^ |
| Novel Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naïve Subjects |
|
2015358566 |
|
Australia |
|
Granted |
|
|
|
|
2015358566 |
|
|
02-Dec-2035* |
| Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naïve Subjects |
|
2968694 |
|
Canada |
|
Granted |
|
|
|
|
2968694 |
|
|
02-Dec-2035* |
| Title |
|
Patent No. |
|
Country |
|
Status |
|
US Patent Date |
|
|
Application No. |
|
|
Estimated/
Anticipated
Expiration
Date |
| Novel Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naïve Subjects |
|
10342791 |
|
US |
|
Issued |
|
7/9/2019 |
|
|
15/532,280 |
|
|
02-Dec-2035^ |
| Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naive Subjects |
|
10888558 |
|
US |
|
Issued |
|
1/12/2021 |
|
|
16/504,533 |
|
|
02-Dec-2035^ |
| Novel Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naïve Subjects |
|
Pending |
|
Singapore |
|
Pending |
|
|
|
|
10201904908Q |
|
|
02-Dec-2035* |
| Novel Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naïve Subjects |
|
Pending |
|
EP |
|
Pending |
|
|
|
|
15865264.4 |
|
|
02-Dec-2035* |
| Novel Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naïve Subjects |
|
Pending |
|
Hong Kong |
|
Pending |
|
|
|
|
18103081.4 |
|
|
02-Dec-2035* |
| Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naive Subjects |
|
11,744,828 |
|
US |
|
Issued |
|
9/5/2023 |
|
|
17/145,530 |
|
|
02-Dec-2035^ |
| Novel Regimens Of Tafenoquine For Prevention Of Malaria In Malaria-Naïve Subjects |
|
Pending |
|
New Zealand |
|
Pending |
|
|
|
|
731813 |
|
|
02-Dec-2035* |
| Regimens of Tafenoquine for Prevention of Malaria in Malaria-Naive Subjects |
|
Pending |
|
US |
|
Pending |
|
|
|
|
18/240,049 |
|
|
02-Dec-2035^ |
| Novel Dosing Regimens Of Celgosivir For The Prevention Of Dengue |
|
2016368580 |
|
Australia |
|
Granted |
|
|
|
|
2016368580 |
|
|
09-Dec-2036* |
| Novel Dosing Regimens Of Celgosivir For The Prevention Of Dengue |
|
Pending |
|
Singapore |
|
Pending |
|
|
|
|
10201912141Y |
|
|
09-Dec-2036* |
| Dosing Regimens Of Celgosivir For The Prevention Of Dengue |
|
11000516 |
|
US |
|
Issued |
|
5/11/2011 |
|
|
16/060,945 |
|
|
09-Dec-2036^ |
| Methods For The Treatment And Prevention Of Lung Infections By Administration Of Tafenoquine |
|
Pending |
|
EP |
|
Pending |
|
|
|
|
21764438.4 |
|
|
02-Mar-2041* |
| Title |
|
Patent No. |
|
Country |
|
Status |
|
US Patent Date |
|
|
Application No. |
|
|
Estimated/
Anticipated
Expiration
Date |
| Methods For The Treatment And Prevention Of Lung Infections By Administration Of Tafenoquine |
|
Pending |
|
China |
|
Pending |
|
|
|
|
202180029643.7 |
|
|
02-Mar-2041* |
| Methods For The Treatment And Prevention Of Lung Infections By Administration Of Tafenoquine |
|
Pending |
|
Australia |
|
Pending |
|
|
|
|
2021231743 |
|
|
02-Mar-2041* |
| Methods For The Treatment And Prevention Of Lung Infections Caused By Gram-Positive Bacteria, Fungus, Or Virus By Administration Of Tafenoquine |
|
Pending |
|
Hong Kong |
|
Pending |
|
|
|
|
62023078645.6 |
|
|
02-Mar-2041* |
| Methods For The Treatment And Prevention Of Lung Infections Caused By Gram-Positive Bacteria, Fungus, Or Virus By Administration Of Tafenoquine |
|
11,633,391 |
|
US |
|
Issued |
|
4/25/2023 |
|
|
17/189,544 |
|
|
05-May-2041^ |
| Methods For The Treatment And Prevention Of Lung Infections Caused By Gram-Positive Bacteria, Fungus, Or Virus By Administration Of Tafenoquine |
|
Pending |
|
US |
|
Pending |
|
|
|
|
18/300,805 |
|
|
02-Mar-2041^ |
| Methods For The Treatment And Prevention Of Lung Infections Caused By Fungus By Administration Of Tafenoquine |
|
Pending |
|
US |
|
Pending |
|
|
|
|
17/683,679 |
|
|
01-Mar-2041^ |
| Methods For The Treatment And Prevention Of Lung Infections Caused By Sars-Cov-2 Virus By Administration Of Tafenoquine |
|
Pending |
|
US |
|
Pending |
|
|
|
|
17/683,718 |
|
|
01-Mar-2041^ |
| Treatment Of Human Coronavirus Infections Using Alpha-Glucosidase Glycoprotein Processing Inhibitors |
|
11369592 |
|
US |
|
Issued |
|
6/28/2022 |
|
|
17/180,140# |
|
|
19-Feb-2041^ |
| Treatment Of Human Coronavirus Infections Using Alpha-Glucosidase Glycoprotein Processing Inhibitors |
|
Pending |
|
US |
|
Pending |
|
|
|
|
17/664,693# |
|
|
19-Feb-2041^ |
| Treatment Of Human Coronavirus Infections Using Alpha-Glucosidase Glycoprotein Processing Inhibitors |
|
Pending |
|
EP |
|
Pending |
|
|
|
|
2021757552# |
|
|
19-Feb-2041* |
| Methods To Treat Respiratory Infection Utilizing Castanospermine Analogs |
|
Pending |
|
PCT |
|
Pending |
|
|
|
|
PCT/US23/26884 |
|
|
05-Jul-2043* |
| Methods To Treat Respiratory Infection Utilizing Castanospermine Analogs |
|
Pending |
|
US |
|
Pending |
|
|
|
|
18/218,202 |
|
|
05-Jul-2043^ |
| Title |
|
Patent No. |
|
Country |
|
Status |
|
US Patent Date |
|
|
Application No. |
|
|
Estimated/
Anticipated
Expiration
Date |
| Methods For The Treatment And Prevention Of Diseases Or Infections With MCP-1 Involvement By Administration Of Tafenoquine |
|
Pending |
|
PCT |
|
Pending |
|
|
|
|
PCT/US23/34169 |
|
|
30-Sep-2043* |
| Methods For The Treatment And Prevention Of Diseases Or Infections With MCP-1 Involvement By Administration Of Tafenoquine |
|
Pending |
|
US |
|
Pending |
|
|
|
|
18/375,070 |
|
|
30-Sep-2043^ |
| Treatment Of Zika Virus Infections Using Alpha Glucosidase Inhibitors |
|
10,328,061+ |
|
US |
|
Issued |
|
|
|
|
15/584,952+ |
|
|
2-May-2037^ |
| Treatment Of Zika Virus Infections Using Alpha Glucosidase Inhibitors |
|
10,561,642+ |
|
US |
|
Issued |
|
|
|
|
15/856,377+ |
|
|
2-May-2037^ |
| Methods For The Treatment And Prevention Of Non-Viral Tick-Borne Diseases And Symptoms Thereof |
|
Pending |
|
PCT |
|
Pending |
|
|
|
|
PCT/US24/25436 |
|
|
19-Apr-2044* |
| Methods For The Treatment And Prevention Of Non-Viral Tick-Borne Diseases And Symptoms Thereof |
|
Pending |
|
PCT |
|
Pending |
|
|
|
|
PCT/US24/25458 |
|
|
19-Apr-2044* |
| Methods For The Treatment And Prevention Of Non-Viral Tick-Borne Diseases And Symptoms Thereof |
|
Pending |
|
PCT |
|
Pending |
|
|
|
|
PCT/US24/25472 |
|
|
19-Apr-2044* |
| Methods For The Treatment And Prevention Of Non-Viral Tick-Borne Diseases And Symptoms Thereof |
|
Pending |
|
US |
|
Pending |
|
|
|
|
18/640,611 |
|
|
19-Apr-2044^ |
| Methods For The Treatment And Prevention Of Non-Viral Tick-Borne Diseases And Symptoms Thereof Of Treating Babesiosis |
|
Pending |
|
US |
|
Pending |
|
|
|
|
18/640,657 |
|
|
19-Apr-2044^ |
| Methods For The Treatment And Prevention Of Non-Viral Tick-Borne Diseases And Symptoms Thereof Of Treating Babesiosis |
|
Pending |
|
US |
|
Pending |
|
|
|
|
18/640,695 |
|
|
19-Apr-2044^ |
| * = |
For foreign patents and applications, the estimated and/or anticipated patent expiration is the date that is twenty years from the PCT filing date. For all issued Australian patents, this estimated date was also confirmed through the Australian patent office web database. |
| ^ = |
For issued U.S. patents, the estimated patent expiration was calculated using information from the front cover of the patent, i.e., 20 years from the date of the nonprovisional filing plus any listed Patent Term Adjustment less any time disclaimed through a Terminal Disclaimer. For pending U.S. applications, the anticipated patent expiration is the date twenty years from the earliest nonprovisional filing date and does not account for possible Patent Term Adjustment (PTA), Patent Term Extension (PTE), or Terminal Disclaimers. |
| & = |
For U.S. provisional applications that are not yet the subject of a nonprovisional or PCT application, the anticipated patent expiration was determined using the assumption that a non-provisional application or PCT will be filed one year after filing the provisional application with a term lasting twenty years from the date of that nonprovisional or PCT filing. This does not account for possible Patent Term Adjustment (PTA), Patent Term Extension (PTE), or Terminal Disclaimers. |
| + = |
60 Degrees Pharmaceuticals, Inc. is not a listed Applicant and Geoffrey S. Dow, Ph.D. is not a listed inventor. |
| # = |
60 Degrees Pharmaceuticals, Inc. is not a listed Applicant, but Geoffrey S. Dow, Ph.D. is a listed inventor. |
All patents not designated with a “+”
list Geoffrey S. Dow, Ph.D. as an inventor.
All patents not designated with a “+”
or a “#” list 60 Degrees Pharmaceuticals, Inc. as an applicant.
All estimated patent expiration dates and anticipated
patent expiration assume payment of any maintenance/annuity fees during the patent term.
Trademarks
| Country |
|
Mark |
|
Status |
|
Application
Number |
|
Date
Filed |
|
Registration
Date |
|
Registration
Number |
|
BIR
Ref Number |
|
Due
Date |
|
Due
Date Description |
| Australia |
|
KODATEF |
|
Registered |
|
1774631 |
|
2-Jun-16 |
|
6/2/2016 |
|
1774631 |
|
0081716-000029 |
|
2-Jun-26 |
|
Renewal
Due |
| Canada |
|
KODATEF |
|
Registered |
|
1785098 |
|
1-Jun-16 |
|
11/26/2019 |
|
TMA1,064,371 |
|
0081716-000028 |
|
26-Nov-29 |
|
Renewal
Due |
| Canada |
|
ARAKODA |
|
Registered |
|
1899317 |
|
15-May-18 |
|
8/20/2020 |
|
TMA1,081,180 |
|
0081716-000053 |
|
20-Aug-30 |
|
Renewal
Due |
| China |
|
KODATEF |
|
Registered |
|
20842242 |
|
2-Aug-16 |
|
9/28/2017 |
|
20842242 |
|
0081716-000035 |
|
27-Sep-27 |
|
Renewal
Due |
| European Union |
|
KODATEF |
|
Registered |
|
15508872 |
|
3-Jun-16 |
|
9/21/2016 |
|
15508872 |
|
0081716-000034 |
|
3-Jun-26 |
|
Renewal
Due |
| European Union |
|
ARAKODA |
|
Registered |
|
17900852 |
|
16-May-18 |
|
9/20/2018 |
|
17900852 |
|
0081716-000054 |
|
16-May-28 |
|
Renewal
Due |
| Israel |
|
KODATEF |
|
Registered |
|
285476 |
|
6-Jun-16 |
|
6/6/2016 |
|
285476 |
|
0081716-000033 |
|
6-Jun-26 |
|
Renewal
Due |
| New Zealand |
|
KODATEF |
|
Registered |
|
1044407 |
|
7-Jun-16 |
|
12/8/2016 |
|
1044407 |
|
0081716-000031 |
|
6-May-26 |
|
Renewal
Due |
| Russian Federation |
|
KODATEF |
|
Registered |
|
2016720181 |
|
6-Jun-16 |
|
7/10/2017 |
|
623174 |
|
0081716-000032 |
|
6-Jun-26 |
|
Renewal
Due |
| Singapore |
|
KODATEF |
|
Registered |
|
40201707950V |
|
2-May-17 |
|
11/8/2017 |
|
40201707950V |
|
0081716-000040 |
|
2-May-27 |
|
Renewal
Due |
| United Kingdom |
|
ARAKODA |
|
Registered |
|
17900852 |
|
16-May-18 |
|
9/20/2018 |
|
UK00917900852 |
|
0081716-000054 |
|
16-May-28 |
|
Renewal
Due |
| United Kingdom |
|
KODATEF |
|
Registered |
|
15508872 |
|
3-Jun-16 |
|
9/21/2016 |
|
UK00915508872 |
|
0081716-000072 |
|
3-Jun-26 |
|
Renewal
Due |
| United States of America |
|
TQ 100
& TABLET DESIGN |
|
Registered |
|
87608493 |
|
14-Sep-17 |
|
9/11/2018 |
|
5562900 |
|
0081716-000037 |
|
11-Sep-24 |
|
Section
8 & 15 Due |
| United States of America |
|
ARAKODA |
|
Registered |
|
87688137 |
|
16-Nov-17 |
|
12/31/2019 |
|
5950691 |
|
0081716-000050 |
|
31-Dec-25 |
|
Section
8 & 15 Due |
| United States of America |
|
KODATEF |
|
Allowed
- 02/16/2021 |
|
90072885 |
|
24-Jul-20 |
|
01/03/2024 |
|
|
|
0081716-000069 |
|
16-Aug-23 |
|
Abandoned |
| United States of America |
|
KODATEF |
|
Pending |
|
98363219 |
|
24-Jan-18 |
|
01/18/2024 |
|
|
|
0081716-000074 |
|
|
|
|
Key Relationships & Licenses
On May 30, 2014, we entered into the Exclusive License Agreement (the
“2014 NUS-SHS Agreement”) with National University of Singapore (“NUS”) and Singapore Health Services Pte Ltd
(“SHS”) in which we were granted a license from NUS and SHS with respect to their share of patent rights regarding “Dosing
Regimen for Use of Celgosivir as an Antiviral Therapeutic for Dengue Virus Infection” to develop, market and sell licensed products.
The 2014 NUS-SHS Agreement continues in force until the expiration of the last to expire of any patents under the patent rights unless
terminated earlier in accordance with the 2014 NUS-SHS Agreement. We are obligated to pay royalties at the rate of 1.5% of gross sales.
On July 15, 2015, we entered into the Exclusive
License Agreement with the U.S. Army Medical Materiel Development Activity (the “U.S. Army”), which was subsequently amended
(the “U.S. Army Agreement”), in which we obtained a license to develop and commercialize the licensed technology with respect
to all therapeutic applications and uses excluding radical cure of symptomatic vivax malaria. This exclusion does not impact our ability
to market Arakoda for the FDA-approved use, which is the prevention of malaria utilizing the indicated dose in asymptomatic individuals
traveling to high-malaria or malaria-prone regions (whereas the license exclusion relates to its use to treat symptomatic vivax malaria
in a patient already presenting with that disease). The term of the U.S. Army Agreement will continue until the expiration of the last
to expire of the patent application or valid claim of the licensed technology, or 20 years from the start date of the U.S. Army Agreement,
unless terminated earlier by the parties. We will be required to make a minimum annual royalty payment of 3% of net sales (as defined
in the U.S. Army Agreement) for net sales < $35 million, and 5% of net sales greater than $35 million, with US government sales excluded
from the definition of net sales. In addition, we must pay fees upon the achievement of certain milestones, including a sales-based milestone
fee of $75,000 once cumulative net sales from all sources exceeds $6 million (which milestone was achieved during the year ended December
31, 2023), $100,000 if we are acquired or merge, and regulatory approval milestone payments once marketing authorizations are achieved
in Canada ($5,000) and Europe ($5,000). Also, we will be required to obtain the U.S. Army Medical Materiel Development Activity’s
consent prior to a change of control of the Company, which consent was obtained on September 2, 2022.
On September 15, 2016, we entered into the Exclusive
License Agreement (the “2016 NUS-SHS Agreement”) with National University of Singapore and Singapore Health Services Pte Ltd
(“SHS”) in which we were granted a license from NUS and SHS with respect to their share of patent rights regarding “Novel
Dosing Regimens of Celgosivir for The Prevention of Dengue” to develop, market and sell licensed products. The 2016 NUS-SHS Agreement
continues in force until the expiration of the last to expire of any patents under the patent rights unless terminated earlier in accordance
with the 2016 NUS-SHS Agreement. We are obligated to pay at the rate of 1.5% of gross sales or minimum annual royalty ($5,000 in 2022
and $15,000 in 2023). In July 2022, we renegotiated the timing of a license fee of $85,000 Singapore Dollars, payable to NUS, such that
payment would be due at the earlier of (i) enrollment of a patient in a Phase II clinical trial involving Celgosivir, (ii) two years from
the agreement date and (iii) an initial public offering.
On February 15, 2021, we entered into the Inter-Institutional
Agreement with FSURF (the “FSURF Agreement”) in which FUSRF granted us the right to manage the licensing of intellectual property
created at FSURF. The term of the FSURF Agreement expires five years from February 15, 2021. After deduction of a 5% administrative fee
by FSURF, capped at $15,000 annually, and reimbursement of patent prosecution expenses, we will receive 20% of license income and FSURF
will receive 80% of license income. Payments of license income shall be paid in U.S. dollars quarterly each year. On February 19, 2021,
we entered into an agreement with FSURF, subsequently amended on February 15, 2023, and again on March 25, 2024, that collectively granted
an option, effective through March 24, 2025, to us to license methods for purifying Castanospermine and its use for the treatment of COVID-19.
On August 19, 2021, we entered into an agreement with FSURF, subsequently amended on February 15, 2023, and again on March 25, 2024, that
collectively granted an option, effective through March 24, 2025, to us to license a patent relating to the use of alpha glucosidase inhibitors
(including Castanospermine and Celgosivir) for treatment of Zika infections.
Ending upon July 12, 2033 or the conversion or
redemption in full of all of the shares of Series A Preferred Stock owned by Knight, we will pay Knight a royalty equal to 3.5% of our
net sales, where “net sales” has the same meaning as in the U.S. Army Agreement. Upon succeeding with the qualified IPO,
at the end of the quarter and each thereafter the royalty will be calculated, and payment will be made within fifteen days.
On February 13, 2024, our majority-owned Australian
subsidiary, 60P Australia Pty Ltd, and Monash University entered into the Research Services Agreement (the “Agreement”) in
which Monash University agreed to provide research services, including among other things, testing the efficacy of Tafenoquine against
candidemia, confirming suitable fungal infection dosage and determining the pharmacokinetics of Tafenoquine following intraperitoneal
drug administration (collectively, the “Services”). The commencement date of the Agreement was effective as of February 5,
2024, and the commencement of experiments was May 2024 and the anticipated completion date is on November 30, 2024. The Company agreed
to pay Monash University $90,167 AUD on April 1, 2024 and $90,167 AUD upon the completion of the Services.
On March 20, 2024, we signed a sponsored research
agreement with North Carolina State University to conduct a pilot study to evaluate the efficacy of Tafenoquine in canine babesiosis.
The research is expected to be completed by March 30, 2026. The Company will retain ownership of all data and inventions related to the
study, subject to retained right of North Carolina State University to utilize study data or research use and publications. For a six-month
period following notification by the University, the Company retains first right of refusal to negotiate a license to utilize any inventions
or data generated by the University relevant to Tafenoquine but not occurring as a direct result of performing the planned studies. The
Company agreed to pay North Carolina State University $12,000 upon contract execution, $8,000 around October 1st, 2024, then
$3,869 around April 1st, 2025 when work is expected to be completed.
On May 10, 2024, we entered into a sponsored research
agreement with North Carolina State University to conduct a study to evaluate the incidence of Babesia infection amongst archived blood
samples from patients with chronic fatigue and neurocognitive problems. The research will be completed by May 31, 2025. The Company retains
the right to use all study data, joint and University-owned inventions for non-commercial purposes and first right of refusal to negotiate
a royalty bearing license for commercial purposes for any university own intellectual property or ownership interest in jointly-owned
intellectual property. The Company agreed to pay North Carolina State University $37,620 upon contract execution, $22,572 after six months,
then $15,048 upon completion of the contract at twelve months.
On May 29, 2024, we signed a clinical trial agreement
with Tufts Medicine, Inc, which specifies the terms on which Tufts will act as a clinical trial site for our Tafenoquine-Babesiosis study.
The Company retains the first right to negotiate an exclusive license to any intellectual property owned by Tufts Medicine, Inc, arising
from the study.
Corporate Structure
60 Degrees Pharmaceuticals, Inc. is a Delaware
corporation that was incorporated on June 1, 2022.
On June 1, 2022, 60 Degrees Pharmaceuticals, LLC,
a District of Columbia limited liability company (“60P LLC”), entered into the Agreement and Plan of Merger with 60 Degrees
Pharmaceuticals, Inc., pursuant to which 60P LLC merged into 60 Degrees Pharmaceuticals, Inc. The value of each outstanding member’s
membership interest in 60P LLC was correspondingly converted into common stock of 60 Degrees Pharmaceuticals, Inc., par value $0.0001
per share, with a cost-basis equal to $5.00 per share.
Our majority-owned subsidiary,
60P Australia Pty Ltd, an Australian proprietary company limited by shares (“60P Australia”), was formed and registered in
Queensland on December 3, 2013, and conducts operations in Australia.
60P Australia previously
solely owned a Singaporean subsidiary company, 60P Singapore Pte. Ltd., which dissolved at our election in the second quarter of 2022.
Going Concern
Our independent auditors have issued a report
raising substantial doubt of our ability to continue as a going concern. We anticipate that we will require additional capital to continue
as a going concern and expand our operations in accordance with our current business plan.
Suppliers
We have quality and contract manufacturing agreements
relating to Arakoda in place with Piramal Enterprises Limited (API, tablets) and PCI Pharma Services (secondary packaging) (“PCI”)
and supply/quality/pharmacovigilance agreements in place with Biocelect Pty Ltd, Scandinavian Biopharma, and Knight Therapeutics Inc.
(to allow supply of Arakoda/Kodatef to Australia, Europe and Canada/Israel/Latin America and Russia, respectively). As of the date of
this prospectus, we have not supplied any of our products to Russia nor do we anticipate supplying any of our products to Russia in the
near future.
Summary Risk Factors
Our business is subject to a number of risks.
You should be aware of these risks before making an investment decision. These risks are discussed more fully in the section of this prospectus
titled “Risk Factors,” which begins on page 18 of this prospectus. These risks include, among others, that:
| |
● |
Our financial statements have been prepared on a going-concern basis and our continued operations are in doubt; |
| |
● |
We have incurred net losses since our inception and if we continue to incur net losses in the foreseeable future, the market price of our common stock may decline; |
| |
● |
There is no assurance that we will be profitable; |
| |
● |
There is no assurance that we will be eligible for Australian government research and development tax rebates; |
| |
● |
Our financial condition and results of operations may be adversely affected by COVID-19; |
| |
● |
If we are not able to successfully develop, obtain FDA approval for,
and provide for the commercialization of non-malaria prevention indications for Tafenoquine (Arakoda or other regimen) or Celgosivir
in a timely manner, we may not be able to expand our business operations; |
| |
● |
Our clinical trials for our product candidates may not yield results that will enable us to further develop our products and obtain regulatory approvals necessary to sell them; |
| |
● |
Our clinical trials for our product candidates
may not recruit sufficient patients, or recruitment of patients may be slower than expected, or we may run out of funds before completion
of our clinical trial. If so, we will not be able to complete the trials necessary for regulatory approval; |
| |
● |
We expect to depend on existing and future collaborations with third parties for the development of some of our product candidates. If those collaborations are not successful, we may not be able to complete the development of these product candidates; |
| |
● |
Even if one of our product candidates receives marketing approval, it may fail to achieve the degree of market acceptance by physicians, patients, third-party payors and others in the medical community necessary for commercial success, in which case we may not generate significant revenues or become profitable; |
| |
● |
Any future clinical trial for Arakoda will require screening for G6PD
deficiency in order to safely administer the product. In the U.S., G6PD testing can be obtained through commercial pathology services
which is associated with delays. The use of a third-party diagnostic provider of point of care testing may be required and we do not
directly control the timing, conduct, expense of such testing or the timing of market entry into the U.S.; |
| |
● |
Our product candidates are subject to extensive regulation, which can be costly and time-consuming, and unsuccessful or delayed regulatory approvals could increase our future development costs or impair our future revenue; |
| |
● |
If our product candidates receive regulatory approval, we would be subject to ongoing regulatory obligations and restrictions, which will continue to change, and which may result in significant expenses and limit our ability to develop and commercialize other potential products; |
| |
● |
We have no manufacturing capacity which puts us at risk of lengthy and costly delays of bringing our products to market; |
| |
● |
We rely on relationships with third-party contract manufacturers and raw material suppliers, which limits our ability to control the availability of, and manufacturing costs for, our product candidates; |
| |
● |
Our future growth depends on our ability to successfully commercialize
Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, Celgosivir and our other product candidates,
and we can provide no assurance that we will successfully commercialize Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria
prevention indications, Celgosivir and other product candidates; |
| |
● |
Health care reform measures could materially and adversely affect our business; |
| |
● |
Our competitors may be better positioned in the marketplace and thereby may be more successful than us at developing, manufacturing and marketing approved products; |
| |
● |
We compete in an industry characterized by extensive research and development efforts and rapid technological progress. New discoveries or commercial developments by our competitors could render our potential products obsolete or non-competitive; |
| |
● |
We would be subject to applicable regulatory approval requirements of the foreign countries in which we market our products, which are costly and may prevent or delay us from marketing our products in those countries; |
| |
● |
Defending against claims relating to improper handling, storage or disposal of hazardous chemicals, radioactive or biological materials could be time consuming and expensive; |
| |
● |
Geopolitical conditions, including direct or indirect acts of war or terrorism could have an adverse effect on our operations and financial results; |
| |
● |
If product liability lawsuits are successfully brought against us,
then we will incur substantial liabilities and may be required to limit commercialization of Arakoda, Celgosivir or other product candidates; |
| |
● |
Our intellectual property rights may not preclude competitors from developing competing products and our business may suffer; |
| |
● |
If the manufacture, use or sale of our products infringe on the intellectual property rights of others, we could face costly litigation, which could cause us to pay substantial damages or licensing fees and limit our ability to sell some or all of our products; |
| |
● |
We may not be able to protect our intellectual property rights throughout the world; |
| |
● |
Patent terms may be inadequate to protect our competitive position on our product candidates for an adequate amount of time; |
| |
● |
The earliest Paragraph IV certification date for Arakoda has passed. Generic companies may file an ANDA at any time, and successful challenge of our malaria use patents would negatively impact our business; |
| |
● |
We may not be able to maintain the listing of our common stock on Nasdaq, which could adversely affect our liquidity and the trading volume and market price of our common stock and decrease or eliminate your investment; |
| |
● |
Any failure to maintain effective internal controls over financial reporting could have an adverse impact on us; |
| |
|
|
| |
● |
If you purchase shares of our common stock in this offering, you may incur immediate and substantial dilution in the book value of your shares if the offering price per share is greater than the as adjusted net tangible book value per share at the time of issuance; and |
| |
● |
We are an “emerging growth company” and a “smaller reporting company” under the JOBS Act, and we cannot be certain if the reduced disclosure requirements applicable to emerging growth companies and smaller reporting companies will make our common stock less attractive to investors. |
Recent Developments
Tafenoquine Babesiosis Clinical Trial.
In April 2024, the FDA provided some questions
and recommendations related to the planned clinical trial that will study the use of Tafenoquine in treating babesiosis.
The Company opened its first clinical site on June 13, 2024, following execution of a clinical agreement with Tufts Medical, Inc on May
29, 2024. The first patient in the trial was enrolled on June 25, 2027.
Nasdaq Notice of Failure to Comply with
Continued Listing Standards
On February 27, 2024, we received a letter from
the Nasdaq Listing Qualifications Staff of The Nasdaq Stock Market LLC (“Nasdaq”) stating that for the 30 consecutive business
day period between January 11, 2024 and February 27, 2024, our common stock had not maintained a minimum closing bid price of $1.00 per
share required for continued listing on The Nasdaq Capital Market pursuant to Nasdaq Listing Rule 5550(a)(2) (the “Bid Price Rule”).
Pursuant to Nasdaq Listing Rule 5810(c)(3)(A), we were provided an initial period of 180 calendar days, or until August 26, 2024 (the
“Compliance Period”), to regain compliance with the Bid Price Rule.
To regain compliance, the closing bid price of
our common stock must meet or exceed $1.00 per share for a minimum of 10 consecutive trading days, unless extended by Nasdaq under
Nasdaq Rule 5810(c)(3)(H), prior to August 26, 2024.
If we do not regain compliance with the Bid Price
Rule by August 26, 2024, we may be eligible for an additional 180-day period to regain compliance if we meet all of the other Nasdaq listing
criteria and if Nasdaq does not believe we will not be able to regain compliance within such 180-day period. If we cannot regain compliance
during the Compliance Period or any subsequently granted compliance period, our common stock will be subject to delisting.
Our common stock continues to be listed on The
Nasdaq Capital Market under the symbol “SXTP.” We are currently evaluating our options for regaining compliance.
The notice from Nasdaq has no immediate effect
on the listing or trading of our common stock on The Nasdaq Capital Market and does not affect our business, operations or reporting requirements
with the SEC.
Monash University
On February 13, 2024, our majority-owned Australian
subsidiary, 60P Australia Pty Ltd, and Monash University entered into the Research Services Agreement (the “Agreement”) in
which Monash University agreed to provide research services, including among other things, testing the efficacy of Tafenoquine against
candidemia, confirming suitable fungal infection dosage and determining the pharmacokinetics of Tafenoquine following intraperitoneal
drug administration (collectively, the “Services”). The commencement date of the Agreement was effective as of February 5,
2024, and the anticipated commencement of experiments and the completion date is in May 2024 and on November 30, 2024, respectively. The
Company agreed to pay Monash University $90,167 AUD on April 1, 2024 and $90,167 AUD upon the completion of the Services.
North Carolina State University
On March 20, 2024, we signed a sponsored
research agreement with North Carolina State University to conduct a pilot study to evaluate the efficacy of Tafenoquine in canine babesiosis.
The research is expected to be completed by March 30, 2026. The Company will retain ownership of all data and inventions related to the
study, subject to retained right of North Carolina State University to utilize study data or research use and publications. For a six-month
period following notification by the University, the Company retains first right of refusal to negotiate a license to utilize any inventions
or data generated by the University relevant to Tafenoquine but not occurring as a direct result of performing the planned studies.
On May 10, 2024, we entered into
a sponsored research agreement with North Carolina State University to conduct a study to evaluate the incidence of Babesia infection
amongst archived blood samples from patients with chronic fatigue and neurocognitive problems. The research will be completed by May 31,
2025. The Company retains the right to use all study data, joint and University-owned inventions for non-commercial purposes and first
right of refusal to negotiate a royalty bearing license for commercial purposes for any university own intellectual property or ownership
interest in jointly-owned intellectual property.
Clinical Trial Agreement with Tufts Medicine,
Inc
On May 29, 2024, we signed a clinical
trial agreement with Tufts Medicine, Inc, which specifies the terms on which Tufts will act as a clinical trial site for our Tafenoquine-Babesiosis
study. The Company retains the first right to negotiate an exclusive license to any intellectual property owned by Tufts Medicine, Inc,
arising from the study.
January 2024 Offering
On January 29, 2024, the Company entered
into an Underwriting Agreement with WallachBeth Capital LLC, as representative of the underwriters listed on Schedule I thereto,
relating to the Company’s public offering (the “Offering”) of 5,260,901 units (the “Units”) at an
offering price of $0.385 per Unit and 999,076 pre-funded units (the “Pre-Funded Units”) at an offering price of $0.375
per Pre-Funded Unit. Each Unit consists of one share of common stock and one warrant exercisable for one share of common stock (the
“Warrant”). Each Warrant has an exercise price of $0.4235 per share, is exercisable immediately upon issuance and
expires five years from the date of issuance. Each Pre-Funded Unit consists of one pre-funded warrant exercisable for one share of
common stock (the “Pre-Funded Warrant”) and one warrant identical to the Warrants included in the Units. The purchase
price of each Pre-Funded Unit is equal to the price per Unit sold to the public in the offering, minus $0.01, and the exercise price
of each Pre-Funded Warrant is $0.01 per share. The Pre-Funded Warrants are immediately exercisable and may be exercised at any time
until all of the Pre-Funded Warrants are exercised in full. WallachBeth Capital LLC partially exercised its
over-allotment option with respect to 818,177 Warrants on January 31, 2024, and purchased an additional 50 shares of common stock at
a purchase price of $0.3750 and 50 Warrants at a purchase price of $0.01 on February 14, 2024.
The Company also issued to WallachBeth Capital
LLC warrants (the “Representative Warrants”) to purchase 375,599 shares of the Company’s common stock, which is equal
to 6% of the common stock sold that were part of the Units and the pre-funded warrants sold that were part of the Pre-Funded Units in
the Offering, at an exercise price of $0.4235 per share (110% of the offering price per Unit). The Representative Warrants may be exercised
beginning on January 31, 2024 until January 31, 2029.
The net proceeds to the Company from the Offering
were approximately $1.9 million, after deducting underwriting discounts and commissions and the payment of other offering expenses associated
with the Offering that were payable by the Company. The Company paid the Underwriter an underwriting discount equal to 8.0% of the gross
proceeds of the Offering and a non-accountable expense fee equal to 1.5% of the gross proceeds of the Offering.
Information Regarding our Capitalization
As of July 12, 2024, we had 12,206,116 shares
of common stock issued and outstanding. Additional information regarding our issued and outstanding securities may be found under “Market
for Common Equity and Related Stockholder Matters” and “Description of Securities.”
Unless otherwise specifically stated, information
throughout this prospectus does not assume the exercise of outstanding options or warrants to purchase shares of our common stock.
Corporate Information
Our principal executive offices are located at
1025 Connecticut Avenue NW Suite 1000, Washington, D.C. 20036. Our corporate website address is 60degreespharma.com. Our telephone
number is (202) 327-5422. The information included on our website is not part of this prospectus.
Implications of Being an Emerging Growth Company
and a Smaller Reporting Company
We are an “emerging growth company,”
as defined in the JOBS Act. We will remain an emerging growth company until the earlier of (i) the last day of the fiscal year following
the fifth anniversary of the date of the first sale of our common stock pursuant to an effective registration statement under the Securities
Act; (ii) the last day of the fiscal year in which we have total annual gross revenues of $1.235 billion or more; (iii) the date on which
we have issued more than $1 billion in nonconvertible debt during the previous three years; or (iv) the date on which we are deemed to
be a large accelerated filer under applicable SEC rules. We expect that we will remain an emerging growth company for the foreseeable
future, but cannot retain our emerging growth company status indefinitely and will no longer qualify as an emerging growth company on
or before the last day of the fiscal year following the fifth anniversary of the date of the first sale of our common stock pursuant to
an effective registration statement under the Securities Act. For so long as we remain an emerging growth company, we are permitted and
intend to rely on exemptions from specified disclosure requirements that are applicable to other public companies that are not emerging
growth companies.
These exemptions include:
| |
● |
being permitted to provide only two years of audited financial statements, in addition to any required unaudited interim financial statements, with correspondingly reduced “Management’s Discussion and Analysis of Financial Condition and Results of Operations” disclosure; |
| |
● |
not being required to comply with the requirement of auditor attestation of our internal controls over financial reporting; |
| |
● |
not being required to comply with any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements; |
| |
● |
reduced disclosure obligations regarding executive compensation; and |
| |
● |
not being required to hold a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved. |
An emerging growth company can take advantage
of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act to comply with new or revised accounting standards.
This allows an emerging growth company to delay the adoption of certain accounting standards until those standards would otherwise apply
to private companies. We have elected to avail ourselves of this extended transition period and, as a result, we will not be required
to adopt new or revised accounting standards on the dates on which adoption of such standards is required for other public reporting companies.
We are also a “smaller reporting company”
as defined in Rule 12b-2 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), and have elected to take
advantage of certain of the scaled disclosure available for smaller reporting companies. We will remain a smaller reporting company until
the end of the fiscal year in which (1) we have a public common equity float of more than $250 million, or (2) we have annual revenues
for the most recently completed fiscal year of more than $100 million and a public common equity float or public float of more than
$700 million. We also would not be eligible for status as a smaller reporting company if we become an investment company, an asset-backed
issuer or a majority-owned subsidiary of a parent company that is not a smaller reporting company.
We have elected to take advantage of certain of
the reduced disclosure obligations in the registration statement of which this prospectus is a part and may elect to take advantage of
other reduced reporting requirements in future filings. As a result, the information that we provide to our stockholders may be different
from what you might receive from other public reporting companies in which you hold equity interests.
THE OFFERING
| Common stock offered by us pursuant to this prospectus supplement |
|
Shares of our common stock having an aggregate offering price of up to $1,253,603. |
| |
|
|
| Common stock to be outstanding after this offering |
|
Up to 15,334,098 shares (as more fully described
in the notes following this table), assuming sales of 3,571,520 shares of our common stock in this offering at an offering price of $0.351
per share, which was the last reported sale price of our common stock on The Nasdaq Capital Market on June 7, 2024. The actual number
of shares issued will vary depending on the sales price under this offering. |
| |
|
|
| Manner of Offering |
|
“At the market offering” as defined in Rule 415(a)(4) under the Securities Act, including sales made directly on or through The Nasdaq Capital Market, or any other existing trading market for our common stock in the United States or to or through a market maker, through the Sales Agent. See “Plan of Distribution” on page S-56 of this prospectus supplement. |
| |
|
|
| Use of Proceeds |
|
We intend to use the net proceeds from this offering for general corporate purposes and working capital. See “Use of Proceeds” on page S-55 of this prospectus supplement. |
| |
|
|
| Risk Factors |
|
An investment in our common stock involves a high degree of risk. See the information contained in or incorporated by reference under “Risk Factors” on page S-22 of this prospectus supplement and under similar headings in the other documents that are incorporated by reference herein, as well as the other information included in or incorporated by reference in this prospectus supplement and the accompanying prospectus. |
| |
|
|
| The Nasdaq Capital Market symbol for its common stock |
|
“SXTP” |
The number of shares of our common stock
to be outstanding after this offering is based on 11,762,578 shares of our common stock outstanding as of March 31, 2024. Unless specifically stated
otherwise, the information in this prospectus supplement is as of March 31, 2024 and excludes:
| |
● |
232,709 shares of common stock reserved for issuance under the 60 Degrees Pharmaceuticals, Inc. 2022 Equity Incentive Plan; (ii) 10,656,690 shares of common stock issuable upon exercise of outstanding warrants at a weighted average exercise price of $2.07 per share; (iii) 37,736 shares of common stock issuable upon the exercise of outstanding options to purchase common stock at a weighted average exercise price of $5.30 per share; (iv) 64,000 shares of common stock issuable pursuant to fully vested restricted stock units which have not yet been issued as of March 31, 2024; (v) 84,906 shares of common stock issuable upon exercise of the representative warrants issued to the representative in our initial public offering at an exercise price equal to 110% of the public offering price of the Units; (vi) 375,599 shares of common stock issuable upon exercise of the representative warrants to the representative in our public offering at an exercise price equal to 110% of the public offering price of the Units; and (vii) shares of common stock issuable upon the conversion of 78,803 shares of Series A Preferred Stock. |
RISK FACTORS
Investing in our common stock involves a high
degree of risk. Before investing in our common stock, you should carefully consider the risks described below, together with all of the
other information contained in this prospectus supplement, including from our most recent Annual Report on Form 10-K and any subsequent
Quarterly Reports on Form 10-Q or Current Reports on Form 8-K, as well as any amendments or update to our risk factors thereto reflected
in subsequent filings with the SEC. Some of these factors relate principally to our business and the industry in which we operate. Other
factors relate principally to your investment in our securities. The risks and uncertainties described therein and below are not the only
risks facing us. Additional risks and uncertainties not presently known to us or that we currently deem immaterial may also materially
and adversely affect our business and operations.
If any of the matters included in the following
risks were to occur, our business, financial condition, results of operations, cash flows or prospects could be materially and adversely
affected. In such case, you may lose all or part of your investment.
Risks Related to Our
Business
Our financial statements
have been prepared on a going-concern basis and our continued operations are in doubt.
The financial statements have been prepared on
a going concern basis under which an entity is considered to be able to realize its assets and satisfy its liabilities in the ordinary
course of business. Our future operations are dependent upon the identification and successful completion of equity or debt financing
and the achievement of profitable operations at an indeterminate time in the future. There can be no assurances that we will be successful
in completing an equity or debt financing or in achieving profitability.
We have incurred
net losses since our inception and if we continue to incur net losses in the foreseeable future, the market price of our common stock
may decline.
To date, we have financed
our operations primarily through the issuance of equity, promissory notes and convertible notes. We incurred annual net losses of $3,813,800 in
2023 and $6,177,784 in 2022 and operating losses were $5,154,583 in 2023 and $1,750,445 in 2022. We had an accumulated deficit
of $32,580,850 as of December 31, 2023 ($28,815,148 as of December 31, 2022). For the three-month period ended March 31, 2024, we
achieved a net gain of $427,985 and an operating loss of $1,331,659. As of March 31, 2024, we had an accumulated deficit
of $32,150,380.
We may not achieve or
maintain profitability in the future. In particular, we expect that our expenses relating to sales and marketing and product development
and support, as well as our general and administrative costs, will increase, requiring us to increase sales in order to achieve and maintain
profitability. If we do not achieve and maintain profitability, our financial condition will be materially and adversely affected. We
would eventually be unable to continue our operations unless we were able to raise additional capital. We may not be able to raise any
necessary capital on commercially reasonable terms or at all. If we fail to achieve or maintain profitability on a quarterly or annual
basis within the timeframe expected by investors, the market price of our common stock may decline.
There is no assurance that we will be profitable.
There is no assurance that we will earn profits
in the future, or that profitability will be sustained. There is no assurance that future revenues will be sufficient to generate the
funds required to continue our business and product development and marketing activities. If we do not have sufficient capital to fund
our operations, we may be required to reduce our sales and marketing efforts or forego certain business opportunities.
There is no assurance we will be eligible
for Australian Government research and development tax credits and eligibility rules might change in a manner that jeopardizes our business.
There is no assurance that the Australian government
will pay research and development rebates on our research activities conducted by our subsidiary, 60P Australia Pty Ltd, in Australia.
There is no assurance that we will be able to demonstrate that our subsidiary will have < $20 million AUD in aggregate turnover amongst
beneficial owners with > 40% beneficial interest. If any of these risks materialize, we might not be able to secure tax rebates on
relevant eligible business in Australia, which would negatively impair our business.
We have limited revenues to date, and any
potential revenues from commercial use may not materialize in the future.
We have earned limited revenues to date from Arakoda.
Any potential revenues from the sale of current approved commercial use may not materialize in the future. There is no guarantee that
we will be able to generate revenue in the future. No assurance can be given that our efforts from sale of current approved products for
commercial use will be successful in the future.
Our financial condition and results of operations
may be adversely affected by COVID-19 or similar global pandemic.
A significant outbreak, epidemic or pandemic of
contagious diseases in any geographic area in which we operate or plan to operate could result in a health crisis adversely affecting
the economies, financial markets and overall demand for our services in such areas. In addition, any preventative or protective actions
that governments implement or that we take in response to a health crisis, such as travel restrictions, quarantines, or site closures,
may interfere with the ability of our employees, suppliers and customers to perform their responsibilities. Such results could have a
material adverse effect on our business.
COVID-19 created significant volatility, uncertainty
and economic disruption. To date, COVID-19 has affected nearly all regions around the world. In the United States, businesses as well
as federal, state and local governments implemented significant actions to mitigate this public health crisis. While we cannot predict
the duration or scope of COVID-19, it may negatively impact our business and such impact could be material to our financial results, condition
and outlook related to:
| |
● |
disruption to our operations or the operations of our suppliers, through the effects of business and facilities closures, worker sickness and COVID-19 related inability to work, social, economic, political or labor instability in affected areas, transportation delays, difficulty in enrolling patients, travel restrictions and changes in operating procedures, including for additional cleaning and safety protocols; |
| |
● |
increased volatility or significant disruption of global financial markets due in part to any future COVID-19 outbreak, which could have a negative impact on our ability to access capital markets and other funding sources, on acceptable terms or at all and impede our ability to comply with debt covenants; and |
| |
● |
the further spread of COVID-19, and the requirements to take action to mitigate the spread of any future COVID-19 outbreak (e.g., hygiene requirements or social distancing or other measures), will impact our ability to carry out our business as usual and may materially adversely impact global economic conditions, our business, results of operations, cash flows and financial condition. |
To the extent COVID-19 or a similar public health
threat has an impact on our business, it is likely to also have the effect of heightening many of the other risks described in this “Risk
Factors” section.
U.S. public sector procurement of Arakoda
might not materialize in the future, which could jeopardize our business.
Sales to the U.S. DoD were important to our revenue
stream in the recent past. Although, as of the date of this prospectus, we are not in discussions with the DoD about additional/future
procurement, we anticipate that if certain conditions/events described in this paragraph occur, our sales to DoD could develop; however,
there is no assurance that such conditions/events will occur. First, the position of Arakoda in the DoD formulary (Tricare, deployed personnel)
needs to be improved from second/third tier to at least equivalency with competing products (as is the case for civilian use as recommended
by the CDC). We believe that changes in pricing or reimbursement structure may be needed to secure that. Second, the shelf-life of the
existing product requires extension, which is known to be technically possible as the shelf-life of Kodatef in Australia is 48 months,
but appropriate data must be generated to meet FDA requirements. Finally, a change in the operational footprint of DoD deployments to
areas with higher malaria attack rates (e.g., the Liberia deployment to manage the Ebola outbreak in 2014) may lead to a rapid reassessment
by DoD of the position of Arakoda in the formulary (advancement of the last approved prophylactic antimalarial to co-equal standard of
care took thirteen years). If none of these events transpire, we would not have the opportunity for revenues and such failure would jeopardize
our business.
Supply chain disruptions across the globe,
including in the U.S., could jeopardize our business and harm our operations.
Global business interruptions
may adversely impact our third-party relationships whom we rely upon in our business as well as manufacturers, suppliers, and makers of
raw materials. If any such parties are adversely impacted by supply chain restrictions, or if they cannot obtain the necessary supplies,
or if such third parties need to prioritize other products or customers over us, we may experience delays or disruptions in our supply
chain, which could have a material and adverse impact on our business. Third-party manufacturers may also need to implement measures and
changes, or deviate from typical requirements because of the COVID-19 pandemic that may otherwise adversely impact our supply chain or
the quality of the resulting products or supplies. Depending on the change, we may need to obtain FDA approval or otherwise provide the
FDA with a notification of the change. As a result, we may not be able to obtain sufficient quantities of certain items, which could impair
our ability to commercialize our products and conduct the post-marketing studies requested by the FDA, in connection with the approval
of our goods. In addition, if there are continued or future disruptions, our third-party manufacturers may not be able to supply our other
potential product candidates, which would adversely affect our research and development activities.
We may lose the
services of key management personnel and may not be able to attract and retain other necessary personnel.
Changes in our management
could have an adverse effect on our business. This is especially an issue while our staff is small. We are dependent upon the active participation
of several key management personnel, including Geoffrey Dow, our President and Chief Executive Officer. We also do not carry key person
life insurance on any of our senior management or other key personnel. Hence, we may suffer if the services of our management were to
become unavailable to us in the future.
We must hire highly skilled
technical personnel as employees and as independent contractors in order to develop our products. As of the date of this prospectus, we
have two full-time employees, and we rely on two independent contractors to provide us with skilled technical support. The competition
for highly skilled technical, managerial and other personnel is intense and we may not be able to retain or recruit such personnel. Our
recruiting and retention success is substantially dependent on our ability to offer competitive salaries and benefits to our employees
and competitive compensation to contractors. We must compete with companies that possess greater financial and other resources than we
do and that may be more attractive to potential employees and contractors. To be competitive, we may have to increase the compensation,
bonuses, stock options and other fringe benefits offered to employees in order to attract and retain such personnel. The costs of retaining
or attracting new personnel may have a material adverse effect on our business and operating results. If we fail to attract and retain
the technical and managerial personnel needed to be successful, our business, operating results and financial condition could be materially
adversely affected.
Cybersecurity risks could adversely affect
our business and disrupt our operations.
The threats to network and data security are increasingly
diverse and sophisticated. Despite our efforts and processes to prevent breaches, our devices, as well as our servers, computer systems,
and those of third parties that we use in our operations are vulnerable to cybersecurity risks, including cyber-attacks such as viruses
and worms, phishing attacks, denial-of-service attacks, physical or electronic break-ins, employee theft or misuse, and similar disruptions
from unauthorized tampering with our servers and computer systems or those of third parties that we use in our operations, which could
lead to interruptions, delays, loss of critical data, and unauthorized access to user data. In addition, we may be the target of email
scams that attempt to acquire personal information or our assets. Despite our efforts to create security barriers to such threats, we
may not be able to entirely mitigate these risks. Any cyber-attack that attempts to obtain our or our users’ data and assets, disrupt
our service, or otherwise access our systems, or those of third parties we use, if successful, could adversely affect our business, operating
results, and financial condition, be expensive to remedy, and damage our reputation. In addition, any such breaches may result in negative
publicity, adversely affect our brand, decrease demand for our products and services, and adversely affect our operating results and financial
condition.
The illegal sale or distribution by third
parties of counterfeit versions of our products could have a negative impact on our business.
Pharmaceutical products are vulnerable to counterfeiting.
Third parties may illegally produce and distribute counterfeit versions of our products that are below the various manufacturing and testing
standards that our products undergo. Counterfeit products are often unsafe, ineffective and potentially life-threatening. As many counterfeit
products may be visually indistinguishable from their authentic versions, the presence of counterfeit products could affect overall consumer
confidence in the authentic product. A public loss of confidence in the integrity of pharmaceutical products in general or in any of our
products in particular due to counterfeiting could have a material adverse effect on our business, prospects, financial condition and
results of operations.
If we encounter difficulties enrolling patients
in any future clinical trials, our future trials could be delayed or otherwise adversely affected. Furthermore, our planned Babesiosis
trial may not necessarily yield sufficient results or patient participants.
If we have difficulty enrolling a sufficient number
of patients in any future clinical trial, including for Babesiosis studies for which the number of cases is unpredictable, we may need
to delay or terminate our trial, which would impair our ability to develop marketable products, and have a negative impact on our business.
Delays in enrolling patients in any future clinical trials would also adversely affect our ability to generate any product, milestone
and royalty revenues under collaboration agreements, if any, and could impose significant additional costs on us or on any future collaborators.
Our clinical trials for our product candidates
may not yield results that will enable us to further develop our products and obtain regulatory approvals necessary to sell them.
We will receive regulatory approval for our product
candidates only if we can demonstrate in carefully designed and conducted clinical trials that the product candidate is safe and effective
for its intended use. We do not know whether any current or future clinical trials for Arakoda, Tafenoquine (Arakoda or other regimen)
for non-malaria prevention indications, Celgosivir, or any other product candidate will demonstrate sufficient safety and efficacy to
obtain the requisite regulatory approvals or will result in marketable products.
Clinical trials are lengthy, complex and expensive
processes with uncertain results. We have spent, and expect to continue to spend, significant amounts of time and money on the clinical
development of our product candidates. The three clinical trials we conducted in the past were managed directly by us but executed by
contract research organizations (“CROs”). While certain of our employees have experience in designing and administering clinical
trials, our experience is limited to three clinical trials conducted by the management team.
The results we obtain in preclinical testing and
early clinical trials may not be predictive of results that are obtained in later studies. We may suffer significant setbacks in advanced
clinical trials, even after seeing promising results in earlier studies. Based on results at any stage of clinical trials, we may decide
to repeat or redesign a trial or discontinue development of one or more of our product candidates. If we fail to adequately demonstrate
the safety and efficacy of our products under development, we will not be able to obtain the required regulatory approvals to commercialize
our product candidates, and our business, results of operations and financial condition would be materially adversely affected.
Administering our product candidates to humans
may produce undesirable side effects. These side effects could interrupt, delay or halt clinical trials of our product candidates and
could result in the FDA or other regulatory authorities denying approval of our product candidates for any or all targeted indications.
If clinical trials for a product candidate are
unsuccessful, we will be unable to commercialize the product candidate. If one or more of our clinical trials are delayed, we will be
unable to meet our anticipated development timelines. Either circumstance could cause the market price of our common stock to decline.
In planning for, and executing, clinical trials,
the targeted standard of care in the United States or other jurisdictions for the therapeutic indication may change, necessitating changes
to the design of such trials. Changes to such design trials will cause delays, and increase costs, thereby rendering us unable to meet
development timelines or complete development programs. The clinical data generated from clinical trials may not be acceptable to regulatory
agencies if changes to the standard of care occurred during trial execution, which may prevent regulatory approval, thereby damaging our
business prospects.
It is possible that our Babesiosis trial might
fail if we cannot recruit sufficient patients, G6PD testing in a hospital setting cannot be conducted quickly enough to allow Tafenoquine
administration with 48 hour of initiation of standard of care therapy, or that Tafenoquine when combined with standard of care azithromycin/atovaquone
does not sufficiently accelerate the time to molecular cure to allow statistical demonstration of clinical benefit within resource constraints.
We will rely on contract research organizations
to conduct substantial portions of our clinical trials, including any future clinical trial of Arakoda Tafenoquine (Arakoda or other regimen)
for non-malaria prevention indications, or Celgosivir, and as a result, we will be unable to directly control the timing, conduct and
expense of all aspects of our clinical trials.
We do not currently have sufficient staff to conduct
our clinical trials ourselves, and therefore, we will rely on third parties to conduct certain aspects of any future clinical trials.
We previously contracted with a CRO to conduct components of our clinical trials and anticipate contracting with a CRO to conduct components
of any future clinical trial for Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, Celgosivir, or
any future clinical trials for our other product candidates. As a result, we will have less control over many details and steps of any
clinical trial, the timing and completion of any clinical trial, the required reporting of adverse events and the management of data developed
through any clinical trial than would be the case if we were relying entirely upon our own staff. Communicating with outside parties can
also be challenging, potentially leading to mistakes as well as difficulties in coordinating activities. Outside parties, such as CROs,
may have staffing difficulties, may undergo changes in priorities or may become financially distressed, adversely affecting their willingness
or ability to conduct our clinical trial. We may experience unexpected cost increases that are beyond our control. Problems with the timeliness
or quality of the work of a CRO may lead us to seek to terminate the relationship and use an alternative service provider. However, making
any change may be costly and may delay ongoing trials, if any, and contractual restrictions may make such a change difficult or impossible.
Additionally, it may be impossible to find a replacement organization that can conduct clinical trials in an acceptable manner and at
an acceptable cost.
Even though we anticipate relying on CROs in the
future, we will likely have to devote substantial resources and rely on the expertise of our employees to manage the work being done by
the CROs. We and our management team only have experience in managing clinical trials being executed on our behalf by CROs based on three
clinical studies. Therefore, we cannot guarantee that our employees will manage such studies effectively in the future.
We expect to depend on existing and future
collaborations with third parties for the development of some of our product candidates. If those collaborations are not successful, we
may not be able to complete the development of these product candidates.
Collaborations involving our product candidates
pose the following risks to us:
| |
● |
collaborators have significant discretion in determining the efforts and resources that they will apply to these collaborations; |
| |
● |
collaborators may not pursue development and commercialization of our product candidates or may elect not to continue or renew development or commercialization programs based on clinical trial results, changes in the collaborator’s strategic focus or available funding, or external factors such as an acquisition that diverts resources or creates competing priorities; |
| |
● |
collaborators may delay clinical trials, provide insufficient funding for a clinical trial program, stop a clinical trial or abandon a product candidate, repeat or conduct new clinical trials or require a new formulation of a product candidate for clinical testing; |
| |
● |
collaborators could independently develop, or develop with third parties, products that compete directly or indirectly with our product candidates if the collaborators believe that competitive products are more likely to be successfully developed or can be commercialized under terms that are more economically attractive than ours; |
| |
● |
collaborators may not properly maintain or defend our intellectual property rights or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our proprietary information or expose us to potential litigation; |
| |
● |
collaborators may not comply with regulatory requirements and as a result their operations may be disrupted or ended until they resolve their regulatory issues with government officials; |
| |
● |
disputes may arise between the collaborators and us that result in the delay or termination of the research, development or commercialization of our product candidates or that result in costly litigation or arbitration that diverts management attention and resources; |
| |
● |
collaborations may be terminated and, if terminated, may result in a need for additional capital to pursue further development or commercialization of the applicable product candidates; |
| |
● |
collaborators may elect to take over manufacturing rather than retain us as manufacturers and may encounter problems in starting up or gaining approval for their manufacturing facility and so be unable to continue development of product candidates; |
| |
● |
we may be required to undertake the expenditure of substantial operational, financial and management resources in connection with any collaboration; |
| |
● |
we may be required to issue equity securities to collaborators that would dilute our existing stockholders’ percentage ownership; |
| |
● |
we may be required to assume substantial actual or contingent liabilities; |
| |
● |
collaborators may not commit adequate resources to the marketing and distribution of our product candidates, limiting our potential revenues from these products; and |
| |
● |
collaborators may experience financial difficulties. |
We face a number of challenges in seeking additional
collaborations. Collaborations are complex and any potential discussions may not result in a definitive agreement for many reasons. For
example, whether we reach a definitive agreement for a collaboration will depend, among other things, upon our assessment of the collaborator’s
resources and expertise, the terms and conditions of the proposed collaboration, and the proposed collaborator’s evaluation of a
number of factors, such as the design or results of our clinical trials, the potential market for our product candidates, the costs and
complexities of manufacturing and delivering our product candidates to patients, the potential of competing products, the existence of
uncertainty with respect to ownership or the coverage of our intellectual property, and industry and market conditions generally. If we
were to determine that additional collaborations for our Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications,
or Celgosivir development program are necessary and were unable to enter into such collaborations on acceptable terms, we might elect
to delay or scale back the development or commercialization of Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention
indications, or Celgosivir in order to preserve our financial resources or to allow us adequate time to develop the required physical
resources and systems and expertise ourselves.
Collaboration agreements may not lead to development
or commercialization of our product candidates in the most efficient manner, or at all. In addition, there have been a significant number
of recent business combinations among large pharmaceutical companies that have resulted in a reduced number of potential future collaborators.
If a present or future collaborator of ours were to be involved in a business combination, the continued pursuit and emphasis on our product
development or commercialization program could be delayed, diminished or terminated.
Any future clinical trial for Tafenoquine
(all regimens) will require screening for G6PD deficiency in order to safely administer the product. In the United States, G6PD testing
can be obtained through commercial pathology services which is associated with delays. The use of a third-party diagnostic provider of
point of care testing may be required and we do not directly control the timing, conduct and expense of such testing.
According to prescribing information for Arakoda,
administration of a test for G6PD deficiency is required before administration in order to prevent the occurrence of hemolytic anemia
that has been observed in some patients who have G6PD deficiency and were inadvertently administered Arakoda in clinical trials. Therefore,
G6PD deficiency is an exclusion criteria in clinical trials involving Tafenoquine (all regimens).
For clinical trials administered in the United
States, G6PD testing is provided through commercial pathology companies including Labcorp and Quest Diagnostics. Such testing, while usually
available with 72-hour turnaround time, may sometimes take much longer. There is a single FDA-approved point of care test (Abbott’s
Binax Now). There is also a 510k approved microfluidics test from Baebies which may be adaptable for point of care use in some research
settings. However, both the Abbott and Baebies tests must be executed in a CLIA-certified setting, which not all clinical trials sites
may have access to.
For many clinical trials, including those involving
Babesiosis, rapid administration of the investigational agent is required to maximize efficacy. Therefore, we will attempt to import and
utilize hand-held point of care tests approved elsewhere in the world in our clinical trials involving Tafenoquine (any regimen). We may
not be successful in this process, which would compromise our ability to recruit patients or result in a lower-than-expected effect of
Tafenoquine (any regimen) in such a trial.
Tafenoquine (all regimens) requires administration
of a G6PD test. The lack of point of care tests may negatively impair sales of Arakoda or other drug regimens containing Tafenoquine.
A G6PD test need only be administered once and
can be recorded in electronic health records for future reference. The commercial providers of G6PD testing in the United States will
usually only commit to at best a 72-hour turn-around time for G6PD testing. Thus, while this does not present a problem in principle for
the existing malaria indication for individuals who travel frequently, or for organizations with organized occupational health and safety
programs where G6PD testing results are held on file, it may be a barrier to use of Arakoda by first time travelers or those planning
to travel, and hence be a barrier to use of Arakoda if prospective patients are unwilling or unable to take the G6PD test.
Several third-party diagnostic test companies
are developing point of care G6PD tests (or platforms that would accommodate them) that utilize finger stick blood samples and which may
be appropriate for use in the United States. One of these tests is approved in Brazil and Australia.39 Another is available
for use in Europe and was recently approved by the FDA in the United States.40 A third test is being developed with the NIH
grant support for the U.S. and ex-U.S. markets and is in clinical development.41 There is no guarantee that these tests will
succeed in clinical development or ever become commercially available to the public. Having to take a test at all, or to go to a third-party
lab in order to take the test, may be a hindrance to the use of Arakoda, which would negatively impact our sales.
| 39 | https://www.sdbiosensor.com/product/product_view?product_no=183. |
| 40 | Baebies Receives FDA 510(k) Clearance
for G6PD Test on FINDER Platform | Baebies. |
| 41 | https://ivd.solutions/grant/. |
Our product candidates are subject to extensive
regulation, which can be costly and time-consuming, and unsuccessful or delayed regulatory approvals could increase our future development
costs or impair our future revenue.
The preclinical and clinical development, testing,
manufacture, safety, efficacy, labeling, storage, recordkeeping, and, if approved, subsequent advertising, promotion, sale, marketing,
and distribution of our product candidates are subject to extensive regulation by the FDA and other regulatory authorities in the United
States and elsewhere. These regulations also vary in important, meaningful ways from country to country. We are not permitted to market
a potential new drug in the United States until we receive approval of an NDA from the FDA for such drug. We have received an NDA approval
for Arakoda for malaria prevention, but have not received approval from the FDA for any non-malaria prevention indications for Tafenoquine
(Arakoda regimen), Tafenoquine (non-Arakoda regimen), or for Celgosivir or any of our other product candidates. There can be no guarantees
with respect to our product candidates that clinical studies will adequately support an NDA, that the products will receive necessary
regulatory approvals, or that they will prove to be commercially successful.
To receive regulatory approval for the commercial
sale of any product candidates, we must demonstrate safety and efficacy in humans to the satisfaction of regulatory authorities through
preclinical studies and adequate and well-controlled clinical trials of the product candidates. This process is expensive and can take
many years, and failure can occur at any stage of the testing. Our failure to adequately demonstrate the safety and efficacy of our product
candidates will prevent regulatory approval and commercialization of such products.
In the event that we or our collaborators conduct
preclinical studies that do not comply with Good Laboratory Practices (“GLP”), or incorrectly design or carry out human clinical
trials in accordance with Good Clinical Practices (“GCP”), or those clinical trials fail to demonstrate clinical significance,
it is unlikely that we will be able to obtain FDA approval for product development candidates. Our inability to successfully initiate
and effectively complete clinical trials for any product candidate on schedule, or at all, will severely harm our business. Significant
delays in clinical development could materially increase product development costs or allow our competitors to bring products to market
before we do, impairing our ability to effectively commercialize any future product candidate. We do not know whether planned clinical
trials will begin on time, will need to be redesigned or will be completed on schedule, if at all. Clinical trials can be delayed for
a variety of reasons, including:
| |
● |
delays or failures in obtaining regulatory authorization to commence a trial because of safety concerns of regulators relating to our product candidates or similar product candidates of our competitors or failure to follow regulatory guidelines; |
| |
● |
delays or failures in obtaining clinical materials and manufacturing sufficient quantities of the product candidates for use in trials; |
| |
● |
delays or failures in reaching agreement on acceptable terms with prospective study sites; |
| |
● |
delays or failures in obtaining approval of our clinical trial protocol from an Institutional Review Board (“IRB”) to conduct a clinical trial at a prospective study site; |
| |
● |
delays in recruiting patients to participate in a clinical trial, which may be due to the size of the patient population, eligibility criteria, protocol design, perceived risks and benefits of the drug, availability of other approved and standard of care therapies or, availability of clinical trial sites; |
| |
● |
other clinical trials seeking to enroll subjects with similar profile; |
| |
● |
failure of our clinical trials and clinical investigators to be in compliance with GCP; |
| |
● |
unforeseen safety issues, including negative results from ongoing preclinical studies; |
| |
● |
inability to monitor patients adequately during or after treatment; |
| |
● |
difficulty recruiting and monitoring multiple study sites; and |
| |
● |
failure of our third-party CROs clinical site organizations and other clinical trial managers, to satisfy their contractual duties, comply with regulations or meet expected deadlines; and |
| |
● |
an insufficient number of patients who have, or are willing to have, a device implanted for monitoring and recording data. |
In addition, any approvals we may obtain may not
cover all of the clinical indications for which we seek approval or permit us to make claims of superiority over currently marketed competitive
products. Also, an approval might contain significant limitations in the form of narrow indications, warnings, precautions or contraindications
with respect to conditions of use. If the FDA determines that a risk evaluation and mitigation strategy (“REMS”) is necessary
to ensure that the benefits of the drug outweigh the risks, we may be required to include as part of the NDA a proposed REMS that may
include a package insert directed to patients, a plan for communication with healthcare providers, restrictions on a drug’s distribution,
or a medication guide to provide better information to consumers about the drug’s risks and benefits. Finally, approval could be
conditioned on our commitment to conduct further clinical trials, which we may not have the resources to conduct or which may negatively
impact our financial situation.
The manufacture and tableting of Arakoda, Tafenoquine
(all regimens), and Celgosivir is, or will be done, by third-party suppliers, who must also meet current Good Manufacturing Practices
(“cGMP”) requirements and pass a pre-approval inspection of their facilities before we obtain marketing approval (now or in
the future). All of our product candidates are prone to the risks of failure inherent in drug development. The results from preclinical
animal testing and early human clinical trials may not be predictive of results obtained in later human clinical trials. Further, although
a new product may show promising results in preclinical or early human clinical trials, it may subsequently prove unfeasible or impossible
to generate sufficient safety and efficacy data to obtain necessary regulatory approvals. The data obtained from preclinical and clinical
studies are susceptible to varying interpretations that may delay, limit or prevent regulatory approval, and the FDA and other regulatory
authorities in the United States and elsewhere exercise substantial discretion in the drug approval process. The numbers, size and design
of preclinical studies and clinical trials that will be required for FDA or other regulatory approval will vary depending on the product
candidate, the disease or condition for which the product candidate is intended to be used and the regulations and guidance documents
applicable to any particular product candidate. The FDA or other regulators can delay, limit or deny approval of any product candidate
for many reasons, including, but not limited to:
| |
● |
defects in the design of clinical trials; |
| |
● |
new understanding related to the pharmacology of other related drug products and their side effects; |
| |
● |
the fact that the FDA or other regulatory officials may not approve our or our third-party manufacturer’s processes or facilities; or |
| |
● |
the fact that new regulations may be enacted by the FDA or other regulators may change their approval policies or adopt new regulations requiring new or different evidence of safety and efficacy for the intended use of a product candidate. |
In light of widely publicized events concerning
the safety of certain drug products, regulatory authorities, members of Congress, the Government Accountability Office, medical professionals
and the general public have raised concerns about potential drug safety issues. These events have resulted in the withdrawal of certain
drug products, revisions to certain drug labeling that further limit use of the drug products and establishment of risk management programs
that may, for instance, restrict distribution of drug products. The increased attention to drug safety issues may result in a more cautious
approach by the FDA to clinical trials and approval. Data from clinical trials may receive greater scrutiny with respect to safety and
the product’s risk/benefit profile, which may make the FDA or other regulatory authorities more likely to terminate clinical trials
before completion, or require longer or additional clinical trials that may result in substantial additional expense, and a delay or failure
in obtaining approval or approval for a more limited indication than originally sought. Aside from issues concerning the quality and sufficiency
of submitted preclinical and clinical data, the FDA may be constrained by limited resources from reviewing and determining the approvability
of an NDA or regulatory supplement for Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications or of a Celgosivir
NDA in a timely manner.
In pursuing clinical development of Tafenoquine
(Arakoda or other regimen) for a non-malaria prevention indication, or Celgosivir for other indications, we will be required to amend
existing prescribing information, or prepare a new NDA as appropriate. The FDA could approve Tafenoquine (Arakoda or other regimen)
or Celgosivir, but without including some or all of the prescribing information that we have requested. For instance, the FDA could approve
Tafenoquine (Arakoda or other regimen) or Celgosivir in a more limited patient population or require additional warnings in the drug’s
label. This, in turn, could substantially and detrimentally impact our ability to successfully commercialize Tafenoquine (Arakoda or other
regimen) or Celgosivir and effectively protect our intellectual property rights in Tafenoquine (Arakoda or other regimen) or Celgosivir.
We plan to proceed with
a revised clinical trial of the Arakoda regimen of Tafenoquine for babesiosis (to include high-risk patients and a clinical primary endpoint
as requested by FDA). At the time of this offering based on written and verbal feedback from FDA, we believe it is likely that our planned
study, if it successfully meets its endpoint, would be pivotal, and therefore support regulatory approval of a supplementary NDA for the
babesiosis treatment indication. There is a risk that in switching to a clinical endpoint as FDA has requested that the sample size of
the study required to reach statistical significance is more than planned. If this risk is realized it may delay the timeline to reach
an interim analysis, thereby harming our business.
It is possible that when
the data from the Tafenoquine Babesiosis study are analyzed at the interim endpoint that (i) we will not achieve statistical significance,
and (ii) the sample size required to reach statistical significance if we enroll more patients exceeds our financial capacity to complete
the study. This would harm our business.
If we are not able to successfully develop,
obtain FDA approval for, and provide for the commercialization of non-malaria prevention indications for Tafenoquine (Arakoda or other
regimen) or Celgosivir in a timely manner, we may not be able to expand our business operations.
We currently have only a single product (Arakoda
for malaria prevention) that has received regulatory approval for commercial sale. The process to develop, obtain regulatory approval
for, and commercialize potential product candidates is long, complex and costly. Any future development of Tafenoquine (Arakoda or other
regimen) for a non-malaria prevention indication, or Celgosivir, including initiating clinical trials, is dependent on obtaining additional
financing, even if we enter into a strategic collaboration.
Failure to demonstrate that a product candidate,
including Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, or, in the future, Celgosivir, is safe and effective,
or significant delays in demonstrating such safety and efficacy, would adversely affect our business. Failure to obtain marketing approval
of Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, or Celgosivir from appropriate regulatory authorities,
or significant delays in obtaining such approval, would also adversely affect our business and could, among other things, preclude us
from completing a strategic transaction or obtaining additional financing necessary to continue as a going concern.
Even if approved for sale, a product candidate
must be successfully commercialized to generate value. Although we plan to undertake limited efforts through a contracts sales organization
to begin commercialization activities for Arakoda for malaria prevention, we do not currently have the capital resources or management
expertise to commercialize Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, or Celgosivir, or any of our
other product candidates and, as a result, will need to complete a strategic transaction, or, alternatively, raise substantial additional
funds to enable commercialization of Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications), or Celgosivir,
or any of our other product candidates, if approved. Failure to successfully provide for the commercialization of Arakoda for its current
malaria prevention application, or Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, or Celgosivir, or any
other product candidate, would damage our business.
If our product candidates receive regulatory
approval, we would be subject to ongoing regulatory obligations and restrictions, and possible litigation exposure, which may result in
significant expenses and limit our ability to develop and commercialize other potential products.
If a product candidate of ours is approved by
the FDA or by another regulatory authority, we would be held to extensive regulatory requirements over product manufacturing, testing,
distribution, labeling, packaging, adverse event reporting and other reporting to regulatory authorities, storage, advertising, marketing,
promotion, distribution, and record keeping. Regulatory approvals may also be subject to significant limitations on the indicated uses
or marketing of the product candidates. Potentially costly follow-up or post-marketing clinical studies may be required as a condition
of approval to further substantiate safety or efficacy, or to investigate specific issues of interest to the regulatory authority. Previously
unknown problems with the product candidate, including adverse events of unanticipated severity or frequency, may result in additional
regulatory controls or restrictions on the marketing or use of the product or the need for post marketing studies, and could include suspension
or withdrawal of the products from the market.
Furthermore, our third-party manufacturers and
the manufacturing facilities that they use to make our product candidates are regulated by the FDA. Quality control and manufacturing
procedures must continue to conform to cGMP after approval. Drug manufacturers and their subcontractors are required to register their
facilities and products manufactured annually with the FDA and certain state agencies and are subject to periodic unannounced inspections
by the FDA, state and/or other foreign authorities. Any subsequent discovery of problems with a product, or a manufacturing or laboratory
facility used by us or our collaborators, may result in restrictions on the product, or on the manufacturing or laboratory facility, including
a withdrawal of the drug from the market or suspension of manufacturing. Any changes to an approved product, including the way it is manufactured
or promoted, often require FDA approval before the product, as modified, can be marketed. We and our third-party manufacturers will also
be subject to ongoing FDA requirements for submission of safety and other post-market information.
The marketing and advertising of our drug products
by our collaborators or us will be regulated by the FDA, certain state agencies or foreign regulatory authorities including the Federal
Trade Commission (“FTC”). Violations of these laws and regulations, including promotion of our products for unapproved “off-label”
uses or failing to disclose risk information, are punishable by criminal and civil sanctions and may result in the issuance of enforcement
letters or other enforcement action by the FDA, U.S. Department of Justice, state agencies, or foreign regulatory authorities that could
jeopardize our ability to market the product.
In addition to these aforementioned FDA, state,
and foreign regulations, the marketing of our drug products by us or our collaborators will be regulated by federal, state or foreign
laws pertaining to health care “fraud and abuse,” such as the federal anti-kickback law prohibiting bribes, kickbacks or other
remuneration for the order or recommendation of items or services reimbursed by federal health care programs. Many states have similar
kickback laws applicable to items or services reimbursed by commercial insurers. Violations of these laws are punishable by criminal and
civil sanctions, including, in some instances, imprisonment and exclusion from participation in federal and state health care programs,
including the Medicare, Medicaid and Veterans Affairs healthcare programs. Because of the far-reaching nature of these laws, we may be
required to discontinue one or more of our practices to be in compliance with these laws. Healthcare fraud and abuse regulations are complex,
and even minor irregularities can potentially give rise to claims that a statute or prohibition has been violated. Any violations of these
laws, or any action against us for violations of these laws, even if we successfully defend against it, could have a material adverse
effect on our business, financial condition and results of operations.
If we, our collaborators or our third-party manufacturers
fail to comply with applicable continuing regulatory requirements, our business could be seriously harmed because a regulatory agency
may:
| ● | issue untitled or warning letters; |
| ● | suspend or withdraw our regulatory
approval for approved products; |
| ● | seize or detain products or
recommend a product recall of a drug or medical device, or issue a mandatory recall of a medical device; |
| ● | refuse import or export of
any of our drug products; |
| ● | refuse to approve pending applications
or supplements to approved applications filed by us; |
| ● | suspend our ongoing clinical
trials; |
| ● | restrict our operations, including
costly new manufacturing requirements, or restrict the sale, marketing and/or distribution of our products; |
| ● | pursue criminal prosecutions; |
| ● | close the facilities of our
contract manufacturers; or |
| ● | impose civil or criminal penalties. |
We could become subject to false claims
litigation under federal statutes, which can lead to civil money penalties, restitution, criminal fines and imprisonment, and exclusion
from participation in Medicare, Medicaid and other federal and state health care programs.
False claims statutes include the False Claims
Act, which allows any person to bring a suit on behalf of the federal government alleging submission of false or fraudulent claims, or
causing the submission of such false or fraudulent claims, under federal programs or contracts claims, or other violations of the statute,
and to share in any amounts paid by the entity to the government in fines or settlement. These suits against pharmaceutical companies
have increased significantly in volume and breadth in recent years. Some of these suits have been brought on the basis of certain sales
practices promoting drug products for unapproved uses. This new growth in litigation has increased the risk that a pharmaceutical company
will have to defend a false claim action, pay fines or restitution, or be excluded from the Medicare, Medicaid, Veterans Affairs and other
federal and state healthcare programs as a result of an investigation arising out of such action. We may become subject to such litigation
and, if we are not successful in defending against such actions, those actions may have a material adverse effect on our business, financial
condition and results of operations.
We could also become subject to false claims litigation
and consumer protection claims under state statutes, which also could lead to civil monetary penalties, restitution, criminal fines and
imprisonment, and exclusion from participation in state health care programs. Of note, over the past few years there has been an increased
focus on the sales and marketing practices of the pharmaceutical industry at both the federal and state level. Additionally, the law or
regulatory policies governing pharmaceuticals may change. New statutory requirements may be enacted or additional regulations may be adopted
that could prevent or delay regulatory approval of our product candidates or limit our ability to commercialize our products. We cannot
predict the likelihood, nature or extent of adverse government regulation that may arise from future legislation or administrative action,
either in the United States or elsewhere.
Failure to be included in formularies developed
by MCOs and other organizations may impact the use of our products.
Managed Care Organizations (“MCOs”)
and other third-party payers try to negotiate the pricing of medical services and products to control their costs. MCOs and pharmacy benefit
managers typically develop formularies to reduce their cost for medications. These formularies can be based on the prices and therapeutic
benefits of the available products. The breadth of the products covered by formularies varies considerably from one MCO to another, and
many formularies include alternative and competitive products for treatment of particular medical conditions. Failure to be included in
such formularies or to achieve favorable formulary status may negatively impact the use of our products. If our products are not included
within an adequate number of formularies, additional coverage criteria are required or if the patient’s cost-sharing obligations
are high, our market share and gross margins could be adversely impacted, which could have a material adverse effect on our business.
Even if we obtain regulatory approvals and
market our products as planned, there is no guarantee of widespread market acceptance and the results of our efforts to commercialize
our products are uncertain.
Even if we are able to obtain and maintain regulatory
approvals for our products, the success of our products depends upon achieving and maintaining market acceptance. Commercializing products
is time-consuming, expensive and unpredictable. Furthermore, the market for products that address unmet medical needs is highly speculative.
If we overestimate the market opportunity for any of our products or candidates, or if we are unsuccessful in gaining market share, these
factors could have a material adverse effect on our business. There can be no assurance that we will be able to successfully commercialize
our products or gain market acceptance for such products, including in new markets. New product candidates that appear promising in development
may fail to reach the market or may have only limited or no commercial success. If any of our products fail to gain, or lose, market acceptance,
our revenues could be adversely impacted, which in turn could have a material adverse effect on our business.
Levels of market acceptance for our products could
be impacted by several factors, some of which are not within our control, including, among others:
| ● | safety, efficacy, convenience
and cost-effectiveness of our products as compared to products of our competitors; |
| ● | scope of approved uses and
marketing approval; |
| ● | availability of patent or regulatory
exclusivity; |
| ● | timing of market approvals
and market entry; |
| ● | availability of alternative
products from our competitors; |
| ● | acceptance of the price of
our products; |
| ● | the shelf life of our products; |
| ● | effectiveness of our sales
forces and promotional efforts; |
| ● | the level of reimbursement
of our products; |
| ● | acceptance of our products
on government and private formularies; |
| ● | ability to market our products
effectively at the retail level or in the appropriate setting of care; and |
| ● | reputation of our products. |
Unexpected safety, efficacy or other concerns,
whether actual or perceived, about our products may arise which could have a material adverse effect on our business and operations.
Unexpected safety or efficacy concerns can arise
with respect to our products, whether or not scientifically justified. These concerns are especially more likely to arise when our products
are used or studied over longer periods of time or used by a wider group of patients, some of whom may be taking other medicines or have
additional underlying health problems. Such developments can potentially result in product recalls, withdrawals (by us of our products
from the market or withdrawal of NDA/marketing authorization by the FDA or other regulators), and/or declining sales, as well as product
liability, consumer fraud and/or other claims, any of which could have a material adverse effect on our business.
Any negative publicity about any of our products,
such as the discovery of safety or efficacy issues, adverse events involving our products or even public rumors about such events, could
have a material adverse effect on our business. In addition, the discovery of one or more significant problems with a product similar
to one of our products that implicates (or are perceived to implicate) an entire class of products, or the withdrawal or recall of such
similar products, could have an adverse effect on the sales of our products. New data about our products, or products similar to our products,
could also cause us reputational harm and could negatively impact demand for our products (or result in product withdrawal), due to real
or perceived side effects or uncertainty regarding safety or efficacy.
Reliance on third parties to commercialize
Arakoda, Tafenoquine (Arakoda or other regimen) Celgosivir or our other product candidates could negatively impact our business. If we
are required to establish a direct sales force in the United States and are unable to do so, our business may be harmed.
We have received FDA approval of Arakoda for malaria
prevention. Arakoda entered the U.S. commercial supply chain in the third quarter of 2019. Sales have been limited due to the impact of
the COVID-19 pandemic, and we accordingly suspended our efforts to build internal sales and marketing capability. Re-establishing such
sales and marketing capability for the malaria indication would require substantial additional resources.
Future commercialization of Tafenoquine (Arakoda
or other regimen) for non-malaria prevention indications, or Celgosivir, or any other product candidate, if approved, particularly the
establishment of a sales organization, will require substantial additional capital resources. We currently intend to pursue a strategic
partnership alternative for the commercialization of Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, or
Celgosivir, if it is approved, and we have suspended our efforts to build internal sales, marketing, and distribution capabilities. If
we elect to rely on third parties to sell Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, or Celgosivir
and any other products, then we may receive less revenue than if we sold such products directly. In addition, we may have little or no
control over the sales efforts of those third parties. If we are unable to complete a strategic transaction, we would be unable to commercialize
Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, or Celgosivir, or any other product candidate
without substantial additional capital. Even if such capital were secured, we would be required to rely on our existing distribution network
in place through prime vendors for sales and marketing and capabilities, since we lack our own internal resources to directly sell and
market Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, or Celgosivir in the United States. None
of our current employees have experience in establishing and managing a sales force.
In the event we are unable to establish an effective
sales channel for Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, or Celgosivir and other selected
product candidates, either directly or through third parties via a strategic transaction, the commercialization of Arakoda, Tafenoquine
(Arakoda or other regimen) for non-malaria prevention indications, or Celgosivir, if approved, may be delayed indefinitely and our revenues
will be impaired.
Off-label use of Arakoda by physicians exposes
us to regulatory scrutiny and safety risks
Physicians in the United States have the legal
authority to prescribe FDA-approved medications in a manner other than described in prescribing information (off-label use). Off-label
use of Arakoda to treat Babesiosis is increasing and might result in increased reports of adverse events or reports of adverse events
not previously observed in clinical trials for the approved indication. The Company does not make efficacy or safety claims regarding
the use of Arakoda for Babesiosis, but does educate the medical community about its research plans for babesiosis and data from the scientific
literature that may facilitate more informed prescribing decisions. We are careful to highlight that Arakoda has not been approved by
FDA for use in the treatment or prevention of Babesiosis. Despite these precautions, it is possible that the FDA might take actions to
attempt to restrict the off-label use of Arakoda, including potentially withdrawing marketing approval, particularly if new, unstudied
safety signals emerge. If this risk materializes it would harm our business.
We may explore new strategic collaborations
that may never materialize or may fail.
We may, in the future, periodically explore a
variety of new strategic collaborations in an effort to gain access to additional product candidates or resources. At the current time,
we cannot predict what form such a strategic collaboration might take. We are likely to face significant competition in seeking appropriate
strategic collaborators, and these strategic collaborations can be complicated and time-consuming to negotiate and document. We may not
be able to negotiate strategic collaborations on acceptable terms, or at all. We are unable to predict when, if ever, we will enter into
any additional strategic collaborations because of the numerous risks and uncertainties associated with establishing strategic collaborations.
We have no manufacturing capacity, which
puts us at risk of lengthy and costly delays of bringing our products to market.
We do not currently operate manufacturing facilities
for clinical or commercial production of our product candidates, including their API. We have no experience in drug formulation or manufacturing,
and we lack the resources and the capabilities to manufacture any of our product candidates on a clinical or commercial scale. We do not
intend to develop Company-owned facilities for the manufacture of product candidates for clinical trials or commercial purposes in the
foreseeable future. We have contracted with Piramal to manufacture the API for Arakoda. For drug product, we previously contracted with
Piramal to manufacture the Arakoda tablets (and placebos) for commercial and clinical use and with PCI in the United States for secondary
packaging. In addition, we contracted with a separate service provider for packaging and distribution of our clinical trial materials.
We may also need to contract with similar manufacturers for similar services in connection with any planned or future clinical trials
of Arakoda and Celgosivir.
Our contract manufacturers may not perform as
agreed or may not remain in the contract manufacturing business for the time required to successfully produce, store and distribute our
products. In addition, these manufacturers may have staffing difficulties, may not be able to manufacture our products on a timely basis
or may become financially distressed. In the event of errors in forecasting production quantities required to meet demand, natural disaster,
equipment malfunctions or failures, technology malfunctions, strikes, lock-outs or work stoppages, regional power outages, product tampering,
war or terrorist activities, actions of regulatory authorities, business failure, strike or other difficulty, we may be unable to find
an alternative third-party manufacturer in a timely manner and the production of our product candidates would be interrupted, resulting
in delays and additional costs, which could impact our ability to commercialize and sell our product candidates. We or our contract manufacturers
may also fail to achieve and maintain required manufacturing standards, which could result in patient injury or death, product recalls
or withdrawals, an order by governmental authorities to halt production, delays or failures in product testing or delivery, stability
testing failures, cost overruns or other problems that could seriously hurt our business.
Contract manufacturers also often encounter difficulties
involving production yields, quality control and quality assurance, as well as shortages of qualified personnel. In addition, our contract
manufacturers are subject to ongoing inspections and regulation by the FDA, the U.S. Drug Enforcement Agency and corresponding foreign
and state agencies and they may fail to meet these agencies’ acceptable standards of compliance. If our contract manufacturers fail
to comply with applicable governmental regulations, such as quality control, quality assurance and the maintenance of records and documentation,
we may not be able to continue production of the API or finished product. If the safety of any API or product supplied is compromised
due to failure to adhere to applicable laws or for other reasons, this may jeopardize our regulatory approval for Arakoda for malaria
prevention, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, or Celgosivir and other product candidates,
and we may be held liable for any injuries sustained as a result. Upon the occurrence of one of the aforementioned events, the ability
to switch manufacturers may be difficult for a number of reasons, including:
| ● | the number of potential manufacturers
is limited and we may not be able to negotiate agreements with alternative manufacturers on commercially reasonable terms, if at all; |
| ● | long lead times are often needed
to manufacture drugs; |
| ● | the manufacturing process is
complex and may require a significant learning curve; and |
| ● | the FDA must approve any replacement
prior to manufacturing, which requires new testing and compliance inspections. |
Our contract manufacturers are subject to
significant regulation with respect to the manufacturing of our products.
All entities involved in the preparation of a
product candidate for clinical trials or commercial sale, including our contract manufacturing organizations used for bulk product manufacturing
and filling and finishing of our bulk product, are subject to extensive regulation. Components of a finished product approved for commercial
sale or used in late-stage clinical trials must be manufactured in accordance with cGMP. These regulations govern manufacturing processes
and procedures, including record keeping, and the implementation and operation of quality systems to control and assure the quality of
investigational products and products approved for sale. The facilities and quality systems of some or all of our third-party contractors
must pass a pre-approval inspection for compliance with the applicable regulations as a condition of any regulatory approval of our product
candidates. In addition, the regulatory authorities may, at any time, audit or inspect a manufacturing facility involved with the preparation
of our product candidates or the associated quality systems for compliance with the regulations applicable to the activities being conducted.
The regulatory authorities also may, at any time
following approval of a product for sale, audit the manufacturing facilities of our third-party contractors or raw material suppliers.
If any such inspection or audit identifies a failure to comply with applicable regulations or if a violation of our product specifications
or applicable regulations occurs independent of such an inspection or audit, the relevant regulatory authority may require remedial measures
that may be costly and time-consuming to implement and that may include the temporary or permanent suspension of a clinical trial or commercial
sales or the temporary or permanent closure of a facility. Our third-party contractors or raw material suppliers may refuse to implement
remedial measures required by regulatory authorities. Any failure to comply with applicable manufacturing regulations or failure to implement
required remedial measures imposed upon third parties with whom we contract could materially harm our business.
We rely on relationships with third-party
contract manufacturers and raw material suppliers, which limits our ability to control the availability of, and manufacturing costs for,
our product candidates.
Problems with any of our contract manufacturers’
or raw material suppliers’ facilities or processes, could prevent or delay the production of adequate supplies of finished products.
This could delay clinical trials or delay and reduce commercial sales and materially harm our business. Any prolonged delay or interruption
in the operations of our collaborators’ facilities or contract manufacturers’ facilities could result in cancellation of shipments,
loss of components in the process of being manufactured or a shortfall in availability of a product candidate or products. A number of
factors could cause interruptions, including, but not limited to:
| ● | the inability of a supplier
to provide raw materials; |
| ● | equipment malfunctions or failures
at the facilities of our collaborators or suppliers; |
| ● | high process failure rates; |
| ● | damage to facilities due to
natural or man-made disasters; |
| ● | changes in regulatory requirements
or standards that require modifications to our or our collaborators’ and suppliers’ manufacturing processes; |
| ● | action by regulatory authorities
or by us that results in the halting or slowdown of production of components or finished product at our facilities or the facilities
of our collaborators or suppliers; |
| ● | problems that delay or prevent
manufacturing technology transfer to another facility, contract manufacturer or collaborator with subsequent delay or inability to start
up a commercial facility; |
| ● | a contract manufacturer or
supplier going out of business, undergoing a capacity shortfall or otherwise failing to produce product as contractually required; |
| ● | employee or contractor misconduct
or negligence; and |
| ● | shipping delays, losses or
interruptions; and other similar factors. |
Because manufacturing processes are complex and
are subject to a lengthy regulatory approval process, alternative qualified production capacity and sufficiently trained or qualified
personnel may not be available on a timely or cost-effective basis or at all. Difficulties or delays in our contract manufacturers’
production of drug substances could delay our clinical trials, increase our costs, damage our reputation and cause us to lose revenue
and market share if we are unable to timely meet market demand for any products that are approved for sale.
The manufacturing process for our product candidates
has several components that are sourced from a single manufacturer. If we utilize an alternative manufacturer or alternative component,
we may be required to demonstrate comparability of the drug product before releasing the product for clinical use and we may not be able
to find an alternative supplier.
Further, if our contract manufacturers are not
in compliance with regulatory requirements at any stage, including post-marketing approval, we may be fined, forced to remove a product
from the market and/or experience other adverse consequences, including delays, which could materially harm our business.
Even if one of our product candidates receives
marketing approval, it may fail to achieve the degree of market acceptance by physicians, patients, third-party payors and others in the
medical community necessary for commercial success, in which case we may not generate significant revenues or become profitable.
Physicians are often reluctant to switch their
patients from existing therapies even when new and potentially more effective or convenient treatments enter the market. Further, patients
often acclimate to the therapy that they are currently taking and do not want to switch unless their physicians recommend switching products
or they are required to switch therapies due to lack of reimbursement for existing therapies.
Efforts to educate the medical community and third-party
payors on the benefits of our product candidates may require significant resources and may not be successful. If any of our product candidates
is approved but does not achieve an adequate level of market acceptance, we may not generate significant revenues and we may not become
profitable. The degree of market acceptance of our product candidates, if approved for commercial sale, will depend on a number of factors,
including:
| ● | the efficacy and safety of
the product; |
| ● | the potential advantages of
the product compared to competitive therapies; |
| ● | the prevalence and severity
of any side effects; |
| ● | the clinical indications for
which the product is approved; |
| ● | whether the product is designated
under physician treatment guidelines as a first-, second- or third-line therapy; |
| ● | the product’s convenience
and ease of administration compared to alternative treatments; |
| ● | the willingness of the target
patient population to try, and of physicians to prescribe, the product; |
| ● | limitations or warnings, including
distribution or use restrictions contained in the product’s approved labeling; |
| ● | the approval of other new products
for the same indications; |
| ● | changes in the standard of
care for the targeted indications for the product; and |
| ● | availability and amount of
coverage and reimbursement from government payors, managed care plans and other third-party payors. |
Our future growth depends on our ability
to successfully commercialize Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, Celgosivir and our
other product candidates, and we can provide no assurance that we will successfully commercialize Arakoda, Tafenoquine (Arakoda or other
regimen) for non-malaria prevention indications, Celgosivir and other product candidates.
Our future growth depends on our ability to successfully
commercialize Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, Celgosivir, and our other product
candidates, including our ability to:
| |
● |
conduct additional clinical trials and develop and obtain regulatory approval for Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, Celgosivir ,or other product candidates; |
| |
● |
successfully partner a companion genetic test (if required by the FDA) with the commercialization of Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, and Celgosivir; |
| |
● |
pursue additional indications for Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, and Celgosivir, and develop other product candidates, including other therapies; and |
| |
● |
obtain commercial quantities of Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, and Celgosivir, or other product candidates at acceptable cost levels. |
Any one of these or other factors could affect
our ability to successfully commercialize products.
If approved by the FDA, Tafenoquine (Arakoda
or other regimen) for non-malaria prevention indications, and Celgosivir, will be entering a competitive marketplace and may not succeed.
Our commercial opportunity may be reduced or eliminated
if competitors develop and commercialize products that are safer, more effective, have fewer side effects, are more convenient or are
less expensive than Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications and Celgosivir. If products with any
of these properties are developed, or any of the existing products are better marketed, then Tafenoquine (Arakoda or other regimen) for
non-malaria prevention indications and Celgosivir could be rendered obsolete and noncompetitive. Further, public announcements regarding
the development of any such competing drugs could adversely affect the market price of our common stock and the value of our assets.
State-specific regulatory activities may
negatively affect our business.
In the United States, individual state governments
regulate certain aspects of the pharmaceutical industry including price transparency, requirements in some cases to obtain state licenses,
compliance with cGMPs, and for environmental stewardship/take-back programs. For distribution of Arakoda, we have employed a “title
model” approach to distribution which limits the extent of state licenses required, and we have contracted with third-party organizations
to ensure we are participating in appropriate stewardship/take programs, and have complied (or have a process in place to comply) with
state licensing/price transparency requirements that we are aware of. However, we cannot guarantee that we will be compliant with all
state regulations, or that we will become aware of and act on any new requirements (which are constantly changing) in time to ensure 100%
compliance at all times. State compliance is expensive and new requirements may impose new costs we were not previously aware of.
Health care reform measures could materially
and adversely affect our business.
The business and financial condition of pharmaceutical
and biotechnology companies are affected by the efforts of governmental and third-party payors to contain or reduce the costs of health
care. The U.S. Congress has enacted legislation to reform the health care system. While we anticipate that this legislation may, over
time, increase the number of patients who have insurance coverage for pharmaceutical products, it also imposes cost containment measures
that may adversely affect the amount of reimbursement for pharmaceutical products. These measures include increasing the minimum rebates
for products covered by Medicaid programs and extending such rebates to drugs dispensed to Medicaid beneficiaries enrolled in Medicaid
managed care organizations as well as expansion of the 340(B) Public Health Services drug discount program. In addition, such legislation
contains a number of provisions designed to generate the revenues necessary to fund the coverage expansion, including new fees or taxes
on certain health-related industries, including medical device manufacturers. Each medical device manufacturer has to pay an excise tax
(or sales tax) in an amount equal to 2.3% of the price for which such manufacturer sells its medical devices. Such excise taxes may impact
any potential sales of the genetic test if it is approved for marketing. On January 22, 2018, legislation was enacted suspending
the medical device tax in 2018 and 2019. In December 2019, a permanent repeal of the medical device tax was enacted. In foreign jurisdictions
there have been, and we expect that there will continue to be, a number of legislative and regulatory proposals aimed at changing the
health care system. For example, in some countries other than the United States, pricing of prescription drugs is subject to government
control and we expect to see continued efforts to reduce healthcare costs in international markets.
In August 2022, the Inflation Reduction Act of
2022 was signed into law. This law requires the federal government to negotiate prices for a small number of high-cost drugs covered under
Medicare, requires drug manufacturers to pay rebates to Medicare if they increase prices faster than inflation for drugs used by Medicare
beneficiaries, and caps Medicare beneficiaries’ out-of-pocket spending under the Medicare Part D benefit. This legislation could
create more demand for negotiated drug prices and further government control of prescription drug pricing. Future legal restrictions regarding
our ability to price our drugs could affect our revenues and our business going forward.
Additionally, federal, state and local governments
continue to consider legislation to limit the growth of healthcare costs, including the cost of prescription drugs and combination products.
Since 2017, several states and local governments have either implemented or are considering implementation of price transparency legislation
that may prevent or limit our ability to take price increases at certain rates or frequencies. If adequate reimbursement levels are not
maintained by government and other third-party payers for our products, our ability to sell our products may be limited and our ability
to establish acceptable pricing levels may be impaired, thereby reducing anticipated revenues and profitability. Further, the pace of
change and varying demands of state requirements may render it very difficult to comply with these various laws, and failure to comply
with these regulations could expose us to substantial financial penalties and the potential for adverse publicity.
Some states are also considering legislation that
would control the prices of drugs, and state Medicaid programs are increasingly requesting manufacturers to pay supplemental rebates and
requiring prior authorization by the state program for use of any drug for which supplemental rebates are not being paid. Managed care
organizations continue to seek price discounts and, in some cases, to impose restrictions on the coverage of particular drugs. Government
efforts to reduce Medicaid expenses may lead to increased use of managed care organizations by Medicaid programs. This may result in managed
care organizations influencing prescription decisions for a larger segment of the population and a corresponding constraint on prices
and reimbursement for drugs. It is likely that federal and state legislatures and health agencies will continue to focus on additional
health care reform in the future although we are unable to predict what additional legislation or regulation, if any, relating to the
health care industry or third-party coverage and reimbursement may be enacted in the future or what effect such legislation or regulation
would have on our business. We or any strategic partner’s ability to commercialize Arakoda, Tafenoquine (Arakoda or other regimen)
for non-malaria indications, Celgosivir, or any other product candidates that we may seek to commercialize, is highly dependent on the
extent to which coverage and reimbursement for these product candidates will be available from government payors, such as Medicare and
Medicaid, private health insurers, including managed care organizations, and other third-party payors, and any change in reimbursement
levels could materially and adversely affect our business. Further, the pendency or approval of future proposals or reforms could result
in a decrease in our stock price or limit our ability to raise capital or to obtain strategic partnerships or licenses.
Our employees may engage in misconduct or
other improper activities, including noncompliance with regulatory standards and requirements, which could have a material adverse effect
on our business.
We are exposed to the risk of employee fraud or
other misconduct. Misconduct by employees could include intentional failures to comply with FDA regulations, provide accurate information
to the FDA, comply with manufacturing standards we have established, comply with federal and state health-care fraud and abuse laws and
regulations, report financial information or data accurately or disclose unauthorized activities to us. In particular, sales, marketing
and business arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, kickbacks,
self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing
and promotion, sales commission, customer incentive programs and other business arrangements.
Employee misconduct could also involve the improper
use of information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to our reputation.
It is not always possible to identify and deter employee misconduct, and the precautions we take to detect and prevent misconduct may
not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions
or lawsuits stemming from a failure to be in compliance with such laws or regulations. If any such actions are instituted against us,
and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business
and results of operations, including the imposition of significant civil, criminal, and administrative penalties, damages, fines, disgorgement,
individual imprisonment, exclusion from governmental funded healthcare programs, such as Medicare and Medicaid, contractual damages, reputational
harm, diminished profits and future earnings, additional reporting obligations and oversight if subject to a corporate integrity agreement
or other agreement to resolve allegations of non-compliance with these laws.
Our competitors may be better positioned
in the marketplace and thereby may be more successful than us at developing, manufacturing and marketing approved products.
Many of our competitors currently have significantly
greater financial resources and expertise in conducting clinical trials, obtaining regulatory approvals, and managing manufacturing and
marketing approved products than us. Other early-stage companies may also prove to be significant competitors, particularly through collaborative
arrangements with large and established companies. In addition, these third parties compete with us in recruiting and retaining qualified
scientific and management personnel, establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring
therapies and therapy licenses complementary to our programs or advantageous to our business. We expect that our ability to compete effectively
will depend upon our ability to:
| ● | successfully and rapidly complete
clinical trials for any product candidates and obtain all requisite regulatory approvals in a cost-effective manner; |
| ● | build an adequate sales and
marketing infrastructure, raise additional funding, or enter into strategic transactions enabling the commercialization of our products; |
| ● | develop competitive formulations
of our product candidates; |
| ● | attract and retain key personnel;
and |
| ● | identify and obtain other product
candidates on commercially reasonable terms. |
We compete in an industry characterized
by extensive research and development efforts and rapid technological progress. New discoveries or commercial developments by our competitors
could render our potential products obsolete or non-competitive.
New developments occur and are expected to continue
to occur at a rapid pace in our industry, and there can be no assurance that discoveries or commercial developments by our competitors
will not render some or all of our potential products obsolete or non-competitive, which could have a material adverse effect on our business,
financial condition and results of operations. New data from commercial and clinical-stage products continue to emerge and it is possible
that these data may alter current standards of care, completely precluding us from further developing our product candidates or preventing
us from getting them approved by regulatory agencies. Further, it is possible that we may initiate a clinical trial or trials for our
product candidates, only to find that data from competing products make it impossible for us to complete enrollment in these trials, resulting
in our inability to file for marketing approval with regulatory agencies. Even if these products are approved for marketing in a particular
indication or indications, they may have limited sales due to particularly intense competition in these markets.
We expect to compete with fully integrated and
well-established pharmaceutical and biotechnology companies in the near- and long-term. Most of these companies have substantially greater
financial, research and development, manufacturing and marketing experience and resources than we do and represent substantial long-term
competition for us. Such companies may succeed in discovering and developing pharmaceutical products more rapidly than we do or pharmaceutical
products that are safer, more effective or less costly than any that we may develop. Such companies also may be more successful than we
are in manufacturing, sales and marketing. Smaller companies may also prove to be significant competitors, particularly through collaborative
arrangements with large pharmaceutical and established biotechnology companies. Academic institutions, governmental agencies and other
public and private research organizations also conduct clinical trials, seek patent protection and establish collaborative arrangements
for the development of product candidates.
We expect competition among products will be based
on product efficacy and safety, the timing and scope of regulatory approvals, availability of supply, marketing and sales capabilities,
reimbursement coverage, price and patent position. There can be no assurance that our competitors will not develop safer and more effective
products, commercialize products earlier than we do, or obtain patent protection or intellectual property rights that limit our ability
to commercialize our products.
There can be no assurance that our issued patents
or pending patent applications, if issued, will not be challenged, invalidated or circumvented or that the rights granted thereunder will
provide us with proprietary protection or a competitive advantage.
We would be subject to applicable regulatory
approval requirements of the foreign countries in which we market our products, which are costly and may prevent or delay us from marketing
our products in those countries.
In addition to regulatory requirements in the
United States, we would be subject to the regulatory approval requirements in each foreign country where we market our products. Also,
we might be required to identify one or more collaborators in these foreign countries to develop, seek approval for and manufacture our
products and any companion genetic test that may be required for Arakoda or Celgosivir. If we decide to pursue regulatory approvals and
commercialization of our product candidates internationally, we may not be able to obtain the required foreign regulatory approvals on
a timely basis, if at all, and any failure to do so may cause us to incur additional costs or prevent us from marketing our products in
foreign countries, which may have a material adverse effect on our business, financial condition and results of operations.
Failure to comply with data protection laws
and regulations could lead to government enforcement actions (which could include civil or criminal penalties), private litigation, and/or
adverse publicity and could negatively affect our operating results and business.
We and our partners may be subject to federal,
state, and foreign data protection laws and regulations (i.e., laws and regulations that address privacy and data security). In the United
States, numerous federal and state laws and regulations, including state data breach notification laws, state health information privacy
laws, and federal and state consumer protection laws and regulations (e.g., Section 5 of the FTC Act of 1914), that govern the collection,
use, disclosure, and protection of health-related and other personal information could apply to our operations or the operations of our
partners. In addition, we may obtain health information from third parties (including research institutions from which we obtain clinical
trial data) that are subject to privacy and security requirements under the Health Insurance Portability and Accountability Act of 1996,
as amended (“HIPAA”). Depending on the facts and circumstances, we could be subject to criminal penalties if we knowingly
obtain, use, or disclose individually identifiable health information maintained by a HIPAA-covered entity in a manner that is not authorized
or permitted by HIPAA.
In addition, the California Consumer Privacy Act,
as amended (“CCPA”), became effective on January 1, 2020. The CCPA gives California residents expanded rights to access
and delete their personal information (subject to certain exceptions), opt out of certain personal information sharing, correct inaccurate
personal information that a business has about them and limit the use and disclosure of sensitive personal information collected about
them and receive detailed information about how their personal information is used by requiring covered companies to provide new disclosures
to California consumers (as that term is broadly defined) and provide such consumers new ways to opt-out of certain sales of personal
information and the right not to be discriminated against for exercising these rights. The CCPA also gives consumers the right to request
disclosure of information collected about them and whether that information has been sold or shared with others.
The CCPA provides for civil penalties for violations,
as well as a private right of action for data breaches that is expected to increase data breach litigation. Although there are limited
exemptions for clinical trial data and the CCPA’s implementation standards and enforcement practices are likely to remain uncertain
for the foreseeable future, the CCPA may increase our compliance costs and potential liability. Many similar privacy laws have been proposed
at the federal level and in other states.
Foreign data protection laws, including, without
limitation, the European Union Directive 95/46/EC, or the Directive, and the European Union’s General Data Protection Regulation
(“GDPR”), that became effective in May 2018, and member state data protection legislation, may also apply to health-related
and other personal information obtained outside of the United States. These laws impose strict obligations on the ability to process health-related
and other personal information of data subjects in the European Union and the United Kingdom, including in relation to use, collection,
analysis, and transfer (including cross-border transfer) of such personal information. These laws include several requirements relating
to the consent of the individuals to whom the personal data relates, limitations on data processing, establishing a legal basis for processing,
notification of data processing obligations or security incidents to appropriate data protection authorities or data subjects, the security
and confidentiality of the personal data and various rights that data subjects may exercise.
The Directive and the GDPR prohibit, without an
appropriate legal basis, the transfer of personal data to countries outside of the European Economic Area (“EEA”), such as
the United States, which are not considered by the European Commission to provide an adequate level of data protection. Switzerland has
adopted similar restrictions. Although there are legal mechanisms to allow for the transfer of personal data from the EEA and Switzerland
to the United States, uncertainty about compliance with European Union data protection laws remains. For example, ongoing legal challenges
in Europe to the mechanisms allowing companies to transfer personal data from the EEA to the United States could result in further limitations
on the ability to transfer personal data across borders, particularly if governments are unable or unwilling to reach new or maintain
existing agreements that support cross-border data transfers, such as the European Union-U.S. and Swiss-U.S. Privacy Shield framework.
Additionally, other countries have passed or are considering passing laws requiring local data residency.
Under the GDPR, regulators may impose substantial
fines and penalties for non-compliance. Companies that violate the GDPR can face fines of up to the greater of 20 million Euros or
4% of their worldwide annual turnover (revenue). The GDPR increases our responsibility and potential liability in relation to personal
data that we process, and we may be required to put in place additional mechanisms to ensure compliance with the GDPR and other EU and
international data protection rules.
Compliance with U.S. and foreign privacy and security
laws, rules and regulations could require us to take on more onerous obligations in our contracts, require us to engage in costly compliance
exercises, restrict our ability to collect, use and disclose data, or in some cases, impact our or our partners’ or suppliers’
ability to operate in certain jurisdictions. Each of these constantly evolving laws can be subject to varying interpretations. Failure
to comply with U.S. and foreign data protection laws and regulations could result in government investigations and enforcement actions
(which could include civil or criminal penalties), fines, private litigation, and/or adverse publicity and could negatively affect our
operating results and business. Moreover, patients about whom we or our partners obtain information, as well as the providers who share
this information with us, may contractually limit our ability to use and disclose the information. Claims that we have violated individuals’
privacy rights, failed to comply with data protection laws, or breached our contractual obligations, even if we are not found liable,
could be expensive and time-consuming to defend and could result in adverse publicity that could harm our business.
Because we have multiple product candidates
in our clinical pipeline and are considering a variety of target indications, we may expend our limited resources to pursue a particular
product candidate or indication and fail to capitalize on product candidates or indications that may be more profitable or for which there
is a greater likelihood of success.
Because we have limited financial and managerial
resources, we must focus our research and development efforts on those product candidates and specific indications that we believe are
the most promising. As a result, we may forego or delay our pursuit of opportunities with other product candidates or other indications
that later prove to have greater commercial potential. Our resource allocation decisions may cause us to fail to capitalize on viable
commercial products or profitable market opportunities. We may in the future spend our resources on other research programs and product
candidates for specific indications that ultimately do not yield any commercially viable products. Furthermore, if we do not accurately
evaluate the commercial potential or target market for a particular product candidate, we may relinquish valuable rights to that product
candidate through collaboration, licensing or other royalty arrangements in cases in which it would have been more advantageous for us
to retain sole development and commercialization rights.
We must meet post-marketing requirements
associated with the Arakoda NDA imposed by the FDA. Failure to complete such requirements, or delays due to lack of resources or other
factors, may negatively impact our business.
When the FDA approved the Arakoda NDA in August
2018, it imposed post-marketing requirements on us, including associated timelines. We have made substantial progress in meeting all such
requirements and recently published data from a clinical trial related to one of them. However, we have experienced delays in our ability
to execute our observational and pediatric study requirements and are in discussion with the FDA regarding future plans relating to our
pediatric program. We may experience new or additional delays in the future on one or more of its post-marketing requirements in the future.
As of the date of this prospectus, we have not received acknowledgement from the FDA that any of the post-marketing requirements are completed
nor been referred for enforcement action due to delays in our post-marketing studies. If we fail to meet FDA requirements, experiences
additional delays or is referred for enforcement action, we might require diversion of managerial and capital resources from planned research
and development to completion of post-marketing requirements, or the FDA might revoke the NDA for Arakoda, and therefore harm the business.
In the future, regulators may impose additional post-marketing requirements for Arakoda for malaria or other indications, or in relation
to our products. This situation would require expensive clinical or non-clinical studies that might damage our financial position.
We are subject to U.S. and certain foreign
export and import controls, sanctions, embargoes, anti-corruption laws, and anti-money laundering laws and regulations. Compliance with
these legal standards could impair our ability to compete in domestic and international markets. We can face criminal liability and other
serious consequences for violations, which can harm our business.
We are subject to export control and import laws
and regulations, including the U.S. Export Administration Regulations, U.S. Customs regulations, various economic and trade sanctions
regulations administered by the U.S. Treasury Department’s Office of Foreign Assets Controls, the U.S. Foreign Corrupt Practices
Act of 1977, as amended, the U.S. domestic bribery statute contained in 18 U.S.C. § 201, the U.S. Travel Act, the USA PATRIOT Act,
and other state and national anti-bribery and anti-money laundering laws in the countries in which we conduct activities. Anti-corruption
laws are interpreted broadly and prohibit companies and their employees, agents, contractors, and other collaborators from authorizing,
promising, offering, or providing, directly or indirectly, improper payments or anything else of value to recipients in the public or
private sector. We may engage third parties for clinical trials outside of the United States, to sell our products abroad once we enter
a commercialization phase, and/or to obtain necessary permits, licenses, patent registrations, and other regulatory approvals. We have
direct or indirect interactions with officials and employees of government agencies or government-affiliated hospitals, universities,
and other organizations. We can be held liable for the corrupt or other illegal activities of our employees, agents, contractors, and
other collaborators, even if we do not explicitly authorize or have actual knowledge of such activities. Any violations of the laws and
regulations described above may result in substantial civil and criminal fines and penalties, imprisonment, the loss of export or import
privileges, debarment, tax reassessments, breach of contract and fraud litigation, reputational harm, and other consequences.
Continued uncertain economic conditions,
including inflation and the risk of a global recession could impair our ability to forecast and may harm our business, operating results,
including our revenue growth and profitability, financial condition and cash flows.
The U.S. economy has recently experienced the
highest rates of inflation since the 1980s. Historically, we have not experienced significant inflation risk in our business. However,
our ability to raise our product prices depends on market conditions and there may be periods during which we are unable to fully recover
increases in our costs. In addition, the global economy suffers from slowing growth and rising interest rates, and many economists believe
that a global recession may begin in the near future. If the global economy slows, our business would likely be adversely affected.
Also, the global credit
and financial markets have recently experienced extreme volatility and disruptions, including severely diminished liquidity and credit
availability, increases in unemployment rates and uncertainty about economic stability. There can be no assurance that further deterioration
in credit and financial markets and confidence in economic conditions will not occur. Our general business strategy may be adversely affected
by any such economic downturn, volatile business environment or continued unpredictable and unstable market conditions.
Geopolitical conditions,
including direct or indirect acts of war or terrorism, could have an adverse effect on our operations and financial results.
Our operations could be disrupted by geopolitical
conditions, political and social instability, acts of war, terrorist activity or other similar events. In February 2022, Russia initiated
significant military action against Ukraine. In response, the U.S. and certain other countries imposed significant sanctions and export
controls against Russia, Belarus and certain individuals and entities connected to Russian or Belarusian political, business, and financial
organizations, and the U.S. and certain other countries could impose further sanctions, trade restrictions, and other retaliatory actions
should the conflict continue or worsen. It is not possible to predict the broader consequences of the conflict, including related geopolitical
tensions, and the measures and retaliatory actions taken by the U.S. and other countries in respect thereof as well as any counter measures
or retaliatory actions by Russia or Belarus in response, including, for example, potential cyberattacks or the disruption of energy exports,
is likely to cause regional instability, geopolitical shifts, and could materially adversely affect global trade, currency exchange rates,
regional economies and the global economy. In addition, the ongoing conflicts in the Middle East may further impact global economic conditions
and market sentiments. This, in turn, could adversely affect the trading price of our shares of common stock and investor interest in
us.
The Russia-Ukraine war and conflicts in the Middle
East remain uncertain, and while it is difficult to predict the impact of any of the foregoing, the conflict and actions taken in response
to the conflict could increase our costs, disrupt our supply chain, reduce our sales and earnings, impair our ability to raise additional
capital when needed on acceptable terms, if at all, or otherwise adversely affect our business, financial condition, and results of operations.
We could be subject to lawsuits.
We may be party to lawsuits, settlement discussions,
mediations, arbitrations and other disputes, including patent and product liability claims, whether brought by companies, individuals
or governmental authorities. These matters may result in a loss of patent protection, reduced revenue, incurrence of significant liabilities
and diversion of our management’s time, attention and resources. Our insurance coverage may not provide adequate protection against
actual losses. In addition, we are subject to the risk that one or more of our insurers may become insolvent and become unable to pay
claims that may be made in the future. Even if we maintain adequate insurance, claims could have a material adverse effect on our financial
condition, liquidity and results of operations and on our ability to obtain suitable, adequate or cost-effective insurance in the future.
Litigation and other disputes, including any adverse outcomes, may have an adverse impact on our business, operations or financial condition.
Even claims without merit could subject us to adverse publicity and require us to incur significant legal fees.
We currently, and may in the future, have
assets held at financial institutions that may exceed the insurance coverage offered by the Federal Deposit Insurance Corporation, the
loss of such assets would have a severe negative affect on our operations and liquidity.
We may maintain our cash assets at certain financial
institutions in the U.S. in amounts that may be in excess of the Federal Deposit Insurance Corporation (“FDIC”) insurance
limit of $250,000. In the event of a failure of any financial institutions where we maintain our deposits or other assets, we may incur
a loss to the extent such loss exceeds the FDIC insurance limitation, which could have a material adverse effect upon our liquidity, financial
condition and our results of operations.
Risks Related to Intellectual Property and
Other Legal Matters
If product liability lawsuits are successfully
brought against us, then we will incur substantial liabilities and may be required to limit commercialization of Arakoda, Tafenoquine
(Arakoda or other regimen) for non-malaria prevention indications, Celgosivir or other product candidates.
We may face product liability exposure related
to the testing of our product candidates in human clinical trials, and may face exposure to claims by an even greater number of persons
once we begin marketing and distributing our products commercially. If we cannot successfully defend against product liability claims,
then we will incur substantial liabilities.
Regardless of merit or eventual outcome, liability
claims may result in:
| |
● |
decreased demand for our products and product candidates; |
| |
● |
injury to our reputation; |
| |
● |
withdrawal of clinical trial participants; |
| |
● |
costs of related litigation; |
| |
● |
substantial monetary awards to patients and others; |
| |
● |
the inability to commercialize our products and product candidates. |
We have obtained limited product liability insurance
coverage. Such coverage, however, may not be adequate or may not continue to be available to us in sufficient amounts or at an acceptable
cost, or at all. We may not be able to obtain commercially reasonable product liability insurance for any product candidate.
Defending against claims relating to improper
handling, storage or disposal of hazardous chemicals, radioactive or biological materials could be time consuming and expensive.
Our research and development of product candidates
may involve the controlled use of hazardous materials, including chemicals, radioactive and biological materials. We cannot eliminate
the risk of accidental contamination or discharge and any resultant injury from the materials. Various laws and regulations govern the
use, manufacture, storage, handling and disposal of hazardous materials. We may be sued or be required to pay fines for any injury or
contamination that results from our use or the use by third parties of these materials. Compliance with environmental laws and regulations
may be expensive, and current or future environmental regulations may impair our research, development and production efforts.
Third parties may own or control patents
or patent applications that we may be required to license to commercialize our product candidates or that could result in litigation that
would be costly and time consuming.
Our or any strategic partner’s ability to
commercialize Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, Celgosivir, and other product candidates
depends upon our ability to develop, manufacture, market and sell these drugs without infringing the proprietary rights of third parties.
A number of pharmaceutical and biotechnology companies, universities and research institutions have or may be granted patents that cover
technologies similar to the technologies owned by or licensed to us. We may choose to seek, or be required to seek, licenses under third-party
patents, which would likely require the payment of license fees or royalties or both. We may also be unaware of existing patents that
may be infringed by Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications or Celgosivir, the genetic
testing we intend to use in connection with Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention indications, Celgosivir
or our other product candidates. Because patent applications can take many years to issue, there may be other currently pending applications
that may later result in issued patents that are infringed by Arakoda, Tafenoquine (Arakoda or other regimen) for non-malaria prevention
indications, Celgosivir or our other product candidates. Moreover, a license may not be available to us on commercially reasonable terms,
or at all.
There is a substantial amount of litigation involving
patent and other intellectual property rights in the biotechnology and biopharmaceutical industries generally. If a third-party claims
that we are infringing on its technology, then our business and results of operations could be harmed by a number of factors, including:
| ● | infringement and other intellectual
property claims, even if without merit, are expensive and time-consuming to litigate and can divert management’s attention from
our core business; |
| ● | monetary damage awards for
past infringement can be substantial; |
| ● | a court may prohibit us from
selling or licensing product candidates unless the patent holder chooses to license the patent to us; and |
| ● | if a license is available from
a patent holder, we may have to pay substantial royalties. |
We may also be forced to bring an infringement
action if we believe that a competitor is infringing our protected intellectual property. Any such litigation will be costly, time-consuming
and divert management’s attention, and the outcome of any such litigation may not be favorable to us.
Our intellectual property rights may not
preclude competitors from developing competing products and our business may suffer.
Our competitive success will depend, in part,
on our ability to obtain and maintain patent protection for our inventions, technologies and discoveries, including intellectual property
that we license. The patent positions of biotechnology companies involve complex legal and factual questions, and we cannot be certain
that our patents and licenses will successfully preclude others from using our technology. Consequently, we cannot be certain that any
of our patents will provide significant market protection or will not be circumvented or challenged and found to be unenforceable or invalid.
In some cases, patent applications in the United States and certain other jurisdictions are maintained in secrecy until patents issue,
and since publication of discoveries in the scientific or patent literature often lags behind actual discoveries, we cannot be certain
of the priority of inventions covered by pending patent applications. An adverse outcome could subject us to significant liabilities to
third parties, require disputed rights to be licensed from third parties or require us to cease using such technology. Regardless of merit,
the listing of patents in the FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations (which is commonly referred
to as the “Orange Book”) for Arakoda, Celgosivir may be challenged as being improperly listed. We may have to defend against
such claims and possible associated antitrust issues. We could also incur substantial costs in seeking to enforce our proprietary rights
against infringement.
We may not be able to effectively protect our
intellectual property rights in some foreign countries, as our patents are limited by jurisdiction and many countries do not offer the
same level of legal protection for intellectual property as the United States.
We require our employees, consultants, business
partners and members of our scientific advisory board to execute confidentiality agreements upon the commencement of employment, consulting
or business relationships with us. These agreements provide that all confidential information developed or made known during the course
of the relationship with us be kept confidential and not disclosed to third parties except in specific circumstances. In the case of employees,
the agreements provide that all inventions resulting from work performed for us, utilizing the property or relating to our business and
conceived or completed by the individual during employment shall be our exclusive property to the extent permitted by applicable law.
Third parties may breach these and other agreements
with us regarding our intellectual property and we may not have adequate remedies for the breach. Third parties could also fail to take
necessary steps to protect our licensed intellectual property, which could seriously harm our intellectual property position.
If we are not able to protect our proprietary technology, trade secrets
and know-how, then our competitors may develop competing products. Any issued patent may not be sufficient to prevent others from competing
with us. Further, we have trade secrets relating to Arakoda, Celgosivir, and such trade secrets may become known or independently discovered.
Our issued patents and those that may issue in the future, or those licensed to us, may be challenged, opposed, invalidated or circumvented,
which could allow competitors to market similar products or limit the patent protection term of our product candidates. All of these factors
may affect our competitive position.
If the manufacture, use or sale of our products
infringe on the intellectual property rights of others, we could face costly litigation, which could cause us to pay substantial damages
or licensing fees and limit our ability to sell some or all of our products.
Extensive litigation regarding patents and other
intellectual property rights has been common in the biopharmaceutical industry. Litigation may be necessary to assert infringement claims,
enforce patent rights, protect trade secrets or know-how and determine the enforceability, scope and validity of certain proprietary rights.
Litigation may even be necessary to defend disputes of inventorship or ownership of proprietary rights. The defense and prosecution of
intellectual property lawsuits, U.S. Patent and Trademark Office interference proceedings, and related legal and administrative proceedings
(e.g., a re-examination, inter partes review, or post-grant review) in the United States and internationally involve complex legal
and factual questions. As a result, such proceedings are costly and time-consuming to pursue, and their outcome is uncertain.
Regardless of merit or outcome, our involvement
in any litigation, interference or other administrative proceedings could cause us to incur substantial expense and could significantly
divert the efforts of our technical and management personnel. Any public announcements related to litigation or interference proceedings
initiated or threatened against us could cause our stock price to decline. Adverse outcomes in patent litigation may potentially subject
us to antitrust litigation which, regardless of the outcome, would adversely affect our business. An adverse determination may subject
us to the loss of our proprietary position or to significant liabilities, or require us to seek licenses that may include substantial
cost and ongoing royalties. Licenses may not be available from third parties, or may not be obtainable on satisfactory terms. An adverse
determination or a failure to obtain necessary licenses may restrict or prevent us from manufacturing and selling our products, if any.
These outcomes could materially harm our business, financial condition and results of operations.
Patent reform legislation could increase
the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents.
Changes in either the patent laws or interpretation
of the patent laws in the United States and Ex-US could increase the uncertainties and costs. Recent patent reform legislation in the
United States and other countries, including the Leahy-Smith America Invents Act (the “Leahy-Smith Act”), signed into law
in the United States on September 16, 2011, could increase those uncertainties and costs surrounding the prosecution of our patent applications
and the enforcement or defense of our issued patents. The Leahy-Smith Act includes a number of significant changes to U.S. patent law.
These include provisions that affect the way patent applications are prosecuted, redefine prior art and provide more efficient and cost-effective
avenues for competitors to challenge the validity of patents. These include allowing third-party submission of prior art to the USPTO
during patent prosecution and additional procedures to attack the validity of a patent by USPTO administered post-grant proceedings, including
post-grant review, inter partes review, and derivation proceedings. After March 2013, under the Leahy-Smith Act, the United States transitioned
to a first inventor to file system in which, assuming that the other statutory requirements are met, the first inventor to file a patent
application will be entitled to the patent on an invention regardless of whether a third-party was the first to invent the claimed invention.
However, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent
applications and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business,
financial condition, results of operations and prospects.
The U.S. Supreme Court has ruled on several patent
cases in recent years, either narrowing the scope of patent protection available in certain circumstances or weakening the rights of patent
owners in certain situations. Depending on future actions by the U.S. Congress, the U.S. courts, the USPTO and the relevant law-making
bodies in other countries, the laws and regulations governing patents could change in unpredictable ways that would weaken our ability
to obtain new patents or to enforce our existing patents and patents that we might obtain in the future.
The patent protection and patent prosecution
for some of our product candidates is dependent or may be dependent in the future on third parties.
While we normally seek and gain the right to fully
prosecute the patents relating to our product candidates, there may be times when platform technology patents or product-specific patents
that relate to our product candidates are controlled by our licensors. In addition, our licensors and/or licensees may have back-up rights
to prosecute patent applications in the event that we do not do so or choose not to do so, and our licensees may have the right to assume
patent prosecution rights after certain milestones are reached. If any of our licensing collaborators fails to appropriately prosecute
and maintain patent protection for patents covering any of our product candidates, our ability to develop and commercialize those product
candidates may be adversely affected and we may not be able to prevent competitors from making, using and selling competing products.
We may not be able to protect our intellectual
property rights throughout the world.
Patents are of national or regional effect, and
filing, prosecuting and defending patents on all of our product candidates throughout the world would be prohibitively expensive. As such,
we may not be able to prevent third parties from practicing our inventions in all countries outside the United States, or from selling
or importing products made using our inventions in and into the United States or other jurisdictions. Further, the legal systems of certain
countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection,
particularly those relating to pharmaceuticals or biologics, which could make it difficult for us to stop the infringement of our patents
or marketing of competing products in violation of our proprietary rights generally. In addition, certain developing countries, including
China and India, have compulsory licensing laws under which a patent owner may be compelled to grant licenses to third parties. In those
countries, we and our licensors may have limited remedies if patents are infringed or if we or our licensors are compelled to grant a
license to a third-party, which could materially diminish the value of those patents. This could limit our potential revenue opportunities.
Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial
advantage from the intellectual property that we develop or license.
Patent terms may be inadequate to protect
our competitive position on our product candidates for an adequate amount of time.
Patent rights are of limited duration. Given the
amount of time required for the development, testing and regulatory review of new product candidates, patents protecting such candidates
might expire before or shortly after such product candidates are commercialized. Even if patents covering our product candidates are obtained,
once the patent life has expired for a product, we may be open to competition from biosimilar or generic products. A patent term extension
based on regulatory delay may be available in the U.S. However, only a single patent can be extended for each marketing approval, and
any patent can be extended only once, for a single product. Moreover, the scope of protection during the period of the patent term extension
does not extend to the full scope of the claim, but instead only to the scope of the product as approved. Laws governing analogous patent
term extensions in foreign jurisdictions vary widely, as do laws governing the ability to obtain multiple patents from a single patent
family. Additionally, we may not receive an extension if we fail to apply within applicable deadlines, fail to apply prior to expiration
of relevant patents or otherwise fail to satisfy applicable requirements. If we are unable to obtain patent term extension or restoration,
or the term of any such extension is less than we request, the period during which we will have the right to exclusively market our product
will be shortened and our competitors may obtain approval of competing products following our patent expiration, and our revenue could
be reduced, possibly materially.
The earliest Paragraph IV certification
date for Arakoda has passed. Generic companies may file an ANDA at any time, and successful challenge of our malaria use patents would
negatively impact our business.
The Prescription Drug User Fee Act date for Arakoda
is August 8, 2018, and the beginning date for marketing exclusivity associated with the product’s API was July 20, 2018. The exclusivity
ending date for Arakoda was July 20, 2023. These dates are relevant because it means that the earliest date a generic company could file
an abbreviated new drug application (“ANDA”), claiming such an application does not infringe our Orange Book listed patents
was July 20, 2022. Any generic company filing such an ANDA with FDA, must notify us within 20 calendar days of receiving acknowledgement
from the FDA of receipt of such an ANDA. Thus, the earliest date we could have received such a notification was August 9, 2022.
As of the date of this prospectus, to the best
of our knowledge, no such notice has been received by us. However, such a notice might be received at any time. Such a notice might require
us to undertake expensive litigation to defend our patents related to Arakoda’s malaria indication, thereby diverting funds away
from critical research and development efforts for Tafenoquine (Arakoda or other regimen) for other indications. This potential litigation
and the related expenditure may harm our business. Additionally, the approval of any ANDA would increase competition and most likely drive
down prices for Arakoda.
Obtaining and maintaining our patent protection
depends on compliance with various procedural, document submissions, fee payment and other requirements imposed by governmental patent
agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
Periodic maintenance fees on any issued patent
are due to be paid to the USPTO and foreign patent agencies in several stages over the lifetime of the patent. The USPTO and various foreign
governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during
the patent application process. While an inadvertent lapse can in many cases be cured by payment of a late fee or by other means in accordance
with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application,
resulting in partial or complete loss of patent rights in the relevant jurisdiction. Non-compliance events that could result in abandonment
or lapse of a patent or patent application include, but are not limited to, failure to respond to official actions within prescribed time
limits, non-payment of fees and failure to properly legalize and submit formal documents. If we fail to maintain the patents and patent
applications covering our product candidates, our competitive position would be adversely affected.
Risks Related to this Offering and Ownership
of our Common Stock
We have a large number of authorized but
unissued shares of our common stock which will dilute your ownership position when issued.
Our authorized capital stock consists of 150,000,000
shares of common stock, of which approximately 126,000,000 remain available for issuance, including shares of common stock issuable upon the exercise
of outstanding warrants. Our management will continue to have broad discretion to issue shares of our common stock in a range of transactions,
including capital-raising transactions, mergers, acquisitions and other transactions, without obtaining stockholder approval, unless stockholder
approval is required under law or, if our common stock is listed on Nasdaq at the time of the transaction, under Nasdaq Rule 5635(b) which
requires stockholder approval for change of control transactions where a stockholder acquires 20% of a Nasdaq-listed company’s common
stock or securities convertible into common stock, calculated on a post-transaction basis. If our management determines to issue shares
of our common stock from the large pool of authorized but unissued shares for any purpose in the future and is not required to obtain
stockholder approval, your ownership position would be diluted without your further ability to vote on that transaction.
Our common stock may be affected by limited
trading volume and price fluctuations, which could adversely impact the value of our common stock.
Our common stock has experienced and is likely
to experience in the future, significant price and volume fluctuations, which could adversely affect the market prices of our common stock
without regard to our operating performance. In addition, we believe that factors such as quarterly fluctuations in our financial results
and changes in the overall economy or the condition of the financial markets could cause the market prices of our common stock to fluctuate
substantially. These fluctuations may also cause short sellers to periodically enter the market in the belief that we will have poor results
in the future. We cannot predict the actions of market participants and, therefore, can offer no assurances that the market for our common
stock will be stable or appreciate over time.
Sales of common stock offered hereby will
be in “at the market offerings,” and investors who buy shares at different times will likely pay different prices.
Investors who purchase shares in this offering
at different times will likely pay different prices, and accordingly may experience different levels of dilution and different outcomes
in their investment results. We will have discretion, subject to market demand, to vary the timing, prices and number of shares sold in
this offering. In addition, subject to the final determination by our Board or any restrictions we may place in any applicable placement
notice delivered to the Sales Agent, there is no minimum or maximum sales price for shares to be sold in this offering. Investors may
experience a decline in the value of the shares they purchase in this offering as a result of sales made at prices lower than the prices
they paid.
The sale of our common stock in this offering
and any future sales of our common stock may depress our stock price and our ability to raise funds in new stock offerings.
We may issue common stock from time to time in
connection with this offering. This issuance from time to time of these new shares of our common stock, or our ability to issue these
shares of common stock in this offering, could result in resales of our common stock by our current stockholders concerned about the potential
dilution of their holdings. In addition, sales of our common stock in the public market following this offering could lower the market
price of our common stock. Sales may also make it more difficult for us to sell equity securities or equity-related securities in the
future at a time and price that our management deems acceptable, or at all. We cannot predict the number of these shares that might be
resold or the effect that future sales of our shares of common stock would have on the market price of our shares of common stock.
It is not possible to predict the aggregate proceeds resulting
from sales made under the Sales Agreement.
Subject to certain limitations in the Sales Agreement
and compliance with applicable law, we have the discretion to deliver a placement notice to the Sales Agent at any time throughout the
term of the Sales Agreement. The number of shares that are sold through the Sales Agent after delivering a placement notice will fluctuate
based on a number of factors, including the market price of our common stock during the sales period, any limits we may set with the Sales
Agent in any applicable placement notice and the demand for our common stock. Because this offering can be terminated at any time and
the price per share of each share sold pursuant to the Sales Agreement will fluctuate over time, it is not currently possible to predict
the aggregate proceeds to be raised in connection with sales under the Sales Agreement.
We have broad discretion in the use of the
net proceeds of this offering and, despite our efforts, we may use the net proceeds in a manner that does not increase the value of your
investment.
We intend to use the net proceeds from this offering
for general corporate purposes and working capital. However, we have not determined the specific allocation of the net proceeds among
these potential uses. Our management will have broad discretion over the use and investment of the net proceeds of this offering, and,
accordingly, investors in this offering will need to rely upon the judgment of our management with respect to the use of proceeds, with
only limited information concerning our specific intentions. These proceeds could be applied in ways that do not improve our operating
results or increase the value of your investment. Please see the section entitled “Use of Proceeds” on page S-19 of
this prospectus or further information.
We may not be able to continue to satisfy
listing requirements of Nasdaq to maintain a listing of our common stock.
Our common stock is currently listed on Nasdaq
and we must meet certain financial and liquidity criteria to maintain such listing. If we violate the maintenance requirements for continued
listing of our common stock, our common stock may be delisted.
On February 27, 2024, we received a letter from
the Nasdaq Listing Qualifications Staff of Nasdaq stating that for the 30 consecutive business day period between January 11, 2024 and
February 27, 2024, our common stock had not maintained a minimum closing bid price of $1.00 per share required for continued listing on
The Nasdaq Capital Market pursuant to Nasdaq Listing Rule 5550(a)(2). Pursuant to Nasdaq Listing Rule 5810(c)(3)(A), we were provided
an initial period of 180 calendar days, or until August 26, 2024, to regain compliance with the Bid Price Rule.
To regain compliance, the closing bid price of
our common stock must meet or exceed $1.00 per share for a minimum of 10 consecutive trading days, unless extended by Nasdaq under
Nasdaq Rule 5810(c)(3)(H), prior to August 26, 2024.
There can be no assurance that we will maintain
compliance with the Bid Price Rule or any of the other Nasdaq continued listing requirements. If the common stock is delisted, it could
be more difficult to buy or sell the common stock or to obtain accurate quotations, and the price of the shares of common stock could
suffer a material decline. Delisting could also impair our ability to raise capital.
In addition, our Board may determine that the
cost of maintaining our listing on a national securities exchange outweighs the benefits of such listing. A delisting of our common stock
from Nasdaq may materially impair our stockholders’ ability to buy and sell our common stock and could have an adverse effect on
the market price of, and the efficiency of the trading market for, our common stock. In addition, the delisting of our common stock could
significantly impair our ability to raise capital.
We currently do not intend to declare dividends
on our common stock in the foreseeable future and, as a result, your returns on your investment may depend solely on the appreciation
of our common stock.
We currently do not expect to declare any dividends
on our common stock in the foreseeable future. Instead, we anticipate that all of our earnings in the foreseeable future will be used
to provide working capital, to support our operations and to finance the growth and development of our business. Any determination to
declare or pay dividends in the future will be at the discretion of our Board, subject to applicable laws and dependent upon a number
of factors, including our earnings, capital requirements and overall financial conditions. In addition, terms of any future debt or preferred securities
may further restrict our ability to pay dividends on our common stock. Accordingly, your only opportunity to achieve a return
on your investment in our common stock may be if the market price of our common stock appreciates and you sell your shares at a profit.
The market price for our common stock may never exceed, and may fall below, the price that you pay for such common stock. See “Dividend
Policy.”
If you purchase shares of our common stock
in this offering, you may incur immediate and substantial dilution in the book value of your shares if the offering price per share is
greater than the as adjusted net tangible book value per share at the time of issuance.
The public offering price of our common stock will vary from time to
time as this offering is an at-the-market offering. Therefore, if you purchase shares of our common stock in this offering at a price
per share that exceeds our as adjusted net tangible book value per share at the time of issuance, you may suffer immediate and substantial
dilution per share in the as adjusted net tangible book value of common stock purchased. Although there is no dilution to investors in
this offering using an assumed offering price of $0.25, which was the last reported sale price of our common stock on Nasdaq on July 11,
2024, investors should be aware of the possibility of dilution. To the extent shares are issued under outstanding options or warrants,
you will incur further dilution.
An investment in our securities is speculative
and there can be no assurance of any return on any such investment.
An investment in our securities is speculative
and there can be no assurance that investors will obtain any return on their investment. Investors may be subject to substantial risks
involved in an investment in the Company, including the risk of losing their entire investment.
We may need, but be unable, to obtain additional
funding on satisfactory terms, which could dilute our stockholders or impose burdensome financial restrictions on our business.
We have relied upon cash from financing activities
and in the future, we hope to rely on revenues generated from operations to fund the cash requirements of our activities. However, there
can be no assurance that we will be able to generate any significant cash from our operating activities in the future. Future financing
may not be available on a timely basis, in sufficient amounts or on terms acceptable to us, if at all. Any debt financing or other financing
of securities senior to the common stock will likely include financial and other covenants that will restrict our flexibility. Any failure
to comply with these covenants would have a material adverse effect on our business, prospects, financial condition and results of operations
because we could lose our existing sources of funding and impair our ability to secure new sources of funding.
The requirements of being a public company
may strain our resources, divert management’s attention and affect our results of operations.
As a public company in the United States, we face
increased legal, accounting, administrative and other costs and expenses. We are subject to the reporting requirements of the Exchange
Act and the Sarbanes-Oxley Act of 2002. The Exchange Act requires, among other things, that we file annual, quarterly and current reports
with respect to our business and financial condition. The Sarbanes-Oxley Act requires, among other things, that we maintain effective
disclosure controls and procedures and internal control over financial reporting. For example, Section 404 requires that our management
report on the effectiveness of our internal controls structure and procedures for financial reporting. Section 404 compliance may divert
internal resources and will take a significant amount of time and effort to complete. If we fail to maintain compliance under Section
404, or if in the future management determines that our internal control over financial reporting are not effective as defined under Section
404, we could be subject to sanctions or investigations by Nasdaq, the SEC or other regulatory authorities. Furthermore, investor perceptions
of our Company may suffer, and this could cause a decline in the market price of our common stock. Any failure of our internal control
over financial reporting could have a material adverse effect on our stated results of operations and harm our reputation. If we are unable
to implement these changes effectively or efficiently, it could harm our operations, financial reporting or financial results and could
result in an adverse opinion on internal controls from our independent auditors. We may need to hire a number of additional employees
with public accounting and disclosure experience in order to meet our ongoing obligations as a public company, particularly if we become
fully subject to Section 404 and its auditor attestation requirements, which will increase costs. We expect these rules and regulations
to increase our legal and financial compliance costs and to make some activities more time consuming and costly, although we are currently
unable to estimate these costs with any degree of certainty. A number of those requirements will require us to carry out activities we
have not done previously. Our management team and other personnel will need to devote a substantial amount of time to new compliance initiatives
and to meeting the obligations that are associated with being a public company, which may divert attention from other business concerns,
which could have a material adverse effect on our business, financial condition and results of operations.
Additionally, the expenses incurred by public
companies generally for reporting and corporate governance purposes have been increasing. These increased costs will require us to divert
a significant amount of money that we could otherwise use to develop our business. If we are unable to satisfy our obligations as a public
company, we could be subject to delisting of our common stock, fines, sanctions and other regulatory action and potentially civil litigation.
New laws, regulations, and standards relating
to corporate governance and public disclosure may create uncertainty for public companies, increasing legal and financial compliance costs
and making some activities more time consuming.
These laws, regulations and standards are subject
to varying interpretations, in many cases due to their lack of specificity, and, as a result, may evolve over time as new guidance is
provided by the courts and other bodies. This could result in continuing uncertainty regarding compliance matters and higher costs necessitated
by ongoing revisions to disclosure and governance practices. If our efforts to comply with new laws, regulations, and standards differ
from the activities intended by regulatory or governing bodies due to ambiguities related to their application and practice, regulatory
authorities may initiate legal proceedings against us and our business may be adversely affected.
As a public company subject to these rules and
regulations, we may find it more expensive for us to obtain director and officer liability insurance, and we may be required to accept
reduced coverage or incur substantially higher costs to obtain coverage. These factors could also make it more difficult in the future
for us to attract and retain qualified members of our Board, particularly to serve on its audit committee and compensation committee,
and qualified executive officers.
If securities or industry analysts do not
publish research or publish inaccurate or unfavorable research about our business, our stock price and trading volume could decline.
The trading market for our common stock will depend
in part on the research and reports that securities or industry analysts publish about us or our business. Several analysts may cover
our stock. If one or more of those analysts downgrade our stock or publish inaccurate or unfavorable research about our business, our
stock price would likely decline. If one or more of these analysts cease coverage of our Company or fail to publish reports on us regularly,
demand for our stock could decrease, which might cause our stock price and trading volume to decline.
If there is no active public market for
our common stock, you may be unable to sell your shares at or above your purchase price.
Although our common stock is listed on Nasdaq,
an active trading market for our shares may not be sustained following the purchase of your common stock. You may be unable to sell your
shares quickly or at the market price if trading in shares of our common stock is not active. Further, an inactive market may also impair
our ability to raise capital by selling shares of our common stock and may impair our ability to enter into strategic partnerships or
acquire companies or products by using our shares of common stock as consideration.
We may be subject to securities litigation,
which is expensive and could divert our management’s attention.
The market price of our securities may be volatile,
and in the past companies that have experienced volatility in the market price of their securities have been subject to securities class
action litigation. We may be the target of this type of litigation in the future. Securities litigation against us could result in substantial
costs and divert our management’s attention from other business concerns.
You should consult your own independent
tax advisor regarding any tax matters arising with respect to the securities offered in connection with this offering.
Participation in this offering could result in
various tax-related consequences for investors. All prospective purchasers of the resold securities are advised to consult their own independent
tax advisors regarding the U.S. federal, state, local and non-U.S. tax consequences relevant to the purchase, ownership and disposition
of the resold securities in their particular situations.
IN ADDITION TO THE ABOVE RISKS, BUSINESSES
ARE OFTEN SUBJECT TO RISKS NOT FORESEEN OR FULLY APPRECIATED BY MANAGEMENT. IN REVIEWING THIS FILING, POTENTIAL INVESTORS SHOULD KEEP
IN MIND THAT OTHER POSSIBLE RISKS MAY ADVERSELY IMPACT THE COMPANY’S BUSINESS OPERATIONS AND THE VALUE OF THE COMPANY’S SECURITIES.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus, the documents incorporated by
reference herein and therein, and other written and oral statements we make from time to time contain certain “forward-looking”
statements within the meaning of Section 27A of the Securities Act and Section 21E of the Securities Exchange Act of 1934, as amended
(the “Exchange Act”). You can identify these forward-looking statements by the fact they use words such as “could,”
“would,” “should,” “expect,” “anticipate,” “estimate,” “target,”
“may,” “project,” “guidance,” “intend,” “plan,” “believe,” “will,”
“potential,” “opportunity,” “future” and other words and terms of similar meaning and expression in
connection with any discussion of future operating or financial performance. You can also identify forward-looking statements by the fact
that they do not relate strictly to historical or current facts. Such forward-looking statements are based on current expectations and
involve inherent risks and uncertainties, including factors that could delay, divert or change any of them, and could cause actual outcomes
to differ materially from current expectations. These statements are likely to relate to, among other things, our business strategy, our
research and development, our product development efforts, our ability to commercialize our product candidates, the activities of our
licensees, our prospects for initiating partnerships or collaborations, the timing of the introduction of products and services, the effect
of new accounting pronouncements, uncertainty regarding our future operating results and our profitability, anticipated sources of funds
as well as our plans, objectives, expectations and intentions.
We have included more detailed descriptions of
these risks and uncertainties and other risks and uncertainties applicable to our business that we believe could cause actual results
to differ materially from any forward-looking statement in the “Risk Factors” sections of this prospectus and the documents
incorporated by reference herein including, but not limited to, the risk factors incorporated by reference from our filings with the SEC.
We encourage you to read those descriptions carefully. Although we believe we have been prudent in our plans and assumptions, no assurance
can be given that any goal or plan set forth in forward-looking statements can be achieved. We caution investors not to place significant
reliance on forward-looking statements; such statements need to be evaluated in light of all the information contained and incorporated
by reference in this prospectus. Furthermore, the statements speak only as of the date of each document, and we undertake no obligation
to update or revise these statements.
USE OF PROCEEDS
We may issue and sell shares of common stock having
aggregate sales proceeds of up to $1,253,603 from time to time, before deducting sales agent commissions and expenses. The amount of proceeds
from this offering will depend upon the number of shares of our common stock sold and the market price at which they are sold. Because
there is no minimum offering amount required as a condition of this offering, the actual total public offering amount, commissions and
proceeds to us, if any, are not determinable at this time. There can be no assurance that we will be able to sell any shares under or
fully utilize the Sales Agreement with the Sales Agent as a source of financing.
We intend to use the net proceeds from this offering
for general corporate purposes, working capital, and executing the Strategy described herein. We may also use a portion of the net proceeds
from this offering to acquire or invest in complementary businesses, technologies, products or other intellectual property, although we
have no present commitments or agreements to do so.
The amounts and timing of our use of the net proceeds
from this offering will depend on a number of factors, such as the timing and progress of our commercialization efforts, research and
development efforts, the timing and progress of any partnering efforts, technological advances and the competitive environment for our
products. As of the date of this prospectus supplement, we cannot specify with certainty all of the particular uses for the net proceeds
to us from the sale of the shares of common stock offered by us hereunder. Accordingly, our management will have broad discretion in the
timing and application of these proceeds. Pending application of the net proceeds as described above, we intend to temporarily invest
the proceeds in short-term, interest-bearing instruments.
DIVIDEND POLICY
We have never declared or paid any cash dividends
on our capital stock. We intend to retain future earnings, if any, to finance the operation of our business and do not anticipate paying
any cash dividends in the foreseeable future. Any future determination related to our dividend policy will be made at the discretion of
our Board after considering our financial condition, results of operations, capital requirements, business prospects and other factors
our Board deems relevant, and subject to the restrictions contained in any future financing instruments.
PLAN OF DISTRIBUTION
We have entered into an At the Market Offering
Agreement, dated July 12, 2024 with WallachBeth Capital LLC under which we may issue and sell shares of our common stock having aggregate
sales proceeds of up to $5,000,000 from time to time through the Sales Agent. Any such sales will be made by any method that is deemed
to be an “at the market offering” as defined in Rule 415(a)(4) under the Securities Act, including sales made directly on
or through The Nasdaq Capital Market or any other existing trading market for our common stock in the United States or to or through a
market maker.
Each time we wish to issue and sell shares of
common stock under the Sales Agreement, we will notify the Sales Agent of the number of shares to be issued, the dates on which such sales
are anticipated to be made, any limitation on the number of shares to be sold in any one day and any minimum price below which sales may
not be made. Once we have so instructed the Sales Agent, subject to the terms and conditions of the Sales Agreement, the Sales Agent has
agreed to use its commercially reasonable efforts consistent with its normal trading and sales practices to sell such shares up to the
amount specified on such terms. The obligations of the Sales Agent under the Sales Agreement to sell our shares of common stock are subject
to a number of conditions that we must meet.
The Sales Agent will provide written confirmation
to us following the close of trading on The Nasdaq Capital Market following each day in which shares of our common stock are sold under
the Sales Agreement. Each confirmation will include the number of shares sold on the day, the aggregate gross sales proceeds, the net
proceeds to us and the compensation payable by us to the Sales Agent with respect to the sales. The settlement of sales of shares between
us and the Sales Agent is generally anticipated to occur on the second trading day following the date on which the sale was made, or such
shorter settlement cycle that is in effect under Exchange Act Rule 15c6-1 from time to time. Sales of our shares of common stock as contemplated
in this prospectus supplement will be settled through the facilities of The Depository Trust Company or by such other means as we and
the Sales Agent may agree. There is no arrangement for funds to be received in an escrow, trust or similar arrangement. We will report
at least quarterly the number of shares of common stock sold through the Sales Agent under the Sales Agreement, the net proceeds to us
and the compensation paid by us to the Sales Agent in connection with the sales of common stock.
We will pay the Sales Agent a commission equal to 4.0% of the aggregate
gross proceeds we receive from each sale of our shares of common stock. Because there is no minimum offering amount required as a condition
of this offering, the actual total public offering amount, commissions and proceeds to us, if any, are not determinable at this time.
In addition, we agreed to reimburse the Sales Agent for the fees and disbursements of its counsel, payable upon execution of the Sales
Agreement, in an amount not to exceed $50,000, in addition to certain ongoing disbursements of its legal counsel up to $2,500 per calendar
quarter. We estimate that the total expenses for the offering, excluding any commissions or expense reimbursement payable to the Sales
Agent under the terms of the Sales Agreement, will be approximately $117,645. The remaining sale proceeds, after deducting any other transaction
fees, will equal our net proceeds from the sale of such shares.
In connection with the sale of the shares of common
stock on our behalf, the Sales Agent may be deemed to be an “underwriter” within the meaning of the Securities Act, and the
compensation of the Sales Agent may be deemed to be underwriting commissions or discounts. We have agreed to indemnify the Sales Agent
against certain civil liabilities, including liabilities under the Securities Act. We have also agreed to contribute to payments the Sales
Agent may be required to make in respect of such liabilities.
The offering of our shares of common stock pursuant
to the Sales Agreement will terminate upon the earlier of (i) the sale of all shares of our common stock subject to the Sales Agreement
and (ii) the termination of the Sales Agreement as permitted therein.
The Sales Agent and its affiliates have provided,
and may in the future provide, various investment banking, commercial banking and other financial services for us and our affiliates,
for which services they have received, or may in the future receive, customary fees. To the extent required by Regulation M promulgated
under the Exchange Act, the Sales Agent will not engage in any market making activities involving our common stock while the offering
is ongoing under this prospectus supplement.
This prospectus supplement and the accompanying
base prospectus in electronic format may be made available on a website maintained by the Sales Agent and the Sales Agent may distribute
this prospectus supplement and the accompanying base prospectus electronically.
LEGAL MATTERS
The validity of the issuance of the securities
offered hereby will be passed upon for us by Sichenzia Ross Ference Carmel LLP located in New York, New York. TroyGould PC, located in
Los Angeles, California, is acting as counsel to WallachBeth Capital LLC in this offering.
EXPERTS
RBSM LLP, an independent registered public accounting
firm, audited our financial statements for the years ended December 31, 2023 and 2022, respectively. We have included our financial statements
in this prospectus and elsewhere in the registration statement in reliance on the reports of RBSM LLP, given their authority as experts
in accounting and auditing.
WHERE YOU CAN FIND MORE INFORMATION
This prospectus is part of the registration statement
on Form S-3 that we filed with the Commission under the Securities Act and does not contain all of the information set forth in the
registration statement. Whenever a reference is made in this prospectus to any of our contracts, agreements or other documents, the reference
may not be complete and you should refer to the exhibits that are part of the registration statement or the exhibits to the reports or
other document incorporated into this prospectus for a copy of such contract agreement or other document. Because we are subject to the
information and reporting requirements under the Exchange Act, we file annual, quarterly and current reports, proxy statements and other
information with the Commission. Our filings with the Commission are available to the public over the Commission’s website at www.sec.gov.
Our Annual Report on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K, including any amendments to those reports,
and other information that we file with or furnish to the Commission pursuant to Section 13(a) or 15(d) of the Exchange Act can also be
accessed free of charge on our website. You may also read and copy any document we file with the SEC at its public reference facility
at 100 F Street, N.E., Washington, D.C., 20549, on official business days during the hours of 10 a.m. to 3 p.m. You may also obtain copies
of the documents at prescribed rates by writing to the Public Reference Section of the SEC at 100 F Street, N.E., Washington, D.C., 20549.
Please call the SEC at 1-800-SEC-0330 for further information on the operation of the public reference facility. In addition, you can
find more information about us on our website at https://60degreespharma.com. Information contained on or accessible through our
website is not a part of this prospectus and is not incorporated by reference herein, and the inclusion of our website address in this
prospectus is an inactive textual reference only.
INCORPORATION OF CERTAIN INFORMATION BY REFERENCE
The SEC allows us to “incorporate by reference”
information that we file with it into this prospectus, which means that we can disclose important information to you by referring you
to those documents. The information incorporated by reference is an important part of this prospectus. The information incorporated by
reference into this prospectus is deemed to be part of this prospectus, and any information filed with the SEC after the date of this
prospectus will automatically be deemed to update and supersede information contained in this prospectus and any accompanying prospectus
supplement.
The following documents previously filed with
the SEC are incorporated by reference in this prospectus:
| |
● |
The Registrant’s Annual Report on Form 10-K for the fiscal year ended December 31, 2023, filed with the SEC on April 1, 2024; |
| |
● |
The Registrant’s Quarterly Report on Form 10-Q for the fiscal quarter ended March 31, 2024, filed with the SEC on May 15, 2024; |
| |
● |
The Registrant’s Preliminary Schedule 14A, filed with the SEC on May 3, 2024, and the Registrant’s Definitive Schedule 14A, filed with the SEC on May 30, 2024; |
| |
● |
The description of the Registrant’s common stock, which is contained in a registration statement on Form 8-A12B filed with the SEC on June 27, 2023, under the Exchange Act, including any amendment or report filed for the purpose of updating such description. |
All filings filed by us pursuant to the Exchange
Act after the date of the initial filing of the registration statement of which this prospectus is a part and prior to effectiveness of
the registration statement shall be deemed to be incorporated by reference into this prospectus.
We also incorporate by reference all additional
documents that we file with the Securities and Exchange Commission under the terms of Sections 13(a), 13(c), 14 or 15(d) of the Exchange
Act that are made after the date of the initial registration statement but prior to effectiveness of the registration statement and after
the date of this prospectus but prior to the termination of the offering of the securities covered by this prospectus. We are not, however,
incorporating, in each case, any documents or information that we are deemed to furnish and not file in accordance with Securities and
Exchange Commission rules.
You should rely only on the information contained
or incorporated by reference in this prospectus. We have not authorized any other person to provide you with different information. If
anyone provides you with different or inconsistent information, you should not rely on it. You should assume that the information appearing
in this prospectus is accurate only as of the date of this prospectus. Our business, financial condition, results of operations and prospects
may have changed since that date.
Any statement contained in a document incorporated
or deemed to be incorporated by reference into this prospectus will be deemed to be modified or superseded for the purposes of this prospectus
to the extent that a statement contained herein, or in any other subsequently filed document which also is or is deemed to be incorporated
by reference herein, modifies or supersedes that statement. The modifying or superseding statement need not state it has modified or superseded
a prior statement or include any other information set forth in the document that it modifies or supersedes. The making of a modifying
or superseding statement is not an admission for any purposes that the modified or superseded statement, when made, constituted a misrepresentation,
an untrue statement of a material fact or an omission to state a material fact that is required to be stated or that is necessary to make
a statement not misleading in light of the circumstances in which it was made. Any statement so modified or superseded will not be deemed,
except as so modified or superseded, to constitute a part of this prospectus.
You may request, and we will provide you with,
a copy of these filings, at no cost, by calling us at (202) 327-5422 or by writing to us at the following address:
60 Degrees Pharmaceuticals, Inc.
1025 Connecticut Avenue NW Suite 1000
Washington, D.C. 20036
Attn: Geoffrey Dow, Chief Executive Officer and
President
$1,253,306
Common Stock

60 Degrees Pharmaceuticals, Inc.
PROSPECTUS SUPPLEMENT
WallachBeth Capital LLC
July 12, 2024
PART II
INFORMATION NOT REQUIRED IN THE PROSPECTUS
Item 14. Other Expenses of Issuance and Distribution.
The following table sets forth an estimate of
the fees and expenses relating to the issuance and distribution of the securities being registered hereby, other than underwriting discounts
and commissions, all of which shall be borne by the Registrant. All of such fees and expenses, except for the Securities and Exchange
Commission (“SEC”) registration fee are estimated:
| SEC registration fee | |
$ | 2,214 | |
| FINRA filing fee | |
| * | |
| Legal fees and expenses | |
| * | |
| Printing fees and expenses | |
| * | |
| Accounting fees and expenses | |
| * | |
| Transfer agent fees and expenses | |
| * | |
| Warrant agent fees and expenses | |
| * | |
| Trustee fees and expenses | |
| * | |
| Miscellaneous fees and expenses | |
| * | |
| Total | |
$ | * | |
| * | These fees and expenses depend
on the securities offered and the number of issuances, and accordingly cannot be estimated at this time and will be reflected in the
applicable prospectus supplement. |
Item 15. Indemnification of Directors and Officers.
Section 102 of the General
Company Law of the State of Delaware (“DGCL”) permits a Company to eliminate the personal liability of directors of a Company
to the Company or its stockholders for monetary damages for a breach of fiduciary duty as a director, except where the director breached
his duty of loyalty, failed to act in good faith, engaged in intentional misconduct or knowingly violated a law, authorized the payment
of a dividend or approved a stock repurchase in violation of Delaware corporate law or obtained an improper personal benefit. Our charter
provides that no director of the Company shall be personally liable to it or its stockholders for monetary damages for any breach of fiduciary
duty as a director, notwithstanding any provision of law imposing such liability, except to the extent that the DGCL prohibits the elimination
or limitation of liability of directors for breaches of fiduciary duty.
Section 145 of the DGCL
provides that a Company has the power to indemnify a director, officer, employee, or agent of the Company, or a person serving at the
request of the Company for another Company, partnership, joint venture, trust or other enterprise in related capacities against expenses
(including attorneys’ fees), judgments, fines and amounts paid in settlement actually and reasonably incurred by the person in connection
with an action, suit or proceeding to which he was or is a party or is threatened to be made a party to any threatened, ending or completed
action, suit or proceeding by reason of such position, if such person acted in good faith and in a manner he reasonably believed to be
in or not opposed to the best interests of the Company, and, in any criminal action or proceeding, had no reasonable cause to believe
his conduct was unlawful, except that, in the case of actions brought by or in the right of the Company, no indemnification shall be made
with respect to any claim, issue or matter as to which such person shall have been adjudged to be liable to the Company unless and only
to the extent that the Court of Chancery or other adjudicating court determines that, despite the adjudication of liability but in view
of all of the circumstances of the case, such person is fairly and reasonably entitled to indemnity for such expenses which the Court
of Chancery or such other court shall deem proper.
If a claim is not paid in full by the Company,
the claimant may at any time thereafter bring suit against the Company to recover the unpaid amount of the claim and, if successful in
whole or in part, the claimant shall also be entitled to be paid the expense of prosecuting such claim. It shall be a defense to any such
action (other than an action brought to enforce a claim for expenses incurred in defending any proceeding in advance of its final disposition
where any undertaking required by the Bylaws has been tendered to the Company) that the claimant has not met the standards of conduct
which make it permissible under the DGCL for the Company to indemnify the claimant for the amount claimed, but the burden of proving such
defense shall be on the Company. Neither the failure of the Company (including its board of directors (“Board”), legal counsel,
or its stockholders) to have made a determination prior to the commencement of such action that indemnification of the claimant is proper
in the circumstances because he or she has met the applicable standard of conduct set forth in the DGCL, nor an actual determination by
the Company (including its Board, legal counsel, or its stockholders) that the claimant has not met such applicable standard of conduct,
shall be a defense to the action or create a presumption that the claimant has not met the applicable standard of conduct. Indemnification
shall include payment by the Company of expenses in defending an action or proceeding in advance of the final disposition of such action
or proceeding upon receipt of an undertaking by the person indemnified to repay such payment if it is ultimately determined that such
person is not entitled to indemnification.
Item 16. Exhibits.
Exhibit
Number |
|
Description |
| 1.1 |
|
At the Market Offering Agreement, dated June 4, 2024, by and between
Wallachbeth Capital, LLC and the Registrant. |
| 3.1 |
|
Certificate of Incorporation (incorporated by reference to the Registrant’s Registration Statement No. 333-269483, filed on January 31, 2023). |
| 3.2 |
|
Certificate of Designation of Series A Preferred Stock (incorporated by reference to the Registrant’s Registration Statement No. 333-269483, filed on January 31, 2023). |
| 3.3 |
|
Certificate of Correction to Certificate of Incorporation of the Registrant (incorporated by reference to the Registrant’s Registration Statement No. 333-269483, filed on January 31, 2023). |
| 3.4 |
|
Amended and Restated Bylaws of the Registrant (incorporated by reference to the Registrant’s Registration Statement No. 333-269483, filed on January 31, 2023). |
| 4.1* |
|
Form of Certificate of Designations, Rights and Preferences of Preferred Stock. |
| 4.2 |
|
Form of Senior Indenture. |
| 4.3 |
|
Form of Subordinated Indenture. |
| 4.4* |
|
Form of Debt Security. |
| 4.5* |
|
Form of Warrant. |
| 4.6* |
|
Form of Warrant Agreement. |
| 4.7* |
|
Form of Unit Agreement. |
| 5.1 |
|
Opinion of Counsel to Registrant relating to the base prospectus. |
| 5.2 |
|
Opinion of Counsel to Registrant relating to the ATM prospectus. |
| 10.1 |
|
Sponsored Project Agreement dated as of May 10, 2024, by and between
the Registrant and North Carolina State University, Raleigh North Carolina. |
| 10.2** |
|
Veterinary Trial Agreement dated as of March 28, 2024, by and between
the Registrant and North Carolina State University, Raleigh North Carolina. |
| 10.3** |
|
Clinical Trial Agreement dated as of May 29, 2024, by and between the
Registrant and Tufts Medicine, Inc. |
| 23.1 |
|
Consent of RBSM LLP dated as of July 12, 2024. |
| 23.2 |
|
Consent of Counsel to Registrant (included in Exhibit 5.1). |
| 24.1 |
|
Power of Attorney (included on the signature page hereto). |
| 25.1* |
|
Statement of Eligibility of Trustee on Form T-1 under the Trust Indenture
Act of 1939, as amended. |
| 107 |
|
Filing Fee Table (filed herewith). |
| |
* |
If applicable, to be filed by amendment or by a report filed under the Exchange Act and incorporated herein by reference. |
| |
** |
Appendixes have been omitted
pursuant to Item 601(b)(5) of Regulation S-K. The Company undertakes to furnish supplemental copies of any of the omitted appendixes
upon request by the SEC. |
Item 17. Undertakings.
| (a) |
The undersigned registrant hereby undertakes: |
| |
(1) |
To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement: |
| |
(i) |
To include any prospectus required by Section 10(a)(3) of the Securities Act of 1933; |
| |
(ii) |
To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in this registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Commission pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than a 20 percent change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement; |
| |
(iii) |
To include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information in this registration statement; |
Provided, however, that paragraphs (a)(1)(i),
(a)(1)(ii) and (a)(1)(iii) of this section do not apply if the registration statement is on Form S-3 or Form F-3 and the information
required to be included in a post-effective amendment by those paragraphs is contained in reports filed with or furnished to the Commission
by the registrant pursuant to section 13 or section 15(d) of the Securities Exchange Act of 1934 that are incorporated by reference in
the registration statement, or is contained in a form of prospectus filed pursuant to Rule 424(b) that is part of the registration statement.
| |
(2) |
That, for the purpose of determining any liability under the Securities Act of 1933, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered herein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
| |
(3) |
To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering. |
| |
(4) |
That, for the purpose of determining liability under the Securities Act of 1933 to any purchaser: |
| |
(i) |
If the registrant is relying on Rule 430B: |
| (A) | Each prospectus filed by the
registrant pursuant to Rule 424(b)(3) shall be deemed to be part of the registration statement as of the date the filed prospectus was
deemed part of and included in the registration statement; and |
| |
(B) |
Each prospectus required to be filed pursuant to Rule 424(b)(2), (b)(5) or (b)(7) as part of a registration statement in reliance on Rule 430B relating to an offering made pursuant to Rule 415 (a)(1)(i), (vii) or (x) for the purpose of providing the information required by section 10(a) of the Securities Act of 1933 shall be deemed to be part of and included in the registration statement as of the earlier of the date such form of prospectus is first used after effectiveness or the date of the first contract of sale of securities in the offering described in the prospectus. As provided in Rule 430B, for liability purposes of the issuer and any person that is at that date an underwriter, such date shall be deemed to be a new effective date of the registration statement relating to the securities in the registration statement to which that prospectus relates, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
Provided, however, that no statement made in a
registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated
by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time
of contract of sale prior to such effective date, supersede or modify any statement that was made in the registration statement or prospectus
that was part of the registration statement or made in any such document immediately prior to such effective date.
| |
(5) |
That, for the purpose of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution of the securities: |
The undersigned registrant undertakes that in
an offering of securities of the undersigned registrant pursuant to this registration statement, regardless of the underwriting method
used to sell the securities to the purchaser, if the securities are offered or sold to such purchaser by means of any of the following
communications, the undersigned registrant will be a seller to the purchaser and will be considered to offer or sell such securities to
such purchaser:
| |
(i) |
Any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424; |
| |
(ii) |
Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by the undersigned registrant; |
| |
(iii) |
The portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant or its securities provided by or on behalf of the undersigned registrant; and |
| |
(iv) |
Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser. |
| (b) |
The undersigned registrant hereby further undertakes that, for the purposes of determining any liability under the Securities Act of 1933, each filing of the registrant’s annual report pursuant to section 13(a) or section 15(d) of the Securities Exchange Act of 1934 (and, where applicable, each filing of an employee benefit plan’s annual report pursuant to section 15(d) of the Securities Exchange Act of 1934) that is incorporated by reference in the registration statement shall be deemed to be a new registration statement relating to the securities offered herein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
| |
|
| (c) |
Insofar as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Securities Act of 1933, and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act of 1933, and will be governed by the final adjudication of such issue. |
| (d) |
The undersigned registrant hereby undertakes to file an application for the purpose of determining the eligibility of the trustee to act under subsection (a) of section 310 of the Trust Indenture Act (“Trust Indenture Act”) in accordance with the rules and regulations prescribed by the Commission under section 305(b)(2) of the Trust Indenture Act. |
SIGNATURES
Pursuant to the requirements of the Securities
Act of 1933, the registrant has duly caused this Registration Statement to be signed on its behalf by the undersigned, thereunto duly
authorized in the City of Washington, District of Columbia, on July 12, 2024.
| |
60 DEGREES PHARMACEUTICALS, INC. |
| |
|
| |
By: |
/s/ Geoffrey Dow |
| |
|
Geoffrey Dow |
| |
|
President and Chief Executive Officer
(Principal Executive Officer) |
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each
person whose signature appears below constitutes and appoints Geoffrey Dow and Tyrone Miller, and each of them (with full power to each
of them to act alone), his or her true and lawful attorneys-in-fact and agents, with full power of substitution and resubstitution, for
him or her and in his or her name, place and stead, in any and all capacities, to sign any or all amendments (including post-effective
amendments) to this registration statement, and to file the same, with all exhibits thereto and other documents in connection therewith,
with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority
to do and perform each and every act and thing requisite and necessary to be done in and about the premises, as fully to all intents and
purposes as he or she might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents, or any
of them, or their substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities
Act of 1933, this Registration Statement has been signed by the following persons in the capacities and on the dates indicated.
| Name |
|
Position |
|
Date |
| |
|
|
|
|
| /s/ Geoffrey
Dow |
|
President, Chief Executive Officer and Director |
|
July 12, 2024 |
| Geoffrey Dow |
|
(Principal Executive Officer) |
|
|
| |
|
|
|
|
| /s/ Tyrone
Miller |
|
Chief Financial Officer |
|
July 12, 2024 |
| Tyrone Miller |
|
(Principal Financial and Accounting Officer) |
|
|
| |
|
|
|
|
| /s/ Charles
Allen |
|
Director |
|
July 12, 2024 |
| Charles Allen |
|
|
|
|
| |
|
|
|
|
| /s/ Cheryl
Xu |
|
Director |
|
July 12, 2024 |
| Cheryl Xu |
|
|
|
|
| |
|
|
|
|
| /s/ Stephen
Toovey |
|
Director |
|
July 12, 2024 |
| Stephen Toovey |
|
|
|
|
| |
|
|
|
|
| /s/ Paul
Field |
|
Director |
|
July 12, 2024 |
| Paul Field |
|
|
|
|
II-5
Exhibit 1.1
60
Degrees Pharmaceuticals, Inc.
Common Stock
(par value $0.0001 per share)
At-The-Market Issuance Sales Agreement
June 4, 2024
WallachBeth Capital LLC
185 Hudson St
Jersey City, NJ 07302
Ladies and Gentlemen:
60 Degrees Pharmaceuticals,
Inc., a Delaware corporation (the “Company”), confirms its agreement (this “Agreement”) with WallachBeth
Capital LLC (the “Agent”), as follows:
1. Issuance and Sale of
Shares. The Company agrees to issue and sell through or to the Agent, shares (the “Placement Shares”) of the Company’s
common stock, par value $0.0001 per share (the “Common Stock”), from time to time during the term of this Agreement
and on the terms set forth in this Agreement; provided however, that in no event will the Company issue or sell through or to
the Agent such dollar amount of Placement Shares that would exceed $5,000,000 in the aggregate, subject to General Instruction I.B.6
of Form S-3 (the “Maximum Amount”). Notwithstanding anything to the contrary contained herein, the parties hereto
agree that compliance with the limitations set forth in this Section 1 on the amount of Placement Shares issued and sold under this Agreement
will be the sole responsibility of the Company and that the Agent will have no obligation in connection with such compliance provided
the Agent follows the trading instructions provided by the Company pursuant to any Placement Notice in all material respects. The issuance
and sale of Placement Shares through or to the Agent will be effected pursuant to the Registration Statement (as defined below) filed
by the Company and declared effective by the U.S. Securities and Exchange Commission (the “SEC”), although nothing
in this Agreement will be construed as requiring the Company to use the Registration Statement to issue Common Stock. Certain capitalized
terms used in this Agreement have the meanings ascribed to them in Section 25.
The Company has filed with
the SEC, in accordance with the provisions of the Securities Act of 1933, as amended (the “Securities Act”), and the
rules and regulations thereunder (the “Securities Act Regulations”), a registration statement on Form S-3 (File No.
333-271030), including a base prospectus, relating to certain securities, including the Placement Shares, to be issued from time to time
by the Company, and which incorporates by reference documents that the Company has filed or will file in accordance with the provisions
of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), and the rules and regulations thereunder.
The Company has prepared or will prepare a prospectus supplement specifically relating to the Placement Shares (the “Prospectus
Supplement”) to the base prospectus included as part of the registration statement. The Company will furnish to the Agent,
for use by them, copies of the prospectus included as part of the registration statement, as supplemented by the Prospectus Supplement.
Except when the context otherwise requires, such registration statement, including all documents filed as part thereof or incorporated
by reference therein, and including any information contained in a Prospectus (as defined below) subsequently filed with the SEC pursuant
to Rule 424(b) under the Securities Act Regulations or deemed to be a part of the registration statement pursuant to Rule 430B
of the Securities Act Regulations, is herein called the “Registration Statement.” The base prospectus, including all
documents incorporated therein by reference, included in the Registration Statement, as it may be supplemented by the Prospectus Supplement,
in the form in which the prospectus and/or Prospectus Supplement have most recently been filed by the Company with the SEC pursuant to
Rule 424(b) under the Securities Act Regulations is herein called the “Prospectus.” Any reference herein to the
Registration Statement, the Prospectus, or any amendment or supplement thereto will be deemed to refer to and include the documents incorporated
by reference therein, and any reference herein to the terms “amend,” “amendment,” or “supplement”
respecting the Registration Statement or the Prospectus will be deemed to refer to and include the filing of any document with the SEC
deemed to be incorporated by reference therein, including in each such case filings made after the execution hereof (any such documents,
collectively, the “Incorporated Documents”).
For purposes of this Agreement,
all references to the Registration Statement, the Prospectus, or to any amendment or supplement thereto will be deemed to include the
most recent copy filed with the SEC pursuant to its Electronic Data Gathering Analysis and Retrieval System, or if applicable, the Interactive
Data Electronic Application system when used by the SEC (collectively, “EDGAR”).
2. Placements. Each
time that the Company wishes to issue and sell Placement Shares hereunder (each, a “Placement”), it will notify the
Agent by email notice (or other method mutually agreed to in writing by the parties) of the number of Placement Shares, the period during
which sales are requested to be made, any limitation on the number of Placement Shares that may be sold in any one day, and any minimum
price below which sales may not be made (a “Placement Notice”), the form of which is attached hereto as Schedule
1. The Placement Notice will originate from any of the individuals from the Company set forth on Schedule 3 (with a copy to
each of the other individuals from the Company listed on the schedule) and will be addressed to each of the individuals from the Agent
that are set forth on Schedule 3, as Schedule 3 may be amended from time to time. The Placement Notice will be effective
unless and until: (a) the Agent declines to accept the terms contained therein for any reason, in its sole discretion, which must
be evidenced by a written notice to the Company, addressed to the individuals set forth on Schedule 3 within two (2) Business
Days after receipt of such Placement Notice; (b) the entire amount of the Placement Shares thereunder have been sold; (c) the
Company suspends or terminates the Placement Notice; or (d) the Agreement has been terminated under the provisions of Section 13.
The amount of any discount, commission, or other compensation to be paid by the Company to the Agent in connection with the sale of the
Placement Shares will be calculated in accordance with the terms set forth in Schedule 2. Neither the Company nor the Agent will
have any obligation whatsoever respecting a Placement or any Placement Shares unless and until the Company delivers a Placement Notice
to the Agent and the Agent does not decline such Placement Notice pursuant to the terms set forth above, and then only upon the terms
specified therein and herein. In the event of a conflict between the terms of Sections 2, 3, and 4 of this Agreement and the terms of
a Placement Notice, the terms of the Placement Notice will control.
3. Sale of Placement Shares
by the Agent. Subject to the terms and conditions of this Agreement, each Agent, at any time it is an Agent, for the period specified
in the Placement Notice, will use its commercially reasonable efforts consistent with its normal trading and sales practices and applicable
state and federal laws, rules, and regulations and the rules of the Nasdaq Stock Market (the “Exchange”), to sell
the Placement Shares in accordance with the terms of such Placement Notice. The Agent will provide written confirmation to the Company
no later than the opening of the Trading Day (as defined below) immediately following the Trading Day on which it has sold Placement
Shares hereunder, setting forth the number of Placement Shares sold on such day, the compensation payable by the Company to the Agent
pursuant to Section 2 for such sales, and the Net Proceeds (as defined below) payable to the Company, with an itemization
of the deductions made by the Agent (as set forth in Section 5(b)) from the gross proceeds that it receives from such sales.
Subject to the terms of the Placement Notice, Placement Shares may be sold hereunder by any method permitted by law deemed to be an “at
the market” offering as defined in Rule 415(a)(1)(x) and Rule 415(a)(4) of the Securities Act Regulations, including sales
made directly on the Exchange or on any other existing trading market for the Common Stock, to or through a market maker or directly
to the Placement Agent as principal in negotiated transactions. Subject to the terms of a Placement Notice, the Agent may also sell Placement
Shares by any other method permitted by law, including in privately negotiated transactions, with the Company’s consent. “Trading
Day” means any day on which Common Stock is purchased and sold on the Exchange.
4. Suspension of Sales.
The Company or the Agent may, upon notice to the other party in writing (including by email correspondence to each of the individuals
of the other party set forth on Schedule 3, if receipt of such correspondence is actually acknowledged by any of the individuals
to whom the notice is sent, other than via auto-reply) or by telephone (confirmed immediately by verifiable email correspondence to each
of the individuals of the other party set forth on Schedule 3), suspend any sale of Placement Shares; provided, however, that
such suspension will not affect or impair any party’s obligations respecting any Placement Shares sold hereunder prior to the receipt
of such notice. Notwithstanding anything herein to the contrary, the obligations under Section 7(l), 7(m), and 7(n) with respect
to delivery of certificates, opinions and comfort letters to the Agent shall not apply while a suspension of sales under this Section
4 is in effect. Each of the parties agrees that no such notice under this Section 4 will be effective against any other party
unless it is made to one of the individuals named on Schedule 3 hereto, as such schedule may be amended from time to time.
5. Sale and Delivery to
the Agent; Settlement.
(a) Sale of
Placement Shares. On the basis of the representations and warranties herein contained and subject to the terms and conditions
herein set forth, upon delivery of a Placement Notice, and unless the sale of the Placement Shares described therein has been declined,
suspended, or otherwise terminated in accordance with the terms of this Agreement, each Agent, at any time it is an Agent, for the period
specified in the Placement Notice, will use its commercially reasonable efforts consistent with its normal trading and sales practices
and applicable state and federal laws, rules and regulations and the rules of the Exchange to sell such Placement Shares up to the amount
specified, and otherwise in accordance with the terms of such Placement Notice. The Company acknowledges and agrees that: (i) there
can be no assurance that any Agent will be successful in selling Placement Shares; (ii) no Agent will incur any liability or obligation
to the Company or any other Person (as defined herein) for a failure to sell Placement Shares for any reason other than a failure by
such Agent to use its commercially reasonable efforts consistent with its normal trading and sales practices and applicable law and regulations
to sell such Placement Shares as required under this Agreement; and (iii) no Agent will be under any obligation to purchase Placement
Shares on a principal basis pursuant to this Agreement, except as otherwise agreed by such Agent and the Company.
(b) Settlement
of Placement Shares. Unless otherwise specified in the applicable Placement Notice, settlement for sales of Placement Shares
will occur on the second Trading Day (or such earlier day as is industry practice for regular-way trading) following the date on which
such sales are made (each, a “Settlement Date”). The Agent shall notify the Company of each sale of Placement Shares
no later than opening day following the Trading Day that the Agent sold Placement Shares. The amount of proceeds to be delivered to the
Company on a Settlement Date against receipt of the Placement Shares sold (the “Net Proceeds”) will be equal to the
aggregate sales price received by the Agent, after deduction for the Agent’s commission, discount, or other compensation for
such sales payable by the Company pursuant to Section 2 hereof.
(c) Delivery
of Placement Shares. Before each Settlement Date, the Company will, or will cause its transfer agent to, electronically transfer
an agreed-upon amount of the Placement Shares being sold by crediting the Agent’s account or its designee’s account (provided
the Agent shall have given the Company written notice of such designee a reasonable period of time prior to the Settlement Date)
at The Depository Trust Company through its Deposit and Withdrawal at Custodian System or by such other means of delivery as may be mutually
agreed upon by the parties hereto, which in all cases will be freely tradable, transferable, registered shares in good deliverable form.
On each Settlement Date, the Agent will deliver the related Net Proceeds in same-day funds to an account designated by the Company on,
or prior to, the Settlement Date. The Agent is not obligated to sell any of the Placement Shares unless they are first deposited in the
Agent’s account or its designee’s account. The Company agrees that if the Company or its transfer agent defaults in its obligation
to deliver Placement Shares before a Settlement Date through no fault of the Agent that, in addition to and in no way limiting the rights
and obligations set forth in Section 11(a) hereto, it will hold the Agent harmless against any loss, claim, damage, or expense
(including reasonable and documented legal fees and expenses) arising out of or in connection with such default by the Company or its
transfer agent (if applicable).
(d) Limitations
on Offering Size. Under no circumstances will the Company cause or request the offer or sale of any Placement Shares if, after
giving effect to the sale of such Placement Shares, the aggregate gross sales proceeds of Placement Shares sold pursuant to this Agreement
would exceed the lesser of: (i) together with all sales of Placement Shares under this Agreement, the Maximum Amount; or (ii) the
amount authorized from time to time to be issued and sold under this Agreement by the Company’s board of directors, a duly authorized
committee thereof, or a duly authorized executive committee, and notified to the Agent in writing. Under no circumstances will the Company
cause or request the offer or sale of any Placement Shares pursuant to this Agreement at a price lower than the minimum price authorized
from time to time by the Company’s board of directors, a duly authorized committee thereof, or a duly authorized executive committee,
and notified to the Agent in writing.
6. Representations and
Warranties of the Company. The Company represents and warrants to, and agrees with, the Agent that as of the date of this Agreement
and as of each Applicable Time (as defined below), unless such representation, warranty or agreement specifies a different date or time:
(a) Registration
Statement and Prospectus. The Company and, assuming no act or omission on the part of the Agent that would make such statement untrue,
the transactions contemplated by this Agreement meet the requirements for and comply with the conditions for the use of Form S-3
under the Securities Act. The Registration Statement has been filed with the SEC and has been declared effective under the Securities
Act. The Prospectus Supplement will name the Agent as the agent in the section entitled “Plan of Distribution.” The Company
has not received, and has no notice of, any order of the SEC preventing or suspending the use of the Registration Statement or threatening
or instituting proceedings for that purpose. The Registration Statement and the offer and sale of Placement Shares as contemplated hereby
meet the requirements of Rule 415 under the Securities Act and comply in all material respects with said Rule. Any statutes, regulations,
contracts, or other documents that are required to be described in the Registration Statement or the Prospectus or to be filed as exhibits
to the Registration Statement have been so described or filed. Copies of the Registration Statement, the Prospectus, and any such amendments
or supplements and all documents incorporated by reference therein that were filed with the SEC on or prior to the date of this Agreement
have been delivered, or are available through EDGAR, to the Agent and its counsel. The Company has not distributed and, prior to the
later to occur of each Settlement Date and completion of the distribution of the Placement Shares, will not distribute any offering material
in connection with the offering or sale of the Placement Shares other than the Registration Statement and the Prospectus and any Issuer
Free Writing Prospectus (as defined below) to which the Agent has consented, which consent shall not be unreasonably withheld or delayed.
The Common Stock is currently listed on the Exchange under the trading symbol “SXPT.” Except as disclosed in the Registration
Statement, including the Incorporated Documents, the Company has not, in the six months preceding the date hereof, received notice from
the Exchange to the effect that the Company is not in compliance with the Exchange’s listing requirements. Except as disclosed
in the Registration Statement, including the Incorporated Documents, or the Prospectus, the Company has no reason to believe that it
will be unable to comply with the Exchange listing requirements.
(b) No Misstatement
or Omission. The Registration Statement, when it became effective, and the Prospectus, and any amendment or supplement thereto, on
the date of such Prospectus or amendment or supplement, conformed and will conform in all material respects with the requirements of
the Securities Act. At each Settlement Date, the Registration Statement and the Prospectus, as of such date, will conform in all material
respects with the requirements of the Securities Act. The Registration Statement, when it became effective, did not contain an untrue
statement of a material fact or omit to state a material fact required to be stated therein or necessary to make the statements therein
not misleading. The Prospectus and any amendment and supplement thereto, on the date thereof and at each Applicable Time (defined below),
did not and will not include an untrue statement of a material fact or omit to state a material fact necessary to make the statements
therein, in light of the circumstances under which they were made, not misleading. The documents incorporated by reference in the Prospectus
or any Prospectus Supplement did not, and any further documents incorporated by reference therein will not, when filed with the SEC,
contain an untrue statement of a material fact or omit to state a material fact required to be stated in such document or necessary to
make the statements in such document, in light of the circumstances under which they were made, not misleading. The foregoing will not
apply to statements in, or omissions from, any such document made in reliance upon, and in conformity with, information furnished to
the Company by the Agent specifically for use in the preparation thereof.
(c) Conformity
with Securities Act and Exchange Act. The Incorporated Documents, when such documents were or are filed with the SEC under the Securities
Act or the Exchange Act, or became or become effective under the Securities Act, as the case may be, conformed or will conform in all
material respects with the requirements of the Securities Act and the Exchange Act, as applicable.
(d) Financial
Information. The consolidated financial statements of the Company included or incorporated by reference in the Registration Statement,
the Prospectus, and the Issuer Free Writing Prospectuses, if any, together with the related notes and schedules, complied as to form
in all material respects with applicable accounting requirements and the published rules and regulations of the SEC with respect thereto
as in effect as of the time of filing. Such financial statements have been prepared in accordance with generally accepted accounting
principles, consistently applied, during the periods involved (except: (i) as may be otherwise indicated in such financial statements
or the notes thereto; or (ii) in the case of unaudited interim statements, to the extent they may exclude footnotes or may be condensed
or summary statements) and fairly present in all material respects the consolidated financial position of the Company and the Subsidiaries
as of the dates indicated and the consolidated results of operations and cash flows of the Company for the periods specified (subject,
in the case of unaudited statements, to normal year-end audit adjustments that will not be material, either individually or in the aggregate);
the other financial and statistical data respecting the Company and the Subsidiaries contained or incorporated by reference in the Registration
Statement, the Prospectus, and the Issuer Free Writing Prospectuses, if any, are accurately and fairly presented and prepared in all
material respects on a basis consistent with the financial statements and books and records of the Company; there are no financial statements
(historical or pro forma) that are required to be included or incorporated by reference in the Registration Statement or the Prospectus
that are not included or incorporated by reference as required; the Company and the Subsidiaries (as defined below) do not have any material
liabilities or obligations, direct or contingent (including any off-balance sheet obligations), not described in the Registration Statement
(including the exhibits thereto) and the Prospectus that are required to be described in the Registration Statement or the Prospectus
(including exhibits thereto and Incorporated Documents); and all disclosures contained or incorporated by reference in the Registration
Statement, the Prospectus, and the Issuer Free Writing Prospectuses, if any, regarding “non-GAAP financial measures” (as
such term is defined by the rules and regulations of the SEC) comply in all material respects with Regulation G of the Exchange Act and
Item 10 of Regulation S-K under the Securities Act, to the extent applicable.
(e) Conformity
with EDGAR Filing. The Prospectus delivered to the Agent for use in connection with the sale of the Placement Shares pursuant to
this Agreement will be identical to the versions of the Prospectus created to be transmitted to the SEC for filing via EDGAR, except
to the extent permitted by Regulation S-T.
(f) Organization.
The Company and each of its Subsidiaries are duly organized, validly existing as a corporation, limited partnership, limited liability
company, or other legal entity, and in good standing under the laws of their respective jurisdictions of organization, except where the
failure to be in good standing would not, individually or in the aggregate, have a Material Adverse Effect (as defined below) or reasonably
be expected to have a Material Adverse Effect. The Company and each of its Subsidiaries are duly qualified as a foreign corporation for
transaction of business and in good standing under the laws of each other jurisdiction in which their respective ownership or lease of
property or the conduct of their respective businesses requires such qualification, and have all corporate power and authority necessary
to own or hold their respective properties and to conduct their respective businesses as described in the Registration Statement and
the Prospectus, except where the failure to be so qualified or in good standing or have such power or authority would not, individually
or in the aggregate, have a material adverse effect or reasonably be expected to have a material adverse effect on the assets, business,
operations, earnings, properties, condition (financial or otherwise), prospects, stockholders’ equity, or results of operations
of the Company and the Subsidiaries (as defined below) taken as a whole, or prevent or materially interfere with consummation of the
transactions contemplated hereby (a “Material Adverse Effect”).
(g) Subsidiaries.
The significant subsidiaries of the Company, as determined pursuant to Rule 1-02(w) of Regulation S-X, are set forth on Schedule 4
(collectively, the “Subsidiaries”). Except as set forth in the Registration Statement and in the Prospectus, the
Company owns, directly or indirectly, all of the equity interests of the Subsidiaries free and clear of any material lien, charge, security
interest, encumbrance, right of first refusal, or other restriction, and all the equity interests of the Subsidiaries are validly issued
and are fully paid, nonassessable, and free of preemptive and similar rights.
(h) No Violation
or Default. Neither the Company nor any of its Subsidiaries is: (i) in violation of its charter or bylaws or similar organizational
documents; (ii) in default, and no event has occurred that, with notice or lapse of time or both, would constitute such a default,
in the due performance or observance of any term, covenant, or condition contained in any indenture, mortgage, deed of trust, loan agreement,
or other agreement or instrument to which the Company or any of its Subsidiaries is a party, by which the Company or any of its Subsidiaries
is bound, or to which any of the property or assets of the Company or any of its Subsidiaries are subject; or (iii) in violation
of any law or statute or any judgment, order, rule, or regulation of any court, arbitrator, or governmental or regulatory authority,
except, in the case of each of clauses (ii) and (iii) above, for any such violation or default that would not, individually or in the
aggregate, reasonably be expected to have a Material Adverse Effect. Except as described in the Prospectus, the Prospectus Supplement,
or the Incorporated Documents, to the Company’s knowledge, no other party under any material contract or other agreement to which
it or any of its Subsidiaries is a party is in default in any respect thereunder where such default would reasonably be expected to have
a Material Adverse Effect.
(i) No Material
Adverse Change. Subsequent to the respective dates as of which information is given in the Registration Statement and the Prospectus
(including any Incorporated Documents), there has not been: (i) any Material Adverse Effect, or any development involving a prospective
Material Adverse Effect, in or affecting the business, properties, management, financial, condition (financial or otherwise), results
of operations, or prospects of the Company and the Subsidiaries taken as a whole; (ii) any transaction that is material to the Company
and the Subsidiaries taken as a whole; (iii) any obligation or liability, direct or contingent (including any off-balance sheet
obligations), incurred by the Company or any Subsidiary, that is material to the Company and the Subsidiaries taken as a whole; (iv) any
material change in the capital stock (other than as a result of the sale of Placement Shares or other than as described in a proxy statement
filed on Schedule 14A or a Registration Statement on Form S-4 and otherwise publicly announced) or outstanding long-term indebtedness
of the Company or any of its Subsidiaries; or (v) any dividend or distribution of any kind declared, paid, or made on the capital
stock of the Company or any Subsidiary, other than in each case above, in the ordinary course of business or as otherwise disclosed in
the Registration Statement or Prospectus (including any document deemed incorporated by reference therein).
(j) Capitalization.
The issued and outstanding shares of capital stock of the Company have been validly issued, are fully paid, and nonassessable. The Company
has an authorized, issued, and outstanding capitalization as set forth in the Registration Statement or the Prospectus as of the dates
referred to therein (other than the grant of shares of Common Stock, options or restricted stock under the Company’s existing equity
incentive plans, or changes in the number of outstanding Common Stock of the Company due to the issuance of shares upon the exercise
or conversion of securities exercisable for, or convertible into, Common Stock outstanding on the date hereof or as a result of the issuance
of Placement Shares), and such authorized capital stock conforms to the description thereof set forth in the Registration Statement and
the Prospectus. The description of the Common Stock in the Registration Statement and the Prospectus is complete and accurate in all
material respects. Except as disclosed in or contemplated by the Registration Statement, the Prospectus, and the Incorporated Documents,
as of the date referred to therein, the Company did not have reserved or available for issuance any shares of Common Stock in respect
of options, any rights or warrants to subscribe for, any securities or obligations convertible into or exchangeable for, or any contracts
or commitments to issue or sell, any shares of capital stock or other securities.
(k) Authorization;
Enforceability. The Company has full legal right, power, and authority to enter into this Agreement and perform the transactions
contemplated hereby. This Agreement has been duly authorized, executed, and delivered by the Company and is a legal, valid, and binding
agreement of the Company enforceable in accordance with its terms, except to the extent that: (i) enforceability may be limited
by bankruptcy, insolvency, reorganization, moratorium, or similar laws affecting creditors’ rights generally and by general equitable
principles; and (ii) the indemnification and contribution provisions of Section 11 hereof may be limited by federal or state
securities laws and public policy considerations in respect thereof.
(l) Authorization
of Placement Shares. The Placement Shares, when issued and delivered pursuant to the terms approved by the board of directors of
the Company or a duly authorized committee thereof, or a duly authorized executive committee, against payment therefor as provided herein,
will be duly and validly authorized and issued and fully paid and nonassessable, free and clear of any pledge, lien, encumbrance, security
interest, or other claim (other than any pledge, lien, encumbrance, security interest, or other claim arising from an act or omission
of any Agent or a purchaser), including any statutory or contractual preemptive rights, resale rights, rights of first refusal, or other
similar rights, and will be registered pursuant to Section 12 of the Exchange Act. The Placement Shares, when issued, will conform in
all material respects to the description thereof set forth in or incorporated into the Prospectus.
(m) No Consents
Required. No consent, approval, authorization, order, registration, or qualification of or with any court or arbitrator or any governmental
or regulatory authority having jurisdiction over the Company is required for the execution, delivery, and performance by the Company
of this Agreement, and the issuance and sale by the Company of the Placement Shares as contemplated hereby, except for such consents,
approvals, authorizations, orders, and registrations or qualifications as may be required under applicable state securities laws or by
the bylaws and rules of the Financial Industry Regulatory Authority (“FINRA”) or the Exchange in connection with the
sale of the Placement Shares by the Agent.
(n) No Preferential
Rights. Except as set forth in the Registration Statement or the Prospectus: (i) no person, as such term is defined in Rule
1-02 of Regulation S-X promulgated under the Securities Act (each, a “Person”), has the right, contractual
or otherwise, to cause the Company to issue or sell to such Person any Common Stock or shares of any other capital stock or other securities
of the Company (other than upon the exercise of options or warrants to purchase Common Stock or upon the exercise of options that may
be granted from time to time under the Company’s stock option plans); (ii) no Person has any preemptive rights, rights of first
refusal, or any other rights (whether pursuant to a “poison pill” provision or otherwise) to purchase any Common Stock or
shares of any other capital stock or other securities of the Company from the Company that have not been duly waived respecting the offering
contemplated hereby; (iii) no Person has the right to act as an underwriter or as a financial advisor to the Company in connection
with the offer and sale of the Placement Shares; and (iv) no Person has the right, contractual or otherwise, to require the Company
to register under the Securities Act any Common Stock or shares of any other capital stock or other securities of the Company, or to
include any such shares or other securities in the Registration Statement or the offering contemplated thereby, whether as a result of
the filing or effectiveness of the Registration Statement or the sale of the Placement Shares as contemplated thereby or otherwise, except
for such rights as have been waived on or prior to the date hereof.
(o) Independent
Public Accountant. RBSM LLP (the “Accountants”), whose reports on the consolidated financial statements of the
Company are filed with the SEC as part of the Company’s most recent Annual Report on Form 10-K and incorporated by reference into
the Registration Statement, is and, during the periods covered by its report, was an independent registered public accounting firm within
the meaning of the Securities Act and the Public Company Accounting Oversight Board (United States). To the Company’s knowledge,
the Accountants are not in violation of the auditor independence requirements of the Sarbanes-Oxley Act of 2002 (the “Sarbanes-Oxley
Act”) respecting the Company.
(p) Enforceability
of Agreements. To the Company’s knowledge, all agreements between the Company and third parties expressly referenced in the
Prospectus, other than such agreements that have expired by their terms or whose termination is disclosed in documents filed by the Company
on EDGAR, are legal, valid, and binding obligations of the Company enforceable in accordance with their respective terms, except to the
extent that: (i) enforceability may be limited by bankruptcy, insolvency, reorganization, moratorium, or similar laws affecting
creditors’ rights generally and by general equitable principles; and (ii) the indemnification provisions of certain agreements
may be limited be federal or state securities laws or public policy considerations in respect thereof, except for any unenforceability
that, individually or in the aggregate, would not reasonably be expected to have a Material Adverse Effect.
(q) No Litigation.
Except as set forth in the Registration Statement or the Prospectus: (i) there are no legal, governmental, or regulatory actions,
suits, or proceedings pending or, to the Company’s knowledge, any legal, governmental, or regulatory investigations to which the
Company, a Subsidiary, or any of their respective directors, officers, or controlling Persons is a party or to which any property of
the Company or any of its Subsidiaries is the subject that, individually or in the aggregate, if determined adversely to the Company
or any of its Subsidiaries, would reasonably be expected to have a Material Adverse Effect or materially and adversely affect the ability
of the Company to perform its obligations under this Agreement; (ii) to the Company’s knowledge, no actions, suits, or proceedings
are threatened or contemplated by any governmental or regulatory authority or threatened by others against the Company, a Subsidiary,
or any of their respective directors, officers, or controlling Persons that, individually or in the aggregate, if determined adversely
to the Company or any of its Subsidiaries, would reasonably be expected to have a Material Adverse Effect; (iii) there are no current
or pending legal, governmental, or regulatory, actions, suits, proceedings or, to the Company’s knowledge, investigations that
are required under the Securities Act to be described in the Prospectus that are not described in the Prospectus; and (iv) there
are no contracts or other documents that are required under the Securities Act to be filed as exhibits to the Registration Statement
that are not so filed.
(r) Licenses
and Permits. Except as set forth in the Registration Statement or the Prospectus, the Company and each of its Subsidiaries possess
or have obtained all licenses, certificates, consents, orders, approvals, permits, and other authorizations issued by, and have made
all declarations and filings with, the appropriate federal, state, local, or foreign governmental or regulatory authorities that are
necessary for the ownership or lease of their respective properties or the conduct of their respective businesses as described in the
Registration Statement and the Prospectus (the “Permits”), except where the failure to possess, obtain, or make the
same would not, individually or in the aggregate, reasonably be expected to have a Material Adverse Effect. Except as disclosed in the
Registration Statement or the Prospectus, neither the Company nor its Subsidiaries have received written notice of any proceeding relating
to revocation or modification of any such Permit or have any reason to believe that such Permit will not be renewed in the ordinary course,
except when the failure to obtain any such renewal would not, individually or in the aggregate, reasonably be expected to have a Material
Adverse Effect.
(s) S-3 Eligibility.
(i) At the time of filing the Registration Statement; and (ii) if applicable, at the time of the most recent amendment thereto
for the purposes of complying with Section 10(a)(3) of the Securities Act (whether such amendment was by post-effective amendment, incorporated
report filed pursuant to Section 13 or 15(d) of the Exchange Act, or form of prospectus), the Company met the then-applicable requirements
for use of Form S-3 under the Securities Act, including compliance with General Instruction I.B.6 of Form S-3, for the sale of up to
the Maximum Amount of Placement Shares.
(t) No Material
Defaults. Except as set forth in the Registration Statement and Prospectus, neither the Company nor any of the Subsidiaries has defaulted
on any installment on indebtedness for borrowed money or on any rental on one or more long-term leases, which defaults, individually
or in the aggregate, would reasonably be expected to have a Material Adverse Effect. Except as set forth in the Registration Statement
and Prospectus, the Company has not filed a report pursuant to Section 13(a) or 15(d) of the Exchange Act since the filing of its last
Annual Report on Form 10-K, indicating that it: (i) has failed to pay any dividend or sinking fund installment on preferred
stock; or (ii) has defaulted on any installment on indebtedness for borrowed money or on any rental on one or more long-term leases,
which defaults, individually or in the aggregate, could reasonably be expected to have a Material Adverse Effect.
(u) Certain
Market Activities. Neither the Company or any of the Subsidiaries, nor, to the Company’s knowledge, any of their respective
directors, officers, or controlling Persons has taken, directly or indirectly, any action designed, or that has constituted or might
reasonably be expected to cause or result in, under the Exchange Act or otherwise, the stabilization or manipulation of the price of
any security of the Company to facilitate the sale or resale of the Placement Shares.
(v) Broker-Dealer
Relationships. Neither the Company nor any of the Subsidiaries or any related entities: (i) are required to register as a “broker”
or “dealer” in accordance with the provisions of the Exchange Act; or (ii) directly or indirectly through one or more
intermediaries, control or are a “person associated with a member” or “associated person of a member” (within
the meaning set forth in the FINRA rules).
(w) No Reliance.
The Company has not relied upon the Agent or legal counsel for the Agent for any legal, tax, or accounting advice in connection with
the offering and sale of the Placement Shares.
(x) Taxes.
The Company and each of its Subsidiaries have filed all federal, state, local, and foreign tax returns that have been required to be
filed and paid all taxes shown thereon through the date hereof, to the extent that such taxes have become due and are not being contested
in good faith, except when the failure to do so would not reasonably be expected to have a Material Adverse Effect. Except as otherwise
disclosed in or contemplated by the Registration Statement or the Prospectus, no tax deficiency has been determined adversely to the
Company or any of its Subsidiaries that has had, or would reasonably be expected to have, individually or in the aggregate, a Material
Adverse Effect. The Company has no knowledge of any federal, state, or other governmental tax deficiency, penalty, or assessment that
has been or might be asserted or threatened against it that could reasonably be expected to have a Material Adverse Effect.
(y) Title to
Real and Personal Property. The Company and each of its Subsidiaries have good title to all of their real and personal property owned
by them that are material to the business of the Company and such Subsidiary, in each case, free and clear of all liens, encumbrances,
and defects, except as described in the Registration Statement and Prospectus or that do not materially affect the value of the properties
of the Company and its Subsidiaries, considered as one enterprise, and do not interfere in any material respect with the use made and
proposed to be made of such properties by the Company and its Subsidiaries, considered as one enterprise; and all of the leases, subleases,
and other rights under which the Company or any of its Subsidiaries holds or uses properties described in the Registration Statement
and Prospectus are in full force and effect, with such exceptions as would not reasonably be expected to have a Material Adverse Effect,
and neither the Company nor any of its Subsidiaries has received any written notice of any material claim of any sort that has been asserted
by anyone adverse to the rights of the Company or its Subsidiaries under any of the leases, subleases, and other rights mentioned above,
or affecting or questioning the rights of the Company or any Subsidiary thereof to the continued possession or use of the leased or subleased
premises or the premises granted by leases, subleases, and other rights. The Company and each of its Subsidiaries have the consents,
easements, rights-of-way, or licenses from any Person as are necessary to enable them to conduct their business in the manner described
in the Registration Statement and the Prospectus, subject to such qualifications as may be set forth in the Registration Statement and
the Prospectus, and except for the consents, easements, rights-of-way, or licenses the lack of which would not have, individually or
in the aggregate, a Material Adverse Effect.
(z) Intellectual
Property. Except as set forth in the Registration Statement or the Prospectus, to the Company’s knowledge, the Company and
its Subsidiaries own or possess adequate enforceable rights to use all patents, patent applications, trademarks (both registered and
unregistered), service marks, trade names, trademark registrations, service mark registrations, copyrights, licenses, and know-how (including
trade secrets and other unpatented and/or unpatentable proprietary or confidential information, systems, or procedures) (collectively,
the “Intellectual Property”), necessary for the conduct of their respective businesses as conducted as of the date
hereof, except to the extent that the failure to own or possess adequate rights to use such Intellectual Property would not, individually
or in the aggregate, reasonably be expected to have a Material Adverse Effect; except as disclosed in writing to the Agent, the Company
and any of its Subsidiaries have not received any written notice of any claim of infringement or conflict that asserted Intellectual
Property rights of others, which infringement or conflict, if the subject of an unfavorable decision, would result in a Material Adverse
Effect; there are no pending, or to the Company’s knowledge, threatened judicial proceedings or interference proceedings against
the Company or its Subsidiaries challenging the Company’s or its Subsidiaries’ rights in or to or the validity of the scope
of any of the Company’s or its Subsidiaries’ material patents, patent applications, or proprietary information; to the Company’s
knowledge, no other entity or individual has any right or claim in any of the Company’s or its Subsidiaries’ owned, material
patents, patent applications, or any patent to be issued therefrom by virtue of any contract, license, or other agreement entered into
between such entity or individual and the Company or a Subsidiary or by any non-contractual obligation of the Company or a Subsidiary,
other than by written licenses granted by the Company or a Subsidiary; the Company and its Subsidiaries have not received any written
notice of any claim challenging the rights of the Company or a Subsidiary in or to any Intellectual Property owned, licensed, or optioned
by the Company or such Subsidiary that, if the subject of an unfavorable decision, would result in a Material Adverse Effect.
(aa) Environmental
Laws. Except as set forth in the Registration Statement or the Prospectus, the Company and its Subsidiaries: (i) are in compliance
with any and all applicable federal, state, local, and foreign laws, rules, regulations, decisions, and orders relating to the protection
of human health and safety, the environment, hazardous or toxic substances or wastes, pollutants, or contaminants (collectively, “Environmental
Laws”); (ii) have received and are in compliance with all permits, licenses, or other approvals required of them under
applicable Environmental Laws to conduct their respective businesses as described in the Registration Statement and the Prospectus; (iii) have
not received written notice of any actual or potential liability for the investigation or remediation of any disposal or release of hazardous
or toxic substances or wastes, pollutants, or contaminants, except, in the case of any of clauses (i), (ii), or (iii) above, for any
such failure to comply or failure to receive required permits, licenses, other approvals or liability as would not, individually or in
the aggregate, reasonably be expected to have a Material Adverse Effect; and (iv) there are no costs or liabilities arising under
Environmental Laws respecting the operation of the Company’s and each of its Subsidiaries’ properties (including any capital
or operating expenditures required for clean-up or closure of the properties, compliance with Environmental Laws, any permit, license,
or approval or any related legal constraints or operating activities, and any potential liabilities of third parties assumed under contract
by the Company or any of its Subsidiaries) that would reasonably be expected, individually or in the aggregate, to have a Material Adverse
Effect.
(bb) Disclosure
Controls. Except as set forth in the Registration Statement or the Prospectus, the Company and each of its Subsidiaries maintains
a system of internal accounting controls designed to provide reasonable assurance that: (i) transactions are executed in accordance
with management’s general or specific authorizations; (ii) transactions are recorded as necessary to permit preparation of
financial statements in conformity with generally accepted accounting principles and to maintain asset accountability; (iii) access
to assets is permitted only in accordance with management’s general or specific authorization; and (iv) the recorded accountability
for assets is compared with the existing assets at reasonable intervals and appropriate action is taken respecting any differences. The
Company is not aware of any material weaknesses in its internal control over financial reporting (other than as set forth in the Prospectus).
Since the date of the latest audited financial statements of the Company included in the Prospectus, there has been no change in the
Company’s internal control over financial reporting that has materially affected, or is reasonably likely to materially affect,
the Company’s internal control over financial reporting (other than as set forth in the Prospectus). Except as set forth in the
Registration Statement or the Prospectus, the Company has established disclosure controls and procedures (as defined in Exchange Act
Rules 13a-15 and 15d-15) for the Company and designed such disclosure controls and procedures to provide reasonable assurance
that material information relating to the Company and each of its Subsidiaries is made known to the certifying officers by others within
those entities, particularly during the period in which the Company’s Annual Report on Form 10-K or Quarterly Report on Form
10-Q, as the case may be, is being prepared. The Company’s certifying officers have evaluated the effectiveness of the Company’s
controls and procedures as of a date within 90 days prior to the filing date of the Form 10-K for the fiscal year most recently ended
(such date, the “Evaluation Date”). The Company will present in its Form 10-K for the fiscal year most recently
ended the conclusions of the certifying officers about the effectiveness of the disclosure controls and procedures based on their evaluations
as of the Evaluation Date. Since the Evaluation Date, there have been no significant changes in the Company’s internal controls
(as such term is defined in Item 307(b) of Regulation S-K under the Securities Act).
(cc) Sarbanes-Oxley.
There is and has been no failure on the part of the Company or, to the knowledge of the Company, any of the Company’s directors
or officers, in their capacities as such, to comply in all material respects with any applicable provisions of the Sarbanes-Oxley Act
and the rules and regulations promulgated thereunder. Each of the principal executive officer and the principal financial officer of
the Company (or each former principal executive officer of the Company and each former principal financial officer of the Company as
applicable) has made all certifications required by Sections 302 and 906 of the Sarbanes-Oxley Act respecting all reports, schedules,
forms, statements, and other documents required to be filed by it or furnished by it to the SEC. For purposes of the preceding sentence,
“principal executive officer” and “principal financial officer” will have the meanings given to such terms in
the Sarbanes-Oxley Act.
(dd) Finder’s
Fees. Neither the Company nor any of the Subsidiaries has incurred any liability for any finder’s fees, brokerage commissions,
or similar payments in connection with the transactions herein contemplated, except as may otherwise exist respecting the Agent pursuant
to this Agreement.
(ee) Labor Disputes.
No labor disturbance by or dispute with employees of the Company or any of its Subsidiaries exists or, to the knowledge of the Company,
is threatened that would reasonably be expected to result in a Material Adverse Effect.
(ff) Investment
Company Act. Neither the Company nor any of the Subsidiaries is or, after giving effect to the offering and sale of the Placement
Shares, will be an “investment company” or an entity “controlled” by an “investment company,” as
such terms are defined in the Investment Company Act of 1940, as amended (the “Investment Company Act”).
(gg) Operations.
The operations of the Company and its Subsidiaries are and have been conducted at all times in compliance with applicable financial recordkeeping
and reporting requirements of the Currency and Foreign Transactions Reporting Act of 1970, as amended, the money laundering statutes
of all jurisdictions to which the Company or its Subsidiaries are subject, the rules and regulations thereunder, and any related or similar
rules, regulations, or guidelines, issued, administered, or enforced by any governmental agency having jurisdiction over the Company
(collectively, the “Money Laundering Laws”), except as would not reasonably be expected to result in a Material Adverse
Effect; and no action, suit, or proceeding by or before any court or governmental agency, authority, or body or any arbitrator involving
the Company or any of its Subsidiaries respecting the Money Laundering Laws is pending or, to the knowledge of the Company, threatened.
(hh) Off-Balance
Sheet Arrangements. There are no transactions, arrangements, and other relationships between and/or among the Company, and/or, to
the knowledge of the Company, any of its affiliates and any unconsolidated entity, including any structural finance, special purpose,
or limited purpose entity (each, an “Off Balance Sheet Transaction”) that could reasonably be expected to affect materially
the Company’s liquidity or the availability of or requirements for its capital resources, including those Off Balance Sheet Transactions
described in the SEC’s Statement about Management’s Discussion and Analysis of Financial Conditions and Results of Operations
(Release Nos. 33-8056; 34-45321; FR-61), required to be described in the Prospectus that have not been described as required.
(jj) Underwriter
Agreements. The Company is not a party to any agreement with an agent or underwriter for any other “at-the-market” or
continuous equity transaction.
(jj) ERISA.
To the knowledge of the Company: (i) each material employee benefit plan, within the meaning of Section 3(3) of the Employee Retirement
Income Security Act of 1974, as amended (“ERISA”), that is maintained, administered, or contributed to by the Company
or any of its Subsidiaries (other than a Multiemployer Plan, within the meaning of Section 3(37) of ERISA) for employees or former employees
of the Company and any of its Subsidiaries has been maintained in material compliance with its terms and the requirements of any applicable
statutes, orders, rules, and regulations, including ERISA and the Internal Revenue Code of 1986, as amended (the “Code”);
(ii) no prohibited transaction, within the meaning of Section 406 of ERISA or Section 4975 of the Code, has occurred respecting
any such plan (excluding transactions effected pursuant to a statutory or administrative exemption); and (iii) for each such plan
that is subject to the funding rules of Section 412 of the Code or Section 302 of ERISA, no “accumulated funding deficiency”
as defined in Section 412 of the Code has been incurred, whether or not waived, and the fair market value of the assets of each such
plan (excluding for these purposes accrued but unpaid contributions) equals or exceeds the present value of all benefits accrued under
such plan determined using reasonable actuarial assumptions, other than, in the case of (i), (ii), and (iii) above, as would not reasonably
be expected to have a Material Adverse Effect.
(kk) Margin
Rules. Neither the issuance, sale, and delivery of the Placement Shares nor the application of the proceeds thereof by the Company
as described in the Registration Statement and the Prospectus will violate Regulation T, U, or X of the Board of Governors of the Federal
Reserve System or any other regulation of such Board of Governors.
(ll) Insurance.
The Company and each of its Subsidiaries carry, or are covered by, insurance in such amounts and covering such risks as the Company and
each of its Subsidiaries reasonably believe are adequate for the conduct of their properties and as is customary for companies of similar
size engaged in similar businesses in similar industries.
(mm) No Improper
Practices. (i) Neither the Company nor, to the Company’s knowledge, the Subsidiaries or any of their respective executive
officers has, in the past five years, made any unlawful contributions to any candidate for any political office (or failed fully to disclose
any contribution in violation of law) or made any contribution or other payment to any official of, or candidate for, any federal, state,
municipal, or foreign office or other Person charged with similar public or quasi-public duty in violation of any law or of the character
required to be disclosed in the Prospectus; (ii) no relationship, direct or indirect, exists between or among the Company or, to
the Company’s knowledge, any Subsidiary or any affiliate of any of them, on the one hand, and the directors, officers and stockholders
of the Company or, to the Company’s knowledge, any Subsidiary, on the other hand, that is required by the Securities Act to be
described in the Registration Statement and the Prospectus that is not so described; (iii) no relationship, direct or indirect,
exists between or among the Company or any Subsidiary or any affiliate of them, on the one hand, and the directors, officers, stockholders,
or directors of the Company or, to the Company’s knowledge, any Subsidiary, on the other hand, that is required by the rules of
FINRA to be described in the Registration Statement and the Prospectus that is not so described; (iv) except as described in the
Prospectus, there are no material outstanding loans or advances or material guarantees of indebtedness by the Company or, to the Company’s
knowledge, any Subsidiary to or for the benefit of any of their respective officers or directors or any of the members of the families
of any of them; and (v) the Company has not offered, or caused any placement agent to offer, Common Stock to any Person with the
intent to influence unlawfully: (1) a customer or supplier of the Company or any Subsidiary to alter the customer’s or supplier’s
level or type of business with the Company or any Subsidiary; or (2) a trade journalist or publication to write or publish favorable
information about the Company or any Subsidiary or any of their respective products or services; and (vi) neither the Company nor
any Subsidiary nor, to the Company’s knowledge, any employee or agent of the Company or any Subsidiary has made any payment of
funds of the Company or any Subsidiary or received or retained any funds in violation of any law, rule, or regulation (including the
Foreign Corrupt Practices Act of 1977), which payment, receipt, or retention of funds is of a character required to be disclosed in the
Registration Statement or the Prospectus.
(nn) Status
under the Securities Act. The Company was not and is not an ineligible issuer as defined in Rule 405 under the Securities Act at
the times specified in Rules 164 and 433 under the Securities Act in connection with the offering of the Placement Shares.
(oo) No Misstatement
or Omission in an Issuer Free Writing Prospectus. Each Issuer Free Writing Prospectus, as of its issue date and as of each Applicable
Time (as defined in Section 25 below), did not, does not and will not include any information that conflicted, conflicts or will
conflict with the information contained in the Registration Statement or the Prospectus, including any incorporated document deemed to
be a part thereof that has not been superseded or modified. The foregoing sentence does not apply to statements in or omissions from
any Issuer Free Writing Prospectus based upon and in conformity with written information furnished to the Company by the Agent specifically
for its therein.
(pp) No Conflicts.
None of the execution of this Agreement; the issuance, offering, or sale of the Placement Shares; the consummation of any of the transactions
contemplated herein; or the compliance by the Company with the terms and provisions hereof will conflict with or result in a breach of
any of the terms and provisions of; constitute or will constitute a default under; or has resulted in or will result in the creation
or imposition of any lien, charge, or encumbrance upon any property or assets of the Company pursuant to the terms of any contract or
other agreement to which the Company may be bound or to which any of the property or assets of the Company is subject, except: (i) such
conflicts, breaches, or defaults as may have been waived; and (ii) such conflicts, breaches, and defaults that would not reasonably
be expected to have a Material Adverse Effect; nor will such action result in any material violation of the provisions of the organizational
or governing documents of the Company or in any material violation of the provisions of any statute or any order, rule, or regulation
applicable to the Company or of any court or of any federal, state, or other regulatory authority or other government body having jurisdiction
over the Company, except where such violation would not reasonably be expected to have a Material Adverse Effect.
(qq) Regulatory
Compliance.
(i) Neither the Company
nor any of its Subsidiaries (each, an “Entity”) nor, to the Company’s knowledge, any director, officer, employee,
agent, affiliate, or representative of the Entity, is a government, individual, or entity that is owned or controlled by any director,
officer, employee, agent, affiliate, or representative of the Entity that is:
(1) the subject
of any sanctions administered or enforced by the U.S. Department of Treasury’s Office of Foreign Assets Control, the United Nations
Security Council, the European Union, Her Majesty’s Treasury, or other relevant sanctions authority (collectively, “Sanctions”);
or
(2) located, organized,
or resident in a country or territory that is the subject of Sanctions (including Burma/Myanmar, Cuba, Iran, North Korea, Sudan and Syria).
(ii) The Company,
on behalf of each Entity, represents and covenants that it will not, directly or indirectly, knowingly use, lend, contribute, or otherwise
make available the proceeds of the offering governed by this Agreement to any subsidiary, joint venture partner, or other director, officer,
employee, agent, affiliate, or representative of the Entity:
(1) to fund or
facilitate any activities or business of or with any Person or in any country or territory that, at the time of such funding or facilitation,
is the subject of Sanctions; or
(2) in any other
manner that will result in a violation of Sanctions by any Person (including any Person participating in the offering, whether as underwriter,
advisor, investor, or otherwise).
(iii) Except as detailed
in the Prospectus, for the past five years, the Entity has not knowingly engaged in, is not now knowingly engaged in, and will not engage
in, any dealings or transactions with any Person, or in any country or territory, that at the time of the dealing or transaction is or
was the subject of Sanctions.
(rr) Stock Transfer
Taxes. On each Settlement Date, all stock transfer or other taxes (other than income taxes) that are required to be paid in connection
with the sale and transfer of the Placement Shares to be sold hereunder will be, or will have been, fully paid or provided for by the
Company and all laws imposing such taxes will be or will have been fully complied with in all material respects.
Any certificate signed by an
officer of the Company and delivered to the Agent or to counsel for the Agent pursuant to or in connection with this Agreement will be
deemed to be a representation and warranty by the Company, as applicable, to the Agent as to the matters set forth therein.
7. Covenants of the Company.
The Company covenants and agrees with the Agent that:
(a) Registration
Statement Amendments. After the date of this Agreement and during any period in which a Prospectus relating to any Placement Shares
is required to be delivered by the Agent under the Securities Act (including in circumstances when such requirement may be satisfied
pursuant to Rule 172 under the Securities Act): (i) the Company will notify the Agent promptly of the time when any subsequent amendment
to the Registration Statement, other than documents incorporated by reference, has been filed with the SEC and/or has become effective
or any subsequent supplement to the Prospectus has been filed and of any request by the SEC for any amendment or supplement to the Registration
Statement or Prospectus or for additional information; (ii) the Company will prepare and file with the SEC, promptly upon the Agent’s
reasonable request, any amendments or supplements to the Registration Statement or Prospectus that, in the Agent’s reasonable opinion,
may be necessary or advisable in connection with the distribution of the Placement Shares by the Agent (provided, however, that
the failure of the Agent to make such request will not relieve the Company of any obligation or liability hereunder, or affect the Agent’s
right to rely on the representations and warranties made by the Company in this Agreement and provided, further, that the only remedy
the Agent will have respecting the failure to make such filing will be to cease making sales under this Agreement until such amendment
or supplement is filed); (iii) the Company will not file any amendment or supplement to the Registration Statement or Prospectus
relating to the sale of Placement Shares or a security convertible into the Placement Shares unless a copy thereof has been submitted
to the Agent within a reasonable period of time before the filing and the Agent has not reasonably objected thereto within two (2) Business
Days (provided, however, that (A) the failure of the Agent to make such objection will not relieve the Company of any obligation
or liability hereunder, or affect the Agent’s right to rely on the representations and warranties made by the Company in this Agreement
and provided, further, that the only remedy the Agent will have respecting the failure by the Company to provide the Agent with such
copy will be to cease making sales under this Agreement and (B) the Company will have no obligation to provide the Agent any advance
copy of such filing or provide to the Agent an opportunity to object to such filing if (i) the filing does not name the Agent and does
not relate to the transactions pursuant hereto or (ii) relates to the termination of this Agreement or the Prospectus Supplement) and
the Company will furnish to the Agent at the time of filing thereof a copy of any document that upon filing is deemed to be incorporated
by reference into the Registration Statement or Prospectus, except for those documents available via EDGAR; and (iv) the Company
will cause each amendment or supplement to the Prospectus to be filed with the SEC as required pursuant to the applicable paragraph of
Rule 424(b) of the Securities Act or, in the case of any document to be incorporated therein by reference, to be filed with the SEC as
required pursuant to the Exchange Act, within the period prescribed (the determination to file or not file any amendment or supplement
with the SEC under this Section 7(a), based on the Company’s reasonable opinion or reasonable objections, will be made exclusively
by the Company).
(b) Notice of
SEC Stop Orders. The Company will advise the Agent, promptly after it receives notice or obtains knowledge thereof, of the issuance
or threatened issuance by the SEC of any stop order suspending the effectiveness of the Registration Statement, of the suspension of
the qualification of the Placement Shares for offering or sale in any jurisdiction, or of the initiation or threatening of any proceeding
for any such purpose; and it will promptly use its commercially reasonable efforts to prevent the issuance of any stop order or to obtain
its withdrawal if such a stop order should be issued. The Company will advise the Agent promptly after it receives any request by the
SEC for any amendments to the Registration Statement or any amendment or supplements to the Prospectus or any Issuer Free Writing Prospectus
or for additional information related to the offering of the Placement Shares or for additional information related to the Registration
Statement, the Prospectus or any Issuer Free Writing Prospectus in connection with the offering of the Placement Shares.
(c) Delivery
of Prospectus; Subsequent Changes. During any period in which a Prospectus relating to the Placement Shares is required to be delivered
by the Agent under the Securities Act respecting the offer and sale of the Placement Shares (including in circumstances where such requirement
may be satisfied pursuant to Rule 172 under the Securities Act) (the “Prospectus Delivery Period”), the Company will
comply with all requirements imposed upon it by the Securities Act, as from time to time in force, and to file on or before their respective
due dates all reports and any definitive proxy or information statements required to be filed by the Company with the SEC pursuant to
Sections 13(a), 13(c), 14, 15(d), or any other provision of or under the Exchange Act. If the Company has omitted any information from
the Registration Statement pursuant to Rule 430A under the Securities Act, it will use its commercially reasonable efforts to comply
with the provisions of and make all requisite filings with the SEC pursuant to said Rule 430A and to notify the Agent promptly of all
such filings. If during the Prospectus Delivery Period any event occurs as a result of which the Prospectus as then amended or supplemented
would include an untrue statement of a material fact or omit to state a material fact necessary to make the statements therein, in the
light of the circumstances then existing, not misleading, or if during the Prospectus Delivery Period it is necessary to amend or supplement
the Registration Statement or Prospectus to comply with the Securities Act, the Company will promptly notify the Agent to suspend the
offering of Placement Shares during such period and the Company will promptly amend or supplement the Registration Statement or Prospectus
(at the expense of the Company) so as to correct such statement or omission or effect such compliance; provided, however, that the Company
may delay any amendment or supplement, if in the sole discretion of the Company, it is in the Company’s best interest to do so.
(d) Listing
of Placement Shares. During the Prospectus Delivery Period, the Company will use its commercially reasonable efforts to cause the
Placement Shares to be listed on the Exchange.
(e) Delivery
of Registration Statement and Prospectus. The Company will furnish to the Agent and its counsel (at the expense of the Company) copies
of the Registration Statement, the Prospectus (including all documents incorporated by reference therein) and all amendments and supplements
to the Registration Statement or Prospectus that are filed with the SEC during the Prospectus Delivery Period (including all documents
filed with the SEC during such period that are deemed to be incorporated by reference therein), in each case as soon as reasonably practicable
and in such quantities as the Agent may from time to time reasonably request and, at the Agent’s request, will also furnish copies
of the Prospectus to each exchange or market on which sales of the Placement Shares may be made; provided, however, that the Company
will not be required to furnish any document (other than the Prospectus) to the Agent to the extent such document is available on EDGAR.
(f) Earnings
Statement. The Company will make generally available to its security holders as soon as practicable, but in any event not later than
15 months after the end of the Company’s current fiscal quarter, an earnings statement covering a 12-month period that satisfies
the provisions of Section 11(a) and Rule 158 of the Securities Act. The Company’s compliance with the reporting requirements of
the Exchange Act shall be deemed to satisfy this Section 7(f).
(g) Use of Proceeds.
The Company will use the Net Proceeds as described in the Prospectus in the section entitled “Use of Proceeds.”
(h) Notice of
Other Sales. Without the prior written notice to the Agent, the Company will not, directly or indirectly, offer to sell, sell, contract
to sell, grant any option to sell or otherwise dispose of any Common Stock (other than the Placement Shares offered pursuant to this
Agreement) or securities convertible into or exchangeable for Common Stock, warrants or any rights to purchase or acquire, Common Stock
during the period beginning on the second Trading Day immediately prior to the date on which any Placement Notice is delivered to the
Agent hereunder and ending on the fifth Trading Day immediately following the final Settlement Date respecting Placement Shares sold
pursuant to such Placement Notice (or, if the Placement Notice has been terminated or suspended prior to the sale of all Placement Shares
covered by a Placement Notice, the date of such suspension or termination); and will not directly or indirectly in any other “at-the-market”
offering sell, contract to sell, grant any option to sell or otherwise dispose of any Common Stock (other than the Placement Shares offered
pursuant to this Agreement) or securities convertible into or exchangeable for Common Stock, warrants or any rights to purchase or acquire,
Common Stock prior to the termination of this Agreement; provided, however, that such restrictions will not be required
in connection with the Company’s issuance or sale of: (1) Common Stock, options to purchase Common Stock or Common Stock issuable
upon the exercise of options or restricted stock units granted pursuant to any employee or director stock option or benefits plan, stock
ownership plan or dividend reinvestment plan (but not Common Stock subject to a waiver to exceed plan limits in its dividend reinvestment
plan) of the Company whether now in effect or hereafter implemented; (2) Common Stock issuable upon conversion of securities or
in respect of dividends accruing thereon or the exercise of warrants, options or other rights in effect or outstanding, and disclosed
in filings by the Company available on EDGAR or otherwise in writing to the Agent; and (3) Common Stock, or securities convertible
into or exercisable for Common Stock, as consideration for mergers, acquisitions, other business combinations, licensing agreements or
strategic alliances, or offered and sold in a privately negotiated transaction to vendors, customers, or strategic partners and otherwise
conducted in a manner so as not to be integrated with the offering of Common Stock hereby.
(i) Change of
Circumstances. The Company will, at any time that a Placement Notice has been issued, advise the Agent promptly after it will have
received notice or obtained knowledge thereof, of any information or fact that would alter or affect in any material respect any opinion,
certificate, letter or other document required to be provided to the Agent pursuant to this Agreement.
(j) Due Diligence
Cooperation. The Company will cooperate with any reasonable due diligence review conducted by the Agent or its representatives in
connection with the transactions contemplated hereby, including providing information and making available documents and senior corporate
officers, during regular business hours and at the Company’s principal offices or such other location mutually agreed to by the
parties, as the Agent may reasonably request.
(k) Disclosure
of Shares Sold. The Company will disclose information regarding the sale of the Placement Shares in compliance with the requirements
of the Exchange Act.
(l) Representation
Dates; Certificate. Each time the Company:
(i) files the Prospectus
relating to the Placement Shares or amends or supplements (other than a prospectus supplement relating solely to an offering of securities
other than the Placement Shares) the Registration Statement or the Prospectus relating to the Placement Shares by means of a post-effective
amendment, sticker, or supplement but not by means of incorporation of documents by reference into the Registration Statement or the
Prospectus relating to the Placement Shares;
(ii) files an annual
report on Form 10-K under the Exchange Act (including any Form 10-K/A that contains restated financial statements);
(iii) files a quarterly
report on Form 10-Q under the Exchange Act; or
(iv) files a current
report on Form 8-K containing amended audited financial information (other than information “furnished” pursuant to Items
2.02 or 7.01 of Form 8-K or to provide disclosure pursuant to Item 8.01 of Form 8-K relating to the reclassification of certain
properties as discontinued operations in accordance with Statement of Financial Accounting Standards No. 144) under the Exchange Act
(each date of filing of one or more of the documents referred to in clauses (i) through (iv) will be a “Representation Date”);
the Company will
furnish the Agent (but in the case of clause (iv) above only if the Agent reasonably determines that the information contained in such
Form 8-K is material) with a certificate, in the form attached hereto as Exhibit A. The requirement to provide a certificate
under this Section 7(l) will be waived for any Representation Date occurring at a time at which no Placement Notice is pending, which
waiver will continue until the earlier to occur of the date the Company delivers a Placement Notice hereunder (which for such calendar
quarter will be considered a Representation Date) and the next occurring Representation Date. Notwithstanding the foregoing, if the Company
subsequently decides to sell Placement Shares following a Representation Date when the Company relied on such waiver and did not provide
the Agent with a certificate under this Section 7(l), then before the Company delivers the Placement Notice or the Agent sells any Placement
Shares, the Company will provide the Agent with a certificate, in the form attached hereto as Exhibit A, dated the date of the
Placement Notice.
(m) Legal Opinion.
On the date of the Registration Statement is effective and within five Trading Days of each Representation Date for which the Company
is obligated to deliver a certificate in the form attached hereto as Exhibit A for which no waiver is applicable, the Company will
cause to be furnished to the Agent written opinions of Sichenzia Ross Ference Carmel LLP
(“Company Counsel”), or other counsel reasonably satisfactory to the Agent, in form and substance reasonably satisfactory
to the Agent and its counsel; provided, however, the Company will be required to furnish to the Agent no more than one opinion
hereunder per calendar quarter; provided, further, that in lieu of such opinions for subsequent periodic filings under the Exchange
Act, Company Counsel may furnish the Agent with a letter (a “Reliance Letter”) to the effect that the Agent may rely
on a prior opinion delivered under this Section 7(m) to the same extent as if it were dated the date of such letter (except that
statements in such prior opinion will be deemed to relate to the Registration Statement and the Prospectus as amended or supplemented
as of the date of the Reliance Letter).
(n) Comfort
Letters. On the date of this Agreement and within five Trading Days of each Representation Date, other than pursuant to Section
7(l)(iii), for which the Company is obligated to deliver a certificate in the form attached hereto as Exhibit A for which
no waiver is applicable, the Company will cause its Accountants to furnish the Agent letters (the “Comfort Letters”),
dated the date the Comfort Letters are delivered, which will meet the requirements set forth in this Section 7(n). The Comfort
Letter from each of the Accountants will be in a form and substance reasonably satisfactory to the Agent: (i) confirming that they
are an independent public accounting firm within the meaning of the Securities Act and the PCAOB; (ii) stating, as of such date,
the conclusions and findings of such firm respecting the financial information and other matters ordinarily covered by accountants’
“comfort letters” to underwriters in connection with registered public offerings (the first such letter, the “Initial
Comfort Letter”); and (iii) updating the Initial Comfort Letter with any information that would have been included in
the Initial Comfort Letter had it been given on such date and modified as necessary to relate to the Registration Statement and the Prospectus,
as amended and supplemented to the date of such letter.
(o) Market Activities.
The Company will not, directly or indirectly: (i) take any action designed to cause or result in, or that constitutes or might reasonably
be expected to constitute, the stabilization or manipulation of the price of any security of the Company to facilitate the sale or resale
of Common Stock; or (ii) sell, bid for, or purchase Common Stock in violation of Regulation M, or pay anyone any compensation for
soliciting purchases of the Placement Shares other than the Agent.
(p) Investment
Company Act. The Company will conduct its affairs in such a manner so as to reasonably ensure that neither it nor any of its Subsidiaries
will be or become, at any time prior to the termination of this Agreement, required to register as an “investment company,”
as such term is defined in the Investment Company Act.
(q) No Offer
to Sell. Other than an Issuer Free Writing Prospectus approved in advance by the Company and the Agent in its capacity as agent hereunder,
neither the Agent nor the Company (including its agents and representatives, other than the Agent in its capacity as such) will make,
use, prepare, authorize, approve, or refer to any written communication (as defined in Rule 405 under the Securities Act), required to
be filed with the SEC, that constitutes an offer to sell or solicitation of an offer to buy Placement Shares hereunder.
(r) Sarbanes-Oxley
Act. The Company and the Subsidiaries shall comply with the applicable provisions of the Sarbanes-Oxley Act in all material respects.
8. Representations and
Covenants of the Agent. The Agent represents and warrants that it is duly registered as a broker-dealer under FINRA, the Exchange
Act, and the applicable statutes and regulations of each state in which the Placement Shares will be offered and sold, except such states
in which such Agent is exempt from registration or such registration is not otherwise required. The Agent will continue, for the term
of this Agreement, to be duly registered as a broker-dealer under FINRA, the Exchange Act, and the applicable statutes and regulations
of each state in which the Placement Shares will be offered and sold, except such states in which such Agent is exempt from registration
or such registration is not otherwise required, during the term of this Agreement. The Agent will comply with all applicable law and
regulations in connection with the Placement Shares, including Regulation M.
9. Payment of Expenses.
The Company will pay all expenses incident to the performance of its obligations under this Agreement, including: (i) the preparation,
filing, including any fees required by the SEC, and printing of the Registration Statement (including financial statements and exhibits)
as originally filed and of each amendment and supplement thereto and each Free Writing Prospectus, in such number as the Agent will reasonably
deem necessary; (ii) the printing and delivery to the Agent of this Agreement and such other documents as may reasonably be required
in connection with the offering, purchase, sale, issuance, or delivery of the Placement Shares; (iii) the preparation, issuance,
and delivery of the certificates, if any, for the Placement Shares to the Agent, including any stock or other transfer taxes and any
capital duties, stamp duties, or other duties or taxes payable upon the sale, issuance, or delivery of the Placement Shares to the Agent;
(iv) the fees and disbursements of the counsel, accountants, and other advisors to the Company; (v) the fees and expenses of
the transfer agent and registrar for the Common Stock; (vi) the filing fees incident to any review by FINRA of the terms of the
sale of the Placement Shares; (vii) the fees and expenses incurred in connection with the listing of the Placement Shares on the
Exchange; (viii) usual and customary transaction, ticket, and similar charges; (ix) $50,000 in due diligence fees and expenses of the
Agent, including Agent’s legal counsel initially and, thereafter, the reasonable fees and expenses of the Agent’s legal counsel
in connection with quarterly and annual bring-downs required hereunder up to a maximum amount of $2,500 for each such bring-down.
10. Conditions to the
Agent’s Obligations. The obligations of the Agent hereunder respecting a Placement will be subject to the continuing accuracy
and completeness of the representations and warranties made by the Company herein in all material respects (except for representations
and warranties qualified by materiality which are required to be accurate and complete in all respects), to the due performance by the
Company of its obligations hereunder in all material respects, to the completion by such Agent of a due diligence review satisfactory
to it in its reasonable judgment, and to the continuing satisfaction (or waiver by such Agent in its sole discretion) of the following
additional conditions:
(a) Registration
Statement Effective. The Company shall at all times maintain in effect the Registration Statement, which will be available for the
sale of all Placement Shares contemplated to be issued by any Placement Notice.
(b) No Material
Notices. None of the following events will have occurred and be continuing: (i) receipt by the Company of any request for additional
information from the SEC or any other federal or state governmental authority during the period of effectiveness of the Registration
Statement the response to which would require any post-effective amendments or supplements to the Registration Statement or the Prospectus
which have not, as of the time of such Placement, been so made; (ii) the issuance by the SEC or any other federal or state governmental
authority of any stop order suspending the effectiveness of the Registration Statement or the initiation of any proceedings for that
purpose; (iii) receipt by the Company of any notification respecting the suspension of the qualification or exemption from qualification
of any of the Placement Shares for sale in any jurisdiction or the initiation or threatening of any proceeding for such purpose; or (iv) the
occurrence of any event that requires the making of any changes in the Registration Statement, the Prospectus or documents so that, in
the case of the Registration Statement, it will not contain any materially untrue statement of a material fact or omit to state any material
fact required to be stated therein or necessary to make the statements therein not misleading and, that in the case of the Prospectus,
it will not contain any materially untrue statement of a material fact or omit to state any material fact required to be stated therein
or necessary to make the statements therein, in the light of the circumstances under which they were made, not misleading.
(c) No Misstatement
or Material Omission. Such Agent will not have advised the Company that the Registration Statement or Prospectus, or any amendment
or supplement thereto, contains an untrue statement of fact that in the Agent’s reasonable opinion is material, or omits to state
a fact that in the Agent’s opinion is material and is required to be stated therein or is necessary to make the statements therein
not misleading, which changes shall not, as of the time of the Placement, have been so made.
(d) Material
Changes. Except as contemplated in the Prospectus, or disclosed in the Company’s reports filed with the SEC, there will not
have been any material adverse change, on a consolidated basis, in the authorized capital stock of the Company or any Material Adverse
Effect, or any development that could reasonably be expected to cause a Material Adverse Effect.
(e) Legal Opinion.
Such Agent will have received the opinion of Company Counsel required to be delivered pursuant Section 7(m) on or before the date
on which such delivery of such opinion is required pursuant to Section 7(m).
(f) Comfort
Letters. Such Agent will have received the Comfort Letter required to be delivered pursuant Section 7(n) on or before the
date on which such delivery of such letter is required pursuant to Section 7(n).
(g) Representation
Certificate. Such Agent will have received the certificate required to be delivered pursuant to Section 7(l) on or before
the date on which delivery of such certificate is required pursuant to Section 7(l).
(h) No Suspension.
Trading in the Common Stock will not have been suspended on the Exchange and the Common Stock will not have been delisted from the Exchange.
(i) Other Materials.
On each date on which the Company is required to deliver a certificate pursuant to Section 7(l), the Company will have furnished
to such Agent such appropriate further information, certificates, and documents as such Agent may reasonably request and that are usually
and customarily furnished by an issuer of securities in connection with a securities offering. All such opinions, certificates, letters,
and other documents will be in compliance with the provisions hereof. The Company will furnish such Agent with such conformed copies
of such opinions, certificates, letters, and other documents as such Agent will reasonably request.
(j) Securities
Act Filings Made. All filings with the SEC required by Rule 424 under the Securities Act to have been filed prior to the issuance
of any Placement Notice hereunder will have been made within the applicable period prescribed for such filing by Rule 424.
(k) Approval
for Listing. To the extent required by continued listing rules of the Exchange, the Placement Shares will either have been approved
for listing on the Exchange, subject only to notice of issuance, or the Company will have filed an application for listing of the Placement
Shares on the Exchange at, or prior to, the issuance of any Placement Notice.
(l) No Termination
Event. No event will have occurred that would permit such Agent to terminate this Agreement pursuant to Section 13(a).
11. Indemnification and
Contribution.
(a) Company
Indemnification. The Company agrees to indemnify and hold harmless the Agent, its partners, members, directors, officers, employees,
and agents and each Person, if any, who controls any Agent within the meaning of Section 15 of the Securities Act or Section 20
of the Exchange Act as follows:
(i) against any and
all loss, liability, claim, damage, and expense whatsoever, as incurred, joint or several, arising out of or based upon any untrue statement
or alleged untrue statement of a material fact contained in the Registration Statement (or any amendment thereto), or the omission or
alleged omission therefrom of a material fact required to be stated therein or necessary to make the statements therein not misleading,
or arising out of any untrue statement or alleged untrue statement of a material fact included in any related Issuer Free Writing Prospectus
or the Prospectus (or any amendment or supplement thereto), or the omission or alleged omission therefrom of a material fact necessary
in order to make the statements therein, in the light of the circumstances under which they were made, not misleading;
(ii) against any
and all loss, liability, claim, damage and expense whatsoever, as incurred, joint or several, to the extent of the aggregate amount paid
in settlement of any litigation, or any investigation or proceeding by any governmental agency or body, commenced or threatened, or of
any claim whatsoever based upon any such untrue statement or omission, or any such alleged untrue statement or omission; provided that
any such settlement is effected with the written consent of the Company, which consent will not unreasonably be delayed or withheld;
and
(iii) against any
and all expense whatsoever, as incurred (including the reasonable fees and disbursements of counsel), reasonably incurred in investigating,
preparing or defending against any litigation, or any investigation or proceeding by any governmental agency or body, commenced or threatened,
or any claim whatsoever based upon any such untrue statement or omission, or any such alleged untrue statement or omission, to the extent
that any such expense is not paid under (i) or (ii) above;
provided,
however, that this indemnity agreement will not apply to any loss, liability, claim, damage, or expense to the extent arising
out of any untrue statement or omission or alleged untrue statement or omission made solely in reliance upon and in conformity with written
information furnished to the Company by the Agent expressly for use in the Registration Statement (or any amendment thereto), or in any
related Issuer Free Writing Prospectus or the Prospectus (or any amendment or supplement thereto).
(b) Agent Indemnification.
The Agent agrees, jointly and severally, to indemnify and hold harmless the Company and its directors and each officer of the Company
who signed the Registration Statement, and each Person, if any, that: (i) controls the Company within the meaning of Section 15
of the Securities Act or Section 20 of the Exchange Act; or (ii) is controlled by or is under common control with the Company against
any and all loss, liability, claim, damage, and expense described in the indemnity contained in Section 11(c), as incurred, but
only respecting untrue statements or omissions, or alleged untrue statements or omissions, made in the Registration Statement (or any
amendments thereto), the Prospectus (or any amendment or supplement thereto) or any Free Writing Prospectus in reliance upon and in conformity
with information furnished to the Company in writing by the Agent expressly for use therein.
(c) Procedure.
(i) Any party that
proposes to assert the right to be indemnified under this Section 11 will, promptly after receipt of notice of commencement of
any action against such party in respect of which a claim is to be made against an indemnifying party or parties under this Section
11, notify each such indemnifying party of the commencement of such action, enclosing a copy of all papers served, but the omission
so to notify such indemnifying party will not relieve the indemnifying party from: (1) any liability that it might have to any indemnified
party otherwise than under this Section 11; and (2) any liability that it may have to any indemnified party under the foregoing
provision of this Section 11 unless, and only to the extent that, such omission results in the forfeiture or material impairment
of substantive rights or defenses by the indemnifying party.
(ii) If any such
action is brought against any indemnified party and it notifies the indemnifying party of its commencement, the indemnifying party will
be entitled to participate in and, to the extent that it elects by delivering written notice to the indemnified party promptly after
receiving notice of the commencement of the action from the indemnified party, jointly with any other indemnifying party similarly notified,
to assume the defense of the action, with counsel reasonably satisfactory to the indemnified party, and after notice from the indemnifying
party to the indemnified party of its election to assume the defense, the indemnifying party will not be liable to the indemnified party
for any legal or other expenses except as provided below and except for the reasonable costs of investigation subsequently incurred by
the indemnified party in connection with the defense.
(iii) The indemnified
party will have the right to employ its own counsel in any such action, but the fees, expenses and other charges of such counsel will
be at the expense of such indemnified party unless: (1) the employment of counsel by the indemnified party has been authorized in
writing by the indemnifying party; (2) the indemnified party has reasonably concluded (based on advice of counsel) that there may
be legal defenses available to it or other indemnified parties that are different from or in addition to those available to the indemnifying
party; (3) a conflict or potential conflict exists (based on advice of counsel to the indemnified party) between the indemnified
party and the indemnifying party (in which case the indemnifying party will not have the right to direct the defense of such action on
behalf of the indemnified party); or (4) the indemnifying party has not in fact employed counsel to assume the defense of such action
within a reasonable time after receiving notice of the commencement of the action, in each of which cases the reasonable fees, disbursements
and other charges of counsel will be at the expense of the indemnifying party or parties.
(iv) It is understood
that the indemnifying party or parties will not, in connection with any proceeding or related proceedings in the same jurisdiction, be
liable for the reasonable fees, disbursements and other charges of more than one separate firm admitted to practice in such jurisdiction
at any one time for all such indemnified party or parties. All such fees, disbursements, and other charges will be reimbursed by the
indemnifying party promptly after the indemnifying party receives a written invoice relating to fees, disbursements and other charges
in reasonable detail.
(v) An indemnifying
party will not, in any event, be liable for any settlement of any action or claim effected without its written consent. No indemnifying
party will, without the prior written consent of each indemnified party, settle or compromise or consent to the entry of any judgment
in any pending or threatened claim, action, or proceeding relating to the matters contemplated by this Section 11 (whether or
not any indemnified party is a party thereto), unless such settlement, compromise or consent: (i) includes an unconditional release
of each indemnified party from all liability arising out of such litigation, investigation, proceeding, or claim; and (ii) does
not include a statement as to or an admission of fault, culpability, or a failure to act by or on behalf of any indemnified party.
(d) Contribution.
In order to provide for just and equitable contribution in circumstances in which the indemnification provided for in the foregoing paragraphs
of this Section 11 is applicable in accordance with its terms but for any reason is held to be unavailable from the Company or
the Agent, the Company and the Agent will contribute to the total losses, claims, liabilities, expenses, and damages (including any investigative,
legal and other expenses reasonably incurred in connection with, and any amount paid in settlement of, any action, suit or proceeding
or any claim asserted, but after deducting any contribution received by the Company from Persons other than the Agent, such as Persons
that control the Company within the meaning of the Securities Act, officers of the Company who signed the Registration Statement and
directors of the Company, who also may be liable for contribution) to which the Company and the Agent may be subject in such proportion
as is appropriate to reflect the relative benefits received by the Company, on the one hand, and the Agent, on the other hand. The relative
benefits received by the Company, on the one hand, and the Agent, on the other hand, will be deemed to be in the same proportion as the
total net proceeds from the sale of the Placement Shares (before deducting expenses) received by the Company bear to the total compensation
received by the Agent (before deducting expenses) from the sale of Placement Shares on behalf of the Company.
If, but only if, the allocation provided
by the foregoing sentence is not permitted by applicable law, the allocation of contribution will be made in such proportion as is appropriate
to reflect not only the relative benefits referred to in the foregoing sentence but also the relative fault of the Company, on the one
hand, and the Agent, on the other hand, respecting the statements or omission that resulted in such loss, claim, liability, expense or
damage, or action in respect thereof, as well as any other relevant equitable considerations respecting such offering. Such relative
fault will be determined by reference to, among other things, whether the untrue or alleged untrue statement of a material fact or omission
or alleged omission to state a material fact relates to information supplied by the Company or the Agent, the intent of the parties and
their relative knowledge, access to information and opportunity to correct or prevent such statement or omission. The Company and the
Agent agree that it would not be just and equitable if contributions pursuant to this Section 11(d) were to be determined by pro
rata allocation or by any other method of allocation that does not take into account the equitable considerations referred to herein.
The amount paid or payable by an indemnified party as a result of the loss, claim, liability, expense, or damage, or action in respect
thereof, referred to above in this Section 11(d) will be deemed to include, for the purpose of this Section 11(d), any
legal or other expenses reasonably incurred by such indemnified party in connection with investigating or defending any such action or
claim to the extent consistent with Section 11(c) hereof. Notwithstanding the foregoing provisions of this Section 11(d), the
Agent will not be required to contribute any amount in excess of the commissions received by it under this Agreement and no Person found
guilty of fraudulent misrepresentation (within the meaning of Section 11(f) of the Securities Act) will be entitled to contribution from
any Person who was not guilty of such fraudulent misrepresentation. For purposes of this Section 11(d), any Person who controls
a party to this Agreement within the meaning of the Securities Act, and any officers, directors, partners, employees or agents of the
Agent, will have the same rights to contribution as that party, and each officer and director of the Company who signed the Registration
Statement will have the same rights to contribution as the Company, subject in each case to the provisions hereof. Any party entitled
to contribution, promptly after receipt of notice of commencement of any action against such party in respect of which a claim for contribution
may be made under this Section 11(d), will notify any such party or parties from whom contribution may be sought, but the omission
to so notify will not relieve that party or parties from whom contribution may be sought from any other obligation it or they may have
under this Section 11(d) except to the extent that the failure to so notify such other party materially prejudiced the substantive
rights or defenses of the party from whom contribution is sought. Except for a settlement entered into pursuant to the last sentence
of Section 11(c) hereof, no party will be liable for contribution respecting any action or claim settled without its written consent
if such consent is required pursuant to Section 11(c) hereof.
12. Representations and
Agreements to Survive Delivery. The indemnity and contribution agreements contained in Section 11 of this Agreement and all
representations and warranties of the Company herein or in certificates delivered pursuant hereto will survive, as of their respective
dates, regardless of: (a) any investigation made by or on behalf of the Agent, any controlling Persons, or the Company (or any of
their respective officers, directors, or controlling Persons); (b) delivery and acceptance of the Placement Shares and payment therefor;
or (c) any termination of this Agreement.
13. Termination.
(a) The Agent may
terminate this Agreement, by written notice to the Company, as hereinafter specified at any time: (i) if there has been, since the
time of execution of this Agreement or since the date as of which information is given in the Prospectus, any Material Adverse Effect,
or any development has occurred that is reasonably likely to have a Material Adverse Effect or in the reasonable judgment of the Agent
makes it impractical or inadvisable to market the Placement Shares or to enforce contracts for the sale of the Placement Shares; (ii) if
there has occurred any material adverse change in the financial markets in the United States or the international financial markets,
any outbreak of hostilities or escalation thereof or other calamity or crisis or any change or development involving a prospective change
in national or international political, financial or economic conditions, in each case the effect of which is such as to make it, in
the judgment of the Agent, impracticable or inadvisable to market the Placement Shares or to enforce contracts for the sale of the Placement
Shares; (iii) if trading in the Common Stock has been suspended or limited by the SEC or the Exchange, or if trading generally on
the Exchange has been suspended or limited, or minimum prices for trading have been fixed on the Exchange; (iv) if a major disruption
of securities settlements or clearance services in the United States will have occurred and be continuing; or (v) if a banking moratorium
has been declared by either U.S. Federal or New York authorities. Any such termination will be without liability of any party to any
other party except that the provisions of Section 9 (Expenses), Section 11 (Indemnification), Section 12 (Survival
of Representations), Section 18 (Applicable Law; Waiver of Jury Trial), Section 19 (Consent to Jurisdiction), and Section
20 (Use of Information) hereof will remain in full force and effect notwithstanding such termination. If the Agent elects to terminate
this Agreement as provided in this Section 13(a), the Agent will provide the required notice as specified in Section 14
(Notices).
(b) (i) The Company
will have the right, by giving 10 days’ notice as hereinafter specified to terminate this Agreement in its sole discretion at any
time after the date of this Agreement.
(ii) If any Agent
decline any commercially reasonable placement notice pursuant to Section 2(a) of this Agreement, then the Company will have the
right to terminate this Agreement with respect to such Agent by giving written notice of termination to such Agent. Any such termination
will be effective immediately upon a delivery of a termination notice by the Company to such Agent.
Any termination
pursuant to Section 13(b) will be without liability of any party to any other party except that the provisions of Section 9,
Section 11, Section 12, Section 18, Section 19, and Section 20 hereof will remain in full force and
effect notwithstanding such termination.
(c) The Agent will
have the right, by giving 10 days’ notice as hereinafter specified to terminate this Agreement in their discretion at any time
after the date of this Agreement. Any such termination will be without liability of any party to any other party except that the provisions
of Section 9, Section 11, Section 12, Section 18, Section 19, and Section 20 hereof will remain
in full force and effect notwithstanding such termination.
(d) Unless earlier
terminated pursuant to this Section 13, this Agreement will automatically terminate upon the earlier to occur of: (i) the two-year
anniversary of the date hereof; or (ii) the issuance and sale of all of the Placement Shares through the Agent on the terms and
subject to the conditions set forth herein, except that, in either such case, the provisions of Section 9, Section 11,
Section 12, Section 18, Section 19, and Section 20 hereof will remain in full force and effect notwithstanding
such termination.
(e) This Agreement
will remain in full force and effect unless terminated pursuant to Sections 13(a), (b), (c), or (d) above
or otherwise by mutual agreement of the parties. Upon termination of this Agreement, the Company will not have any liability to any Agent
for any discount, commission, or other compensation respecting any Placement Shares not otherwise sold by an Agent under this Agreement.
(f) Any termination
of this Agreement will be effective on the date specified in such notice of termination; provided, however, that such termination
will not be effective until the close of business on the date of receipt of such notice by the Agent or the Company, as the case may
be; provided further, that the Agent shall suspend any ongoing Placement as soon as practicable following receipt of the notice of termination
(and in any event by the close of business on the date of receipt). If such termination will occur prior to the Settlement Date for any
sale of Placement Shares, such Placement Shares will settle in accordance with the provisions of this Agreement.
14. Notices.
(a) All notices
or other communications required or permitted to be given by any party to any other party pursuant to the terms of this Agreement will
be in writing, unless otherwise specified, and if sent to WallachBeth Capital LLC, will be delivered to:
WallachBeth Capital LLC
185 Hudson St
Jersey City, NJ 07302
Attn: Eric
Schweitzer
EMAIL
with a copy to:
TroyGould PC
1801 Century Park East, Suite 1600
Los Angeles, CA 90067
EMAIL
and if to the Company,
will be delivered to:
60 Degrees Pharmaceuticals,
Inc.
1025 Connecticut Avenue
NW Suite 1000
Washington, D.C. 20036
Attn: Geoffrey Dow,
CEO
EMAIL
with a copy to:
Sichenzia Ross Ference Carmel LLP
1185 Avenue of the Americas, 31st Floor
New York, New York 10036
Attn.:
EMAIL
Notice to any other Agent
shall be sent to its address set forth in Annex 1 hereto.
(b) Each such notice
or other communication will be deemed given: (i) when delivered personally on or before 4:30 p.m., New York City time, on a
Business Day or, if such day is not a Business Day, on the next succeeding Business Day; or (ii) on the next Business Day after
timely delivery to a nationally recognized overnight courier. For purposes of this Agreement, “Business Day” will
mean any day on which the Exchange and commercial banks in the City of New York are open for business. An Electronic Notice needs to
be sent for every communication between the parties to this Agreement.
(c) An electronic
communication (“Electronic Notice”) will be deemed written notice for purposes of this Section 14 if sent to
the electronic mail address set forth above or specified by the receiving party under separate cover. Electronic Notice will be deemed
received at the time the party sending Electronic Notice receives confirmation of receipt by the receiving party. Any party receiving
Electronic Notice may request and will be entitled to receive the notice on paper, in a nonelectronic form (“Nonelectronic Notice”),
which will be sent to the requesting party within 10 days of receipt of the written request for Nonelectronic Notice.
(d) Each party
to this Agreement may change such address for notices by sending to the parties to this Agreement written notice of a new address for
such purpose.
15. Successors and Assigns.
This Agreement will inure to the benefit of and be binding upon the Company and the Agent and their respective successors and the affiliates,
controlling persons, partners, members, officers, directors, employees, and agents referred to in Section 11 hereof. References
to any of the parties contained in this Agreement will be deemed to include the successors and permitted assigns of such party. Nothing
in this Agreement, express or implied, is intended to confer upon any party other than the parties hereto or their respective successors
and permitted assigns any rights, remedies, obligations or liabilities under or by reason of this Agreement, except as expressly provided
in this Agreement. Neither party may assign its rights or obligations under this Agreement without the prior written consent of the other
party.
16. Adjustments for Stock
Splits. The parties acknowledge and agree that all share-related numbers contained in this Agreement will be adjusted to take into
account any share consolidation, stock split, stock dividend, corporate domestication or similar event effected respecting the Placement
Shares.
17. Entire Agreement;
Amendment; Severability. This Agreement (including all schedules and exhibits attached hereto and Placement Notices issued pursuant
hereto), by and between the Company and the Agent constitutes the entire agreement of the parties respecting the subject matter hereof
and thereof and supersedes all other prior and contemporaneous agreements and undertakings, both written and oral, among the parties
hereto with regard to the subject matter hereof and thereof. Neither this Agreement nor any term hereof may be amended except pursuant
to a written instrument executed by the Company and the Agent. In the event that any one or more of the provisions contained herein,
or the application thereof in any circumstance, is held invalid, illegal, or unenforceable as written by a court of competent jurisdiction,
then such provision will be given full force and effect to the fullest possible extent that it is valid, legal, and enforceable, and
the remainder of the terms and provisions herein will be construed as if such invalid, illegal, or unenforceable term or provision was
not contained herein, but only to the extent that giving effect to such provision and the remainder of the terms and provisions hereof
will be in accordance with the intent of the parties as reflected in this Agreement.
18. APPLICABLE LAW;
WAIVER OF JURY TRIAL. THIS AGREEMENT WILL BE GOVERNED BY AND CONSTRUED IN ACCORDANCE WITH THE LAWS OF THE STATE OF NEW YORK WITHOUT
REGARD TO THE PRINCIPLES OF CONFLICTS OF LAWS. SPECIFIED TIMES OF DAY REFER TO NEW YORK CITY TIME. THE COMPANY HEREBY IRREVOCABLY WAIVES,
TO THE FULLEST EXTENT PERMITTED BY APPLICABLE LAW, ANY AND ALL RIGHT TO TRIAL BY JURY IN ANY LEGAL PROCEEDING ARISING OUT OF OR RELATING
TO THIS AGREEMENT OR THE TRANSACTIONS CONTEMPLATED HEREBY.
19. CONSENT TO JURISDICTION.
EACH PARTY HEREBY IRREVOCABLY SUBMITS TO THE NON-EXCLUSIVE JURISDICTION OF THE STATE AND FEDERAL COURTS SITTING IN THE CITY OF NEW YORK,
BOROUGH OF MANHATTAN, FOR THE ADJUDICATION OF ANY DISPUTE HEREUNDER OR IN CONNECTION WITH ANY TRANSACTION CONTEMPLATED HEREBY, AND HEREBY
IRREVOCABLY WAIVES, AND AGREES NOT TO ASSERT IN ANY SUIT, ACTION OR PROCEEDING, ANY CLAIM THAT IT IS NOT PERSONALLY SUBJECT TO THE JURISDICTION
OF ANY SUCH COURT, THAT SUCH SUIT, ACTION OR PROCEEDING IS BROUGHT IN AN INCONVENIENT FORUM OR THAT THE VENUE OF SUCH SUIT, ACTION OR
PROCEEDING IS IMPROPER. EACH PARTY HEREBY IRREVOCABLY WAIVES PERSONAL SERVICE OF PROCESS AND CONSENTS TO PROCESS BEING SERVED IN ANY
SUCH SUIT, ACTION OR PROCEEDING BY MAILING A COPY THEREOF (CERTIFIED OR REGISTERED MAIL, RETURN RECEIPT REQUESTED) TO SUCH PARTY AT THE
ADDRESS IN EFFECT FOR NOTICES TO IT UNDER THIS AGREEMENT AND AGREES THAT SUCH SERVICE WILL CONSTITUTE GOOD AND SUFFICIENT SERVICE OF
PROCESS AND NOTICE THEREOF. NOTHING CONTAINED HEREIN WILL BE DEEMED TO LIMIT IN ANY WAY ANY RIGHT TO SERVE PROCESS IN ANY MANNER PERMITTED
BY LAW.
20. Use of Information.
The Agent may not use or disclose any information gained in connection with this Agreement and the transactions contemplated by this
Agreement, including due diligence, for any purpose except in connection with entering into this Agreement and providing services as
distribution agent hereunder.
21. Counterparts.
This Agreement may be executed in two or more counterparts, each of which will be deemed an original, but all of which together will
constitute one and the same instrument. Delivery of an executed Agreement by one party to the other may be made by facsimile transmission.
22. Effect of Headings.
The section and exhibit headings herein are for convenience only and will not affect the construction hereof.
23. Permitted Free Writing
Prospectuses. The Company represents, warrants, and agrees that, unless it obtains the prior consent of the Agent, which consent
shall not be unreasonably withheld, conditioned or delayed, and each Agent represents, warrants and agrees that, unless it obtains the
prior consent of the Company, which consent shall not be unreasonably withheld, conditioned or delayed, it has not made and will not
make any offer relating to the Placement Shares that would constitute an Issuer Free Writing Prospectus, or that would otherwise constitute
a “free writing prospectus,” as defined in Rule 405, required to be filed with the SEC. Any such free writing prospectus
consented to by the Agent or by the Company, as the case may be, is hereinafter referred to as a “Permitted Free Writing Prospectus.”
The Company represents and warrants that it has treated and agrees that it will treat each Permitted Free Writing Prospectus as an “issuer
free writing prospectus,” as defined in Rule 433, and has complied and will comply with the requirements of Rule 433
applicable to any Permitted Free Writing Prospectus, including timely filing with the SEC where required, legending, and recordkeeping.
For the purposes of clarity, the parties hereto agree that all free writing prospectuses, if any, listed in Exhibit B hereto
are Permitted Free Writing Prospectuses.
24. Absence of Fiduciary
Relationship. The Company acknowledges and agrees that:
(a) Each Agent
is acting solely as agent in connection with the public offering of the Placement Shares and in connection with each transaction contemplated
by this Agreement and the process leading to such transactions, and no fiduciary or advisory relationship between the Company or any
of its respective affiliates, stockholders (or other equity holders), creditors or employees or any other party, on the one hand, and
such Agent, on the other hand, has been or will be created in respect of any of the transactions contemplated by this Agreement, irrespective
of whether or not such Agent has advised or is advising the Company on other matters, and such Agent has no obligation to the Company
respecting the transactions contemplated by this Agreement except the obligations expressly set forth in this Agreement;
(b) it is capable
of evaluating and understanding, and understands and accepts, the terms, risks and conditions of the transactions contemplated by this
Agreement;
(c) the Agent has
not provided any legal, accounting, regulatory or tax advice respecting the transactions contemplated by this Agreement and it has consulted
its own legal, accounting, regulatory and tax advisors to the extent it has deemed appropriate;
(d) it is aware
that the Agent and its affiliates are engaged in a broad range of transactions that may involve interests that differ from those of the
Company and the Agent has no obligation to disclose such interests and transactions to the Company by virtue of any fiduciary, advisory
or agency relationship or otherwise; provided that the Agent hereby agrees not to engage in any such transaction that would cause its
interests to be in direct conflict with the best interests of the Company; and
(e) it waives,
to the fullest extent permitted by law, any claims it may have against any Agent for breach of fiduciary duty or alleged breach of fiduciary
duty in connection with the sale of Placement Shares under this Agreement and agrees that no Agent will have any liability (whether direct
or indirect, in contract, tort or otherwise) to it in respect of such a fiduciary duty claim or to any Person asserting a fiduciary duty
claim on its behalf or in right of it or the Company, employees or creditors of Company, other than in respect of such Agent’s
obligations under this Agreement and to keep information provided by the Company to the Agent’s and the Agents’ counsel confidential
to the extent not otherwise publicly available.
25. Definitions. As
used in this Agreement, the following terms have the respective meanings set forth below:
(a) “Applicable
Time” means: (i) each Representation Date; and (ii) the time of each sale of any Placement Shares pursuant to this
Agreement.
(b) “Company’s
knowledge,” “knowledge of the Company” and similar expressions mean the actual knowledge of an executive
officer of the Company as of the date to which the expression relates.
(c) “Agent”
means, as of any given time, an Agent that the Company has designated as sales agent to sell Placement Shares pursuant to the terms of
this Agreement, which shall initially be WallachBeth Capital LLC.
(d) “Issuer
Free Writing Prospectus” means any “issuer free writing prospectus,” as defined in Rule 433, relating to the
Placement Shares.
(e) “Rule
164,” “Rule 172,” “Rule 405,” “Rule 415,” “Rule 424,”
“Rule 424(b),” “Rule 430A,” “Rule 430B,” and “Rule 433”
refer to such rules under the Securities Act Regulations.
(f) All references
in this Agreement to financial statements and schedules and other information that is “contained,” “included,”
or “stated” in the Registration Statement or the Prospectus (and all other references of like import) will be deemed to mean
and include all such financial statements and schedules and other information that is incorporated by reference in the Registration Statement
or the Prospectus, as the case may be.
(g) All references
in this Agreement to the Registration Statement, the Prospectus or any amendment or supplement to any of the foregoing will be deemed
to include the copy filed with the SEC pursuant to EDGAR; all references in this Agreement to any Issuer Free Writing Prospectus (other
than any Issuer Free Writing Prospectuses that, pursuant to Rule 433, are not required to be filed with the SEC) will be deemed
to include the copy thereof filed with the SEC pursuant to EDGAR; and all references in this Agreement to “supplements” to
the Prospectus will include any supplements, “wrappers,” or similar materials prepared in connection with any offering, sale,
or private placement of any Placement Shares by the Agent outside of the United States.
(h)
[Signature Page Follows]
If the foregoing correctly
sets forth the understanding between the Company and the Agent, please so indicate in the space provided below for that purpose, whereupon
this letter will constitute a binding agreement between the Company and the Agent.
| |
Very truly yours, |
| |
|
| |
60
Degrees Pharmaceuticals, Inc. |
| |
|
| |
By: |
/s/
Geoffrey Dow |
| |
Name: |
Geoffrey Dow |
| |
Title: |
Chief Executive Officer |
ACCEPTED as of the date first-above
written:
| |
WALLACHBETH CAPITAL, LLC |
| |
|
| |
By: |
/s/ Eric Schweitzer |
| |
Name: |
Eric Schweitzer |
| |
Title: |
Chief Compliance Officer |
SCHEDULE 1
FORM OF PLACEMENT NOTICE
| | From: | 60 Degrees Pharmaceuticals,
Inc. |
| | | |
| To: | WallachBeth Capital, LLC |
| | | Attention: Eric Schweitzer |
| Subject: | At-The-Market Issuance--Placement Notice |
Gentlemen:
Pursuant to the terms and
subject to the conditions contained in the At-The-Market Issuance Sales Agreement between 60 Degrees Pharmaceuticals, Inc., a Delaware
corporation (the “Company”) and the Agent party thereto (“WallachBeth Capital LLC”), dated June [●],
2024, the Company hereby requests that the Agent sell up to $____________ of the Company’s Common Stock, par value $0.0001 per
share, at a minimum market price of $_______ per share, during the period beginning [month, day, time] and ending [month, day, time],
not to exceed $______ in a single Trading Day.
SCHEDULE 2
Compensation
The Company will pay to the
Agent in cash, upon each sale of Placement Shares pursuant to this Agreement, an amount equal to up to Four percent (4.0%) of the gross
proceeds from each sale of Placement Shares.
SCHEDULE 3
Notice Parties
The Company:
WallachBeth Capital LLC
Eric Schweitzer [email]
SCHEDULE 4
Subsidiaries
Subsidiaries of 60 Degrees Pharmaceuticals,
Inc.
| Name of Subsidiary |
|
Jurisdiction of Incorporation |
| 60P Australia Pty Ltd |
|
Australia |
Exhibit A
Form of Representation Date
Certificate
This Officer’s Certificate
(this “Certificate”) is executed and delivered in connection with Section 7(l) of the At-The-Market Issuance Sales
Agreement (the “Agreement”), dated June [●], 2024, and entered into between 60 Degrees Pharmaceuticals, Inc.
(the “Company”) and the Agent party thereto. All capitalized terms used but not defined herein shall have the meanings
given to such terms in the Agreement
The undersigned, a duly appointed
and authorized officer of the Company, having made all necessary inquiries to establish the accuracy of the statements below and having
been authorized by the Company to execute this certificate, hereby certifies, in his capacity as such officer and not in his individual
capacity, as follows:
| 1. | As of the date of this Certificate, neither
the Registration Statement nor the Prospectus contain any untrue statement of a material
fact or omit to state a material fact required to be stated therein or necessary in order
to make the statements therein, in light of the circumstances under which they were made,
not misleading and (iii) no event has occurred as a result of which it is necessary to amend
or supplement the Prospectus in order to make the statements therein not untrue or misleading. |
| 2. | Each of the representations and warranties
of the Company contained in Section 6 of the Agreement: |
| (a) | to the extent such representations and
warranties are subject to qualifications and exceptions contained therein relating to materiality
or Material Adverse Effect, are true and correct on and as of the date hereof with the same
force and effect as if expressly made on and as of the date hereof, except for those representations
and warranties that speak solely as of a specific date, which were true and correct as of
such date; and |
| (b) | to the extent such representations and
warranties are not subject to any qualifications or exceptions, are true and correct in all
material respects as of the date hereof as if made on and as of the date hereof with the
same force and effect as if expressly made on and as of the date hereof, except for those
representations and warranties that speak solely as of a specific date, which were true and
correct as of such date. |
| 3. | Each of the covenants required to be
performed by the Company in the Agreement on or prior to the date of the Agreement, this
Representation Date, and each such other date as set forth in the Agreement, has been duly,
timely and fully performed in all material respects and each condition required to be complied
with by the Company on or prior to the date of the Agreement, this Representation Date, and
each such other date as set forth in the Agreement has been duly, timely and fully complied
with in all material respects. |
| 4. | Subsequent to the date of the most recent
financial statements included in or incorporated by reference into the Prospectus, there
has been no Material Adverse Effect. |
| 5. | No stop order suspending the effectiveness
of the Registration Statement or of any part thereof has been issued, and, to the Company’s
knowledge, no proceedings for that purpose have been instituted or are pending or threatened
by any securities or other governmental authority (including, without limitation, the SEC). |
The undersigned has executed
this Officer’s Certificate as of the date first written above.
| |
60 Degrees Pharmaceuticals, Inc. |
| |
|
| |
By: |
|
| |
Name: |
|
| |
Title: |
|
Exhibit 4.2
60 DEGREES PHARMACEUTICALS, INC.
as the Company
and
as Trustee
Senior Indenture
Dated as of
, 20
TABLE OF CONTENTS
| |
PAGE |
| ARTICLE 1 |
|
| DEFINITIONS AND INCORPORATION BY REFERENCE |
1 |
| |
|
| Section 1.01. Definitions |
1 |
| Section 1.02. Other Definitions |
5 |
| Section 1.03. Incorporation by Reference of Trust Indenture Act |
5 |
| Section 1.04. Rules of Construction |
6 |
| |
|
| ARTICLE 2 |
|
| THE SECURITIES |
6 |
| |
|
| Section 2.01. Form and Dating |
6 |
| Section 2.02. Execution And Authentication |
6 |
| Section 2.03. Amount Unlimited; Issuable in Series |
7 |
| Section 2.04. Denomination and Date of Securities; Payments of Interest |
9 |
| Section 2.05. Registrar and Paying Agent; Agents Generally |
10 |
| Section 2.06. Paying Agent to Hold Money in Trust |
10 |
| Section 2.07. Transfer and Exchange |
10 |
| Section 2.08. Replacement Securities |
12 |
| Section 2.09. Outstanding Securities |
13 |
| Section 2.10. Temporary Securities |
13 |
| Section 2.11. Cancellation |
14 |
| Section 2.12. CUSIP Numbers |
14 |
| Section 2.13. Defaulted Interest |
14 |
| Section 2.14. Series May Include Tranches |
14 |
| |
|
| ARTICLE 3 |
|
| REDEMPTION |
14 |
| |
|
| Section 3.01. Applicability of Article |
14 |
| Section 3.02. Notice of Redemption; Partial Redemptions |
15 |
| Section 3.03. Payment Of Securities Called For Redemption |
16 |
| Section 3.04. Exclusion of Certain Securities from Eligibility for Selection for Redemption |
16 |
| Section 3.05. Mandatory and Optional Sinking Funds |
16 |
| |
|
| ARTICLE 4 |
|
| COVENANTS |
18 |
| |
|
| Section 4.01. Payment of Securities |
18 |
| Section 4.02. Maintenance of Office or Agency |
18 |
| Section 4.03. Securityholders’ Lists |
19 |
| Section 4.04. Certificate to Trustee |
19 |
| Section 4.05. Reports by the Company |
19 |
| Section 4.06. Additional Amounts |
19 |
| ARTICLE 5 |
|
| SUCCESSOR CORPORATION |
20 |
| |
|
| Section 5.01. When Company May Merge, Etc |
20 |
| Section 5.02. Successor Substituted |
20 |
| |
|
| ARTICLE 6 |
|
| DEFAULT AND REMEDIES |
20 |
| |
|
| Section 6.01. Events of Default |
20 |
| Section 6.02. Acceleration |
21 |
| Section 6.03. Other Remedies |
21 |
| Section 6.04. Waiver of Past Defaults |
22 |
| Section 6.05. Control by Majority |
22 |
| Section 6.06. Limitation on Suits |
22 |
| Section 6.07. Rights of Holders to Receive Payment |
22 |
| Section 6.08. Collection Suit by Trustee |
23 |
| Section 6.09. Trustee May File Proofs of Claim |
23 |
| Section 6.10. Application of Proceeds |
23 |
| Section 6.11. Restoration of Rights and Remedies |
24 |
| Section 6.12. Undertaking for Costs |
24 |
| Section 6.13. Rights and Remedies Cumulative |
24 |
| Section 6.14. Delay or Omission not Waiver |
24 |
| |
|
| ARTICLE 7 |
|
| TRUSTEE |
24 |
| |
|
| Section 7.01. General |
24 |
| Section 7.02. Certain Rights of Trustee |
24 |
| Section 7.03. Individual Rights of Trustee |
25 |
| Section 7.04. Trustee’s Disclaimer |
25 |
| Section 7.05. Notice of Default |
26 |
| Section 7.06. Reports by Trustee to Holders |
26 |
| Section 7.07. Compensation and Indemnity |
26 |
| Section 7.08. Replacement of Trustee |
26 |
| Section 7.09. Acceptance of Appointment by Successor |
27 |
| Section 7.10. Successor Trustee By Merger, Etc |
28 |
| Section 7.11. Eligibility |
28 |
| Section 7.12. Money Held in Trust |
28 |
| |
|
| ARTICLE 8 |
|
| SATISFACTION AND DISCHARGE OF INDENTURE; UNCLAIMED MONEYS |
28 |
| |
|
| Section 8.01. Satisfaction and Discharge of Indenture |
28 |
| Section 8.02. Application by Trustee of Funds Deposited for Payment of Securities |
29 |
| Section 8.03. Repayment of Moneys Held by Paying Agent |
29 |
| Section 8.04. Return of Moneys Held by Trustee and Paying Agent Unclaimed for Two Years |
29 |
| Section 8.05. Defeasance and Discharge of Indenture |
29 |
| Section 8.06. Defeasance of Certain Obligations |
30 |
| Section 8.07. Reinstatement |
31 |
| Section 8.08. Indemnity |
31 |
| Section 8.09. Excess Funds |
31 |
| Section 8.10. Qualifying Trustee |
31 |
| |
|
| ARTICLE 9 |
|
| AMENDMENTS, SUPPLEMENTS AND WAIVERS |
31 |
| |
|
| Section 9.01. Without Consent of Holders |
31 |
| Section 9.02. With Consent of Holders |
32 |
| Section 9.03. Revocation and Effect of Consent |
33 |
| Section 9.04. Notation on or Exchange of Securities |
33 |
| Section 9.05. Trustee to Sign Amendments, Etc |
33 |
| Section 9.06. Conformity with Trust Indenture Act |
33 |
| |
|
| ARTICLE 10 |
|
| MISCELLANEOUS |
33 |
| |
|
| Section 10.01. Trust Indenture Act of 1939 |
33 |
| Section 10.02. Notices |
34 |
| Section 10.03. Certificate and Opinion as to Conditions Precedent |
34 |
| Section 10.04. Statements Required in Certificate or Opinion |
34 |
| Section 10.05. Evidence of Ownership |
35 |
| Section 10.06. Rules by Trustee, Paying Agent or Registrar |
35 |
| Section 10.07. Payment Date Other Than a Business Day |
35 |
| Section 10.08. Governing Law |
35 |
| Section 10.09. No Adverse Interpretation of Other Agreements |
35 |
| Section 10.10. Successors |
36 |
| Section 10.11. Duplicate Originals |
36 |
| Section 10.12. Separability |
36 |
| Section 10.13. Table of Contents, Headings, Etc |
36 |
| Section 10.14. Incorporators, Stockholders, Officers and Directors of Company Exempt from Individual Liability |
36 |
| Section 10.15. Judgment Currency |
36 |
SENIOR INDENTURE, dated as
of ,
20 , between 60 Degrees Pharmaceuticals, Inc., a Delaware corporation, as the Company, and
, as Trustee.
RECITALS OF THE COMPANY
WHEREAS, the Company has
duly authorized the issue from time to time of its senior debentures, notes or other evidences of indebtedness to be issued in one or
more series (the “Securities”) up to such principal amount or amounts as may from time to time be authorized in accordance
with the terms of this Indenture and to provide, among other things, for the authentication, delivery and administration thereof, the
Company has duly authorized the execution and delivery of this Indenture; and
WHEREAS, all things necessary
to make this Indenture a valid indenture and agreement according to its terms have been done;
NOW, THEREFORE:
In consideration of the premises
and the purchases of the Securities by the holders thereof, the Company and the Trustee mutually covenant and agree for the equal and
proportionate benefit of the respective holders from time to time of the Securities or of any and all series thereof and of the coupons,
if any, appertaining thereto as follows:
ARTICLE 1
DEFINITIONS
AND INCORPORATION BY REFERENCE
Section 1.01. Definitions.
“Affiliate”
of any Person means any other Person directly or indirectly controlling or controlled by or under direct or indirect common control with
such Person. For the purposes of this definition, “control” (including, with correlative meanings, the terms “controlling”,
“controlled by” and “under common control with”) when used with respect to any Person means the possession, directly
or indirectly, of the power to direct or cause the direction of the management and policies of such Person, whether through the ownership
of voting securities, by contract or otherwise.
“Agent”
means any Registrar, Paying Agent, transfer agent or Authenticating Agent.
“Authorized Newspaper”
means a newspaper (which, in the case of The City of New York, will, if practicable, be The Wall Street Journal (Eastern Edition) and
in the case of London, will, if practicable, be the Financial Times (London Edition) and published in an official language of the country
of publication customarily published at least once a day for at least five days in each calendar week and of general circulation in The
City of New York or London, as applicable. If it shall be impractical in the opinion of the Trustee to make any publication of any notice
required hereby in an Authorized Newspaper, any publication or other notice in lieu thereof which is made or given with the approval of
the Trustee shall constitute a sufficient publication of such notice.
“Board Resolution”
means one or more resolutions of the board of directors of the Company or any authorized committee thereof, certified by the secretary
or an assistant secretary to have been duly adopted and to be in full force and effect on the date of certification, and delivered to
the Trustee.
“Business Day”
means any day, other than a Saturday or Sunday, that is neither a legal holiday nor a day on which banking institutions are authorized
or required by law or regulation to close in The City of New York, with respect to any Security the interest on which is based on the
offered quotations in the interbank Eurodollar market for dollar deposits in London, or with respect to Securities denominated in a specified
currency other than United States dollars, in the principal financial center of the country of the specified currency.
“Capital Lease”
means, with respect to any Person, any lease of any property which, in conformity with GAAP, is required to be capitalized on the balance
sheet of such Person.
“Commission”
means the Securities and Exchange Commission, as from time to time constituted, created under the Exchange Act or, if at any time after
the execution of this instrument such Commission is not existing and performing the duties now assigned to it under the Trust Indenture
Act, then the body performing such duties at such time.
“Company”
means the party named as such in the first paragraph of this Indenture until a successor replaces it pursuant to Article 5 of this Indenture
and thereafter means the successor.
“Corporate Trust
Office” means the office of the Trustee at which the corporate trust business of the Trustee shall, at any particular time,
be administered, which office is, at the date of this Indenture, located at Attention: .
“Currency Agreement”
means, with respect to any Person, any foreign exchange contract, currency swap agreement or other similar agreement or arrangement designed
to protect such Person or any of its Subsidiaries against fluctuations in currency values to or under which such Person or any of its
Subsidiaries is a party or a beneficiary on the date hereof or becomes a party or a beneficiary thereafter.
“Debt”
means, with respect to any Person at any date of determination (without duplication), (i) all indebtedness of such Person for borrowed
money, (ii) all obligations of such Person evidenced by bonds, debentures, notes or other similar instruments, (iii) all obligations of
such Person in respect of letters of credit or bankers’ acceptance or other similar instruments (or reimbursement obligations with
respect thereto), (iv) all obligations of such Person to pay the deferred purchase price of property or services, except Trade Payables,
(v) all obligations of such Person as lessee under Capital Leases, (vi) all Debt of others secured by a Lien on any asset of such Person,
whether or not such Debt is assumed by such Person; provided that, for purposes of determining the amount of any Debt of the type described
in this clause, if recourse with respect to such Debt is limited to such asset, the amount of such Debt shall be limited to the lesser
of the fair market value of such asset or the amount of such Debt, (vii) all Debt of others Guaranteed by such Person to the extent such
Debt is Guaranteed by such Person, (viii) all redeemable stock valued at the greater of its voluntary or involuntary liquidation preference
plus accrued and unpaid dividends and (ix) to the extent not otherwise included in this definition, all obligations of such Person under
Currency Agreements and Interest Rate Agreements.
“Default”
means any event that is, or after notice or passage of time or both would be, an Event of Default.
“Depositary”
means, with respect to the Securities of any series issuable or issued in the form of one or more Registered Global Securities, the Person
designated as Depositary by the Company pursuant to Section 2.03 until a successor Depositary shall have become such pursuant to the applicable
provisions of this Indenture, and thereafter “Depositary” shall mean or include each Person who is then a Depositary
hereunder, and if at any time there is more than one such Person, “Depositary” as used with respect to the Securities
of any such series shall mean the Depositary with respect to the Registered Global Securities of that series.
“Exchange Act”
means the Securities Exchange Act of 1934, as amended.
“GAAP”
means generally accepted accounting principles in the U.S. as in effect as of the date hereof applied on a basis consistent with the principles,
methods, procedures and practices employed in the preparation of the Company’s audited financial statements, including, without
limitation, those set forth in the opinions and pronouncements of the Accounting Principles Board of the American Institute of Certified
Public Accountants and statements and pronouncements of the Financial Accounting Standards Board or in such other statements by such other
entity as is approved by a significant segment of the accounting profession.
“Guarantee”
means any obligation, contingent or otherwise, of any Person directly or indirectly guaranteeing any Debt or other obligation of any other
Person and, without limiting the generality of the foregoing, any obligation, direct or indirect, contingent or otherwise, of such Person
(i) to purchase or pay (or advance or supply funds for the purchase or payment of) such Debt or other obligation of such other Person
(whether arising by virtue of partnership arrangements, or by agreement to keep well, to purchase assets, goods, securities or services,
to take-or-pay, or to maintain financial statement conditions or otherwise) or (ii) entered into for purposes of assuring in any other
manner the obligee of such Debt or other obligation of the payment thereof or to protect such obligee against loss in respect thereof
(in whole or in part); provided that the term “Guarantee” shall not include endorsements for collection or deposit
in the ordinary course of business. The term “Guarantee” used as a verb has a corresponding meaning.
“Holder”
or “Securityholder” means the registered holder of any Security with respect to Registered Securities and the bearer
of any Unregistered Security or any coupon appertaining thereto, as the case may be.
“Indenture”
means this Indenture as originally executed and delivered or as it may be amended or supplemented from time to time by one or more indentures
supplemental to this Indenture entered into pursuant to the applicable provisions of this Indenture and shall include the forms and terms
of the Securities of each series established as contemplated pursuant to Sections 2.01 and 2.03.
“Interest Rate Agreement”
means, with respect to any Person, any interest rate protection agreement, interest rate future agreement, interest rate option agreement,
interest rate swap agreement, interest rate cap agreement, interest rate collar agreement, interest rate hedge agreement or other similar
agreement or arrangement designed to protect such Person or any of its Subsidiaries against fluctuations in interest rates to or under
which such Person or any of its Subsidiaries is a party or a beneficiary on the date hereof or becomes a party or a beneficiary thereafter.
“Lien”
means, with respect to any property, any mortgage, lien, pledge, charge, security interest or encumbrance of any kind in respect of such
property. For purposes of this Indenture, the Company shall be deemed to own subject to a Lien any property which it has acquired or holds
subject to the interest of a vendor or lessor under any conditional sale agreement, capital lease or other title retention agreement relating
to such property.
“Officer”
means, with respect to the Company, the president, the chief executive officer, the chief financial officer or the secretary.
“Officers’
Certificate” means a certificate signed in the name of the Company (i) by the president or chief executive officer and (ii)
by the chief financial officer or the secretary, and delivered to the Trustee. Each such certificate shall comply with Section 314 of
the Trust Indenture Act, if applicable, and include (except as otherwise expressly provided in this Indenture) the statements provided
in Section 10.04, if applicable.
“Opinion of Counsel”
means a written opinion signed by legal counsel, who may be an employee of or counsel to the Company, satisfactory to the Trustee. Each
such opinion shall comply with Section 314 of the Trust Indenture Act, if applicable, and include the statements provided in Section 10.04,
if and to the extent required thereby.
“Original issue
date” of any Security (or portion thereof) means the earlier of (a) the date of authentication of such Security or (b) the date
of any Security (or portion thereof) for which such Security was issued (directly or indirectly) on registration of transfer, exchange
or substitution.
“Original Issue
Discount Security” means any Security that provides for an amount less than the principal amount thereof to be due and payable
upon a declaration of acceleration of the maturity thereof pursuant to Section 6.02.
“Periodic Offering”
means an offering of Securities of a series from time to time, the specific terms of which Securities, including, without limitation,
the rate or rates of interest, if any, thereon, the stated maturity or maturities thereof and the redemption provisions, if any, with
respect thereto, are to be determined by the Company or its agents upon the issuance of such Securities.
“Person”
means an individual, a corporation, a partnership, a limited liability company, an association, a trust or any other entity or organization,
including a government or political subdivision or an agency or instrumentality thereof.
“Principal”
of a Security means the principal amount of, and, unless the context indicates otherwise, includes any premium payable on, the Security.
“Registered Global
Security” means a Security evidencing all or a part of a series of Registered Securities, issued to the Depositary for such
series in accordance with Section 2.02, and bearing the legend prescribed in Section 2.02.
“Registered Security”
means any Security registered on the Security Register (as defined in Section 2.05).
“Responsible Officer”
when used with respect to the Trustee, shall mean an officer of the Trustee in the Corporate Trust Office, having direct responsibility
for the administration of this Indenture, and also, with respect to a particular matter, any other officer to whom such matter is referred
because of such officer’s knowledge of and familiarity with the particular subject.
“Securities”
means any of the securities, as defined in the first paragraph of the recitals hereof, that are authenticated and delivered under this
Indenture and, unless the context indicates otherwise, shall include any coupon appertaining thereto.
“Securities Act”
means the Securities Act of 1933, as amended.
“Subsidiary”
means, with respect to any Person, any corporation, association or other business entity of which a majority of the capital stock or other
ownership interests having ordinary voting power to elect a majority of the board of directors or other persons performing similar functions
are at the time directly or indirectly owned by such Person.
“Trade Payables”
means, with respect to any Person, any accounts payable or any other indebtedness or monetary obligation to trade creditors created, assumed
or Guaranteed by such Person or any of its Subsidiaries arising in the ordinary course of business in connection with the acquisition
of goods or services.
“Trustee”
means the party named as such in the first paragraph of this Indenture until a successor replaces it in accordance with the provisions
of Article 7 and thereafter shall mean or include each Person who is then a Trustee hereunder, and if at any time there is more than one
such Person, “Trustee” as used with respect to the Securities of any series shall mean the Trustee with respect to Securities
of that series.
“Trust Indenture
Act” means the Trust Indenture Act of 1939, as amended (15 U.S. Code §§ 77aaa-77bbbb), as it may be amended from time
to time.
“Unregistered Security”
means any Security other than a Registered Security.
“U.S. Government
Obligations” means securities that are (i) direct obligations of the United States of America for the payment of which its full
faith and credit is pledged or (ii) obligations of an agency or instrumentality of the United States of America the payment of which is
unconditionally guaranteed as a full faith and credit obligation by the United States of America, and shall also include a depository
receipt issued by a bank or trust company as custodian with respect to any such U.S. Government Obligation or a specific payment of interest
on or principal of any such U.S. Government Obligation held by such custodian for the account of the holder of a depository receipt; provided
that (except as required by law) such custodian is not authorized to make any deduction from the amount payable to the holder of such
depository receipt from any amount received by the custodian in respect of the U.S. Government Obligation or the specific payment of interest
on or principal of the U.S. Government Obligation evidenced by such depository receipt.
“Yield to Maturity”
means, as the context may require, the yield to maturity (i) on a series of Securities or (ii) if the Securities of a series are issuable
from time to time, on a Security of such series, calculated at the time of issuance of such series in the case of clause (i) or at the
time of issuance of such Security of such series in the case of clause (ii), or, if applicable, at the most recent redetermination of
interest on such series or on such Security, and calculated in accordance with the constant interest method or such other accepted financial
practice as is specified in the terms of such Security.
Section 1.02. Other
Definitions. Each of the following terms is defined in the section set forth opposite such term:
| Term | |
Section | |
| Authenticating Agent | |
| 2.02 | |
| Cash Transaction | |
| 7.03 | |
| Dollars | |
| 4.02 | |
| Event of Default | |
| 6.01 | |
| Judgment Currency | |
| 10.15 | (a) |
| mandatory sinking fund payment | |
| 3.05 | |
| optional sinking fund payment | |
| 3.05 | |
| Paying Agent | |
| 2.05 | |
| record date | |
| 2.04 | |
| Registrar | |
| 2.05 | |
| Required Currency | |
| 10.15 | (a) |
| Security Register | |
| 2.05 | |
| self-liquidating paper | |
| 7.03 | |
| sinking fund payment date | |
| 3.05 | |
| tranche | |
| 2.14 | |
Section 1.03. Incorporation
by Reference of Trust Indenture Act. Whenever this Indenture refers to a provision of the Trust Indenture Act, the provision is incorporated
by reference in and made a part of this Indenture. The following terms used in this Indenture that are defined by the Trust Indenture
Act have the following meanings:
“indenture securities”
means the Securities;
“indenture security
holder” means a Holder or a Securityholder;
“indenture to be
qualified” means this Indenture;
“indenture trustee”
or “institutional trustee” means the Trustee; and
“obligor”
on the indenture securities means the Company or any other obligor on the Securities.
All other terms used in this
Indenture that are defined by the Trust Indenture Act, defined by reference in the Trust Indenture Act to another statute or defined by
a rule of the Commission and not otherwise defined herein have the meanings assigned to them therein.
Section 1.04. Rules
of Construction. Unless the context otherwise requires:
(a) an accounting term not
otherwise defined has the meaning assigned to it in accordance with GAAP;
(b) words in the singular
include the plural, and words in the plural include the singular;
(c) “herein,”
“hereof” and other words of similar import refer to this Indenture as a whole and not to any particular Article, Section or
other subdivision;
(d) all references to Sections
or Articles refer to Sections or Articles of this Indenture unless otherwise indicated; and
(e) use of masculine, feminine
or neuter pronouns should not be deemed a limitation, and the use of any such pronouns should be construed to include, where appropriate,
the other pronouns.
ARTICLE 2
THE SECURITIES
Section 2.01. Form
and Dating. The Securities of each series shall be substantially in such form or forms (not inconsistent with this Indenture) as shall
be established by or pursuant to one or more Board Resolutions or in one or more indentures supplemental hereto, in each case with such
appropriate insertions, omissions, substitutions and other variations as are required or permitted by this Indenture and may have imprinted
or otherwise reproduced thereon such legend or legends or endorsements, not inconsistent with the provisions of this Indenture, as may
be required to comply with any law, or with any rules of any securities exchange or usage, all as may be determined by the officers executing
such Securities as evidenced by their execution of the Securities. Unless otherwise so established, Unregistered Securities shall have
coupons attached.
Section 2.02. Execution
And Authentication. Two Officers shall execute the Securities and one Officer shall execute the coupons appertaining thereto for the
Company by facsimile or manual signature in the name and on behalf of the Company. The seal of the Company, if any, shall be reproduced
on the Securities. If an Officer whose signature is on a Security or coupon appertaining thereto no longer holds that office at the time
the Security is authenticated, the Security and such coupon shall nevertheless be valid.
The Trustee, at the expense
of the Company, may appoint an authenticating agent (the “Authenticating Agent”) to authenticate Securities. The Authenticating
Agent may authenticate Securities whenever the Trustee may do so. Each reference in this Indenture to authentication by the Trustee includes
authentication by such Authenticating Agent.
A Security and the coupons
appertaining thereto shall not be valid until the Trustee or Authenticating Agent manually signs the certificate of authentication on
the Security or on the Security to which such coupon appertains by an authorized officer. The signature shall be conclusive evidence that
the Security or the Security to which the coupon appertains has been authenticated under this Indenture.
At any time and from time
to time after the execution and delivery of this Indenture, the Company may deliver Securities of any series having attached thereto appropriate
coupons, if any, executed by the Company to the Trustee for authentication together with the applicable documents referred to below in
this Section, and the Trustee shall thereupon authenticate and deliver such Securities to or upon the written order of the Company. In
authenticating any Securities of a series, the Trustee shall be entitled to receive prior to the authentication of any Securities of such
series, and (subject to Article 7) shall be fully protected in relying upon, unless and until such documents have been superseded or revoked:
(a) any Board Resolution
and/or executed supplemental indenture referred to in Sections 2.01 and 2.03 by or pursuant to which the forms and terms of the Securities
of that series were established;
(b) an Officers’ Certificate
setting forth the form or forms and terms of the Securities, stating that the form or forms and terms of the Securities of such series
have been, or, in the case of a Periodic Offering, will be when established in accordance with such procedures as shall be referred to
therein, established in compliance with this Indenture; and
(c) an Opinion of Counsel
substantially to the effect that the form or forms and terms of the Securities of such series have been, or, in the case of a Periodic
Offering, will be when established in accordance with such procedures as shall be referred to therein, established in compliance with
this Indenture and that the supplemental indenture, to the extent applicable, and Securities have been duly authorized and, if executed
and authenticated in accordance with the provisions of the Indenture and delivered to and duly paid for by the purchasers thereof on the
date of such opinion, would be entitled to the benefits of the Indenture and would be valid and binding obligations of the Company, enforceable
against the Company in accordance with their respective terms, subject to bankruptcy, insolvency, reorganization, receivership, moratorium
and other similar laws affecting creditors’ rights generally, general principles of equity, and covering such other matters as shall
be specified therein and as shall be reasonably requested by the Trustee.
The Trustee shall not be
required to authenticate such Securities if the issue of such Securities pursuant to this Indenture will affect the Trustee’s own
rights, duties or immunities under the Securities and this Indenture or otherwise in a manner which is not reasonably acceptable to the
Trustee.
Notwithstanding the provisions
of Sections 2.01 and 2.02, if, in connection with a Periodic Offering, all Securities of a series are not to be originally issued at one
time, it shall not be necessary to deliver the Board Resolution otherwise required pursuant to Section 2.01 or the written order, Officers’
Certificate and Opinion of Counsel otherwise required pursuant to Section 2.02 at or prior to the authentication of each Security of such
series if such documents are delivered at or prior to the authentication upon original issuance of the first Security of such series to
be issued.
With respect to Securities
of a series offered in a Periodic Offering, the Trustee may rely, as to the authorization by the Company of any of such Securities, the
forms and terms thereof and the legality, validity, binding effect and enforceability thereof, upon the Opinion of Counsel and the other
documents delivered pursuant to Sections 2.01 and 2.02, as applicable, in connection with the first authentication of Securities of such
series.
If the Company shall establish
pursuant to Section 2.03 that the Securities of a series or a portion thereof are to be issued in the form of one or more Registered Global
Securities, then the Company shall execute and the Trustee shall authenticate and deliver one or more Registered Global Securities that
(i) shall represent and shall be denominated in an amount equal to the aggregate principal amount of all of the Securities of such series
issued in such form and not yet cancelled, (ii) shall be registered in the name of the Depositary for such Registered Global Security
or Securities or the nominee of such Depositary, (iii) shall be delivered by the Trustee to such Depositary or its custodian or pursuant
to such Depositary’s instructions and (iv) shall bear a legend substantially to the following effect: “Unless and until it
is exchanged in whole or in part for Securities in definitive registered form, this Security may not be transferred except as a whole
by the Depositary to the nominee of the Depositary or by a nominee of the Depositary to the Depositary or another nominee of the Depositary
or by the Depositary or any such nominee to a successor Depositary or a nominee of such successor Depositary.”
Section 2.03. Amount
Unlimited; Issuable in Series. The aggregate principal amount of Securities which may be authenticated and delivered under this Indenture
is unlimited.
The Securities may be
issued in one or more series. There shall be established in or pursuant to Board Resolution or one or more indentures supplemental
hereto, prior to the initial issuance of Securities of any series, subject to the last sentence of this Section 2.03,
(a) the
designation of the Securities of the series, which shall distinguish the Securities of the series from the Securities of all other
series;
(b) any limit upon the aggregate
principal amount of the Securities of the series that may be authenticated and delivered under this Indenture and any limitation on the
ability of the Company to increase such aggregate principal amount after the initial issuance of the Securities of that series (except
for Securities authenticated and delivered upon registration of transfer of, or in exchange for, or in lieu of, or upon redemption of,
other Securities of the series pursuant hereto);
(c) the date or dates on
which the principal of the Securities of the series is payable (which date or dates may be fixed or extendible);
(d) the rate or rates (which
may be fixed or variable) per annum at which the Securities of the series shall bear interest, if any, the date or dates from which such
interest shall accrue, on which such interest shall be payable and (in the case of Registered Securities) on which a record shall be taken
for the determination of Holders to whom interest is payable and/or the method by which such rate or rates or date or dates shall be determined;
(e) if other than as provided
in Section 4.02, the place or places where the principal of and any interest on Securities of the series shall be payable, any Registered
Securities of the series may be surrendered for exchange, notices, demands to or upon the Company in respect of the Securities of the
series and this Indenture may be served and notice to Holders may be published;
(f) the right, if any, of
the Company to redeem Securities of the series, in whole or in part, at its option and the period or periods within which, the price or
prices at which and any terms and conditions upon which Securities of the series may be so redeemed, pursuant to any sinking fund or otherwise;
(g) the obligation, if any,
of the Company to redeem, purchase or repay Securities of the series pursuant to any mandatory redemption, sinking fund or analogous provisions
or at the option of a Holder thereof and the price or prices at which and the period or periods within which and any of the terms and
conditions upon which Securities of the series shall be redeemed, purchased or repaid, in whole or in part, pursuant to such obligation;
(h) if other than denominations
of $1,000 and any integral multiple thereof, the denominations in which Securities of the series shall be issuable;
(i) if other than the principal
amount thereof, the portion of the principal amount of Securities of the series which shall be payable upon declaration of acceleration
of the maturity thereof;
(j) if other than the coin
or currency in which the Securities of the series are denominated, the coin or currency in which payment of the principal of or interest
on the Securities of the series shall be payable or if the amount of payments of principal of and/or interest on the Securities of the
series may be determined with reference to an index based on a coin or currency other than that in which the Securities of the series
are denominated, the manner in which such amounts shall be determined;
(k) if other than the currency
of the United States of America, the currency or currencies, including composite currencies, in which payment of the Principal of and
interest on the Securities of the series shall be payable, and the manner in which any such currencies shall be valued against other currencies
in which any other Securities shall be payable;
(l) whether the Securities
of the series or any portion thereof will be issuable as Registered Securities (and if so, whether such Securities will be issuable as
Registered Global Securities) or Unregistered Securities (with or without coupons) (and if so, whether such Securities will be issued
in temporary or permanent global form), or any combination of the foregoing, any restrictions applicable to the offer, sale or delivery
of Unregistered Securities or the payment of interest thereon and, if other than as provided herein, the terms upon which Unregistered
Securities of any series may be exchanged for Registered Securities of such series and vice versa;
(m) whether and under what
circumstances the Company will pay additional amounts on the Securities of the series held by a person who is not a U.S. person in respect
of any tax, assessment or governmental charge withheld or deducted and, if so, whether the Company will have the option to redeem such
Securities rather than pay such additional amounts;
(n) if the Securities of
the series are to be issuable in definitive form (whether upon original issue or upon exchange of a temporary Security of such series)
only upon receipt of certain certificates or other documents or satisfaction of other conditions, the form and terms of such certificates,
documents or conditions;
(o) any trustees, depositaries,
authenticating or paying agents, transfer agents or the registrar or any other agents with respect to the Securities of the series;
(p) provisions, if any, for
the defeasance of the Securities of the series (including provisions permitting defeasance of less than all Securities of the series),
which provisions may be in addition to, in substitution for, or in modification of (or any combination of the foregoing) the provisions
of Article 8;
(q) if the Securities of
the series are issuable in whole or in part as one or more Registered Global Securities or Unregistered Securities in global form, the
identity of the Depositary or common Depositary for such Registered Global Security or Securities or Unregistered Securities in global
form;
(r) any other Events of Default
or covenants with respect to the Securities of the series; and
(s) any other terms of the
Securities of the series (which terms shall not be inconsistent with the provisions of this Indenture).
All Securities of any one
series and coupons, if any, appertaining thereto shall be substantially identical, except in the case of Registered Securities as to date
and denomination, except in the case of any Periodic Offering and except as may otherwise be provided by or pursuant to the Board Resolution
referred to above or as set forth in any such indenture supplemental hereto. All Securities of any one series need not be issued at the
same time and may be issued from time to time, consistent with the terms of this Indenture, if so provided by or pursuant to such Board
Resolution or in any such indenture supplemental hereto and any forms and terms of Securities to be issued from time to time may be completed
and established from time to time prior to the issuance thereof by procedures described in such Board Resolution or supplemental indenture.
Unless otherwise expressly
provided with respect to a series of Securities, the aggregate principal amount of a series of Securities may be increased and additional
Securities of such series may be issued up to the maximum aggregate principal amount authorized with respect to such series as increased.
Section 2.04. Denomination
and Date of Securities; Payments of Interest. The Securities of each series shall be issuable as Registered Securities or Unregistered
Securities in denominations established as contemplated by Section 2.03 or, if not so established with respect to Securities of any series,
in denominations of $1,000 and any integral multiple thereof. The Securities of each series shall be numbered, lettered or otherwise distinguished
in such manner or in accordance with such plan as the Officers of the Company executing the same may determine, as evidenced by their
execution thereof.
Unless otherwise specified
with respect to a series of Securities, each Security shall be dated the date of its authentication. The Securities of each series shall
bear interest, if any, from the date, and such interest and shall be payable on the dates, established as contemplated by Section 2.03.
The person in whose name
any Registered Security of any series is registered at the close of business on any record date applicable to a particular series with
respect to any interest payment date for such series shall be entitled to receive the interest, if any, payable on such interest payment
date notwithstanding any transfer or exchange of such Registered Security subsequent to the record date and prior to such interest payment
date, except if and to the extent the Company shall default in the payment of the interest due on such interest payment date for such
series, in which case the provisions of Section 2.13 shall apply. The term “record date” as used with respect to any
interest payment date (except a date for payment of defaulted interest) for the Securities of any series shall mean the date specified
as such in the terms of the Registered Securities of such series established as contemplated by Section 2.03, or, if no such date is so
established, the fifteenth day next preceding such interest payment date, whether or not such record date is a Business Day.
Section 2.05. Registrar
and Paying Agent; Agents Generally. The Company shall maintain an office or agency where Securities may be presented for registration,
registration of transfer or for exchange (the “Registrar”) and an office or agency where Securities may be presented
for payment (the “Paying Agent”), which shall be in the Borough of Manhattan, The City of New York. The Company shall
cause the Registrar to keep a register of the Registered Securities and of their registration, transfer and exchange (the “Security
Register”). The Company may have one or more additional Paying Agents or transfer agents with respect to any series.
The Company shall enter into
an appropriate agency agreement with any Agent not a party to this Indenture. The agreement shall implement the provisions of this Indenture
and the Trust Indenture Act that relate to such Agent. The Company shall give prompt written notice to the Trustee of the name and address
of any Agent and any change in the name or address of an Agent. If the Company fails to maintain a Registrar or Paying Agent, the Trustee
shall act as such. The Company may remove any Agent upon written notice to such Agent and the Trustee; provided that
no such removal shall become effective until (i) the acceptance of an appointment by a successor Agent to such Agent as evidenced by an
appropriate agency agreement entered into by the Company and such successor Agent and delivered to the Trustee or (ii) notification to
the Trustee that the Trustee shall serve as such Agent until the appointment of a successor Agent in accordance with clause (i) of this
proviso. The Company or any affiliate of the Company may act as Paying Agent or Registrar; provided that neither the
Company nor an affiliate of the Company shall act as Paying Agent in connection with the defeasance of the Securities or the discharge
of this Indenture under Article 8.
The Company initially appoints
the Trustee as Registrar, Paying Agent and Authenticating Agent. If, at any time, the Trustee is not the Registrar, the Registrar shall
make available to the Trustee ten days prior to each interest payment date and at such other times as the Trustee may reasonably request
the names and addresses of the Holders as they appear in the Security Register.
Section 2.06. Paying
Agent to Hold Money in Trust. Not later than 10:00 a.m. New York City time on each due date or, in the case of Unregistered Securities,
10:00 a.m. New York City time on the Business Day prior to the due date, of any Principal or interest on any Securities, the Company shall
deposit with the Paying Agent money in immediately available funds sufficient to pay such Principal or interest. The Company shall require
each Paying Agent other than the Trustee to agree in writing that such Paying Agent shall hold in trust for the benefit of the Holders
of such Securities or the Trustee all money held by the Paying Agent for the payment of Principal of and interest on such Securities and
shall promptly notify the Trustee of any default by the Company in making any such payment. The Company at any time may require a Paying
Agent to pay all money held by it to the Trustee and account for any funds disbursed, and the Trustee may at any time during the continuance
of any payment default, upon written request to a Paying Agent, require such Paying Agent to pay all money held by it to the Trustee and
to account for any funds disbursed. Upon doing so, the Paying Agent shall have no further liability for the money so paid over to the
Trustee. If the Company or any affiliate of the Company acts as Paying Agent, it will, on or before each due date of any Principal of
or interest on any Securities, segregate and hold in a separate trust fund for the benefit of the Holders thereof a sum of money sufficient
to pay such Principal or interest so becoming due until such sum of money shall be paid to such Holders or otherwise disposed of as provided
in this Indenture, and will promptly notify the Trustee in writing of its action or failure to act as required by this Section.
Section 2.07. Transfer
and Exchange. Unregistered Securities (except for any temporary global Unregistered Securities) and coupons (except for coupons attached
to any temporary global Unregistered Securities) shall be transferable by delivery.
At the option of the Holder
thereof, Registered Securities of any series (other than a Registered Global Security, except as set forth below) may be exchanged for
a Registered Security or Registered Securities of such series and tenor having authorized denominations and an equal aggregate principal
amount, upon surrender of such Registered Securities to be exchanged at the agency of the Company that shall be maintained for such purpose
in accordance with Section 2.05 and upon payment, if the Company shall so require, of the charges hereinafter provided. If the Securities
of any series are issued in both registered and unregistered form, except as otherwise established pursuant to Section 2.03, at the option
of the Holder thereof, Unregistered Securities of any series may be exchanged for Registered Securities of such series and tenor having
authorized denominations and an equal aggregate principal amount, upon surrender of such Unregistered Securities to be exchanged at the
agency of the Company that shall be maintained for such purpose in accordance with Section 4.02, with, in the case of Unregistered Securities
that have coupons attached, all unmatured coupons and all matured coupons in default thereto appertaining, and upon payment, if the Company
shall so require, of the charges hereinafter provided. At the option of the Holder thereof, if Unregistered Securities of any series,
maturity date, interest rate and original issue date are issued in more than one authorized denomination, except as otherwise established
pursuant to Section 2.03, such Unregistered Securities may be exchanged for Unregistered Securities of such series and tenor having authorized
denominations and an equal aggregate principal amount, upon surrender of such Unregistered Securities to be exchanged at the agency of
the Company that shall be maintained for such purpose in accordance with Section 4.02, with, in the case of Unregistered Securities that
have coupons attached, all unmatured coupons and all matured coupons in default thereto appertaining, and upon payment, if the Company
shall so require, of the charges hereinafter provided. Registered Securities of any series may not be exchanged for Unregistered Securities
of such series. Whenever any Securities are so surrendered for exchange, the Company shall execute, and the Trustee shall authenticate
and deliver, the Securities which the Holder making the exchange is entitled to receive.
Upon surrender for registration
of transfer of any Registered Security of a series at the agency of the Company that shall be maintained for that purpose in accordance
with Section 2.05 and upon payment, if the Company shall so require, of the charges hereinafter provided, the Company shall execute, and
the Trustee shall authenticate and deliver, in the name of the designated transferee or transferees, one or more new Registered Securities
of the same series, of any authorized denominations and of like tenor and aggregate principal amount.
All Registered Securities
presented for registration of transfer, exchange, redemption or payment shall be duly endorsed by, or be accompanied by a written instrument
or instruments of transfer in form satisfactory to the Company and the Trustee duly executed by, the holder or his attorney duly authorized
in writing.
The Company may require payment
of a sum sufficient to cover any tax or other governmental charge that may be imposed in connection with any exchange or registration
of transfer of Securities. No service charge shall be made for any such transaction.
Notwithstanding any other
provision of this Section 2.07, unless and until it is exchanged in whole or in part for Securities in definitive registered form, a Registered
Global Security representing all or a portion of the Securities of a series may not be transferred except as a whole by the Depositary
for such series to a nominee of such Depositary or by a nominee of such Depositary to such Depositary or another nominee of such Depositary
or by such Depositary or any such nominee to a successor Depositary for such series or a nominee of such successor Depositary.
If at any time the Depositary
for any Registered Global Securities of any series notifies the Company that it is unwilling or unable to continue as Depositary for such
Registered Global Securities or if at any time the Depositary for such Registered Global Securities shall no longer be eligible under
applicable law, the Company shall appoint a successor Depositary eligible under applicable law with respect to such Registered Global
Securities. If a successor Depositary eligible under applicable law for such Registered Global Securities is not appointed by the Company
within 90 days after the Company receives such notice or becomes aware of such ineligibility, the Company will execute, and the Trustee,
upon receipt of the Company’s order for the authentication and delivery of definitive Registered Securities of such series and tenor,
will authenticate and deliver Registered Securities of such series and tenor, in any authorized denominations, in an aggregate principal
amount equal to the principal amount of such Registered Global Securities, in exchange for such Registered Global Securities.
The Company may at any time
and in its sole discretion and subject to the procedures of the Depositary determine that any Registered Global Securities of any series
shall no longer be maintained in global form. In such event the Company will execute, and the Trustee, upon receipt of the Company’s
order for the authentication and delivery of definitive Registered Securities of such series and tenor, will authenticate and deliver,
Registered Securities of such series and tenor in any authorized denominations, in an aggregate principal amount equal to the principal
amount of such Registered Global Securities, in exchange for such Registered Global Securities.
Any time the Registered Securities
of any series are not in the form of Registered Global Securities pursuant to the preceding two paragraphs, the Company agrees to supply
the Trustee with a reasonable supply of certificated Registered Securities without the legend required by Section 2.02 and the Trustee
agrees to hold such Registered Securities in safekeeping until authenticated and delivered pursuant to the terms of this Indenture.
If established by the Company
pursuant to Section 2.03 with respect to any Registered Global Security, the Depositary for such Registered Global Security may surrender
such Registered Global Security in exchange in whole or in part for Registered Securities of the same series and tenor in definitive registered
form on such terms as are acceptable to the Company and such Depositary. Thereupon, the Company shall execute, and the Trustee shall authenticate
and deliver, without service charge,
(a) to the Person specified
by such Depositary new Registered Securities of the same series and tenor, of any authorized denominations as requested by such Person,
in an aggregate principal amount equal to and in exchange for such Person’s beneficial interest in the Registered Global Security;
and
(b) to such Depositary a
new Registered Global Security in a denomination equal to the difference, if any, between the principal amount of the surrendered Registered
Global Security and the aggregate principal amount of Registered Securities authenticated and delivered pursuant to clause (a) above.
Registered Securities issued
in exchange for a Registered Global Security pursuant to this Section 2.07 shall be registered in such names and in such authorized denominations
as the Depositary for such Registered Global Security, pursuant to instructions from its direct or indirect participants or otherwise,
shall instruct the Trustee or an agent of the Company or the Trustee. The Trustee or such agent shall deliver such Securities to or as
directed by the Persons in whose names such Securities are so registered.
All Securities issued upon
any transfer or exchange of Securities shall be valid obligations of the Company, evidencing the same debt, and entitled to the same benefits
under this Indenture, as the Securities surrendered upon such transfer or exchange.
Notwithstanding anything
herein or in the forms or terms of any Securities to the contrary, none of the Company, the Trustee or any agent of the Company or the
Trustee shall be required to exchange any Unregistered Security for a Registered Security if such exchange would result in adverse Federal
income tax consequences to the Company (such as, for example, the inability of the Company to deduct from its income, as computed for
Federal income tax purposes, the interest payable on the Unregistered Securities) under then applicable United States Federal income tax
laws. The Trustee and any such agent shall be entitled to rely on an Officers’ Certificate or an Opinion of Counsel in determining
such result.
The Registrar shall not be
required (i) to issue, authenticate, register the transfer of or exchange Securities of any series for a period of 15 days before a selection
of such Securities to be redeemed or (ii) to register the transfer of or exchange any Security selected for redemption in whole or in
part.
Section 2.08. Replacement
Securities. If any mutilated Security or a Security with a mutilated coupon appertaining to it is surrendered to the Trustee, the
Company shall execute and the Trustee shall authenticate and deliver, in exchange for such mutilated Security or in exchange for the Security
to which a mutilated coupon appertains, a new Security of the same series and of like tenor and principal amount and bearing a number
not contemporaneously outstanding, with coupons corresponding to the coupons, if any, appertaining to such mutilated Security or to the
Security to which such mutilated coupon appertains.
If there shall be delivered
to the Company and the Trustee (i) evidence to their satisfaction of the destruction, loss or theft of any Security or coupon and (ii)
such security or indemnity as may be required by them to save each of them and any agent of any of them harmless, then, in the absence
of notice to the Company or the Trustee that such Security or coupon has been acquired by a bona fide purchaser, the Company shall execute
and the Trustee shall authenticate and deliver, in lieu of any such destroyed, lost or stolen Security or in exchange for the Security
to which a destroyed, lost or stolen coupon appertains (with all appurtenant coupons not destroyed, lost or stolen), a new Security of
the same series and of like tenor and principal amount and bearing a number not contemporaneously outstanding, with coupons corresponding
to the coupons, if any, appertaining to such destroyed, lost or stolen Security or to the Security to which such destroyed, lost or stolen
coupon appertains.
In case any such mutilated,
destroyed, lost or stolen Security or coupon has become or is about to become due and payable, the Company in its discretion may, instead
of issuing a new Security, pay such Security or coupon (without surrender thereof except in the case of a mutilated Security or coupon)
if the applicant for such payment shall furnish to the Company and the Trustee such security or indemnity as may be required by them to
save each of them and any agent of any of them harmless, and in the case of destruction, loss or theft, evidence satisfactory to the Company
and the Trustee and any agent of them of the destruction, loss or theft of such Security and the ownership thereof; provided,
however, that the Principal of and any interest on Unregistered Securities shall, except as otherwise provided in Section 4.02, be
payable only at an office or agency located outside the United States.
Upon the issuance of any
new Security under this Section, the Company may require payment of a sum sufficient to cover any tax or other governmental charge that
may be imposed in relation thereto and any other expenses (including the fees and expenses of the Trustee) connected therewith.
Every new Security of any
series, with its coupons, if any, issued pursuant to this Section in lieu of any destroyed, lost or stolen Security or in exchange for
any mutilated Security, or in exchange for a Security to which a mutilated, destroyed, lost or stolen coupon appertains, shall constitute
an original additional contractual obligation of the Company, whether or not the mutilated, destroyed, lost or stolen Security and its
coupons, if any, or the mutilated, destroyed, lost or stolen coupon shall be at any time enforceable by anyone, and any such new Security
and coupons, if any, shall be entitled to all the benefits of this Indenture equally and proportionately with any and all other Securities
of that series and their coupons, if any, duly issued hereunder.
The provisions of this Section
are exclusive and shall preclude (to the extent lawful) any other rights and remedies with respect to the replacement or payment of mutilated,
destroyed, lost or stolen Securities or coupons.
Section 2.09. Outstanding
Securities. Securities outstanding at any time are all Securities that have been authenticated by the Trustee except for those cancelled
by it, those delivered to it for cancellation, those described in this Section as not outstanding and those that have been defeased pursuant
to Section 8.05.
If a Security is replaced
pursuant to Section 2.08, it ceases to be outstanding unless and until the Trustee and the Company receive proof satisfactory to them
that the replaced Security is held by a holder in due course.
If the Paying Agent (other
than the Company or an affiliate of the Company) holds on the maturity date or any redemption date or date for repurchase of the Securities
money sufficient to pay Securities payable or to be redeemed or repurchased on that date, then on and after that date such Securities
cease to be outstanding and interest on them shall cease to accrue.
A Security does not cease
to be outstanding because the Company or one of its affiliates holds such Security, provided, however, that, in determining
whether the Holders of the requisite principal amount of the outstanding Securities have given any request, demand, authorization, direction,
notice, consent or waiver hereunder, Securities owned by the Company or any affiliate of the Company shall be disregarded and deemed not
to be outstanding, except that, in determining whether the Trustee shall be protected in relying upon any such request, demand, authorization,
direction, notice, consent or waiver, only Securities as to which a Responsible Officer of the Trustee has received written notice to
be so owned shall be so disregarded. Any Securities so owned which are pledged by the Company, or by any affiliate of the Company, as
security for loans or other obligations, otherwise than to another such affiliate of the Company, shall be deemed to be outstanding, if
the pledgee is entitled pursuant to the terms of its pledge agreement and is free to exercise in its or his discretion the right to vote
such securities, uncontrolled by the Company or by any such affiliate.
Section 2.10. Temporary
Securities. Until definitive Securities of any series are ready for delivery, the Company may prepare and the Trustee shall authenticate
temporary Securities of such series. Temporary Securities of any series shall be substantially in the form of definitive Securities of
such series but may have insertions, substitutions, omissions and other variations determined to be appropriate by the Officers executing
the temporary Securities, as evidenced by their execution of such temporary Securities. If temporary Securities of any series are issued,
the Company will cause definitive Securities of such series to be prepared without unreasonable delay. After the preparation of definitive
Securities of any series, the temporary Securities of such series shall be exchangeable for definitive Securities of such series and tenor
upon surrender of such temporary Securities at the office or agency of the Company designated for such purpose pursuant to Section 4.02,
without charge to the Holder. Upon surrender for cancellation of any one or more temporary Securities of any series the Company shall
execute and the Trustee shall authenticate and deliver in exchange therefor a like principal amount of definitive Securities of such series
and tenor and authorized denominations. Until so exchanged, the temporary Securities of any series shall be entitled to the same benefits
under this Indenture as definitive Securities of such series.
Section 2.11. Cancellation. The
Company at any time may deliver to the Trustee for cancellation any Securities previously authenticated and delivered hereunder which
the Company may have acquired in any manner whatsoever, and may deliver to the Trustee for cancellation any Securities previously authenticated
hereunder which the Company has not issued and sold. The Registrar, any transfer agent and the Paying Agent shall forward to the Trustee
any Securities surrendered to them for transfer, exchange or payment. The Trustee shall cancel and dispose of in accordance with its customary
procedures all Securities surrendered for transfer, exchange, payment or cancellation and shall deliver a certificate of disposition to
the Company. The Company may not issue new Securities to replace Securities it has paid in full or delivered to the Trustee for cancellation.
Section 2.12. CUSIP
Numbers. The Company in issuing the Securities may use “CUSIP” and “CINS” numbers (if then generally
in use), and the Trustee shall use CUSIP numbers or CINS numbers, as the case may be, in notices of redemption or exchange as a convenience
to Holders and no representation shall be made as to the correctness of such numbers either as printed on the Securities or as contained
in any notice of redemption or exchange.
Section 2.13. Defaulted
Interest. If the Company defaults in a payment of interest on the Registered Securities, it shall pay, or shall deposit with
the Paying Agent money in immediately available funds sufficient to pay, the defaulted interest plus (to the extent lawful) any interest
payable on the defaulted interest (as may be specified in the terms thereof, established pursuant to Section 2.03) to the Persons who
are Holders on a subsequent special record date, which shall mean the 15th day next preceding the date fixed by the Company for the payment
of defaulted interest, whether or not such day is a Business Day. At least 15 days before such special record date, the Company shall
mail to each Holder of such Registered Securities and to the Trustee a notice that states the special record date, the payment date and
the amount of defaulted interest to be paid.
Section 2.14. Series
May Include Tranches. A series of Securities may include one or more tranches (each a “tranche”) of Securities,
including Securities issued in a Periodic Offering. The Securities of different tranches may have one or more different terms, including
authentication dates and public offering prices, but all the Securities within each such tranche shall have identical terms, including
authentication date and public offering price. Notwithstanding any other provision of this Indenture, with respect to Sections 2.02 (other
than the fourth, sixth and seventh paragraphs thereof) through 2.04, 2.07, 2.08, 2.10, 3.01 through 3.05, 4.02, 6.01 through 6.14, 8.01
through 8.07, 9.02 and Section 10.07, if any series of Securities includes more than one tranche, all provisions of such sections applicable
to any series of Securities shall be deemed equally applicable to each tranche of any series of Securities in the same manner as though
originally designated a series unless otherwise provided with respect to such series or tranche pursuant to Section 2.03. In particular,
and without limiting the scope of the next preceding sentence, any of the provisions of such sections which provide for or permit action
to be taken with respect to a series of Securities shall also be deemed to provide for and permit such action to be taken instead only
with respect to Securities of one or more tranches within that series (and such provisions shall be deemed satisfied thereby), even if
no comparable action is taken with respect to Securities in the remaining tranches of that series.
ARTICLE 3
REDEMPTION
Section 3.01. Applicability
of Article. The provisions of this Article shall be applicable to the Securities of any series which are redeemable before their
maturity or to any sinking fund for the retirement of Securities of a series except as otherwise specified as contemplated by Section
2.03 for Securities of such series.
Section 3.02. Notice
of Redemption; Partial Redemptions. Notice of redemption to the Holders of Registered Securities of any series to be redeemed
as a whole or in part at the option of the Company shall be given by mailing notice of such redemption by first class mail, postage prepaid,
at least 30 days and not more than 60 days prior to the date fixed for redemption to such Holders of Registered Securities of such series
at their last addresses as they shall appear upon the registry books. Notice of redemption to the Holders of Unregistered Securities of
any series to be redeemed as a whole or in part who have filed their names and addresses with the Trustee pursuant to Section 313(c)(2)
of the Trust Indenture Act, shall be given by mailing notice of such redemption, by first class mail, postage prepaid, at least 30 days
and not more than 60 days prior to the date fixed for redemption, to such Holders at such addresses as were so furnished to the Trustee
(and, in the case of any such notice given by the Company, the Trustee shall make such information available to the Company for such purpose).
Notice of redemption to all other Holders of Unregistered Securities of any series to be redeemed as a whole or in part shall be published
in an Authorized Newspaper in The City of New York or with respect to any Security the interest on which is based on the offered quotations
in the interbank Eurodollar market for dollar deposits in an Authorized Newspaper in London, in each case, once in each of three successive
calendar weeks, the first publication to be not less than 30 days nor more than 60 days prior to the date fixed for redemption. Any notice
which is mailed or published in the manner herein provided shall be conclusively presumed to have been duly given, whether or not the
Holder receives the notice. Failure to give notice by mail, or any defect in the notice to the Holder of any Security of a series designated
for redemption as a whole or in part shall not affect the validity of the proceedings for the redemption of any other Security of such
series.
The notice of redemption
to each such Holder shall specify the principal amount of each Security of such series held by such Holder to be redeemed, the CUSIP numbers
of the Securities to be redeemed, the date fixed for redemption, the redemption price, or if not then ascertainable, the manner of calculation
thereof, the place or places of payment, that payment will be made upon presentation and surrender of such Securities and, in the case
of Securities with coupons attached thereto, of all coupons appertaining thereto maturing after the date fixed for redemption, that such
redemption is pursuant to the mandatory or optional sinking fund, or both, if such be the case, that interest accrued to the date fixed
for redemption will be paid as specified in such notice and that on and after said date interest thereon or on the portions thereof to
be redeemed will cease to accrue. In case any Security of a series is to be redeemed in part only, the notice of redemption shall state
the portion of the principal amount thereof to be redeemed and shall state that on and after the date fixed for redemption, upon surrender
of such Security, a new Security or Securities of such series and tenor in principal amount equal to the unredeemed portion thereof will
be issued.
The notice of redemption
of Securities of any series to be redeemed at the option of the Company shall be given by the Company or, at the Company’s request,
by the Trustee in the name and at the expense of the Company.
On or before 10:00 a.m. New
York City time on the redemption date or, in the case of Unregistered Securities, on or before 10:00 a.m. New York City time on the Business
Day prior to the redemption date specified in the notice of redemption given as provided in this Section, the Company will deposit with
the Trustee or with one or more Paying Agents (or, if the Company is acting as its own Paying Agent, set aside, segregate and hold in
trust as provided in Section 2.06) an amount of money sufficient to redeem on the redemption date all the Securities of such series so
called for redemption at the appropriate redemption price, together with accrued interest to the date fixed for redemption. If all of
the outstanding Securities of a series are to be redeemed, the Company will deliver to the Trustee at least 10 days prior to the last
date on which notice of redemption may be given to Holders pursuant to the first paragraph of this Section 3.02 (or such shorter period
as shall be acceptable to the Trustee) an Officers’ Certificate stating that all such Securities are to be redeemed. If less than
all the outstanding Securities of a series are to be redeemed, the Company will deliver to the Trustee at least 15 days prior to the last
date on which notice of redemption may be given to Holders pursuant to the first paragraph of this Section 3.02 (or such shorter period
as shall be acceptable to the Trustee) an Officers’ Certificate stating the aggregate principal amount of such Securities to be
redeemed. In the case of any redemption of Securities (a) prior to the expiration of any restriction on such redemption provided in the
terms of such Securities or elsewhere in this Indenture, or (b) pursuant to an election of the Company which is subject to a condition
specified in the terms of such Securities or elsewhere in this Indenture, the Company shall deliver to the Trustee, prior to the giving
of any notice of redemption to Holders pursuant to this Section, an Officers’ Certificate evidencing compliance with such restriction
or condition.
If less than all the Securities
of a series are to be redeemed, the Trustee shall select, pro rata, by lot or in such manner as it shall deem appropriate and fair, Securities
of such series to be redeemed in whole or in part. Securities may be redeemed in part in principal amounts equal to authorized denominations
for Securities of such series. The Trustee shall promptly notify the Company in writing of the Securities of such series selected for
redemption and, in the case of any Securities of such series selected for partial redemption, the principal amount thereof to be redeemed.
For all purposes of this Indenture, unless the context otherwise requires, all provisions relating to the redemption of Securities shall
relate, in the case of any Security redeemed or to be redeemed only in part, to the portion of the principal amount of such Security which
has been or is to be redeemed.
Section 3.03. Payment
Of Securities Called For Redemption. If notice of redemption has been given as above provided, the Securities or portions of Securities
specified in such notice shall become due and payable on the date and at the place stated in such notice at the applicable redemption
price, together with interest accrued to the date fixed for redemption, and on and after such date (unless the Company shall default in
the payment of such Securities at the redemption price, together with interest accrued to such date) interest on the Securities or portions
of Securities so called for redemption shall cease to accrue, and the unmatured coupons, if any, appertaining thereto shall be void and,
except as provided in Sections 7.12 and 8.02, such Securities shall cease from and after the date fixed for redemption to be entitled
to any benefit under this Indenture, and the Holders thereof shall have no right in respect of such Securities except the right to receive
the redemption price thereof and unpaid interest to the date fixed for redemption. On presentation and surrender of such Securities at
a place of payment specified in said notice, together with all coupons, if any, appertaining thereto maturing after the date fixed for
redemption, said Securities or the specified portions thereof shall be paid and redeemed by the Company at the applicable redemption price,
together with interest accrued thereon to the date fixed for redemption; provided that payment of interest becoming due on or prior to
the date fixed for redemption shall be payable in the case of Securities with coupons attached thereto, to the Holders of the coupons
for such interest upon surrender thereof, and in the case of Registered Securities, to the Holders of such Registered Securities registered
as such on the relevant record date subject to the terms and provisions of Sections 2.04 and 2.13 hereof.
If any Security called for
redemption shall not be so paid upon surrender thereof for redemption, the principal shall, until paid or duly provided for, bear interest
from the date fixed for redemption at the rate of interest or Yield to Maturity (in the case of an Original Issue Discount Security) borne
by such Security.
If any Security with coupons
attached thereto is surrendered for redemption and is not accompanied by all appurtenant coupons maturing after the date fixed for redemption,
the surrender of such missing coupon or coupons may be waived by the Company and the Trustee, if there be furnished to each of them such
security or indemnity as they may require to save each of them harmless.
Upon presentation of any
Security of any series redeemed in part only, the Company shall execute and the Trustee shall authenticate and deliver to or on the order
of the Holder thereof, at the expense of the Company, a new Security or Securities of such series and tenor (with any unmatured coupons
attached), of authorized denominations, in principal amount equal to the unredeemed portion of the Security so presented.
Section 3.04. Exclusion
of Certain Securities from Eligibility for Selection for Redemption. Securities shall be excluded from eligibility for selection for
redemption if they are identified by registration and certificate number in a written statement signed by an authorized officer of the
Company and delivered to the Trustee at least 40 days prior to the last date on which notice of redemption may be given as being owned
of record and beneficially by, and not pledged or hypothecated by, either (a) the Company or (b) an entity specifically identified in
such written statement as directly or indirectly controlling or controlled by or under direct or indirect common control with the Company.
Section 3.05. Mandatory
and Optional Sinking Funds. The minimum amount of any sinking fund payment provided for by the terms of Securities of any series
is herein referred to as a “mandatory sinking fund payment”, and any payment in excess of such minimum amount provided
for by the terms of the Securities of any series is herein referred to as an “optional sinking fund payment”. The date
on which a sinking fund payment is to be made is herein referred to as the “sinking fund payment date”.
In lieu of making all or
any part of any mandatory sinking fund payment with respect to any series of Securities in cash, the Company may at its option (a) deliver
to the Trustee Securities of such series theretofore purchased or otherwise acquired (except through a mandatory sinking fund payment)
by the Company or receive credit for Securities of such series (not previously so credited) theretofore purchased or otherwise acquired
(except as aforesaid) by the Company and delivered to the Trustee for cancellation pursuant to Section 2.11, (b) receive credit for optional
sinking fund payments (not previously so credited) made pursuant to this Section, or (c) receive credit for Securities of such series
(not previously so credited) redeemed by the Company at the option of the Company pursuant to the terms of such Securities or through
any optional sinking fund payment. Securities so delivered or credited shall be received or credited by the Trustee at the sinking fund
redemption price specified in such Securities.
On or before the sixtieth
day next preceding each sinking fund payment date for any series, or such shorter period as shall be acceptable to the Trustee, the Company
will deliver to the Trustee an Officers’ Certificate (a) specifying the portion of the mandatory sinking fund payment to be satisfied
by payment of cash and the portion to be satisfied by credit of specified Securities of such series and the basis for such credit, (b)
stating that none of the specified Securities of such series has theretofore been so credited, (c) stating that no defaults in the payment
of interest or Events of Default with respect to such series have occurred (which have not been waived or cured) and are continuing and
(d) stating whether or not the Company intends to exercise its right to make an optional sinking fund payment with respect to such series
and, if so, specifying the amount of such optional sinking fund payment which the Company intends to pay on or before the next succeeding
sinking fund payment date. Any Securities of such series to be credited and required to be delivered to the Trustee in order for the Company
to be entitled to credit therefor as aforesaid which have not theretofore been delivered to the Trustee shall be delivered for cancellation
pursuant to Section 2.11 to the Trustee with such Officers’ Certificate (or reasonably promptly thereafter if acceptable to the
Trustee). Such Officers’ Certificate shall be irrevocable and upon its receipt by the Trustee the Company shall become unconditionally
obligated to make all the cash payments or delivery of Securities therein referred to, if any, on or before the next succeeding sinking
fund payment date. Failure of the Company, on or before any such sixtieth day, to deliver such Officer’s Certificate and Securities
specified in this paragraph, if any, shall not constitute a default but shall constitute, on and as of such date, the irrevocable election
of the Company (i) that the mandatory sinking fund payment for such series due on the next succeeding sinking fund payment date shall
be paid entirely in cash without the option to deliver or credit Securities of such series in respect thereof and (ii) that the Company
will make no optional sinking fund payment with respect to such series as provided in this Section.
If the sinking fund payment
or payments (mandatory or optional or both) to be made in cash on the next succeeding sinking fund payment date plus any unused balance
of any preceding sinking fund payments made in cash shall exceed $50,000 (or a lesser sum if the Company shall so request with respect
to the Securities of any series), such cash shall be applied on the next succeeding sinking fund payment date to the redemption of Securities
of such series at the sinking fund redemption price thereof together with accrued interest thereon to the date fixed for redemption. If
such amount shall be $50,000 (or such lesser sum) or less and the Company makes no such request then it shall be carried over until a
sum in excess of $50,000 (or such lesser sum) is available. The Trustee shall select, in the manner provided in Section 3.02, for redemption
on such sinking fund payment date a sufficient principal amount of Securities of such series to absorb said cash, as nearly as may be,
and shall (if requested in writing by the Company) inform the Company of the serial numbers of the Securities of such series (or portions
thereof) so selected. Securities shall be excluded from eligibility for redemption under this Section if they are identified by registration
and certificate number in an Officers’ Certificate delivered to the Trustee at least 60 days prior to the sinking fund payment date
as being owned of record and beneficially by, and not pledged or hypothecated by either (a) the Company or (b) an entity specifically
identified in such Officers’ Certificate as directly or indirectly controlling or controlled by or under direct or indirect common
control with the Company. The Trustee, in the name and at the expense of the Company (or the Company, if it shall so request the Trustee
in writing) shall cause notice of redemption of the Securities of such series to be given in substantially the manner provided in Section
3.02 (and with the effect provided in Section 3.03) for the redemption of Securities of such series in part at the option of the Company.
The amount of any sinking fund payments not so applied or allocated to the redemption of Securities of such series shall be added to the
next cash sinking fund payment for such series and, together with such payment, shall be applied in accordance with the provisions of
this Section. Any and all sinking fund moneys held on the stated maturity date of the Securities of any particular series (or earlier,
if such maturity is accelerated), which are not held for the payment or redemption of particular Securities of such series shall be applied,
together with other moneys, if necessary, sufficient for the purpose, to the payment of the Principal of, and interest on, the Securities
of such series at maturity.
On or before 10:00 a.m. New
York City time on each sinking fund payment date or, in the case of Unregistered Securities, 10:00 a.m. New York City time on the Business
Day prior to the sinking fund payment date, the Company shall pay to the Trustee in cash or shall otherwise provide for the payment of
all interest accrued to the date fixed for redemption on Securities to be redeemed on the next following sinking fund payment date.
The Trustee shall not redeem
or cause to be redeemed any Securities of a series with sinking fund moneys or mail any notice of redemption of Securities of such series
by operation of the sinking fund during the continuance of a Default in payment of interest on such Securities or of any Event of Default
except that, where the mailing of notice of redemption of any Securities shall theretofore have been made, the Trustee shall redeem or
cause to be redeemed such Securities, provided that it shall have received from the Company a sum sufficient for such redemption. Except
as aforesaid, any moneys in the sinking fund for such series at the time when any such Default or Event of Default shall occur, and any
moneys thereafter paid into the sinking fund, shall, during the continuance of such Default or Event of Default, be deemed to have been
collected under Article 6 and held for the payment of all such Securities. In case such Event of Default shall have been waived as provided
in Section 6.04 or the Default cured on or before the sixtieth day preceding the sinking fund payment date in any year, such moneys shall
thereafter be applied on the next succeeding sinking fund payment date in accordance with this Section to the redemption of such Securities.
ARTICLE 4
COVENANTS
Section 4.01. Payment
of Securities. The Company shall pay the Principal of and interest on the Securities on the dates and in the manner provided
in the Securities and this Indenture. The interest on Securities with coupons attached (together with any additional amounts payable pursuant
to the terms of such Securities) shall be payable only upon presentation and surrender of the several coupons for such interest installments
as are evidenced thereby as they severally mature. The interest on any temporary Unregistered Securities (together with any additional
amounts payable pursuant to the terms of such Securities) shall be paid, as to the installments of interest evidenced by coupons attached
thereto, if any, only upon presentation and surrender thereof, and, as to the other installments of interest, if any, only upon presentation
of such Unregistered Securities for notation thereon of the payment of such interest. The interest on Registered Securities (together
with any additional amounts payable pursuant to the terms of such Securities) shall be payable only to the Holders thereof (subject to
Section 2.04) and at the option of the Company may be paid by mailing checks for such interest payable to or upon the written order of
such Holders at their last addresses as they appear on the Security Register of the Company.
Notwithstanding any provisions
of this Indenture and the Securities of any series to the contrary, if the Company and a Holder of any Registered Security so agree, payments
of interest on, and any portion of the Principal of, such Holder’s Registered Security (other than interest payable at maturity
or on any redemption or repayment date or the final payment of Principal on such Security) shall be made by the Paying Agent, upon receipt
from the Company of immediately available funds by 11:00 a.m., New York City time (or such other time as may be agreed to between the
Company and the Paying Agent), directly to the Holder of such Security (by Federal funds wire transfer or otherwise) if the Holder has
delivered written instructions to the Trustee 15 days prior to such payment date requesting that such payment will be so made and designating
the bank account to which such payments shall be so made and in the case of payments of Principal, surrenders the same to the Trustee
in exchange for a Security or Securities aggregating the same principal amount as the unredeemed principal amount of the Securities surrendered.
The Trustee shall be entitled to rely on the last instruction delivered by the Holder pursuant to this Section 4.01 unless a new instruction
is delivered 15 days prior to a payment date. The Company will indemnify and hold each of the Trustee and any Paying Agent harmless against
any loss, liability or expense (including attorneys’ fees) resulting from any act or omission to act on the part of the Company
or any such Holder in connection with any such agreement or from making any payment in accordance with any such agreement.
The Company shall pay interest
on overdue Principal, and interest on overdue installments of interest, to the extent lawful, at the rate per annum specified in the Securities.
Section 4.02. Maintenance
of Office or Agency. The Company will maintain in the Borough of Manhattan, The City of New York an office or agency where Securities
may be surrendered for registration of transfer or exchange or for presentation for payment and where notices and demands to or upon the
Company in respect of the Securities and this Indenture may be served. The Company hereby initially designates the Corporate Trust Office
of the Trustee, located in the Borough of Manhattan, The City of New York, as such office or agency of the Company. The Company will give
prompt written notice to the Trustee of the location, and any change in the location, of such office or agency. If at any time the Company
shall fail to maintain any such required office or agency or shall fail to furnish the Trustee with the address thereof, such presentations,
surrenders, notices and demands may be made or served at the address of the Trustee set forth in Section 10.02.
The Company will maintain
one or more agencies in a city or cities located outside the United States (including any city in which such an agency is required to
be maintained under the rules of any stock exchange on which the Securities of any series are listed) where the Unregistered Securities,
if any, of each series and coupons, if any, appertaining thereto may be presented for payment. No payment on any Unregistered Security
or coupon will be made upon presentation of such Unregistered Security or coupon at an agency of the Company within the United States
nor will any payment be made by transfer to an account in, or by mail to an address in, the United States unless, pursuant to applicable
United States laws and regulations then in effect, such payment can be made without adverse tax consequences to the Company. Notwithstanding
the foregoing, if full payment in United States Dollars (“Dollars”) at each agency maintained by the Company outside
the United States for payment on such Unregistered Securities or coupons appertaining thereto is illegal or effectively precluded by exchange
controls or other similar restrictions, payments in Dollars of Unregistered Securities of any series and coupons appertaining thereto
which are payable in Dollars may be made at an agency of the Company maintained in the Borough of Manhattan, The City of New York.
The Company may also from
time to time designate one or more other offices or agencies where the Securities of any series may be presented or surrendered for any
or all such purposes and may from time to time rescind such designations; provided that no such designation or rescission shall in any
manner relieve the Company of its obligation to maintain an office or agency in the Borough of Manhattan, The City of New York, for such
purposes. The Company will give prompt written notice to the Trustee of any such designation or rescission and of any change in the location
of any such other office or agency.
Section 4.03. Securityholders’
Lists. The Company will furnish or cause to be furnished to the Trustee a list in such form as the Trustee may reasonably require
of the names and addresses of the holders of the Securities pursuant to Section 312 of the Trust Indenture Act (a) semi-annually not more
than 15 days after each record date for the payment of semi-annual interest on the Securities, as hereinabove specified, as of such record
date, and (b) at such other times as the Trustee may request in writing, within thirty days after receipt by the Company of any such request
as of a date not more than 15 days prior to the time such information is furnished.
Section 4.04. Certificate
to Trustee. The Company will furnish to the Trustee annually, on or before a date not more than four months after the end of
its fiscal year (which, on the date hereof, is a calendar year), a brief certificate (which need not contain the statements required by
Section 10.04) from its principal executive, financial or accounting officer as to his or her knowledge of the compliance of the Company
with all conditions and covenants under this Indenture (such compliance to be determined without regard to any period of grace or requirement
of notice provided under this Indenture) which certificate shall comply with the requirements of the Trust Indenture Act.
Section 4.05. Reports
by the Company. The Company covenants to file with the Trustee, within 15 days after the Company is required to file the same
with the Commission, copies of the annual reports and of the information, documents, and other reports which the Company may be required
to file with the Commission pursuant to Section 13 or Section 15(d) of the Exchange Act.
Section 4.06. Additional
Amounts. If the Securities of a series provide for the payment of additional amounts, at least 10 days prior to the first interest
payment date with respect to that series of Securities and at least 10 days prior to each date of payment of Principal of or interest
on the Securities of that series if there has been a change with respect to the matters set forth in the below-mentioned Officers’
Certificate, the Company shall furnish to the Trustee and the principal paying agent, if other than the Trustee, an Officers’ Certificate
instructing the Trustee and such paying agent whether such payment of Principal of or interest on the Securities of that series shall
be made to Holders of the Securities of that series without withholding or deduction for or on account of any tax, assessment or other
governmental charge described in the Securities of that series. If any such withholding or deduction shall be required, then such Officers’
Certificate shall specify by country the amount, if any, required to be withheld or deducted on such payments to such Holders and shall
certify the fact that additional amounts will be payable and the amounts so payable to each Holder, and the Company shall pay to the Trustee
or such paying agent the additional amounts required to be paid by this Section. The Company covenants to indemnify the Trustee and any
paying agent for, and to hold them harmless against, any loss, liability or expense reasonably incurred without negligence or bad faith
on their part arising out of or in connection with actions taken or omitted by any of them in reliance on any Officers’ Certificate
furnished pursuant to this Section.
Whenever in this Indenture
there is mentioned, in any context, the payment of the Principal of or interest or any other amounts on, or in respect of, any Security
of any series, such mention shall be deemed to include mention of the payment of additional amounts provided by the terms of such series
established hereby or pursuant hereto to the extent that, in such context, additional amounts are, were or would be payable in respect
thereof pursuant to such terms, and express mention of the payment of additional amounts (if applicable) in any provision hereof shall
not be construed as excluding the payment of additional amounts in those provisions hereof where such express mention is not made.
ARTICLE 5
SUCCESSOR
CORPORATION
Section 5.01. When
Company May Merge, Etc. The Company shall not consolidate with, merge with or into, or sell, convey, transfer, lease or otherwise
dispose of all or substantially all of its property and assets (in one transaction or a series of related transactions) to, any Person
unless either (x) the Company shall be the continuing Person or (y) the Person (if other than the Company) formed by such consolidation
or into which the Company is merged or to which properties and assets of the Company shall be sold, conveyed, transferred or leased shall
be a corporation organized and validly existing under the laws of the United States of America or any jurisdiction thereof and shall expressly
assume, by a supplemental indenture, executed and delivered to the Trustee, all of the obligations of the Company on all of the Securities
and under this Indenture and the Company in the case of clauses (x) and (y) shall have delivered to the Trustee (A) an Opinion of Counsel
stating that such consolidation, merger or sale, conveyance, transfer or lease and such supplemental indenture (if any) complies with
this provision and that all conditions precedent provided for herein relating to such transaction have been complied with and that such
supplemental indenture (if any) constitutes the legal, valid and binding obligation of the Company and such successor enforceable against
such entity in accordance with its terms, subject to customary exceptions and (B) an Officers’ Certificate to the effect that immediately
after giving effect to such transaction, no Default shall have occurred and be continuing.
Section 5.02. Successor
Substituted. Upon any consolidation or merger, or any sale, conveyance, transfer, lease or other disposition of all or substantially
all of the property and assets of the Company in accordance with Section 5.01 of this Indenture, the successor Person formed by such consolidation
or into which the Company is merged or to which such sale, conveyance, transfer, lease or other disposition is made shall succeed to,
and be substituted for, and may exercise every right and power of, the Company under this Indenture with the same effect as if such successor
Person had been named as the Company herein and thereafter the predecessor Person, except in the case of a lease, shall be relieved of
all obligations and covenants under this Indenture and the Securities.
ARTICLE 6
DEFAULT
AND REMEDIES
Section 6.01. Events
of Default. An “Event of Default” shall occur with respect to the Securities of any series if:
(a) the Company defaults
in the payment of the Principal of any Security of such series when the same becomes due and payable at maturity, upon acceleration, redemption
or mandatory repurchase, including as a sinking fund installment, or otherwise;
(b) the Company defaults
in the payment of interest on any Security of such series when the same becomes due and payable, and such default continues for a period
of 30 days;
(c) the Company defaults
in the performance of or breaches any other covenant or agreement of the Company in this Indenture with respect to any Security of such
series or in the Securities of such series and such default or breach continues for a period of 30 consecutive days after written notice
to the Company by the Trustee or to the Company and the Trustee by the Holders of 25% or more in aggregate principal amount of the Securities
of all series affected thereby specifying such default or breach and requiring it to be remedied and stating that such notice is a “Notice
of Default” hereunder;
(d) a court having jurisdiction
in the premises shall enter a decree or order for relief in respect of the Company in an involuntary case under any applicable bankruptcy,
insolvency or other similar law now or hereafter in effect, or appointing a receiver, liquidator, assignee, custodian, trustee, sequestrator
(or similar official) of the Company or for any substantial part of its property or ordering the winding up or liquidation of its affairs,
and such decree or order shall remain unstayed and in effect for a period of 60 consecutive days;
(e) the Company (i) commences
a voluntary case under any applicable bankruptcy, insolvency or other similar law now or hereafter in effect, or consents to the entry
of an order for relief in an involuntary case under any such law, (ii) consents to the appointment of or taking possession by a receiver,
liquidator, assignee, custodian, trustee, sequestrator or similar official of the Company or for all or substantially all of the property
and assets of the Company or (iii) effects any general assignment for the benefit of creditors; or
(f) any other Event of Default
established pursuant to Section 2.03 with respect to the Securities of such series occurs.
Section 6.02. Acceleration.
(a) If an Event of Default other than as described in clauses (d) or (e) of Section 6.01 with respect to the Securities of any series
then outstanding occurs and is continuing, then, and in each and every such case, except for any series of Securities the principal of
which shall have already become due and payable, either the Trustee or the Holders of not less than 25% in aggregate principal amount
of the Securities of any such series then outstanding hereunder (each such series treated as a separate class) by notice in writing to
the Company (and to the Trustee if given by Securityholders), may declare the entire principal (or, if the Securities of any such series
are Original Issue Discount Securities, such portion of the principal amount as may be specified in the terms of such series established
pursuant to Section 2.03) of all Securities of such series, and the interest accrued thereon, if any, to be due and payable immediately,
and upon any such declaration the same shall become immediately due and payable.
(b) If an Event of Default
described in clause (d) or (e) of Section 6.01 occurs and is continuing, then the principal amount (or, if any Securities are Original
Issue Discount Securities, such portion of the principal as may be specified in the terms thereof established pursuant to Section 2.03)
of all the Securities then outstanding and interest accrued thereon, if any, shall be and become immediately due and payable, without
any notice or other action by any Holder or the Trustee, to the full extent permitted by applicable law.
The foregoing provisions,
however, are subject to the condition that if, at any time after the principal (or, if the Securities are Original Issue Discount Securities,
such portion of the principal as may be specified in the terms thereof established pursuant to Section 2.03) of the Securities of any
series (or of all the Securities, as the case may be) shall have been so declared or become due and payable, and before any judgment or
decree for the payment of the moneys due shall have been obtained or entered as hereinafter provided, the Company shall pay or shall deposit
with the Trustee a sum sufficient to pay all matured installments of interest upon all the Securities of each such series (or of all the
Securities, as the case may be) and the principal of any and all Securities of each such series (or of all the Securities, as the case
may be) which shall have become due otherwise than by acceleration (with interest upon such principal and, to the extent that payment
of such interest is enforceable under applicable law, on overdue installments of interest, at the same rate as the rate of interest or
Yield to Maturity (in the case of Original Issue Discount Securities) specified in the Securities of each such series to the date of such
payment or deposit) and such amount as shall be sufficient to cover all amounts owing the Trustee under Section 7.07, and if any and all
Events of Default under the Indenture, other than the non-payment of the principal of Securities which shall have become due by acceleration,
shall have been cured, waived or otherwise remedied as provided herein, then and in every such case the Holders of a majority in aggregate
principal amount of all the then outstanding Securities of all such series that have been accelerated (voting as a single class), by written
notice to the Company and to the Trustee, may waive all defaults with respect to all such series (or with respect to all the Securities,
as the case may be) and rescind and annul such declaration and its consequences, but no such waiver or rescission and annulment shall
extend to or shall affect any subsequent default or shall impair any right consequent thereon.
For all purposes under this
Indenture, if a portion of the principal of any Original Issue Discount Securities shall have been accelerated and declared or become
due and payable pursuant to the provisions hereof, then, from and after such declaration, unless such declaration has been rescinded and
annulled, the principal amount of such Original Issue Discount Securities shall be deemed, for all purposes hereunder, to be such portion
of the principal thereof as shall be due and payable as a result of such acceleration, and payment of such portion of the principal thereof
as shall be due and payable as a result of such acceleration, together with interest, if any, thereon and all other amounts owing thereunder,
shall constitute payment in full of such Original Issue Discount Securities.
Section 6.03. Other
Remedies. If a payment default or an Event of Default with respect to the Securities of any series occurs and is continuing, the Trustee
may pursue, in its own name or as trustee of an express trust, any available remedy by proceeding at law or in equity to collect the payment
of Principal of and interest on the Securities of such series or to enforce the performance of any provision of the Securities of such
series or this Indenture.
The Trustee may maintain
a proceeding even if it does not possess any of the Securities or does not produce any of them in the proceeding.
Section 6.04. Waiver
of Past Defaults. Subject to Sections 6.02, 6.07 and 9.02, the Holders of at least a majority in principal amount (or, if the Securities
are Original Issue Discount Securities, such portion of the principal as is then accelerable under Section 6.02) of the outstanding Securities
of all series affected (voting as a single class), by notice to the Trustee, may waive an existing Default or Event of Default with respect
to the Securities of such series and its consequences, except a Default in the payment of Principal of or interest on any Security as
specified in clauses (a) or (b) of Section 6.01 or in respect of a covenant or provision of this Indenture which cannot be modified or
amended without the consent of the Holder of each outstanding Security affected. Upon any such waiver, such Default shall cease to exist,
and any Event of Default with respect to the Securities of such series arising therefrom shall be deemed to have been cured, for every
purpose of this Indenture; but no such waiver shall extend to any subsequent or other Default or Event of Default or impair any right
consequent thereto.
Section 6.05. Control
by Majority. Subject to Sections 7.01 and 7.02(e), the Holders of at least a majority in aggregate principal amount (or, if any Securities
are Original Issue Discount Securities, such portion of the principal as is then accelerable under Section 6.02) of the outstanding Securities
of all series affected (voting as a single class) may direct the time, method and place of conducting any proceeding for any remedy available
to the Trustee or exercising any trust or power conferred on the Trustee with respect to the Securities of such series by this Indenture;
provided, that the Trustee may refuse to follow any direction that conflicts with law or this Indenture, that may involve the Trustee
in personal liability or that the Trustee determines in good faith may be unduly prejudicial to the rights of Holders not joining in the
giving of such direction; and provided further, that the Trustee may take any other action it deems proper that is not inconsistent with
any directions received from Holders of Securities pursuant to this Section 6.05.
Section 6.06. Limitation
on Suits. No Holder of any Security of any series may institute any proceeding, judicial or otherwise, with respect to this Indenture
or the Securities of such series, or for the appointment of a receiver or trustee, or for any other remedy hereunder, unless:
(a) such Holder has previously
given to the Trustee written notice of a continuing Event of Default with respect to the Securities of such series;
(b) the Holders of at least
25% in aggregate principal amount of outstanding Securities of all such series affected shall have made written request to the Trustee
to institute proceedings in respect of such Event of Default in its own name as Trustee hereunder;
(c) such Holder or Holders
have offered to the Trustee indemnity reasonably satisfactory to the Trustee against any costs, liabilities or expenses to be incurred
in compliance with such request;
(d) the Trustee for 60 days
after its receipt of such notice, request and offer of indemnity has failed to institute any such proceeding; and
(e) during such 60-day period,
the Holders of a majority in aggregate principal amount of the outstanding Securities of all such affected series have not given the Trustee
a direction that is inconsistent with such written request.
A Holder may not use this
Indenture to prejudice the rights of another Holder or to obtain a preference or priority over such other Holder.
Section 6.07. Rights
of Holders to Receive Payment. Notwithstanding any other provision of this Indenture, the right of any Holder of a Security to receive
payment of Principal of or interest, if any, on such Holder’s Security on or after the respective due dates expressed on such Security,
or to bring suit for the enforcement of any such payment on or after such respective dates, shall not be impaired or affected without
the consent of such Holder.
Section 6.08. Collection
Suit by Trustee. If an Event of Default with respect to the Securities of any series in payment of Principal or interest specified
in clause (a) or (b) of Section 6.01 occurs and is continuing, the Trustee may recover judgment in its own name and as trustee of an express
trust against the Company for the whole amount (or such portion thereof as specified in the terms established pursuant to Section 2.03
of Original Issue Discount Securities) of Principal of, and accrued interest remaining unpaid on, together with interest on overdue Principal
of, and, to the extent that payment of such interest is lawful, interest on overdue installments of interest on, the Securities of such
series, in each case at the rate or Yield to Maturity (in the case of Original Issue Discount Securities) specified in such Securities,
and such further amount as shall be sufficient to cover all amounts owing the Trustee under Section 7.07.
Section 6.09. Trustee
May File Proofs of Claim. The Trustee may file such proofs of claim and other papers or documents as may be necessary or advisable
in order to have the claims of the Trustee (including any claim for amounts due the Trustee under Section 7.07) and the Holders allowed
in any judicial proceedings relative to the Company (or any other obligor on the Securities), its creditors or its property and shall
be entitled and empowered to collect and receive any moneys, securities or other property payable or deliverable upon conversion or exchange
of the Securities or upon any such claims and to distribute the same, and any custodian, receiver, assignee, trustee, liquidator, sequestrator
or other similar official in any such judicial proceeding is hereby authorized by each Holder to make such payments to the Trustee and,
in the event that the Trustee shall consent to the making of such payments directly to the Holders, to pay to the Trustee any amount due
to it under Section 7.07. Nothing herein contained shall be deemed to empower the Trustee to authorize or consent to, or accept or adopt
on behalf of any Holder, any plan of reorganization, arrangement, adjustment or composition affecting the Securities or the rights of
any Holder thereof, or to authorize the Trustee to vote in respect of the claim of any Holder in any such proceeding.
Section 6.10. Application
of Proceeds. Any moneys collected by the Trustee pursuant to this Article in respect of the Securities of any series shall be applied
in the following order at the date or dates fixed by the Trustee and, in case of the distribution of such moneys on account of Principal
or interest, upon presentation of the several Securities and coupons appertaining to such Securities in respect of which moneys have been
collected and noting thereon the payment, or issuing Securities of such series and tenor in reduced principal amounts in exchange for
the presented Securities of such series and tenor if only partially paid, or upon surrender thereof if fully paid:
FIRST: To
the payment of all amounts due the Trustee under Section 7.07 applicable to the Securities of such series in respect of which moneys have
been collected;
SECOND:
In case the principal of the Securities of such series in respect of which moneys have been collected shall not have become and be then
due and payable, to the payment of interest on the Securities of such series in default in the order of the maturity of the installments
of such interest, with interest (to the extent that such interest has been collected by the Trustee) upon the overdue installments of
interest at the same rate as the rate of interest or Yield to Maturity (in the case of Original Issue Discount Securities) specified in
such Securities, such payments to be made ratably to the persons entitled thereto, without discrimination or preference;
THIRD: In
case the principal of the Securities of such series in respect of which moneys have been collected shall have become and shall be then
due and payable, to the payment of the whole amount then owing and unpaid upon all the Securities of such series for Principal and interest,
with interest upon the overdue Principal, and (to the extent that such interest has been collected by the Trustee) upon overdue installments
of interest at the same rate as the rate of interest or Yield to Maturity (in the case of Original Issue Discount Securities) specified
in the Securities of such series; and in case such moneys shall be insufficient to pay in full the whole amount so due and unpaid upon
the Securities of such series, then to the payment of such Principal and interest or Yield to Maturity, without preference or priority
of Principal over interest or Yield to Maturity, or of interest or Yield to Maturity over Principal, or of any installment of interest
over any other installment of interest, or of any Security of such series over any other Security of such series, ratably to the aggregate
of such Principal and accrued and unpaid interest or Yield to Maturity; and
FOURTH:
To the payment of the remainder, if any, to the Company or any other person lawfully entitled thereto.
Section 6.11. Restoration
of Rights and Remedies. If the Trustee or any Holder has instituted any proceeding to enforce any right or remedy under this Indenture
and such proceeding has been discontinued or abandoned for any reason, or has been determined adversely to the Trustee or to such Holder,
then, and in every such case, subject to any determination in such proceeding, the Company, the Trustee and the Holders shall be restored
to their former positions hereunder and thereafter all rights and remedies of the Company, Trustee and the Holders shall continue as though
no such proceeding had been instituted.
Section 6.12. Undertaking
for Costs. In any suit for the enforcement of any right or remedy under this Indenture or in any suit against the Trustee for any
action taken or omitted by it as Trustee, in either case in respect to the Securities of any series, a court may require any party litigant
in such suit (other than the Trustee) to file an undertaking to pay the costs of the suit, and the court may assess reasonable costs,
including reasonable attorneys’ fees, against any party litigant (other than the Trustee) in the suit having due regard to the merits
and good faith of the claims or defenses made by the party litigant. This Section 6.12 does not apply to a suit by a Holder pursuant to
Section 6.07, a suit instituted by the Trustee or a suit by Holders of more than 10% in principal amount of the outstanding Securities
of such series.
Section 6.13. Rights
and Remedies Cumulative. Except as otherwise provided with respect to the replacement or payment of mutilated, destroyed, lost or
wrongfully taken Securities in Section 2.08, no right or remedy herein conferred upon or reserved to the Trustee or to the Holders is
intended to be exclusive of any other right or remedy, and every right and remedy shall, to the extent permitted by law, be cumulative
and in addition to every other right and remedy given hereunder or now or hereafter existing at law or in equity or otherwise. The assertion
or employment of any right or remedy hereunder, or otherwise, shall not prevent the concurrent assertion or employment of any other appropriate
right or remedy.
Section 6.14. Delay
or Omission not Waiver. No delay or omission of the Trustee or of any Holder to exercise any right or remedy accruing upon any Event
of Default shall impair any such right or remedy or constitute a waiver of any such Event of Default or an acquiescence therein. Every
right and remedy given by this Article 6 or by law to the Trustee or to the Holders may be exercised from time to time, and as often as
may be deemed expedient, by the Trustee or by the Holders, as the case may be.
ARTICLE 7
TRUSTEE
Section 7.01. General. The
duties and responsibilities of the Trustee shall be as provided by the Trust Indenture Act and as set forth herein. Notwithstanding the
foregoing, no provision of this Indenture shall require the Trustee to expend or risk its own funds or otherwise incur any financial liability
in the performance of any of its duties hereunder, or in the exercise of any of its rights or powers, unless it receives indemnity satisfactory
to it against any loss, liability or expense. Whether or not therein expressly so provided, every provision of this Indenture relating
to the conduct or affecting the liability of or affording protection to the Trustee shall be subject to the provisions of this Article
7.
Section 7.02. Certain
Rights of Trustee. Subject to Trust Indenture Act Sections 315(a) through (d):
(a) the Trustee may rely
and shall be protected in acting or refraining from acting upon any resolution, certificate, Officers’ Certificate, Opinion of Counsel
(or both), statement, instrument, opinion, report, notice, request, direction, consent, order, bond, debenture, note, other evidence of
indebtedness or other paper or document believed by it to be genuine and to have been signed or presented by the proper person or persons.
The Trustee need not investigate any fact or matter stated in the document, but the Trustee, in its discretion, may make such further
inquiry or investigation into such facts or matters as it may see fit;
(b) before the Trustee acts
or refrains from acting, it may require an Officers’ Certificate and/or an Opinion of Counsel, which shall conform to Section 10.04
and shall cover such other matters as the Trustee may reasonably request. The Trustee shall not be liable for any action it takes or omits
to take in good faith in reliance on such certificate or opinion. Subject to Sections 7.01 and 7.02, whenever in the administration of
the trusts of this Indenture the Trustee shall deem it necessary or desirable that a matter be proved or established prior to taking or
suffering or omitting any action hereunder, such matter (unless other evidence in respect thereof be herein specifically prescribed) may,
in the absence of negligence or bad faith on the part of the Trustee, be deemed to be conclusively proved and established by an Officers’
Certificate delivered to the Trustee, and such certificate, in the absence of negligence or bad faith on the part of the Trustee, shall
be full warrant to the Trustee for any action taken, suffered or omitted by it under the provisions of this Indenture upon the faith thereof;
(c) the Trustee may act through
its attorneys and agents not regularly in its employ and shall not be responsible for the misconduct or negligence of any agent or attorney
appointed with due care;
(d) any request, direction,
order or demand of the Company mentioned herein shall be sufficiently evidenced by an Officers’ Certificate (unless other evidence
in respect thereof be herein specifically prescribed); and any Board Resolution may be evidenced to the Trustee by a copy thereof certified
by the Secretary or an Assistant Secretary of the Company;
(e) the Trustee shall be
under no obligation to exercise any of the rights or powers vested in it by this Indenture at the request, order or direction of any of
the Holders, unless such Holders shall have offered to the Trustee reasonable security or indemnity against the costs, expenses and liabilities
that might be incurred by it in compliance with such request or direction;
(f) the Trustee shall not
be liable for any action it takes or omits to take in good faith that it believes to be authorized or within its rights or powers or for
any action it takes or omits to take in accordance with the direction of the Holders in accordance with Section 6.05 relating to the time,
method and place of conducting any proceeding for any remedy available to the Trustee, or exercising any trust or power conferred upon
the Trustee, under this Indenture;
(g) the Trustee may consult
with counsel and the written advice of such counsel or any Opinion of Counsel shall be full and complete authorization and protection
in respect of any action taken, suffered or omitted by it hereunder in good faith and in reliance thereon; and
(h) prior to the occurrence
of an Event of Default hereunder and after the curing or waiving of all Events of Default, the Trustee shall not be bound to make any
investigation into the facts or matters stated in any resolution, certificate, Officers’ Certificate, Opinion of Counsel, Board
Resolution, statement, instrument, opinion, report, notice, request, consent, order, approval, appraisal, bond, debenture, note, coupon,
security, or other paper or document unless requested in writing so to do by the Holders of not less than a majority in aggregate principal
amount of the Securities of all series affected then outstanding; provided that, if the payment within a reasonable time to the Trustee
of the costs, expenses or liabilities likely to be incurred by it in the making of such investigation is, in the opinion of the Trustee,
not reasonably assured to the Trustee by the security afforded to it by the terms of this Indenture, the Trustee may require reasonable
indemnity against such expenses or liabilities as a condition to proceeding.
Section 7.03. Individual
Rights of Trustee. The Trustee, in its individual or any other capacity, may become the owner or pledgee of Securities and may otherwise
deal with the Company or its Affiliates with the same rights it would have if it were not the Trustee. Any Agent may do the same with
like rights. However, the Trustee is subject to Trust Indenture Act Sections 310(b) and 311. For purposes of Trust Indenture Act Section
311(b)(4) and (6), the following terms shall mean:
(a) “cash transaction”
means any transaction in which full payment for goods or securities sold is made within seven days after delivery of the goods or securities
in currency or in checks or other orders drawn upon banks or bankers and payable upon demand; and
(b) “self-liquidating
paper” means any draft, bill of exchange, acceptance or obligation which is made, drawn, negotiated or incurred by the Company
for the purpose of financing the purchase, processing, manufacturing, shipment, storage or sale of goods, wares or merchandise and which
is secured by documents evidencing title to, possession of, or a lien upon, the goods, wares or merchandise or the receivables or proceeds
arising from the sale of the goods, wares or merchandise previously constituting the security, provided the security is received by the
Trustee simultaneously with the creation of the creditor relationship with the Company arising from the making, drawing, negotiating or
incurring of the draft, bill of exchange, acceptance or obligation.
Section 7.04. Trustee’s
Disclaimer. The recitals contained herein and in the Securities (except the Trustee’s certificate of authentication) shall be
taken as statements of the Company and not of the Trustee and the Trustee assumes no responsibility for the correctness of the same. Neither
the Trustee nor any of its agents (a) makes any representation as to the validity or adequacy of this Indenture or the Securities and
(b) shall be accountable for the Company’s use or application of the proceeds from the Securities.
Section 7.05. Notice
of Default. If any Default with respect to the Securities of any series occurs and is continuing and if such Default is known to the
actual knowledge of a Responsible Officer with the Corporate Trust Department of the Trustee, the Trustee shall give to each Holder of
Securities of such series notice of such Default within 90 days after it occurs (a) if any Unregistered Securities of such series are
then outstanding, to the Holders thereof, by publication at least once in an Authorized Newspaper in the Borough of Manhattan, The City
of New York and at least once in an Authorized Newspaper in London and (b) to all Holders of Securities of such series in the manner and
to the extent provided in Section 313(c) of the Trust Indenture Act, unless such Default shall have been cured or waived before the mailing
or publication of such notice; provided, however, that, except in the case of a Default in the payment of the Principal of or interest
on any Security, the Trustee shall be protected in withholding such notice if the Trustee in good faith determines that the withholding
of such notice is in the interests of the Holders.
Section 7.06. Reports
by Trustee to Holders. The Trustee shall transmit to Holders such reports concerning the Trustee and its actions under this Indenture
as may be required pursuant to the Trust Indenture Act at the times and in the manner provided pursuant thereto. If required by Section
313(a) of the Trust Indenture Act, the Trustee shall, within 60 days after each May 15 following the date of this Indenture, deliver to
Holders a brief report, dated as of such May 15, which complies with the provisions of such Section 313(a).
A copy of each such report
shall, at the time of such transmission to Holders, be filed by the Trustee with each stock exchange upon which any Securities are listed,
with the Commission and with the Company. The Company will promptly notify the Trustee when any Securities are listed on any stock exchange.
Section 7.07. Compensation
and Indemnity. The Company shall pay to the Trustee such compensation as shall be agreed upon in writing from time to time for its
services. The compensation of the Trustee shall not be limited by any law on compensation of a Trustee of an express trust. The Company
shall reimburse the Trustee and any predecessor Trustee upon request for all reasonable out-of-pocket expenses, disbursements and advances
incurred or made by the Trustee or such predecessor Trustee. Such expenses shall include the reasonable compensation and expenses of the
Trustee’s or such predecessor Trustee’s agents, counsel and other persons not regularly in their employ.
The Company shall indemnify
the Trustee and any predecessor Trustee for, and hold them harmless against, any loss or liability or expense incurred by them without
negligence or bad faith on their part arising out of or in connection with the acceptance or administration of this Indenture and the
Securities or the issuance of the Securities or of series thereof or the trusts hereunder and the performance of duties under this Indenture
and the Securities, including the costs and expenses of defending themselves against or investigating any claim or liability and of complying
with any process served upon them or any of their officers in connection with the exercise or performance of any of their powers or duties
under this Indenture and the Securities.
To secure the Company’s
payment obligations in this Section 7.07, the Trustee shall have a lien prior to the Securities on all money or property held or collected
by the Trustee, in its capacity as Trustee, except money or property held in trust to pay Principal of, and interest on particular Securities.
The obligations of the Company
under this Section to compensate and indemnify the Trustee and each predecessor Trustee and to pay or reimburse the Trustee and each predecessor
Trustee for expenses, disbursements and advances shall constitute additional indebtedness hereunder and shall survive the satisfaction
and discharge of this Indenture or the rejection or termination of this Indenture under bankruptcy law. Such additional indebtedness shall
be a senior claim to that of the Securities upon all property and funds held or collected by the Trustee as such, except funds held in
trust for the benefit of the Holders of particular Securities or coupons, and the Securities are hereby subordinated to such senior claim.
Without prejudice to any other rights available to the Trustee under applicable law, if the Trustee renders services and incurs expenses
following an Event of Default under Section 6.01(d) or Section 6.01(e) hereof, the parties hereto and the holders by their acceptance
of the Securities hereby agree that such expenses are intended to constitute expenses of administration under any bankruptcy law.
Section 7.08. Replacement
of Trustee. A resignation or removal of the Trustee as Trustee with respect to the Securities of any series and appointment of a successor
Trustee as Trustee with respect to the Securities of any series shall become effective only upon the successor Trustee’s acceptance
of appointment as provided in this Section 7.08.
The Trustee may resign as
Trustee with respect to the Securities of any series at any time by so notifying the Company in writing. The Holders of a majority in
principal amount of the outstanding Securities of any series may remove the Trustee as Trustee with respect to the Securities of such
series by so notifying the Trustee in writing and may appoint a successor Trustee with respect thereto with the consent of the Company.
The Company may remove the Trustee as Trustee with respect to the Securities of any series if: (i) the Trustee is no longer eligible under
Section 7.11 of this Indenture; (ii) the Trustee is adjudged a bankrupt or insolvent; (iii) a receiver or other public officer takes charge
of the Trustee or its property; or (iv) the Trustee becomes incapable of acting.
If the Trustee resigns or
is removed as Trustee with respect to the Securities of any series, or if a vacancy exists in the office of Trustee with respect to the
Securities of any series for any reason, the Company shall promptly appoint a successor Trustee with respect thereto. Within one year
after the successor Trustee takes office, the Holders of a majority in principal amount of the outstanding Securities of such series may
appoint a successor Trustee in respect of such Securities to replace the successor Trustee appointed by the Company. If the successor
Trustee with respect to the Securities of any series does not deliver its written acceptance required by Section 7.09 within 30 days after
the retiring Trustee resigns or is removed, the retiring Trustee, the Company or the Holders of a majority in principal amount of the
outstanding Securities of such series may petition any court of competent jurisdiction for the appointment of a successor Trustee with
respect thereto.
The Company shall give notice
of any resignation and any removal of the Trustee with respect to the Securities of any series and each appointment of a successor Trustee
in respect of the Securities of such series to all Holders of Securities of such series. Each notice shall include the name of the successor
Trustee and the address of its Corporate Trust Office.
Notwithstanding replacement
of the Trustee with respect to the Securities of any series pursuant to this Section 7.08 and Section 7.09, the Company’s obligations
under Section 7.07 shall continue for the benefit of the retiring Trustee.
Section 7.09. Acceptance
of Appointment by Successor. In case of the appointment hereunder of a successor Trustee with respect to all Securities, every such
successor Trustee so appointed shall execute, acknowledge and deliver to the Company and to the retiring Trustee an instrument accepting
such appointment, and thereupon the resignation or removal of the retiring Trustee shall become effective and such successor Trustee,
without any further act, deed or conveyance, shall become vested with all the rights, powers, trusts and duties of the retiring Trustee;
but, on the request of the Company or the successor Trustee, such retiring Trustee shall, upon payment of its charges and subject to the
lien provided for in Section 7.07, execute and deliver an instrument transferring to such successor Trustee all the rights, powers and
trusts of the retiring Trustee and shall duly assign, transfer and deliver to such successor Trustee all property and money held by such
retiring Trustee hereunder.
In case of the appointment
hereunder of a successor Trustee with respect to the Securities of one or more (but not all) series, the Company, the retiring Trustee
and each successor Trustee with respect to the Securities of one or more series shall execute and deliver an indenture supplemental hereto
wherein each successor Trustee shall accept such appointment and which (1) shall contain such provisions as shall be necessary or desirable
to transfer and confirm to, and to vest in, each successor Trustee all the rights, powers, trusts and duties of the retiring Trustee with
respect to the Securities of that or those series to which the appointment of such successor Trustee relates, (2) if the retiring Trustee
is not retiring with respect to all Securities, shall contain such provisions as shall be deemed necessary or desirable to confirm that
all the rights, powers, trusts and duties of the retiring Trustee with respect to the Securities of that or those series as to which the
retiring Trustee is not retiring shall continue to be vested in the retiring Trustee, and (3) shall add to or change any of the provisions
of this Indenture as shall be necessary to provide for or facilitate the administration of the trusts hereunder by more than one Trustee,
it being understood that nothing herein or in such supplemental indenture shall constitute such Trustees co-trustees of the same trust
and that each such Trustee shall be trustee of a trust or trusts hereunder separate and apart from any trust or trusts hereunder administered
by any other such Trustee; and upon the execution and delivery of such supplemental indenture the resignation or removal of the retiring
Trustee shall become effective to the extent provided therein and each such successor Trustee, without any further act, deed or conveyance,
shall become vested with all the rights, powers, trusts and duties of the retiring Trustee with respect to the Securities of that or those
series to which the appointment of such successor Trustee relates; but, on request of the Company or any successor Trustee, such retiring
Trustee shall duly assign, transfer and deliver to such successor Trustee all property and money held by such retiring Trustee hereunder
with respect to the Securities of that or those series to which the appointment of such successor Trustee relates.
Upon request of any such
successor Trustee, the Company shall execute any and all instruments for more fully and certainly vesting in and confirming to such successor
Trustee all such rights, powers and trusts referred to in the first or second preceding paragraph, as the case may be.
No successor Trustee shall
accept its appointment unless at the time of such acceptance such successor Trustee shall be eligible under this Article and qualified
under Section 310(b) of the Trust Indenture Act.
Section 7.10. Successor
Trustee By Merger, Etc. If the Trustee consolidates with, merges or converts into, or transfers all or substantially all of its corporate
trust business to, another corporation or national banking association, the resulting, surviving or transferee corporation or national
banking association without any further act shall be the successor Trustee with the same effect as if the successor Trustee had been named
as the Trustee herein.
Section 7.11. Eligibility.
This Indenture shall always have a Trustee who satisfies the requirements of Trust Indenture Act Section 310(a). The Trustee shall have
a combined capital and surplus of at least $25,000,000 as set forth in its most recent published annual report of condition.
Section 7.12. Money
Held in Trust. The Trustee shall not be liable for interest on any money received by it except as the Trustee may agree in writing
with the Company. Money held in trust by the Trustee need not be segregated from other funds except to the extent required by law and
except for money held in trust under Article 8 of this Indenture.
ARTICLE 8
SATISFACTION
AND DISCHARGE OF INDENTURE; UNCLAIMED MONEYS
Section 8.01. Satisfaction
and Discharge of Indenture. If at any time (a) the Company shall have paid or caused to be paid the Principal of and interest
on all the Securities of any series outstanding hereunder (other than Securities of such series which have been destroyed, lost or stolen
and which have been replaced or paid as provided in Section 2.08) as and when the same shall have become due and payable, or (b) the Company
shall have delivered to the Trustee for cancellation all Securities of any series theretofore authenticated (other than any Securities
of such series which shall have been destroyed, lost or stolen and which shall have been replaced or paid as provided in Section 2.08)
or (c) (i) all the securities of such series not theretofore delivered to the Trustee for cancellation shall have become due and payable,
or are by their terms to become due and payable within one year or are to be called for redemption within one year under arrangements
satisfactory to the Trustee for the giving of notice of redemption, and (ii) the Company shall have irrevocably deposited or caused to
be deposited with the Trustee as trust funds the entire amount in cash (other than moneys repaid by the Trustee or any paying agent to
the Company in accordance with Section 8.04) or U.S. Government Obligations, maturing as to principal and interest in such amounts and
at such times as will insure (without consideration of the reinvestment of such interest) the availability of cash, or a combination thereof,
sufficient to pay at maturity or upon redemption all Securities of such series (other than any Securities of such series which shall have
been destroyed, lost or stolen and which shall have been replaced or paid as provided in Section 2.08) not theretofore delivered to the
Trustee for cancellation, including principal and interest due or to become due on or prior to such date of maturity or redemption as
the case may be, and if, in any such case, the Company shall also pay or cause to be paid all other sums payable hereunder by the Company
with respect to Securities of such series, then this Indenture shall cease to be of further effect with respect to Securities of such
series (except as to (i) rights of registration of transfer and exchange of securities of such series, and the Company’s right of
optional redemption, if any, (ii) substitution of mutilated, defaced, destroyed, lost or stolen Securities, (iii) rights of holders to
receive payments of principal thereof and interest thereon, upon the original stated due dates therefor (but not upon acceleration) and
remaining rights of the holders to receive mandatory sinking fund payments, if any, (iv) the rights, obligations and immunities of the
Trustee hereunder and (v) the rights of the Securityholders of such series as beneficiaries hereof with respect to the property so deposited
with the Trustee payable to all or any of them), and the Trustee, on demand of the Company accompanied by an Officers’ Certificate
and an Opinion of Counsel and at the cost and expense of the Company, shall execute proper instruments acknowledging such satisfaction
of and discharging this Indenture with respect to such series; provided, that the rights of Holders of the Securities to receive amounts
in respect of Principal of and interest on the Securities held by them shall not be delayed longer than required by then-applicable mandatory
rules or policies of any securities exchange upon which the Securities are listed. The Company agrees to reimburse the Trustee for any
costs or expenses thereafter reasonably and properly incurred and to compensate the Trustee for any services thereafter reasonably and
properly rendered by the Trustee in connection with this Indenture or the Securities of such series.
Section 8.02. Application
by Trustee of Funds Deposited for Payment of Securities. Subject to Section 8.04, all moneys (including U.S. Government Obligations
and the proceeds thereof) deposited with the Trustee pursuant to Section 8.01, Section 8.05 or Section 8.06 shall be held in trust and
applied by it to the payment, either directly or through any paying agent to the Holders of the particular Securities of such series for
the payment or redemption of which such moneys have been deposited with the Trustee, of all sums due and to become due thereon for Principal
and interest; but such money need not be segregated from other funds except to the extent required by law.
Section 8.03. Repayment
of Moneys Held by Paying Agent. In connection with the satisfaction and discharge of this Indenture with respect to Securities
of any series, all moneys then held by any paying agent under the provisions of this Indenture with respect to such series of Securities
shall, upon demand of the Company, be repaid to it or paid to the Trustee and thereupon such paying agent shall be released from all further
liability with respect to such moneys.
Section 8.04. Return
of Moneys Held by Trustee and Paying Agent Unclaimed for Two Years. Any moneys deposited with or paid to the Trustee or any paying
agent for the payment of the Principal of or interest on any Security of any series and not applied but remaining unclaimed for two years
after the date upon which such Principal or interest shall have become due and payable, shall, upon the written request of the Company
and unless otherwise required by mandatory provisions of applicable escheat or abandoned or unclaimed property law, be repaid to the Company
by the Trustee for such series or such paying agent, and the Holder of the Security of such series shall, unless otherwise required by
mandatory provisions of applicable escheat or abandoned or unclaimed property laws, thereafter look only to the Company for any payment
which such Holder may be entitled to collect, and all liability of the Trustee or any paying agent with respect to such moneys shall thereupon
cease.
Section 8.05. Defeasance
and Discharge of Indenture. The Company shall be deemed to have paid and shall be discharged from any and all obligations in
respect of the Securities of any series, on the 123rd day after the deposit referred to in clause (i) hereof has been made, and the provisions
of this Indenture shall no longer be in effect with respect to the Securities of such series (and the Trustee, at the expense of the Company,
shall execute proper instruments acknowledging the same), except as to: (a) rights of registration of transfer and exchange, and the Company’s
right of optional redemption, (b) substitution of apparently mutilated, defaced, destroyed, lost or stolen Securities, (c) rights of holders
to receive payments of principal thereof and interest thereon, upon the original stated due dates therefor (but not upon acceleration),
(d) the rights, obligations and immunities of the Trustee hereunder and (e) the rights of the Securityholders of such series as beneficiaries
hereof with respect to the property so deposited with the Trustee payable to all or any of them; provided that the following conditions
shall have been satisfied:
(i) with reference
to this provision the Company has deposited or caused to be irrevocably deposited with the Trustee (or another qualifying trustee satisfying
the requirements of Section 7.11) as trust funds in trust, specifically pledged as security for, and dedicated solely to, the benefit
of the Holders of the Securities of such series, (A) money in an amount, or (B) U.S. Government Obligations which through the payment
of interest and principal in respect thereof in accordance with their terms will provide not later than one day before the due date of
any payment referred to in subclause (x) or (y) of this clause (i) money in an amount, or (C) a combination thereof, sufficient, in the
opinion of a nationally recognized firm of independent public accountants expressed in a written certification thereof delivered to the
Trustee, to pay and discharge without consideration of the reinvestment of such interest and after payment of all federal, state and local
taxes or other charges and assessments in respect thereof payable by the Trustee (x) the principal of, premium, if any, and each installment
of interest on the outstanding Securities of such series on the due dates thereof and (y) any mandatory sinking fund payments or analogous
payments applicable to the Securities of such series on the day on which such payments are due and payable in accordance with the terms
of Securities of such series and the Indenture with respect to the Securities of such series;
(ii) the Company
has delivered to the Trustee (A) either (x) an Opinion of Counsel to the effect that Holders of Securities of such series will not recognize
income, gain or loss for federal income tax purposes as a result of the Company’s exercise of its option under this Section 8.05
and will be subject to federal income tax on the same amount and in the same manner and at the same times as would have been the case
if such deposit, defeasance and discharge had not occurred, which Opinion of Counsel must be based upon a ruling of the Internal Revenue
Service to the same effect or a change in applicable federal income tax law or related treasury regulations after the date of this Indenture
or (y) a ruling directed to the Trustee received from the Internal Revenue Service to the same effect as the aforementioned Opinion of
Counsel and (B) an Opinion of Counsel to the effect that the creation of the defeasance trust does not violate the Investment Company
Act of 1940, as amended, and after the passage of 123 days following the deposit, the trust fund will not be subject to the effect of
Section 547 of the U.S. Bankruptcy Code or Section 15 of the New York Debtor and Creditor Law;
(iii) immediately
after giving effect to such deposit on a pro forma basis, no Event of Default, or event that after the giving of notice or lapse of time
or both would become an Event of Default, shall have occurred and be continuing on the date of such deposit or during the period ending
on the 123rd day after the date of such deposit, and such deposit shall not result in a breach or violation of, or constitute a default
under, any other agreement or instrument to which the Company is a party or by which the Company is bound;
(iv) if at such
time the Securities of such series are listed on a national securities exchange, the Company has delivered to the Trustee an Opinion of
Counsel to the effect that the Securities of such series will not be delisted as a result of such deposit, defeasance and discharge;
(v) the Company
shall have delivered to the Trustee an Officers’ Certificate and an Opinion of Counsel, each stating that all conditions precedent
to the defeasance and discharge under this Section have been complied with; and
(vi) if the Securities
of such series are to be redeemed prior to the final maturity thereof (other than from mandatory sinking fund payments or analogous payments),
notice of such redemption shall have been duly given pursuant to this Indenture or provision therefor satisfactory to the Trustee shall
have been made.
Section 8.06. Defeasance
of Certain Obligations. The Company may omit to comply with any term, provision or condition set forth in, and this Indenture
will no longer be in effect with respect to, any covenant established pursuant to Section 2.03(r) and clause (c) (with respect to any
covenants established pursuant to Section 2.03(r)) and clause (f) of Section 6.01 shall be deemed not to be an Event of Default, if
(a) with reference to this
Section 8.06, the Company has deposited or caused to be irrevocably deposited with the Trustee (or another qualifying trustee satisfying
the requirements of Section 7.11) as trust funds in trust, specifically pledged as security for, and dedicated solely to, the benefit
of the Holders of the Securities of such series and the Indenture with respect to the Securities of such series, (i) money in an amount
or (ii) U.S. Government Obligations which through the payment of interest and principal in respect thereof in accordance with their terms
will provide not later than one day before the due dates thereof or earlier redemption (irrevocably provided for under agreements satisfactory
to the Trustee), as the case may be, of any payment referred to in subclause (x) or (y) of this clause (a) money in an amount, or (iii)
a combination thereof, sufficient, in the opinion of a nationally recognized firm of independent public accountants expressed in a written
certification thereof delivered to the Trustee, to pay and discharge without consideration of the reinvestment of such interest and after
payment of all federal, state and local taxes or other charges and assessments in respect thereof payable by the Trustee (x) the principal
of, premium, if any, and each installment of interest on the outstanding Securities on the due date thereof or earlier redemption (irrevocably
provided for under arrangements satisfactory to the Trustee), as the case may be, and (y) any mandatory sinking fund payments or analogous
payments applicable to the Securities of such series and the Indenture with respect to the Securities of such series on the day on which
such payments are due and payable in accordance with the terms of the Indenture and of Securities of such series and the Indenture with
respect to the Securities of such series;
(b) the Company has delivered
to the Trustee (i) an Opinion of Counsel to the effect that Holders of Securities of such series will not recognize income, gain or loss
for federal income tax purposes as a result of the Company’s exercise of its option under this Section 8.06 and will be subject
to federal income tax on the same amount and in the same manner and at the same times as would have been the case if such deposit and
defeasance had not occurred and (ii) an Opinion of Counsel to the effect that the creation of the defeasance trust does not violate the
Investment Company Act of 1940, as amended, and after the passage of 123 days following the deposit, the trust fund will not be subject
to the effect of Section 547 of the U.S. Bankruptcy Code or Section 15 of the New York Debtor and Creditor Law;
(c) immediately after giving
effect to such deposit on a pro forma basis, no Event of Default, or event that after the giving of notice or lapse of time or both would
become an Event of Default, shall have occurred and be continuing on the date of such deposit or during the period ending on the 123rd
day after the date of such deposit, and such deposit shall not result in a breach or violation of, or constitute a default under, any
other agreement or instrument to which the Company is a party or by which the Company is bound;
(d) if at such time the Securities
of such series are listed on a national securities exchange, the Company has delivered to the Trustee an Opinion of Counsel to the effect
that the Securities of such series will not be delisted as a result of such deposit, defeasance and discharge; and
(e) the Company shall have
delivered to the Trustee an Officers’ Certificate and an Opinion of Counsel, each stating that all conditions precedent to the defeasance
under this Section have been complied with.
Section 8.07. Reinstatement. If
the Trustee or paying agent is unable to apply any monies or U.S. Government Obligations in accordance with Article 8 by reason of any
legal proceeding or by reason of any order or judgment of any court or governmental authority enjoining, restraining or otherwise prohibiting
such application, the Company’s obligations under this Indenture and the Securities shall be revived and reinstated as though no
deposit had occurred pursuant to this Article until such time as the Trustee or paying agent is permitted to apply all such monies or
U.S. Government Obligations in accordance with Article 8; provided, however, that if the Company has made any
payment of Principal of or interest on any Securities because of the reinstatement of its obligations, the Company shall be subrogated
to the rights of the Holders of such Securities to receive such payment from the monies or U.S. Government Obligations held by the Trustee
or paying agent.
Section 8.08. Indemnity. The
Company shall pay and indemnify the Trustee (or other qualifying trustee, collectively for purposes of this Section 8.08 and Section 8.02,
the “Trustee”) against any tax, fee or other charge, imposed on or assessed against the U.S. Government Obligations deposited
pursuant to Section 8.01, 8.05 or 8.06 or the principal or interest received in respect thereof other than any such tax, fee or other
charge which by law is for the account of the Holders of the Securities and any coupons appertaining thereto.
Section 8.09. Excess
Funds. Anything in this Article 8 to the contrary notwithstanding, the Trustee shall deliver or pay to the Company from time
to time upon request of the Company, any money or U.S. Government Obligations (or other property and any proceeds therefrom) held by it
as provided in Section 8.01, 8.05 or 8.06 which, in the opinion of a nationally recognized firm of independent public accountants expressed
in a written certification thereof delivered to the Trustee, are in excess of the amount thereof which would then be required to be deposited
to effect a discharge or defeasance, as applicable, in accordance with this Article 8.
Section 8.10. Qualifying
Trustee. Any trustee appointed pursuant to Section 8.05 or 8.06 for the purpose of holding money or U.S. Government Obligations
deposited pursuant to such Sections shall be appointed under an agreement in form acceptable to the Trustee and shall provide to the Trustee
a certificate, upon which certificate the Trustee shall be entitled to conclusively rely, that all conditions precedent provided for herein
to the related defeasance have been complied with. In no event shall the Trustee be liable for any acts or omissions of said trustee.
ARTICLE 9
AMENDMENTS,
SUPPLEMENTS AND WAIVERS
Section 9.01. Without
Consent of Holders. The Company and the Trustee may amend or supplement this Indenture or the Securities of any series without
notice to or the consent of any Holder:
(a) to cure any ambiguity,
defect or inconsistency in this Indenture; provided that such amendments or supplements shall not materially and adversely affect the
interests of the Holders;
(b) to comply with Article
5;
(c) to comply with any requirements
of the Commission in connection with the qualification of this Indenture under the Trust Indenture Act;
(d) to evidence and provide
for the acceptance of appointment hereunder with respect to the Securities of any or all series by a successor Trustee and to add to or
change any of the provisions of this Indenture as shall be necessary to provide for or facilitate the administration of the trusts hereunder
by more than one Trustee, pursuant to the requirements of Section 7.09;
(e) to establish the form
or forms or terms of Securities of any series or of the coupons appertaining to such Securities as permitted by Section 2.03;
(f) to provide for uncertificated
or Unregistered Securities and to make all appropriate changes for such purpose; and
(g) to make any change that
does not materially and adversely affect the rights of any Holder.
Section 9.02. With
Consent of Holders. Subject to Sections 6.04 and 6.07, without prior notice to any Holders, the Company and the Trustee may amend
this Indenture and the Securities of any series with the written consent of the Holders of a majority in principal amount of the outstanding
Securities of all series affected by such amendment (all such series voting as a separate class), and the Holders of a majority in principal
amount of the outstanding Securities of all series affected thereby (all such series voting as a separate class) by written notice to
the Trustee may waive future compliance by the Company with any provision of this Indenture or the Securities of such series.
Notwithstanding the provisions
of this Section 9.02, without the consent of each Holder affected thereby, an amendment or waiver, including a waiver pursuant to Section
6.04, may not:
(a) change the stated maturity
of the Principal of, or any sinking fund obligation or any installment of interest on, such Holder’s Security;
(b) reduce the Principal
amount thereof or the rate of interest thereon (including any amount in respect of original issue discount);
(c) reduce the above stated
percentage of outstanding Securities the consent of whose holders is necessary to modify or amend the Indenture with respect to the Securities
of the relevant series; and
(d) reduce the percentage
in principal amount of outstanding Securities of the relevant series the consent of whose Holders is required for any supplemental indenture
or for any waiver of compliance with certain provisions of this Indenture or certain Defaults and their consequences provided for in this
Indenture.
A supplemental indenture
which changes or eliminates any covenant or other provision of this Indenture which has expressly been included solely for the benefit
of one or more particular series of Securities, or which modifies the rights of Holders of Securities of such series with respect to such
covenant or provision, shall be deemed not to affect the rights under this Indenture of the Holders of Securities of any other series
or of the coupons appertaining to such Securities.
It shall not be necessary
for the consent of any Holder under this Section 9.02 to approve the particular form of any proposed amendment, supplement or waiver,
but it shall be sufficient if such consent approves the substance thereof.
After an amendment, supplement
or waiver under this Section 9.02 becomes effective, the Company shall give to the Holders affected thereby a notice briefly describing
the amendment, supplement or waiver. The Company will mail supplemental indentures to Holders upon request. Any failure of the Company
to mail such notice, or any defect therein, shall not, however, in any way impair or affect the validity of any such supplemental indenture
or waiver.
Section 9.03. Revocation
and Effect of Consent. Until an amendment or waiver becomes effective, a consent to it by a Holder is a continuing consent by
the Holder and every subsequent Holder of a Security or portion of a Security that evidences the same debt as the Security of the consenting
Holder, even if notation of the consent is not made on any Security. However, any such Holder or subsequent Holder may revoke the consent
as to its Security or portion of its Security. Such revocation shall be effective only if the Trustee receives the notice of revocation
before the date the amendment, supplement or waiver becomes effective. An amendment, supplement or waiver shall become effective with
respect to any Securities affected thereby on receipt by the Trustee of written consents from the requisite Holders of outstanding Securities
affected thereby.
The Company may, but shall
not be obligated to, fix a record date (which may be not less than five nor more than 60 days prior to the solicitation of consents) for
the purpose of determining the Holders of the Securities of any series affected entitled to consent to any amendment, supplement or waiver.
If a record date is fixed, then, notwithstanding the immediately preceding paragraph, those Persons who were such Holders at such record
date (or their duly designated proxies) and only those Persons shall be entitled to consent to such amendment, supplement or waiver or
to revoke any consent previously given, whether or not such Persons continue to be such Holders after such record date. No such consent
shall be valid or effective for more than 90 days after such record date.
After an amendment, supplement
or waiver becomes effective with respect to the Securities of any series affected thereby, it shall bind every Holder of such Securities
unless it is of the type described in any of clauses (a) through (d) of Section 9.02. In case of an amendment or waiver of the type described
in clauses (a) through (d) of Section 9.02, the amendment or waiver shall bind each such Holder who has consented to it and every subsequent
Holder of a Security that evidences the same indebtedness as the Security of the consenting Holder.
Section 9.04. Notation
on or Exchange of Securities. If an amendment, supplement or waiver changes the terms of any Security, the Trustee may require
the Holder thereof to deliver it to the Trustee. The Trustee may place an appropriate notation on the Security about the changed terms
and return it to the Holder and the Trustee may place an appropriate notation on any Security of such series thereafter authenticated.
Alternatively, if the Company or the Trustee so determines, the Company in exchange for the Security shall issue and the Trustee shall
authenticate a new Security of the same series and tenor that reflects the changed terms.
Section 9.05. Trustee
to Sign Amendments, Etc. The Trustee shall be entitled to receive, and shall be fully protected in relying upon, an Opinion of
Counsel stating that the execution of any amendment, supplement or waiver authorized pursuant to this Article 9 is authorized or permitted
by this Indenture, stating that all requisite consents have been obtained or that no consents are required and stating that such supplemental
indenture constitutes the legal, valid and binding obligation of the Company, enforceable against the Company in accordance with its terms,
subject to customary exceptions. The Trustee may, but shall not be obligated to, execute any such amendment, supplement or waiver that
affects the Trustee’s own rights, duties or immunities under this Indenture or otherwise.
Section 9.06. Conformity
with Trust Indenture Act. Every supplemental indenture executed pursuant to this Article 9 shall conform to the requirements
of the Trust Indenture Act as then in effect.
ARTICLE 10
MISCELLANEOUS
Section 10.01. Trust
Indenture Act of 1939. This Indenture shall incorporate and be governed by the provisions of the Trust Indenture Act that are required
to be part of and to govern indentures qualified under the Trust Indenture Act.
Section 10.02. Notices. Any
notice or communication by the Company or the Trustee to the other, or by a Holder to the Company or the Trustee, is duly given if in
writing and delivered in person or mailed by first-class mail (registered or certified, return receipt requested), email or overnight
air courier guaranteeing next day delivery, to the others’ address:
if to the Company:
| |
60 Degrees Pharmaceuticals, Inc.
1025 Connecticut Ave. NW, Suite 1000
Washington, DC 20036 |
| |
Phone: (202) 327-5422 |
| |
Attention: Chief Executive Officer and President |
if to the Trustee:
| |
[Name of Trustee] |
| |
[Address] |
| |
Phone: |
| |
Attention: |
The Company or the Trustee
by written notice to the other may designate additional or different addresses for subsequent notices or communications.
Any notice or communication
shall be sufficiently given to Holders of any Unregistered Securities, by publication at least once in an Authorized Newspaper in The
City of New York, or with respect to any Security the interest on which is based on the offered quotations in the interbank Eurodollar
market for dollar deposits at least once in an Authorized Newspaper in London, and by mailing to the Holders thereof who have filed their
names and addresses with the Trustee pursuant to Section 313(c)(2) of the Trust Indenture Act at such addresses as were so furnished to
the Trustee and to Holders of Registered Securities by mailing to such Holders at their addresses as they shall appear on the Security
Register. Notice mailed shall be sufficiently given if so mailed within the time prescribed. Copies of any such communication or notice
to a Holder shall also be mailed to the Trustee and each Agent at the same time.
Failure to mail a notice
or communication to a Holder or any defect in it shall not affect its sufficiency with respect to other Holders. Except as otherwise provided
in this Indenture, if a notice or communication is mailed in the manner provided in this Section 10.02, it is duly given, whether or not
the addressee receives it.
Where this Indenture provides
for notice in any manner, such notice may be waived in writing by the Person entitled to receive such notice, either before or after the
event, and such waiver shall be the equivalent of such notice. Waivers of notice by Holders shall be filed with the Trustee, but such
filing shall not be a condition precedent to the validity of any action taken in reliance upon such waiver.
In case it shall be impracticable
to give notice as herein contemplated, then such notification as shall be made with the approval of the Trustee shall constitute a sufficient
notification for every purpose hereunder.
Section 10.03. Certificate
and Opinion as to Conditions Precedent. Upon any request or application by the Company to the Trustee to take any action under
this Indenture, the Company shall furnish to the Trustee:
(a) an Officers’ Certificate
stating that, in the opinion of the signers, all conditions precedent, if any, provided for in this Indenture relating to the proposed
action have been complied with; and
(b) an Opinion of Counsel
stating that, in the opinion of such counsel, all such conditions precedent have been complied with.
Section 10.04. Statements
Required in Certificate or Opinion. Each certificate or opinion with respect to compliance with a condition or covenant provided
for in this Indenture (other than the certificate required by Section 4.04) shall include:
(a) a statement that each
person signing such certificate or opinion has read such covenant or condition and the definitions herein relating thereto;
(b) a brief statement as
to the nature and scope of the examination or investigation upon which the statement or opinion contained in such certificate or opinion
is based;
(c) a statement that, in
the opinion of each such person, he has made such examination or investigation as is necessary to enable him to express an informed opinion
as to whether or not such covenant or condition has been complied with; and
(d) a statement as to whether
or not, in the opinion of each such person, such condition or covenant has been complied with; provided, however, that, with
respect to matters of fact, an Opinion of Counsel may rely on an Officers’ Certificate or certificates of public officials.
Section 10.05. Evidence
of Ownership. The Company, the Trustee and any agent of the Company or the Trustee may deem and treat the Holder of any Unregistered
Security and the Holder of any coupon as the absolute owner of such Unregistered Security or coupon (whether or not such Unregistered
Security or coupon shall be overdue) for the purpose of receiving payment thereof or on account thereof and for all other purposes, and
neither the Company, the Trustee, nor any agent of the Company or the Trustee shall be affected by any notice to the contrary. The fact
of the holding by any Holder of an Unregistered Security, and the identifying number of such Security and the date of his holding the
same, may be proved by the production of such Security or by a certificate executed by any trust company, bank, banker or recognized securities
dealer wherever situated satisfactory to the Trustee, if such certificate shall be deemed by the Trustee to be satisfactory. Each such
certificate shall be dated and shall state that on the date thereof a Security bearing a specified identifying number was deposited with
or exhibited to such trust company, bank, banker or recognized securities dealer by the person named in such certificate. Any such certificate
may be issued in respect of one or more Unregistered Securities specified therein. The holding by the person named in any such certificate
of any Unregistered Securities specified therein shall be presumed to continue for a period of one year from the date of such certificate
unless at the time of any determination of such holding (1) another certificate bearing a later date issued in respect of the same Securities
shall be produced or (2) the Security specified in such certificate shall be produced by some other Person, or (3) the Security specified
in such certificate shall have ceased to be outstanding. Subject to Article 7, the fact and date of the execution of any such instrument
and the amount and numbers of Securities held by the Person so executing such instrument may also be proven in accordance with such reasonable
rules and regulations as may be prescribed by the Trustee or in any other manner which the Trustee may deem sufficient.
The Company, the Trustee
and any agent of the Company or the Trustee may deem and treat the person in whose name any Registered Security shall be registered upon
the Security Register for such series as the absolute owner of such Registered Security (whether or not such Registered Security shall
be overdue and notwithstanding any notation of ownership or other writing thereon) for the purpose of receiving payment of or on account
of the Principal of and, subject to the provisions of this Indenture, interest on such Registered Security and for all other purposes;
and neither the Company nor the Trustee nor any agent of the Company or the Trustee shall be affected by any notice to the contrary.
Section 10.06. Rules
by Trustee, Paying Agent or Registrar. The Trustee may make reasonable rules for action by or at a meeting of Holders. The Paying
Agent or Registrar may make reasonable rules for its functions.
Section 10.07. Payment
Date Other Than a Business Day. Except as otherwise provided with respect to a series of Securities, if any date for payment
of Principal or interest on any Security shall not be a Business Day at any place of payment, then payment of Principal of or interest
on such Security, as the case may be, need not be made on such date, but may be made on the next succeeding Business Day at any place
of payment with the same force and effect as if made on such date and no interest shall accrue in respect of such payment for the period
from and after such date.
Section 10.08. Governing
Law. The laws of the State of New York shall govern this Indenture and the Securities.
Section 10.09. No
Adverse Interpretation of Other Agreements. This Indenture may not be used to interpret another indenture or loan or debt agreement
of the Company or any Subsidiary of the Company. Any such indenture or agreement may not be used to interpret this Indenture.
Section 10.10. Successors. All
agreements of the Company in this Indenture and the Securities shall bind its successors. All agreements of the Trustee in this Indenture
shall bind its successors.
Section 10.11. Duplicate
Originals. The parties may sign any number of copies of this Indenture. Each signed copy shall be an original, but all of them
together represent the same agreement.
Section 10.12. Separability. In
case any provision in this Indenture or in the Securities shall be invalid, illegal or unenforceable, the validity, legality and enforceability
of the remaining provisions shall not in any way be affected or impaired thereby.
Section 10.13. Table
of Contents, Headings, Etc. The Table of Contents and headings of the Articles and Sections of this Indenture have been inserted
for convenience of reference only, are not to be considered a part hereof and shall in no way modify or restrict any of the terms and
provisions hereof.
Section 10.14. Incorporators,
Stockholders, Officers and Directors of Company Exempt from Individual Liability. No recourse under or upon any obligation, covenant
or agreement contained in this Indenture or any indenture supplemental hereto, or in any Security or any coupons appertaining thereto,
or because of any indebtedness evidenced thereby, shall be had against any incorporator, as such or against any past, present or future
stockholder, officer, director or employee, as such, of the Company or of any successor, either directly or through the Company or any
successor, under any rule of law, statute or constitutional provision or by the enforcement of any assessment or by any legal or equitable
proceeding or otherwise, all such liability being expressly waived and released by the acceptance of the Securities and the coupons appertaining
thereto by the holders thereof and as part of the consideration for the issue of the Securities and the coupons appertaining thereto.
Section 10.15. Judgment
Currency. The Company agrees, to the fullest extent that it may effectively do so under applicable law, that (a) if for the purpose
of obtaining judgment in any court it is necessary to convert the sum due in respect of the Principal of or interest on the Securities
of any series (the “Required Currency”) into a currency in which a judgment will be rendered (the “Judgment
Currency”), the rate of exchange used shall be the rate at which in accordance with normal banking procedures the Trustee could
purchase in The City of New York the Required Currency with the Judgment Currency on the day on which final unappealable judgment is entered,
unless such day is not a Business Day, then, to the extent permitted by applicable law, the rate of exchange used shall be the rate at
which in accordance with normal banking procedures the Trustee could purchase in The City of New York the Required Currency with the Judgment
Currency on the Business Day preceding the day on which final unappealable judgment is entered and (b) its obligations under this Indenture
to make payments in the Required Currency (i) shall not be discharged or satisfied by any tender, or any recovery pursuant to any judgment
(whether or not entered in accordance with subsection (a)), in any currency other than the Required Currency, except to the extent that
such tender or recovery shall result in the actual receipt, by the payee, of the full amount of the Required Currency expressed to be
payable in respect of such payments, (ii) shall be enforceable as an alternative or additional cause of action for the purpose of recovering
in the Required Currency the amount, if any, by which such actual receipt shall fall short of the full amount of the Required Currency
so expressed to be payable and (iii) shall not be affected by judgment being obtained for any other sum due under this Indenture.
SIGNATURES
IN WITNESS WHEREOF, the parties
hereto have caused this Indenture to be duly executed, all as of the date first written above.
| |
60 DEGREES PHARMACEUTICALS, INC., as the Company |
| |
|
|
| |
By: |
|
| |
Name: |
|
| |
Title: |
|
| |
|
_________________, as the Trustee |
37
Exhibit 4.3
60 DEGREES PHARMACEUTICALS, INC.
as the Company
and
as Trustee
Subordinated Indenture
Dated as of
, 20
TABLE OF CONTENTS
| |
PAGE |
| ARTICLE 1 |
|
| DEFINITIONS AND INCORPORATION BY REFERENCE |
1 |
| |
|
| Section 1.01. Definitions |
1 |
| Section 1.02. Other Definitions |
5 |
| Section 1.03. Incorporation by Reference of Trust Indenture Act |
5 |
| Section 1.04. Rules of Construction |
6 |
| |
|
| ARTICLE 2 |
|
| THE SECURITIES |
6 |
| |
|
| Section 2.01. Form and Dating |
6 |
| Section 2.02. Execution And Authentication |
6 |
| Section 2.03. Amount Unlimited; Issuable in Series |
7 |
| Section 2.04. Denomination and Date of Securities; Payments of Interest |
9 |
| Section 2.05. Registrar and Paying Agent; Agents Generally |
10 |
| Section 2.06. Paying Agent to Hold Money in Trust |
10 |
| Section 2.07. Transfer and Exchange |
10 |
| Section 2.08. Replacement Securities |
12 |
| Section 2.09. Outstanding Securities |
13 |
| Section 2.10. Temporary Securities |
13 |
| Section 2.11. Cancellation |
14 |
| Section 2.12. CUSIP Numbers |
14 |
| Section 2.13. Defaulted Interest |
14 |
| Section 2.14. Series May Include Tranches |
14 |
| |
|
| ARTICLE 3 |
|
| REDEMPTION |
14 |
| |
|
| Section 3.01. Applicability of Article |
14 |
| Section 3.02. Notice of Redemption; Partial Redemptions |
15 |
| Section 3.03. Payment Of Securities Called For Redemption |
16 |
| Section 3.04. Exclusion of Certain Securities from Eligibility for Selection for Redemption |
16 |
| Section 3.05. Mandatory and Optional Sinking Funds |
16 |
| ARTICLE 4 |
|
| COVENANTS |
18 |
| |
|
| Section 4.01. Payment of Securities |
18 |
| Section 4.02. Maintenance of Office or Agency |
18 |
| Section 4.03. Securityholders’ Lists |
19 |
| Section 4.04. Certificate to Trustee |
19 |
| Section 4.05. Reports by the Company |
19 |
| Section 4.06. Additional Amounts |
19 |
| |
|
| ARTICLE 5 |
|
| SUCCESSOR CORPORATION |
20 |
| |
|
| Section 5.01. When Company May Merge, Etc |
20 |
| Section 5.02. Successor Substituted |
20 |
| |
|
| ARTICLE 6 |
|
| DEFAULT AND REMEDIES |
20 |
| |
|
| Section 6.01. Events of Default |
20 |
| Section 6.02. Acceleration |
21 |
| Section 6.03. Other Remedies |
21 |
| Section 6.04. Waiver of Past Defaults |
22 |
| Section 6.05. Control by Majority |
22 |
| Section 6.06. Limitation on Suits |
22 |
| Section 6.07. Rights of Holders to Receive Payment |
22 |
| Section 6.08. Collection Suit by Trustee |
23 |
| Section 6.09. Trustee May File Proofs of Claim |
23 |
| Section 6.10. Application of Proceeds |
23 |
| Section 6.11. Restoration of Rights and Remedies |
24 |
| Section 6.12. Undertaking for Costs |
24 |
| Section 6.13. Rights and Remedies Cumulative |
24 |
| Section 6.14. Delay or Omission not Waiver |
24 |
| |
|
| ARTICLE 7 |
|
| TRUSTEE |
24 |
| |
|
| Section 7.01. General |
24 |
| Section 7.02. Certain Rights of Trustee |
24 |
| Section 7.03. Individual Rights of Trustee |
25 |
| Section 7.04. Trustee’s Disclaimer |
25 |
| Section 7.05. Notice of Default |
26 |
| Section 7.06. Reports by Trustee to Holders |
26 |
| Section 7.07. Compensation and Indemnity |
26 |
| Section 7.08. Replacement of Trustee |
26 |
| Section 7.09. Acceptance of Appointment by Successor |
27 |
| Section 7.10. Successor Trustee By Merger, Etc |
28 |
| Section 7.11. Eligibility |
28 |
| Section 7.12. Money Held in Trust |
28 |
| |
|
| ARTICLE 8 |
|
| SATISFACTION AND DISCHARGE OF INDENTURE; UNCLAIMED MONEYS |
28 |
| |
|
| Section 8.01. Satisfaction and Discharge of Indenture |
28 |
| Section 8.02. Application by Trustee of Funds Deposited for Payment of Securities |
29 |
| Section 8.03. Repayment of Moneys Held by Paying Agent |
29 |
| Section 8.04. Return of Moneys Held by Trustee and Paying Agent Unclaimed for Two Years |
29 |
| Section 8.05. Defeasance and Discharge of Indenture |
29 |
| Section 8.06. Defeasance of Certain Obligations |
30 |
| Section 8.07. Reinstatement |
31 |
| Section 8.08. Indemnity |
31 |
| Section 8.09. Excess Funds |
31 |
| Section 8.10. Qualifying Trustee |
31 |
| |
|
| ARTICLE 9 |
|
| AMENDMENTS, SUPPLEMENTS AND WAIVERS |
31 |
| |
|
| Section 9.01. Without Consent of Holders |
31 |
| Section 9.02. With Consent of Holders |
32 |
| Section 9.03. Revocation and Effect of Consent |
33 |
| Section 9.04. Notation on or Exchange of Securities |
33 |
| Section 9.05. Trustee to Sign Amendments, Etc |
33 |
| Section 9.06. Conformity with Trust Indenture Act |
33 |
| |
|
| ARTICLE 10 |
|
| MISCELLANEOUS |
33 |
| |
|
| Section 10.01. Trust Indenture Act of 1939 |
33 |
| Section 10.02. Notices |
34 |
| Section 10.03. Certificate and Opinion as to Conditions Precedent |
34 |
| Section 10.04. Statements Required in Certificate or Opinion |
34 |
| Section 10.05. Evidence of Ownership |
35 |
| Section 10.06. Rules by Trustee, Paying Agent or Registrar |
35 |
| Section 10.07. Payment Date Other Than a Business Day |
35 |
| Section 10.08. Governing Law |
35 |
| Section 10.09. No Adverse Interpretation of Other Agreements |
35 |
| Section 10.10. Successors |
35 |
| Section 10.11. Duplicate Originals |
35 |
| Section 10.12. Separability |
36 |
| Section 10.13. Table of Contents, Headings, Etc. |
36 |
| Section 10.14. Incorporators, Stockholders, Officers, and Directors of Company Exempt from Individual Liability |
36 |
| Section 10.15. Judgment Currency |
36 |
| |
|
| ARTICLE 11 |
|
| SUBORDINATION OF SECURITIES |
37 |
| |
|
| Section 11.01. Agreement to Subordinate |
37 |
| Section 11.02. Payments to Securityholders |
37 |
| Section 11.03. Subrogation of Securities |
38 |
| Section 11.04. Authorization by Securityholders |
38 |
| Section 11.05. Notice to Trustee |
39 |
| Section 11.06. Trustee’s Relation to Senior Indebtedness |
39 |
| Section 11.07. No Impairment of Subordination |
39 |
SUBORDINATED INDENTURE, dated
as of , 20 , between 60 Degrees Pharmaceuticals, Inc., a Delaware
corporation, as the Company, and , as Trustee.
RECITALS OF THE COMPANY
WHEREAS, the Company has
duly authorized the issue from time to time of its subordinated debentures, notes or other evidences of indebtedness to be issued in one
or more series (the “Securities”) up to such principal amount or amounts as may from time to time be authorized in
accordance with the terms of this Indenture and to provide, among other things, for the authentication, delivery and administration thereof,
the Company has duly authorized the execution and delivery of this Indenture; and
WHEREAS, all things necessary
to make this Indenture a valid indenture and agreement according to its terms have been done;
NOW, THEREFORE:
In consideration of the premises
and the purchases of the Securities by the holders thereof, the Company and the Trustee mutually covenant and agree for the equal and
proportionate benefit of the respective holders from time to time of the Securities or of any and all series thereof and of the coupons,
if any, appertaining thereto as follows:
ARTICLE 1
DEFINITIONS
AND INCORPORATION BY REFERENCE
Section 1.01. Definitions.
“Affiliate”
of any Person means any other Person directly or indirectly controlling or controlled by or under direct or indirect common control with
such Person. For the purposes of this definition, “control” (including, with correlative meanings, the terms “controlling”,
“controlled by” and “under common control with”) when used with respect to any Person means the possession, directly
or indirectly, of the power to direct or cause the direction of the management and policies of such Person, whether through the ownership
of voting securities, by contract or otherwise.
“Agent”
means any Registrar, Paying Agent, transfer agent or Authenticating Agent.
“Authorized Newspaper”
means a newspaper (which, in the case of The City of New York, will, if practicable, be The Wall Street Journal (Eastern Edition) and
in the case of London, will, if practicable, be the Financial Times (London Edition) and published in an official language of the country
of publication customarily published at least once a day for at least five days in each calendar week and of general circulation in The
City of New York or London, as applicable. If it shall be impractical in the opinion of the Trustee to make any publication of any notice
required hereby in an Authorized Newspaper, any publication or other notice in lieu thereof which is made or given with the approval of
the Trustee shall constitute a sufficient publication of such notice.
“Board Resolution”
means one or more resolutions of the board of directors of the Company or any authorized committee thereof, certified by the secretary
or an assistant secretary to have been duly adopted and to be in full force and effect on the date of certification, and delivered to
the Trustee.
“Business Day”
means any day, other than a Saturday or Sunday, that is neither a legal holiday nor a day on which banking institutions are authorized
or required by law or regulation to close in The City of New York, with respect to any Security the interest on which is based on the
offered quotations in the interbank Eurodollar market for dollar deposits in London, or with respect to Securities denominated in a specified
currency other than United States dollars, in the principal financial center of the country of the specified currency.
“Capital Lease”
means, with respect to any Person, any lease of any property which, in conformity with GAAP, is required to be capitalized on the balance
sheet of such Person.
“Commission”
means the Securities and Exchange Commission, as from time to time constituted, created under the Exchange Act or, if at any time after
the execution of this instrument such Commission is not existing and performing the duties now assigned to it under the Trust Indenture
Act, then the body performing such duties at such time.
“Company”
means the party named as such in the first paragraph of this Indenture until a successor replaces it pursuant to Article 5 of this Indenture
and thereafter means the successor.
“Corporate Trust
Office” means the office of the Trustee at which the corporate trust business of the Trustee shall, at any particular time,
be administered, which office is, at the date of this Indenture, located at Attention: .
“Currency Agreement”
means, with respect to any Person, any foreign exchange contract, currency swap agreement or other similar agreement or arrangement designed
to protect such Person or any of its Subsidiaries against fluctuations in currency values to or under which such Person or any of its
Subsidiaries is a party or a beneficiary on the date hereof or becomes a party or a beneficiary thereafter.
“Debt”
means, with respect to any Person at any date of determination (without duplication), (i) all indebtedness of such Person for borrowed
money, (ii) all obligations of such Person evidenced by bonds, debentures, notes or other similar instruments, (iii) all obligations of
such Person in respect of letters of credit or bankers’ acceptance or other similar instruments (or reimbursement obligations with
respect thereto), (iv) all obligations of such Person to pay the deferred purchase price of property or services, except Trade Payables,
(v) all obligations of such Person as lessee under Capital Leases, (vi) all Debt of others secured by a Lien on any asset of such Person,
whether or not such Debt is assumed by such Person; provided that, for purposes of determining the amount of any Debt of the type described
in this clause, if recourse with respect to such Debt is limited to such asset, the amount of such Debt shall be limited to the lesser
of the fair market value of such asset or the amount of such Debt, (vii) all Debt of others Guaranteed by such Person to the extent such
Debt is Guaranteed by such Person, (viii) all redeemable stock valued at the greater of its voluntary or involuntary liquidation preference
plus accrued and unpaid dividends and (ix) to the extent not otherwise included in this definition, all obligations of such Person under
Currency Agreements and Interest Rate Agreements.
“Default”
means any event that is, or after notice or passage of time or both would be, an Event of Default.
“Depositary”
means, with respect to the Securities of any series issuable or issued in the form of one or more Registered Global Securities, the Person
designated as Depositary by the Company pursuant to Section 2.03 until a successor Depositary shall have become such pursuant to the applicable
provisions of this Indenture, and thereafter “Depositary” shall mean or include each Person who is then a Depositary
hereunder, and if at any time there is more than one such Person, “Depositary” as used with respect to the Securities
of any such series shall mean the Depositary with respect to the Registered Global Securities of that series.
“Exchange Act”
means the Securities Exchange Act of 1934, as amended.
“GAAP”
means generally accepted accounting principles in the U.S. as in effect as of the date hereof applied on a basis consistent with the principles,
methods, procedures and practices employed in the preparation of the Company’s audited financial statements, including, without
limitation, those set forth in the opinions and pronouncements of the Accounting Principles Board of the American Institute of Certified
Public Accountants and statements and pronouncements of the Financial Accounting Standards Board or in such other statements by such other
entity as is approved by a significant segment of the accounting profession.
“Guarantee”
means any obligation, contingent or otherwise, of any Person directly or indirectly guaranteeing any Debt or other obligation of any other
Person and, without limiting the generality of the foregoing, any obligation, direct or indirect, contingent or otherwise, of such Person
(i) to purchase or pay (or advance or supply funds for the purchase or payment of) such Debt or other obligation of such other Person
(whether arising by virtue of partnership arrangements, or by agreement to keep well, to purchase assets, goods, securities or services,
to take-or-pay, or to maintain financial statement conditions or otherwise) or (ii) entered into for purposes of assuring in any other
manner the obligee of such Debt or other obligation of the payment thereof or to protect such obligee against loss in respect thereof
(in whole or in part); provided that the term “Guarantee” shall not include endorsements for collection or deposit
in the ordinary course of business. The term “Guarantee” used as a verb has a corresponding meaning.
“Holder”
or “Securityholder” means the registered holder of any Security with respect to Registered Securities and the bearer
of any Unregistered Security or any coupon appertaining thereto, as the case may be.
“Indenture”
means this Indenture as originally executed and delivered or as it may be amended or supplemented from time to time by one or more indentures
supplemental to this Indenture entered into pursuant to the applicable provisions of this Indenture and shall include the forms and terms
of the Securities of each series established as contemplated pursuant to Sections 2.01 and 2.03.
“Interest Rate Agreement”
means, with respect to any Person, any interest rate protection agreement, interest rate future agreement, interest rate option agreement,
interest rate swap agreement, interest rate cap agreement, interest rate collar agreement, interest rate hedge agreement or other similar
agreement or arrangement designed to protect such Person or any of its Subsidiaries against fluctuations in interest rates to or under
which such Person or any of its Subsidiaries is a party or a beneficiary on the date hereof or becomes a party or a beneficiary thereafter.
“Lien”
means, with respect to any property, any mortgage, lien, pledge, charge, security interest or encumbrance of any kind in respect of such
property. For purposes of this Indenture, the Company shall be deemed to own subject to a Lien any property which it has acquired or holds
subject to the interest of a vendor or lessor under any conditional sale agreement, capital lease or other title retention agreement relating
to such property.
“Officer”
means, with respect to the Company, the president, the chief executive officer the chief financial officer or the secretary.
“Officers’
Certificate” means a certificate signed in the name of the Company (i) by the president or chief executive officer and (ii)
by the chief financial officer or the secretary, and delivered to the Trustee. Each such certificate shall comply with Section 314 of
the Trust Indenture Act, if applicable, and include (except as otherwise expressly provided in this Indenture) the statements provided
in Section 10.04, if applicable.
“Opinion of Counsel”
means a written opinion signed by legal counsel, who may be an employee of or counsel to the Company, satisfactory to the Trustee. Each
such opinion shall comply with Section 314 of the Trust Indenture Act, if applicable, and include the statements provided in Section 10.04,
if and to the extent required thereby.
“Original issue
date” of any Security (or portion thereof) means the earlier of (a) the date of authentication of such Security or (b) the date
of any Security (or portion thereof) for which such Security was issued (directly or indirectly) on registration of transfer, exchange
or substitution.
“Original Issue
Discount Security” means any Security that provides for an amount less than the principal amount thereof to be due and payable
upon a declaration of acceleration of the maturity thereof pursuant to Section 6.02.
“Periodic Offering”
means an offering of Securities of a series from time to time, the specific terms of which Securities, including, without limitation,
the rate or rates of interest, if any, thereon, the stated maturity or maturities thereof and the redemption provisions, if any, with
respect thereto, are to be determined by the Company or its agents upon the issuance of such Securities.
“Person”
means an individual, a corporation, a partnership, a limited liability company, an association, a trust or any other entity or organization,
including a government or political subdivision or an agency or instrumentality thereof.
“Principal”
of a Security means the principal amount of, and, unless the context indicates otherwise, includes any premium payable on, the Security.
“Registered Global
Security” means a Security evidencing all or a part of a series of Registered Securities, issued to the Depositary for such
series in accordance with Section 2.02, and bearing the legend prescribed in Section 2.02.
“Registered Security”
means any Security registered on the Security Register (as defined in Section 2.05).
“Responsible Officer”
when used with respect to the Trustee, shall mean an officer of the Trustee in the Corporate Trust Office, having direct responsibility
for the administration of this Indenture, and also, with respect to a particular matter, any other officer to whom such matter is referred
because of such officer’s knowledge of and familiarity with the particular subject.
“Securities”
means any of the securities, as defined in the first paragraph of the recitals hereof, that are authenticated and delivered under this
Indenture and, unless the context indicates otherwise, shall include any coupon appertaining thereto.
“Securities Act”
means the Securities Act of 1933, as amended.
“Senior Indebtedness”
means the principal of (and premium, if any) and interest on all Debt of the Company whether created, incurred or assumed before, on or
after the date of this Indenture; provided that such Senior Indebtedness shall not include (i) Debt of the Company that, when incurred
and without respect to any election under Section 1111(b) of Title 11, U.S. Code, was without recourse and (ii) any other Debt of the
Company which by the terms of the instrument creating or evidencing the same are specifically designated as not being senior in right
of payment to the Securities; provided that Senior Indebtedness does not include any obligation to the Company or any Subsidiary.
“Subsidiary”
means, with respect to any Person, any corporation, association or other business entity of which a majority of the capital stock or other
ownership interests having ordinary voting power to elect a majority of the board of directors or other persons performing similar functions
are at the time directly or indirectly owned by such Person.
“Trade Payables”
means, with respect to any Person, any accounts payable or any other indebtedness or monetary obligation to trade creditors created, assumed
or Guaranteed by such Person or any of its Subsidiaries arising in the ordinary course of business in connection with the acquisition
of goods or services.
“Trustee”
means the party named as such in the first paragraph of this Indenture until a successor replaces it in accordance with the provisions
of Article 7 and thereafter shall mean or include each Person who is then a Trustee hereunder, and if at any time there is more than one
such Person, “Trustee” as used with respect to the Securities of any series shall mean the Trustee with respect to Securities
of that series.
“Trust Indenture
Act” means the Trust Indenture Act of 1939, as amended (15 U.S. Code §§ 77aaa-77bbbb), as it may be amended from time
to time.
“Unregistered Security”
means any Security other than a Registered Security.
“U.S. Government
Obligations” means securities that are (i) direct obligations of the United States of America for the payment of which its full
faith and credit is pledged or (ii) obligations of an agency or instrumentality of the United States of America the payment of which is
unconditionally guaranteed as a full faith and credit obligation by the United States of America, and shall also include a depository
receipt issued by a bank or trust company as custodian with respect to any such U.S. Government Obligation or a specific payment of interest
on or principal of any such U.S. Government Obligation held by such custodian for the account of the holder of a depository receipt; provided
that (except as required by law) such custodian is not authorized to make any deduction from the amount payable to the holder of such
depository receipt from any amount received by the custodian in respect of the U.S. Government Obligation or the specific payment of interest
on or principal of the U.S. Government Obligation evidenced by such depository receipt.
“Yield to Maturity”
means, as the context may require, the yield to maturity (i) on a series of Securities or (ii) if the Securities of a series are issuable
from time to time, on a Security of such series, calculated at the time of issuance of such series in the case of clause (i) or at the
time of issuance of such Security of such series in the case of clause (ii), or, if applicable, at the most recent redetermination of
interest on such series or on such Security, and calculated in accordance with the constant interest method or such other accepted financial
practice as is specified in the terms of such Security.
Section 1.02. Other
Definitions. Each of the following terms is defined in the section set forth opposite such term:
| Term | |
Section | |
| Authenticating Agent | |
| 2.02 | |
| Cash Transaction | |
| 7.03 | |
| Dollars | |
| 4.02 | |
| Event of Default | |
| 6.01 | |
| Judgment Currency | |
| 10.15 | (a) |
| mandatory sinking fund payment | |
| 3.05 | |
| optional sinking fund payment | |
| 3.05 | |
| Paying Agent | |
| 2.05 | |
| record date | |
| 2.04 | |
| Registrar | |
| 2.05 | |
| Required Currency | |
| 10.15 | (a) |
| Security Register | |
| 2.05 | |
| self-liquidating paper | |
| 7.03 | |
| sinking fund payment date | |
| 3.05 | |
| tranche | |
| 2.14 | |
Section 1.03. Incorporation
by Reference of Trust Indenture Act. Whenever this Indenture refers to a provision of the Trust Indenture Act, the provision is incorporated
by reference in and made a part of this Indenture. The following terms used in this Indenture that are defined by the Trust Indenture
Act have the following meanings:
“indenture securities”
means the Securities;
“indenture security
holder” means a Holder or a Securityholder;
“indenture to be
qualified” means this Indenture;
“indenture trustee”
or “institutional trustee” means the Trustee; and
“obligor”
on the indenture securities means the Company or any other obligor on the Securities.
All other terms used in this
Indenture that are defined by the Trust Indenture Act, defined by reference in the Trust Indenture Act to another statute or defined by
a rule of the Commission and not otherwise defined herein have the meanings assigned to them therein.
Section 1.04. Rules
of Construction. Unless the context otherwise requires:
(a) an accounting term not
otherwise defined has the meaning assigned to it in accordance with GAAP;
(b) words in the singular
include the plural, and words in the plural include the singular;
(c) “herein,”
“hereof” and other words of similar import refer to this Indenture as a whole and not to any particular Article, Section or
other subdivision;
(d) all references to Sections
or Articles refer to Sections or Articles of this Indenture unless otherwise indicated; and
(e) use of masculine, feminine
or neuter pronouns should not be deemed a limitation, and the use of any such pronouns should be construed to include, where appropriate,
the other pronouns.
ARTICLE 2
THE SECURITIES
Section 2.01. Form
and Dating. The Securities of each series shall be substantially in such form or forms (not inconsistent with this Indenture) as shall
be established by or pursuant to one or more Board Resolutions or in one or more indentures supplemental hereto, in each case with such
appropriate insertions, omissions, substitutions and other variations as are required or permitted by this Indenture and may have imprinted
or otherwise reproduced thereon such legend or legends or endorsements, not inconsistent with the provisions of this Indenture, as may
be required to comply with any law, or with any rules of any securities exchange or usage, all as may be determined by the officers executing
such Securities as evidenced by their execution of the Securities. Unless otherwise so established, Unregistered Securities shall have
coupons attached.
Section 2.02. Execution
And Authentication. Two Officers shall execute the Securities and one Officer shall execute the coupons appertaining thereto for the
Company by facsimile or manual signature in the name and on behalf of the Company. The seal of the Company, if any, shall be reproduced
on the Securities. If an Officer whose signature is on a Security or coupon appertaining thereto no longer holds that office at the time
the Security is authenticated, the Security and such coupon shall nevertheless be valid.
The Trustee, at the expense
of the Company, may appoint an authenticating agent (the “Authenticating Agent”) to authenticate Securities. The Authenticating
Agent may authenticate Securities whenever the Trustee may do so. Each reference in this Indenture to authentication by the Trustee includes
authentication by such Authenticating Agent.
A Security and the coupons
appertaining thereto shall not be valid until the Trustee or Authenticating Agent manually signs the certificate of authentication on
the Security or on the Security to which such coupon appertains by an authorized officer. The signature shall be conclusive evidence that
the Security or the Security to which the coupon appertains has been authenticated under this Indenture.
At any time and from time
to time after the execution and delivery of this Indenture, the Company may deliver Securities of any series having attached thereto appropriate
coupons, if any, executed by the Company to the Trustee for authentication together with the applicable documents referred to below in
this Section, and the Trustee shall thereupon authenticate and deliver such Securities to or upon the written order of the Company. In
authenticating any Securities of a series, the Trustee shall be entitled to receive prior to the authentication of any Securities of such
series, and (subject to Article 7) shall be fully protected in relying upon, unless and until such documents have been superseded or revoked:
(a) any Board Resolution
and/or executed supplemental indenture referred to in Sections 2.01 and 2.03 by or pursuant to which the forms and terms of the Securities
of that series were established;
(b) an Officers’ Certificate
setting forth the form or forms and terms of the Securities, stating that the form or forms and terms of the Securities of such series
have been, or, in the case of a Periodic Offering, will be when established in accordance with such procedures as shall be referred to
therein, established in compliance with this Indenture; and
(c) an Opinion of Counsel
substantially to the effect that the form or forms and terms of the Securities of such series have been, or, in the case of a Periodic
Offering, will be when established in accordance with such procedures as shall be referred to therein, established in compliance with
this Indenture and that the supplemental indenture, to the extent applicable, and Securities have been duly authorized and, if executed
and authenticated in accordance with the provisions of the Indenture and delivered to and duly paid for by the purchasers thereof on the
date of such opinion, would be entitled to the benefits of the Indenture and would be valid and binding obligations of the Company, enforceable
against the Company in accordance with their respective terms, subject to bankruptcy, insolvency, reorganization, receivership, moratorium
and other similar laws affecting creditors’ rights generally, general principles of equity, and covering such other matters as shall
be specified therein and as shall be reasonably requested by the Trustee.
The Trustee shall not be
required to authenticate such Securities if the issue of such Securities pursuant to this Indenture will affect the Trustee’s own
rights, duties or immunities under the Securities and this Indenture or otherwise in a manner which is not reasonably acceptable to the
Trustee.
Notwithstanding the provisions
of Sections 2.01 and 2.02, if, in connection with a Periodic Offering, all Securities of a series are not to be originally issued at one
time, it shall not be necessary to deliver the Board Resolution otherwise required pursuant to Section 2.01 or the written order, Officers’
Certificate and Opinion of Counsel otherwise required pursuant to Section 2.02 at or prior to the authentication of each Security of such
series if such documents are delivered at or prior to the authentication upon original issuance of the first Security of such series to
be issued.
With respect to Securities
of a series offered in a Periodic Offering, the Trustee may rely, as to the authorization by the Company of any of such Securities, the
forms and terms thereof and the legality, validity, binding effect and enforceability thereof, upon the Opinion of Counsel and the other
documents delivered pursuant to Sections 2.01 and 2.02, as applicable, in connection with the first authentication of Securities of such
series.
If the Company shall establish
pursuant to Section 2.03 that the Securities of a series or a portion thereof are to be issued in the form of one or more Registered Global
Securities, then the Company shall execute and the Trustee shall authenticate and deliver one or more Registered Global Securities that
(i) shall represent and shall be denominated in an amount equal to the aggregate principal amount of all of the Securities of such series
issued in such form and not yet cancelled, (ii) shall be registered in the name of the Depositary for such Registered Global Security
or Securities or the nominee of such Depositary, (iii) shall be delivered by the Trustee to such Depositary or its custodian or pursuant
to such Depositary’s instructions and (iv) shall bear a legend substantially to the following effect: “Unless and until it
is exchanged in whole or in part for Securities in definitive registered form, this Security may not be transferred except as a whole
by the Depositary to the nominee of the Depositary or by a nominee of the Depositary to the Depositary or another nominee of the Depositary
or by the Depositary or any such nominee to a successor Depositary or a nominee of such successor Depositary.”
Section 2.03. Amount
Unlimited; Issuable in Series. The aggregate principal amount of Securities which may be authenticated and delivered under this Indenture
is unlimited.
The Securities may be issued
in one or more series and shall be subordinated to the Senior Indebtedness pursuant to the provisions of Article 11 hereof. There shall
be established in or pursuant to Board Resolution or one or more indentures supplemental hereto, prior to the initial issuance of Securities
of any series, subject to the last sentence of this Section 2.03,
(a) the designation of the
Securities of the series, which shall distinguish the Securities of the series from the Securities of all other series;
(b) any limit upon the aggregate
principal amount of the Securities of the series that may be authenticated and delivered under this Indenture and any limitation on the
ability of the Company to increase such aggregate principal amount after the initial issuance of the Securities of that series (except
for Securities authenticated and delivered upon registration of transfer of, or in exchange for, or in lieu of, or upon redemption of,
other Securities of the series pursuant hereto);
(c) the date or dates on
which the principal of the Securities of the series is payable (which date or dates may be fixed or extendible);
(d) the rate or rates (which
may be fixed or variable) per annum at which the Securities of the series shall bear interest, if any, the date or dates from which such
interest shall accrue, on which such interest shall be payable and (in the case of Registered Securities) on which a record shall be taken
for the determination of Holders to whom interest is payable and/or the method by which such rate or rates or date or dates shall be determined;
(e) if other than as provided
in Section 4.02, the place or places where the principal of and any interest on Securities of the series shall be payable, any Registered
Securities of the series may be surrendered for exchange, notices, demands to or upon the Company in respect of the Securities of the
series and this Indenture may be served and notice to Holders may be published;
(f) the right, if any, of
the Company to redeem Securities of the series, in whole or in part, at its option and the period or periods within which, the price or
prices at which and any terms and conditions upon which Securities of the series may be so redeemed, pursuant to any sinking fund or otherwise;
(g) the obligation, if any,
of the Company to redeem, purchase or repay Securities of the series pursuant to any mandatory redemption, sinking fund or analogous provisions
or at the option of a Holder thereof and the price or prices at which and the period or periods within which and any of the terms and
conditions upon which Securities of the series shall be redeemed, purchased or repaid, in whole or in part, pursuant to such obligation;
(h) if other than denominations
of $1,000 and any integral multiple thereof, the denominations in which Securities of the series shall be issuable;
(i) if other than the principal
amount thereof, the portion of the principal amount of Securities of the series which shall be payable upon declaration of acceleration
of the maturity thereof;
(j) if other than the coin
or currency in which the Securities of the series are denominated, the coin or currency in which payment of the principal of or interest
on the Securities of the series shall be payable or if the amount of payments of principal of and/or interest on the Securities of the
series may be determined with reference to an index based on a coin or currency other than that in which the Securities of the series
are denominated, the manner in which such amounts shall be determined;
(k) if other than the currency
of the United States of America, the currency or currencies, including composite currencies, in which payment of the Principal of and
interest on the Securities of the series shall be payable, and the manner in which any such currencies shall be valued against other currencies
in which any other Securities shall be payable;
(l) whether the Securities
of the series or any portion thereof will be issuable as Registered Securities (and if so, whether such Securities will be issuable as
Registered Global Securities) or Unregistered Securities (with or without coupons) (and if so, whether such Securities will be issued
in temporary or permanent global form), or any combination of the foregoing, any restrictions applicable to the offer, sale or delivery
of Unregistered Securities or the payment of interest thereon and, if other than as provided herein, the terms upon which Unregistered
Securities of any series may be exchanged for Registered Securities of such series and vice versa;
(m) whether and under what
circumstances the Company will pay additional amounts on the Securities of the series held by a person who is not a U.S. person in respect
of any tax, assessment or governmental charge withheld or deducted and, if so, whether the Company will have the option to redeem such
Securities rather than pay such additional amounts;
(n) if the Securities of
the series are to be issuable in definitive form (whether upon original issue or upon exchange of a temporary Security of such series)
only upon receipt of certain certificates or other documents or satisfaction of other conditions, the form and terms of such certificates,
documents or conditions;
(o) any trustees, depositaries,
authenticating or paying agents, transfer agents or the registrar or any other agents with respect to the Securities of the series;
(p) provisions, if any, for
the defeasance of the Securities of the series (including provisions permitting defeasance of less than all Securities of the series),
which provisions may be in addition to, in substitution for, or in modification of (or any combination of the foregoing) the provisions
of Article 8;
(q) if the Securities of
the series are issuable in whole or in part as one or more Registered Global Securities or Unregistered Securities in global form, the
identity of the Depositary or common Depositary for such Registered Global Security or Securities or Unregistered Securities in global
form;
(r) any other Events of Default
or covenants with respect to the Securities of the series; and
(s) any other terms of the
Securities of the series (which terms shall not be inconsistent with the provisions of this Indenture).
All Securities of any one
series and coupons, if any, appertaining thereto shall be substantially identical, except in the case of Registered Securities as to date
and denomination, except in the case of any Periodic Offering and except as may otherwise be provided by or pursuant to the Board Resolution
referred to above or as set forth in any such indenture supplemental hereto. All Securities of any one series need not be issued at the
same time and may be issued from time to time, consistent with the terms of this Indenture, if so provided by or pursuant to such Board
Resolution or in any such indenture supplemental hereto and any forms and terms of Securities to be issued from time to time may be completed
and established from time to time prior to the issuance thereof by procedures described in such Board Resolution or supplemental indenture.
Unless otherwise expressly
provided with respect to a series of Securities, the aggregate principal amount of a series of Securities may be increased and additional
Securities of such series may be issued up to the maximum aggregate principal amount authorized with respect to such series as increased.
Section 2.04. Denomination
and Date of Securities; Payments of Interest. The Securities of each series shall be issuable as Registered Securities or Unregistered
Securities in denominations established as contemplated by Section 2.03 or, if not so established with respect to Securities of any series,
in denominations of $1,000 and any integral multiple thereof. The Securities of each series shall be numbered, lettered or otherwise distinguished
in such manner or in accordance with such plan as the Officers of the Company executing the same may determine, as evidenced by their
execution thereof.
Unless otherwise specified
with respect to a series of Securities, each Security shall be dated the date of its authentication. The Securities of each series shall
bear interest, if any, from the date, and such interest and shall be payable on the dates, established as contemplated by Section 2.03.
The person in whose name
any Registered Security of any series is registered at the close of business on any record date applicable to a particular series with
respect to any interest payment date for such series shall be entitled to receive the interest, if any, payable on such interest payment
date notwithstanding any transfer or exchange of such Registered Security subsequent to the record date and prior to such interest payment
date, except if and to the extent the Company shall default in the payment of the interest due on such interest payment date for such
series, in which case the provisions of Section 2.13 shall apply. The term “record date” as used with respect to any
interest payment date (except a date for payment of defaulted interest) for the Securities of any series shall mean the date specified
as such in the terms of the Registered Securities of such series established as contemplated by Section 2.03, or, if no such date is so
established, the fifteenth day next preceding such interest payment date, whether or not such record date is a Business Day.
Section 2.05. Registrar
and Paying Agent; Agents Generally. The Company shall maintain an office or agency where Securities may be presented for registration,
registration of transfer or for exchange (the “Registrar”) and an office or agency where Securities may be presented
for payment (the “Paying Agent”), which shall be in the Borough of Manhattan, The City of New York. The Company shall
cause the Registrar to keep a register of the Registered Securities and of their registration, transfer and exchange (the “Security
Register”). The Company may have one or more additional Paying Agents or transfer agents with respect to any series.
The Company shall enter into
an appropriate agency agreement with any Agent not a party to this Indenture. The agreement shall implement the provisions of this Indenture
and the Trust Indenture Act that relate to such Agent. The Company shall give prompt written notice to the Trustee of the name and address
of any Agent and any change in the name or address of an Agent. If the Company fails to maintain a Registrar or Paying Agent, the Trustee
shall act as such. The Company may remove any Agent upon written notice to such Agent and the Trustee; provided that
no such removal shall become effective until (i) the acceptance of an appointment by a successor Agent to such Agent as evidenced by an
appropriate agency agreement entered into by the Company and such successor Agent and delivered to the Trustee or (ii) notification to
the Trustee that the Trustee shall serve as such Agent until the appointment of a successor Agent in accordance with clause (i) of this
proviso. The Company or any affiliate of the Company may act as Paying Agent or Registrar; provided that neither the
Company nor an affiliate of the Company shall act as Paying Agent in connection with the defeasance of the Securities or the discharge
of this Indenture under Article 8.
The Company initially appoints
the Trustee as Registrar, Paying Agent and Authenticating Agent. If, at any time, the Trustee is not the Registrar, the Registrar shall
make available to the Trustee ten days prior to each interest payment date and at such other times as the Trustee may reasonably request
the names and addresses of the Holders as they appear in the Security Register.
Section 2.06. Paying
Agent to Hold Money in Trust. Not later than 10:00 a.m. New York City time on each due date or, in the case of Unregistered Securities,
10:00 a.m. New York City time on the Business Day prior to the due date, of any Principal or interest on any Securities, the Company shall
deposit with the Paying Agent money in immediately available funds sufficient to pay such Principal or interest. The Company shall require
each Paying Agent other than the Trustee to agree in writing that such Paying Agent shall hold in trust for the benefit of the Holders
of such Securities or the Trustee all money held by the Paying Agent for the payment of Principal of and interest on such Securities and
shall promptly notify the Trustee of any default by the Company in making any such payment. The Company at any time may require a Paying
Agent to pay all money held by it to the Trustee and account for any funds disbursed, and the Trustee may at any time during the continuance
of any payment default, upon written request to a Paying Agent, require such Paying Agent to pay all money held by it to the Trustee and
to account for any funds disbursed. Upon doing so, the Paying Agent shall have no further liability for the money so paid over to the
Trustee. If the Company or any affiliate of the Company acts as Paying Agent, it will, on or before each due date of any Principal of
or interest on any Securities, segregate and hold in a separate trust fund for the benefit of the Holders thereof a sum of money sufficient
to pay such Principal or interest so becoming due until such sum of money shall be paid to such Holders or otherwise disposed of as provided
in this Indenture, and will promptly notify the Trustee in writing of its action or failure to act as required by this Section.
Section 2.07. Transfer
and Exchange. Unregistered Securities (except for any temporary global Unregistered Securities) and coupons (except for coupons attached
to any temporary global Unregistered Securities) shall be transferable by delivery.
At the option of the Holder
thereof, Registered Securities of any series (other than a Registered Global Security, except as set forth below) may be exchanged for
a Registered Security or Registered Securities of such series and tenor having authorized denominations and an equal aggregate principal
amount, upon surrender of such Registered Securities to be exchanged at the agency of the Company that shall be maintained for such purpose
in accordance with Section 2.05 and upon payment, if the Company shall so require, of the charges hereinafter provided. If the Securities
of any series are issued in both registered and unregistered form, except as otherwise established pursuant to Section 2.03, at the option
of the Holder thereof, Unregistered Securities of any series may be exchanged for Registered Securities of such series and tenor having
authorized denominations and an equal aggregate principal amount, upon surrender of such Unregistered Securities to be exchanged at the
agency of the Company that shall be maintained for such purpose in accordance with Section 4.02, with, in the case of Unregistered Securities
that have coupons attached, all unmatured coupons and all matured coupons in default thereto appertaining, and upon payment, if the Company
shall so require, of the charges hereinafter provided. At the option of the Holder thereof, if Unregistered Securities of any series,
maturity date, interest rate and original issue date are issued in more than one authorized denomination, except as otherwise established
pursuant to Section 2.03, such Unregistered Securities may be exchanged for Unregistered Securities of such series and tenor having authorized
denominations and an equal aggregate principal amount, upon surrender of such Unregistered Securities to be exchanged at the agency of
the Company that shall be maintained for such purpose in accordance with Section 4.02, with, in the case of Unregistered Securities that
have coupons attached, all unmatured coupons and all matured coupons in default thereto appertaining, and upon payment, if the Company
shall so require, of the charges hereinafter provided. Registered Securities of any series may not be exchanged for Unregistered Securities
of such series. Whenever any Securities are so surrendered for exchange, the Company shall execute, and the Trustee shall authenticate
and deliver, the Securities which the Holder making the exchange is entitled to receive.
Upon surrender for registration
of transfer of any Registered Security of a series at the agency of the Company that shall be maintained for that purpose in accordance
with Section 2.05 and upon payment, if the Company shall so require, of the charges hereinafter provided, the Company shall execute, and
the Trustee shall authenticate and deliver, in the name of the designated transferee or transferees, one or more new Registered Securities
of the same series, of any authorized denominations and of like tenor and aggregate principal amount.
All Registered Securities
presented for registration of transfer, exchange, redemption or payment shall be duly endorsed by, or be accompanied by a written instrument
or instruments of transfer in form satisfactory to the Company and the Trustee duly executed by, the holder or his attorney duly authorized
in writing.
The Company may require payment
of a sum sufficient to cover any tax or other governmental charge that may be imposed in connection with any exchange or registration
of transfer of Securities. No service charge shall be made for any such transaction.
Notwithstanding any other
provision of this Section 2.07, unless and until it is exchanged in whole or in part for Securities in definitive registered form, a Registered
Global Security representing all or a portion of the Securities of a series may not be transferred except as a whole by the Depositary
for such series to a nominee of such Depositary or by a nominee of such Depositary to such Depositary or another nominee of such Depositary
or by such Depositary or any such nominee to a successor Depositary for such series or a nominee of such successor Depositary.
If at any time the Depositary
for any Registered Global Securities of any series notifies the Company that it is unwilling or unable to continue as Depositary for such
Registered Global Securities or if at any time the Depositary for such Registered Global Securities shall no longer be eligible under
applicable law, the Company shall appoint a successor Depositary eligible under applicable law with respect to such Registered Global
Securities. If a successor Depositary eligible under applicable law for such Registered Global Securities is not appointed by the Company
within 90 days after the Company receives such notice or becomes aware of such ineligibility, the Company will execute, and the Trustee,
upon receipt of the Company’s order for the authentication and delivery of definitive Registered Securities of such series and tenor,
will authenticate and deliver Registered Securities of such series and tenor, in any authorized denominations, in an aggregate principal
amount equal to the principal amount of such Registered Global Securities, in exchange for such Registered Global Securities.
The Company may at any time
and in its sole discretion and subject to the procedures of the Depositary determine that any Registered Global Securities of any series
shall no longer be maintained in global form. In such event the Company will execute, and the Trustee, upon receipt of the Company’s
order for the authentication and delivery of definitive Registered Securities of such series and tenor, will authenticate and deliver,
Registered Securities of such series and tenor in any authorized denominations, in an aggregate principal amount equal to the principal
amount of such Registered Global Securities, in exchange for such Registered Global Securities.
Any time the Registered Securities
of any series are not in the form of Registered Global Securities pursuant to the preceding two paragraphs, the Company agrees to supply
the Trustee with a reasonable supply of certificated Registered Securities without the legend required by Section 2.02 and the Trustee
agrees to hold such Registered Securities in safekeeping until authenticated and delivered pursuant to the terms of this Indenture.
If established by the Company
pursuant to Section 2.03 with respect to any Registered Global Security, the Depositary for such Registered Global Security may surrender
such Registered Global Security in exchange in whole or in part for Registered Securities of the same series and tenor in definitive registered
form on such terms as are acceptable to the Company and such Depositary. Thereupon, the Company shall execute, and the Trustee shall authenticate
and deliver, without service charge,
(a) to the Person specified
by such Depositary new Registered Securities of the same series and tenor, of any authorized denominations as requested by such Person,
in an aggregate principal amount equal to and in exchange for such Person’s beneficial interest in the Registered Global Security;
and
(b) to such Depositary a
new Registered Global Security in a denomination equal to the difference, if any, between the principal amount of the surrendered Registered
Global Security and the aggregate principal amount of Registered Securities authenticated and delivered pursuant to clause (a) above.
Registered Securities issued
in exchange for a Registered Global Security pursuant to this Section 2.07 shall be registered in such names and in such authorized denominations
as the Depositary for such Registered Global Security, pursuant to instructions from its direct or indirect participants or otherwise,
shall instruct the Trustee or an agent of the Company or the Trustee. The Trustee or such agent shall deliver such Securities to or as
directed by the Persons in whose names such Securities are so registered.
All Securities issued upon
any transfer or exchange of Securities shall be valid obligations of the Company, evidencing the same debt, and entitled to the same benefits
under this Indenture, as the Securities surrendered upon such transfer or exchange.
Notwithstanding anything
herein or in the forms or terms of any Securities to the contrary, none of the Company, the Trustee or any agent of the Company or the
Trustee shall be required to exchange any Unregistered Security for a Registered Security if such exchange would result in adverse Federal
income tax consequences to the Company (such as, for example, the inability of the Company to deduct from its income, as computed for
Federal income tax purposes, the interest payable on the Unregistered Securities) under then applicable United States Federal income tax
laws. The Trustee and any such agent shall be entitled to rely on an Officers’ Certificate or an Opinion of Counsel in determining
such result.
The Registrar shall not be
required (i) to issue, authenticate, register the transfer of or exchange Securities of any series for a period of 15 days before a selection
of such Securities to be redeemed or (ii) to register the transfer of or exchange any Security selected for redemption in whole or in
part.
Section 2.08. Replacement
Securities. If any mutilated Security or a Security with a mutilated coupon appertaining to it is surrendered to the Trustee, the
Company shall execute and the Trustee shall authenticate and deliver, in exchange for such mutilated Security or in exchange for the Security
to which a mutilated coupon appertains, a new Security of the same series and of like tenor and principal amount and bearing a number
not contemporaneously outstanding, with coupons corresponding to the coupons, if any, appertaining to such mutilated Security or to the
Security to which such mutilated coupon appertains.
If there shall be delivered
to the Company and the Trustee (i) evidence to their satisfaction of the destruction, loss or theft of any Security or coupon and (ii)
such security or indemnity as may be required by them to save each of them and any agent of any of them harmless, then, in the absence
of notice to the Company or the Trustee that such Security or coupon has been acquired by a bona fide purchaser, the Company shall execute
and the Trustee shall authenticate and deliver, in lieu of any such destroyed, lost or stolen Security or in exchange for the Security
to which a destroyed, lost or stolen coupon appertains (with all appurtenant coupons not destroyed, lost or stolen), a new Security of
the same series and of like tenor and principal amount and bearing a number not contemporaneously outstanding, with coupons corresponding
to the coupons, if any, appertaining to such destroyed, lost or stolen Security or to the Security to which such destroyed, lost or stolen
coupon appertains.
In case any such mutilated,
destroyed, lost or stolen Security or coupon has become or is about to become due and payable, the Company in its discretion may, instead
of issuing a new Security, pay such Security or coupon (without surrender thereof except in the case of a mutilated Security or coupon)
if the applicant for such payment shall furnish to the Company and the Trustee such security or indemnity as may be required by them to
save each of them and any agent of any of them harmless, and in the case of destruction, loss or theft, evidence satisfactory to the Company
and the Trustee and any agent of them of the destruction, loss or theft of such Security and the ownership thereof; provided,
however, that the Principal of and any interest on Unregistered Securities shall, except as otherwise provided in Section 4.02, be
payable only at an office or agency located outside the United States.
Upon the issuance of any
new Security under this Section, the Company may require payment of a sum sufficient to cover any tax or other governmental charge that
may be imposed in relation thereto and any other expenses (including the fees and expenses of the Trustee) connected therewith.
Every new Security of any
series, with its coupons, if any, issued pursuant to this Section in lieu of any destroyed, lost or stolen Security or in exchange for
any mutilated Security, or in exchange for a Security to which a mutilated, destroyed, lost or stolen coupon appertains, shall constitute
an original additional contractual obligation of the Company, whether or not the mutilated, destroyed, lost or stolen Security and its
coupons, if any, or the mutilated, destroyed, lost or stolen coupon shall be at any time enforceable by anyone, and any such new Security
and coupons, if any, shall be entitled to all the benefits of this Indenture equally and proportionately with any and all other Securities
of that series and their coupons, if any, duly issued hereunder.
The provisions of this Section
are exclusive and shall preclude (to the extent lawful) any other rights and remedies with respect to the replacement or payment of mutilated,
destroyed, lost or stolen Securities or coupons.
Section 2.09. Outstanding
Securities. Securities outstanding at any time are all Securities that have been authenticated by the Trustee except for those cancelled
by it, those delivered to it for cancellation, those described in this Section as not outstanding and those that have been defeased pursuant
to Section 8.05.
If a Security is replaced
pursuant to Section 2.08, it ceases to be outstanding unless and until the Trustee and the Company receive proof satisfactory to them
that the replaced Security is held by a holder in due course.
If the Paying Agent (other
than the Company or an affiliate of the Company) holds on the maturity date or any redemption date or date for repurchase of the Securities
money sufficient to pay Securities payable or to be redeemed or repurchased on that date, then on and after that date such Securities
cease to be outstanding and interest on them shall cease to accrue.
A Security does not cease
to be outstanding because the Company or one of its affiliates holds such Security, provided, however, that, in determining
whether the Holders of the requisite principal amount of the outstanding Securities have given any request, demand, authorization, direction,
notice, consent or waiver hereunder, Securities owned by the Company or any affiliate of the Company shall be disregarded and deemed not
to be outstanding, except that, in determining whether the Trustee shall be protected in relying upon any such request, demand, authorization,
direction, notice, consent or waiver, only Securities as to which a Responsible Officer of the Trustee has received written notice to
be so owned shall be so disregarded. Any Securities so owned which are pledged by the Company, or by any affiliate of the Company, as
security for loans or other obligations, otherwise than to another such affiliate of the Company, shall be deemed to be outstanding, if
the pledgee is entitled pursuant to the terms of its pledge agreement and is free to exercise in its or his discretion the right to vote
such securities, uncontrolled by the Company or by any such affiliate.
Section 2.10. Temporary
Securities. Until definitive Securities of any series are ready for delivery, the Company may prepare and the Trustee shall authenticate
temporary Securities of such series. Temporary Securities of any series shall be substantially in the form of definitive Securities of
such series but may have insertions, substitutions, omissions and other variations determined to be appropriate by the Officers executing
the temporary Securities, as evidenced by their execution of such temporary Securities. If temporary Securities of any series are issued,
the Company will cause definitive Securities of such series to be prepared without unreasonable delay. After the preparation of definitive
Securities of any series, the temporary Securities of such series shall be exchangeable for definitive Securities of such series and tenor
upon surrender of such temporary Securities at the office or agency of the Company designated for such purpose pursuant to Section 4.02,
without charge to the Holder. Upon surrender for cancellation of any one or more temporary Securities of any series the Company shall
execute and the Trustee shall authenticate and deliver in exchange therefor a like principal amount of definitive Securities of such series
and tenor and authorized denominations. Until so exchanged, the temporary Securities of any series shall be entitled to the same benefits
under this Indenture as definitive Securities of such series.
Section 2.11. Cancellation. The
Company at any time may deliver to the Trustee for cancellation any Securities previously authenticated and delivered hereunder which
the Company may have acquired in any manner whatsoever, and may deliver to the Trustee for cancellation any Securities previously authenticated
hereunder which the Company has not issued and sold. The Registrar, any transfer agent and the Paying Agent shall forward to the Trustee
any Securities surrendered to them for transfer, exchange or payment. The Trustee shall cancel and dispose of in accordance with its customary
procedures all Securities surrendered for transfer, exchange, payment or cancellation and shall deliver a certificate of disposition to
the Company. The Company may not issue new Securities to replace Securities it has paid in full or delivered to the Trustee for cancellation.
Section 2.12. CUSIP
Numbers. The Company in issuing the Securities may use “CUSIP” and “CINS” numbers (if then generally
in use), and the Trustee shall use CUSIP numbers or CINS numbers, as the case may be, in notices of redemption or exchange as a convenience
to Holders and no representation shall be made as to the correctness of such numbers either as printed on the Securities or as contained
in any notice of redemption or exchange.
Section 2.13. Defaulted
Interest. If the Company defaults in a payment of interest on the Registered Securities, it shall pay, or shall deposit with
the Paying Agent money in immediately available funds sufficient to pay, the defaulted interest plus (to the extent lawful) any interest
payable on the defaulted interest (as may be specified in the terms thereof, established pursuant to Section 2.03) to the Persons who
are Holders on a subsequent special record date, which shall mean the 15th day next preceding the date fixed by the Company for the payment
of defaulted interest, whether or not such day is a Business Day. At least 15 days before such special record date, the Company shall
mail to each Holder of such Registered Securities and to the Trustee a notice that states the special record date, the payment date and
the amount of defaulted interest to be paid.
Section 2.14. Series
May Include Tranches. A series of Securities may include one or more tranches (each a “tranche”) of Securities,
including Securities issued in a Periodic Offering. The Securities of different tranches may have one or more different terms, including
authentication dates and public offering prices, but all the Securities within each such tranche shall have identical terms, including
authentication date and public offering price. Notwithstanding any other provision of this Indenture, with respect to Sections 2.02 (other
than the fourth, sixth and seventh paragraphs thereof) through 2.04, 2.07, 2.08, 2.10, 3.01 through 3.05, 4.02, 6.01 through 6.14, 8.01
through 8.07, 9.02 and Section 10.07, if any series of Securities includes more than one tranche, all provisions of such sections applicable
to any series of Securities shall be deemed equally applicable to each tranche of any series of Securities in the same manner as though
originally designated a series unless otherwise provided with respect to such series or tranche pursuant to Section 2.03. In particular,
and without limiting the scope of the next preceding sentence, any of the provisions of such sections which provide for or permit action
to be taken with respect to a series of Securities shall also be deemed to provide for and permit such action to be taken instead only
with respect to Securities of one or more tranches within that series (and such provisions shall be deemed satisfied thereby), even if
no comparable action is taken with respect to Securities in the remaining tranches of that series.
ARTICLE 3
REDEMPTION
Section 3.01. Applicability
of Article. The provisions of this Article shall be applicable to the Securities of any series which are redeemable before their
maturity or to any sinking fund for the retirement of Securities of a series except as otherwise specified as contemplated by Section
2.03 for Securities of such series.
Section 3.02. Notice
of Redemption; Partial Redemptions. Notice of redemption to the Holders of Registered Securities of any series to be redeemed
as a whole or in part at the option of the Company shall be given by mailing notice of such redemption by first class mail, postage prepaid,
at least 30 days and not more than 60 days prior to the date fixed for redemption to such Holders of Registered Securities of such series
at their last addresses as they shall appear upon the registry books. Notice of redemption to the Holders of Unregistered Securities of
any series to be redeemed as a whole or in part who have filed their names and addresses with the Trustee pursuant to Section 313(c)(2)
of the Trust Indenture Act, shall be given by mailing notice of such redemption, by first class mail, postage prepaid, at least 30 days
and not more than 60 days prior to the date fixed for redemption, to such Holders at such addresses as were so furnished to the Trustee
(and, in the case of any such notice given by the Company, the Trustee shall make such information available to the Company for such purpose).
Notice of redemption to all other Holders of Unregistered Securities of any series to be redeemed as a whole or in part shall be published
in an Authorized Newspaper in The City of New York or with respect to any Security the interest on which is based on the offered quotations
in the interbank Eurodollar market for dollar deposits in an Authorized Newspaper in London, in each case, once in each of three successive
calendar weeks, the first publication to be not less than 30 days nor more than 60 days prior to the date fixed for redemption. Any notice
which is mailed or published in the manner herein provided shall be conclusively presumed to have been duly given, whether or not the
Holder receives the notice. Failure to give notice by mail, or any defect in the notice to the Holder of any Security of a series designated
for redemption as a whole or in part shall not affect the validity of the proceedings for the redemption of any other Security of such
series.
The notice of redemption
to each such Holder shall specify the principal amount of each Security of such series held by such Holder to be redeemed, the CUSIP numbers
of the Securities to be redeemed, the date fixed for redemption, the redemption price, or if not then ascertainable, the manner of calculation
thereof, the place or places of payment, that payment will be made upon presentation and surrender of such Securities and, in the case
of Securities with coupons attached thereto, of all coupons appertaining thereto maturing after the date fixed for redemption, that such
redemption is pursuant to the mandatory or optional sinking fund, or both, if such be the case, that interest accrued to the date fixed
for redemption will be paid as specified in such notice and that on and after said date interest thereon or on the portions thereof to
be redeemed will cease to accrue. In case any Security of a series is to be redeemed in part only, the notice of redemption shall state
the portion of the principal amount thereof to be redeemed and shall state that on and after the date fixed for redemption, upon surrender
of such Security, a new Security or Securities of such series and tenor in principal amount equal to the unredeemed portion thereof will
be issued.
The notice of redemption
of Securities of any series to be redeemed at the option of the Company shall be given by the Company or, at the Company’s request,
by the Trustee in the name and at the expense of the Company.
On or before 10:00 a.m. New
York City time on the redemption date or, in the case of Unregistered Securities, on or before 10:00 a.m. New York City time on the Business
Day prior to the redemption date specified in the notice of redemption given as provided in this Section, the Company will deposit with
the Trustee or with one or more Paying Agents (or, if the Company is acting as its own Paying Agent, set aside, segregate and hold in
trust as provided in Section 2.06) an amount of money sufficient to redeem on the redemption date all the Securities of such series so
called for redemption at the appropriate redemption price, together with accrued interest to the date fixed for redemption. If all of
the outstanding Securities of a series are to be redeemed, the Company will deliver to the Trustee at least 10 days prior to the last
date on which notice of redemption may be given to Holders pursuant to the first paragraph of this Section 3.02 (or such shorter period
as shall be acceptable to the Trustee) an Officers’ Certificate stating that all such Securities are to be redeemed. If less than
all the outstanding Securities of a series are to be redeemed, the Company will deliver to the Trustee at least 15 days prior to the last
date on which notice of redemption may be given to Holders pursuant to the first paragraph of this Section 3.02 (or such shorter period
as shall be acceptable to the Trustee) an Officers’ Certificate stating the aggregate principal amount of such Securities to be
redeemed. In the case of any redemption of Securities (a) prior to the expiration of any restriction on such redemption provided in the
terms of such Securities or elsewhere in this Indenture, or (b) pursuant to an election of the Company which is subject to a condition
specified in the terms of such Securities or elsewhere in this Indenture, the Company shall deliver to the Trustee, prior to the giving
of any notice of redemption to Holders pursuant to this Section, an Officers’ Certificate evidencing compliance with such restriction
or condition.
If less than all the Securities
of a series are to be redeemed, the Trustee shall select, pro rata, by lot or in such manner as it shall deem appropriate and fair, Securities
of such series to be redeemed in whole or in part. Securities may be redeemed in part in principal amounts equal to authorized denominations
for Securities of such series. The Trustee shall promptly notify the Company in writing of the Securities of such series selected for
redemption and, in the case of any Securities of such series selected for partial redemption, the principal amount thereof to be redeemed.
For all purposes of this Indenture, unless the context otherwise requires, all provisions relating to the redemption of Securities shall
relate, in the case of any Security redeemed or to be redeemed only in part, to the portion of the principal amount of such Security which
has been or is to be redeemed.
Section 3.03. Payment
Of Securities Called For Redemption. If notice of redemption has been given as above provided, the Securities or portions of Securities
specified in such notice shall become due and payable on the date and at the place stated in such notice at the applicable redemption
price, together with interest accrued to the date fixed for redemption, and on and after such date (unless the Company shall default in
the payment of such Securities at the redemption price, together with interest accrued to such date) interest on the Securities or portions
of Securities so called for redemption shall cease to accrue, and the unmatured coupons, if any, appertaining thereto shall be void and,
except as provided in Sections 7.12 and 8.02, such Securities shall cease from and after the date fixed for redemption to be entitled
to any benefit under this Indenture, and the Holders thereof shall have no right in respect of such Securities except the right to receive
the redemption price thereof and unpaid interest to the date fixed for redemption. On presentation and surrender of such Securities at
a place of payment specified in said notice, together with all coupons, if any, appertaining thereto maturing after the date fixed for
redemption, said Securities or the specified portions thereof shall be paid and redeemed by the Company at the applicable redemption price,
together with interest accrued thereon to the date fixed for redemption; provided that payment of interest becoming due on or prior to
the date fixed for redemption shall be payable in the case of Securities with coupons attached thereto, to the Holders of the coupons
for such interest upon surrender thereof, and in the case of Registered Securities, to the Holders of such Registered Securities registered
as such on the relevant record date subject to the terms and provisions of Sections 2.04 and 2.13 hereof.
If any Security called for
redemption shall not be so paid upon surrender thereof for redemption, the principal shall, until paid or duly provided for, bear interest
from the date fixed for redemption at the rate of interest or Yield to Maturity (in the case of an Original Issue Discount Security) borne
by such Security.
If any Security with coupons
attached thereto is surrendered for redemption and is not accompanied by all appurtenant coupons maturing after the date fixed for redemption,
the surrender of such missing coupon or coupons may be waived by the Company and the Trustee, if there be furnished to each of them such
security or indemnity as they may require to save each of them harmless.
Upon presentation of any
Security of any series redeemed in part only, the Company shall execute and the Trustee shall authenticate and deliver to or on the order
of the Holder thereof, at the expense of the Company, a new Security or Securities of such series and tenor (with any unmatured coupons
attached), of authorized denominations, in principal amount equal to the unredeemed portion of the Security so presented.
Section 3.04. Exclusion
of Certain Securities from Eligibility for Selection for Redemption. Securities shall be excluded from eligibility for selection for
redemption if they are identified by registration and certificate number in a written statement signed by an authorized officer of the
Company and delivered to the Trustee at least 40 days prior to the last date on which notice of redemption may be given as being owned
of record and beneficially by, and not pledged or hypothecated by, either (a) the Company or (b) an entity specifically identified in
such written statement as directly or indirectly controlling or controlled by or under direct or indirect common control with the Company.
Section 3.05. Mandatory
and Optional Sinking Funds. The minimum amount of any sinking fund payment provided for by the terms of Securities of any series
is herein referred to as a “mandatory sinking fund payment”, and any payment in excess of such minimum amount provided
for by the terms of the Securities of any series is herein referred to as an “optional sinking fund payment”. The date
on which a sinking fund payment is to be made is herein referred to as the “sinking fund payment date”.
In lieu of making all or
any part of any mandatory sinking fund payment with respect to any series of Securities in cash, the Company may at its option (a) deliver
to the Trustee Securities of such series theretofore purchased or otherwise acquired (except through a mandatory sinking fund payment)
by the Company or receive credit for Securities of such series (not previously so credited) theretofore purchased or otherwise acquired
(except as aforesaid) by the Company and delivered to the Trustee for cancellation pursuant to Section 2.11, (b) receive credit for optional
sinking fund payments (not previously so credited) made pursuant to this Section, or (c) receive credit for Securities of such series
(not previously so credited) redeemed by the Company at the option of the Company pursuant to the terms of such Securities or through
any optional sinking fund payment. Securities so delivered or credited shall be received or credited by the Trustee at the sinking fund
redemption price specified in such Securities.
On or before the sixtieth
day next preceding each sinking fund payment date for any series, or such shorter period as shall be acceptable to the Trustee, the Company
will deliver to the Trustee an Officers’ Certificate (a) specifying the portion of the mandatory sinking fund payment to be satisfied
by payment of cash and the portion to be satisfied by credit of specified Securities of such series and the basis for such credit, (b)
stating that none of the specified Securities of such series has theretofore been so credited, (c) stating that no defaults in the payment
of interest or Events of Default with respect to such series have occurred (which have not been waived or cured) and are continuing and
(d) stating whether or not the Company intends to exercise its right to make an optional sinking fund payment with respect to such series
and, if so, specifying the amount of such optional sinking fund payment which the Company intends to pay on or before the next succeeding
sinking fund payment date. Any Securities of such series to be credited and required to be delivered to the Trustee in order for the Company
to be entitled to credit therefor as aforesaid which have not theretofore been delivered to the Trustee shall be delivered for cancellation
pursuant to Section 2.11 to the Trustee with such Officers’ Certificate (or reasonably promptly thereafter if acceptable to the
Trustee). Such Officers’ Certificate shall be irrevocable and upon its receipt by the Trustee the Company shall become unconditionally
obligated to make all the cash payments or delivery of Securities therein referred to, if any, on or before the next succeeding sinking
fund payment date. Failure of the Company, on or before any such sixtieth day, to deliver such Officer’s Certificate and Securities
specified in this paragraph, if any, shall not constitute a default but shall constitute, on and as of such date, the irrevocable election
of the Company (i) that the mandatory sinking fund payment for such series due on the next succeeding sinking fund payment date shall
be paid entirely in cash without the option to deliver or credit Securities of such series in respect thereof and (ii) that the Company
will make no optional sinking fund payment with respect to such series as provided in this Section.
If the sinking fund payment
or payments (mandatory or optional or both) to be made in cash on the next succeeding sinking fund payment date plus any unused balance
of any preceding sinking fund payments made in cash shall exceed $50,000 (or a lesser sum if the Company shall so request with respect
to the Securities of any series), such cash shall be applied on the next succeeding sinking fund payment date to the redemption of Securities
of such series at the sinking fund redemption price thereof together with accrued interest thereon to the date fixed for redemption. If
such amount shall be $50,000 (or such lesser sum) or less and the Company makes no such request then it shall be carried over until a
sum in excess of $50,000 (or such lesser sum) is available. The Trustee shall select, in the manner provided in Section 3.02, for redemption
on such sinking fund payment date a sufficient principal amount of Securities of such series to absorb said cash, as nearly as may be,
and shall (if requested in writing by the Company) inform the Company of the serial numbers of the Securities of such series (or portions
thereof) so selected. Securities shall be excluded from eligibility for redemption under this Section if they are identified by registration
and certificate number in an Officers’ Certificate delivered to the Trustee at least 60 days prior to the sinking fund payment date
as being owned of record and beneficially by, and not pledged or hypothecated by either (a) the Company or (b) an entity specifically
identified in such Officers’ Certificate as directly or indirectly controlling or controlled by or under direct or indirect common
control with the Company. The Trustee, in the name and at the expense of the Company (or the Company, if it shall so request the Trustee
in writing) shall cause notice of redemption of the Securities of such series to be given in substantially the manner provided in Section
3.02 (and with the effect provided in Section 3.03) for the redemption of Securities of such series in part at the option of the Company.
The amount of any sinking fund payments not so applied or allocated to the redemption of Securities of such series shall be added to the
next cash sinking fund payment for such series and, together with such payment, shall be applied in accordance with the provisions of
this Section. Any and all sinking fund moneys held on the stated maturity date of the Securities of any particular series (or earlier,
if such maturity is accelerated), which are not held for the payment or redemption of particular Securities of such series shall be applied,
together with other moneys, if necessary, sufficient for the purpose, to the payment of the Principal of, and interest on, the Securities
of such series at maturity.
On or before 10:00 a.m. New
York City time on each sinking fund payment date or, in the case of Unregistered Securities, 10:00 a.m. New York City time on the Business
Day prior to the sinking fund payment date, the Company shall pay to the Trustee in cash or shall otherwise provide for the payment of
all interest accrued to the date fixed for redemption on Securities to be redeemed on the next following sinking fund payment date.
The Trustee shall not redeem
or cause to be redeemed any Securities of a series with sinking fund moneys or mail any notice of redemption of Securities of such series
by operation of the sinking fund during the continuance of a Default in payment of interest on such Securities or of any Event of Default
except that, where the mailing of notice of redemption of any Securities shall theretofore have been made, the Trustee shall redeem or
cause to be redeemed such Securities, provided that it shall have received from the Company a sum sufficient for such redemption. Except
as aforesaid, any moneys in the sinking fund for such series at the time when any such Default or Event of Default shall occur, and any
moneys thereafter paid into the sinking fund, shall, during the continuance of such Default or Event of Default, be deemed to have been
collected under Article 6 and held for the payment of all such Securities. In case such Event of Default shall have been waived as provided
in Section 6.04 or the Default cured on or before the sixtieth day preceding the sinking fund payment date in any year, such moneys shall
thereafter be applied on the next succeeding sinking fund payment date in accordance with this Section to the redemption of such Securities.
ARTICLE 4
COVENANTS
Section 4.01. Payment
of Securities. The Company shall pay the Principal of and interest on the Securities on the dates and in the manner provided
in the Securities and this Indenture. The interest on Securities with coupons attached (together with any additional amounts payable pursuant
to the terms of such Securities) shall be payable only upon presentation and surrender of the several coupons for such interest installments
as are evidenced thereby as they severally mature. The interest on any temporary Unregistered Securities (together with any additional
amounts payable pursuant to the terms of such Securities) shall be paid, as to the installments of interest evidenced by coupons attached
thereto, if any, only upon presentation and surrender thereof, and, as to the other installments of interest, if any, only upon presentation
of such Unregistered Securities for notation thereon of the payment of such interest. The interest on Registered Securities (together
with any additional amounts payable pursuant to the terms of such Securities) shall be payable only to the Holders thereof (subject to
Section 2.04) and at the option of the Company may be paid by mailing checks for such interest payable to or upon the written order of
such Holders at their last addresses as they appear on the Security Register of the Company.
Notwithstanding any provisions
of this Indenture and the Securities of any series to the contrary, if the Company and a Holder of any Registered Security so agree, payments
of interest on, and any portion of the Principal of, such Holder’s Registered Security (other than interest payable at maturity
or on any redemption or repayment date or the final payment of Principal on such Security) shall be made by the Paying Agent, upon receipt
from the Company of immediately available funds by 11:00 a.m., New York City time (or such other time as may be agreed to between the
Company and the Paying Agent), directly to the Holder of such Security (by Federal funds wire transfer or otherwise) if the Holder has
delivered written instructions to the Trustee 15 days prior to such payment date requesting that such payment will be so made and designating
the bank account to which such payments shall be so made and in the case of payments of Principal, surrenders the same to the Trustee
in exchange for a Security or Securities aggregating the same principal amount as the unredeemed principal amount of the Securities surrendered.
The Trustee shall be entitled to rely on the last instruction delivered by the Holder pursuant to this Section 4.01 unless a new instruction
is delivered 15 days prior to a payment date. The Company will indemnify and hold each of the Trustee and any Paying Agent harmless against
any loss, liability or expense (including attorneys’ fees) resulting from any act or omission to act on the part of the Company
or any such Holder in connection with any such agreement or from making any payment in accordance with any such agreement.
The Company shall pay interest
on overdue Principal, and interest on overdue installments of interest, to the extent lawful, at the rate per annum specified in the Securities.
Section 4.02. Maintenance
of Office or Agency. The Company will maintain in the Borough of Manhattan, The City of New York an office or agency where Securities
may be surrendered for registration of transfer or exchange or for presentation for payment and where notices and demands to or upon the
Company in respect of the Securities and this Indenture may be served. The Company hereby initially designates the Corporate Trust Office
of the Trustee, located in the Borough of Manhattan, The City of New York, as such office or agency of the Company. The Company will give
prompt written notice to the Trustee of the location, and any change in the location, of such office or agency. If at any time the Company
shall fail to maintain any such required office or agency or shall fail to furnish the Trustee with the address thereof, such presentations,
surrenders, notices and demands may be made or served at the address of the Trustee set forth in Section 10.02.
The Company will maintain
one or more agencies in a city or cities located outside the United States (including any city in which such an agency is required to
be maintained under the rules of any stock exchange on which the Securities of any series are listed) where the Unregistered Securities,
if any, of each series and coupons, if any, appertaining thereto may be presented for payment. No payment on any Unregistered Security
or coupon will be made upon presentation of such Unregistered Security or coupon at an agency of the Company within the United States
nor will any payment be made by transfer to an account in, or by mail to an address in, the United States unless, pursuant to applicable
United States laws and regulations then in effect, such payment can be made without adverse tax consequences to the Company. Notwithstanding
the foregoing, if full payment in United States Dollars (“Dollars”) at each agency maintained by the Company outside
the United States for payment on such Unregistered Securities or coupons appertaining thereto is illegal or effectively precluded by exchange
controls or other similar restrictions, payments in Dollars of Unregistered Securities of any series and coupons appertaining thereto
which are payable in Dollars may be made at an agency of the Company maintained in the Borough of Manhattan, The City of New York.
The Company may also from
time to time designate one or more other offices or agencies where the Securities of any series may be presented or surrendered for any
or all such purposes and may from time to time rescind such designations; provided that no such designation or rescission shall in any
manner relieve the Company of its obligation to maintain an office or agency in the Borough of Manhattan, The City of New York for such
purposes. The Company will give prompt written notice to the Trustee of any such designation or rescission and of any change in the location
of any such other office or agency.
Section 4.03. Securityholders’
Lists. The Company will furnish or cause to be furnished to the Trustee a list in such form as the Trustee may reasonably require
of the names and addresses of the holders of the Securities pursuant to Section 312 of the Trust Indenture Act (a) semi-annually not more
than 15 days after each record date for the payment of semi-annual interest on the Securities, as hereinabove specified, as of such record
date, and (b) at such other times as the Trustee may request in writing, within thirty days after receipt by the Company of any such request
as of a date not more than 15 days prior to the time such information is furnished.
Section 4.04. Certificate
to Trustee. The Company will furnish to the Trustee annually, on or before a date not more than four months after the end of
its fiscal year (which, on the date hereof, is a calendar year), a brief certificate (which need not contain the statements required by
Section 10.04) from its principal executive, financial or accounting officer as to his or her knowledge of the compliance of the Company
with all conditions and covenants under this Indenture (such compliance to be determined without regard to any period of grace or requirement
of notice provided under this Indenture) which certificate shall comply with the requirements of the Trust Indenture Act.
Section 4.05. Reports
by the Company. The Company covenants to file with the Trustee, within 15 days after the Company is required to file the same
with the Commission, copies of the annual reports and of the information, documents, and other reports which the Company may be required
to file with the Commission pursuant to Section 13 or Section 15(d) of the Exchange Act.
Section 4.06. Additional
Amounts. If the Securities of a series provide for the payment of additional amounts, at least 10 days prior to the first interest
payment date with respect to that series of Securities and at least 10 days prior to each date of payment of Principal of or interest
on the Securities of that series if there has been a change with respect to the matters set forth in the below-mentioned Officers’
Certificate, the Company shall furnish to the Trustee and the principal paying agent, if other than the Trustee, an Officers’ Certificate
instructing the Trustee and such paying agent whether such payment of Principal of or interest on the Securities of that series shall
be made to Holders of the Securities of that series without withholding or deduction for or on account of any tax, assessment or other
governmental charge described in the Securities of that series. If any such withholding or deduction shall be required, then such Officers’
Certificate shall specify by country the amount, if any, required to be withheld or deducted on such payments to such Holders and shall
certify the fact that additional amounts will be payable and the amounts so payable to each Holder, and the Company shall pay to the Trustee
or such paying agent the additional amounts required to be paid by this Section. The Company covenants to indemnify the Trustee and any
paying agent for, and to hold them harmless against, any loss, liability or expense reasonably incurred without negligence or bad faith
on their part arising out of or in connection with actions taken or omitted by any of them in reliance on any Officers’ Certificate
furnished pursuant to this Section.
Whenever in this Indenture
there is mentioned, in any context, the payment of the Principal of or interest or any other amounts on, or in respect of, any Security
of any series, such mention shall be deemed to include mention of the payment of additional amounts provided by the terms of such series
established hereby or pursuant hereto to the extent that, in such context, additional amounts are, were or would be payable in respect
thereof pursuant to such terms, and express mention of the payment of additional amounts (if applicable) in any provision hereof shall
not be construed as excluding the payment of additional amounts in those provisions hereof where such express mention is not made.
ARTICLE 5
SUCCESSOR
CORPORATION
Section 5.01. When
Company May Merge, Etc. The Company shall not consolidate with, merge with or into, or sell, convey, transfer, lease or otherwise
dispose of all or substantially all of its property and assets (in one transaction or a series of related transactions) to, any Person
unless either (x) the Company shall be the continuing Person or (y) the Person (if other than the Company) formed by such consolidation
or into which the Company is merged or to which properties and assets of the Company shall be sold, conveyed, transferred or leased shall
be a corporation organized and validly existing under the laws of the United States of America or any jurisdiction thereof and shall expressly
assume, by a supplemental indenture, executed and delivered to the Trustee, all of the obligations of the Company on all of the Securities
and under this Indenture and the Company in the case of clauses (x) and (y) shall have delivered to the Trustee (A) an Opinion of Counsel
stating that such consolidation, merger or sale, conveyance, transfer or lease and such supplemental indenture (if any) complies with
this provision and that all conditions precedent provided for herein relating to such transaction have been complied with and that such
supplemental indenture (if any) constitutes the legal, valid and binding obligation of the Company and such successor enforceable against
such entity in accordance with its terms, subject to customary exceptions and (B) an Officers’ Certificate to the effect that immediately
after giving effect to such transaction, no Default shall have occurred and be continuing.
Section 5.02. Successor
Substituted. Upon any consolidation or merger, or any sale, conveyance, transfer, lease or other disposition of all or substantially
all of the property and assets of the Company in accordance with Section 5.01 of this Indenture, the successor Person formed by such consolidation
or into which the Company is merged or to which such sale, conveyance, transfer, lease or other disposition is made shall succeed to,
and be substituted for, and may exercise every right and power of, the Company under this Indenture with the same effect as if such successor
Person had been named as the Company herein and thereafter the predecessor Person, except in the case of a lease, shall be relieved of
all obligations and covenants under this Indenture and the Securities.
ARTICLE 6
DEFAULT
AND REMEDIES
Section 6.01. Events
of Default. An “Event of Default” shall occur with respect to the Securities of any series if:
(a) the Company defaults
in the payment of the Principal of any Security of such series when the same becomes due and payable at maturity, upon acceleration, redemption
or mandatory repurchase, including as a sinking fund installment, or otherwise;
(b) the Company defaults
in the payment of interest on any Security of such series when the same becomes due and payable, and such default continues for a period
of 30 days;
(c) the Company defaults
in the performance of or breaches any other covenant or agreement of the Company in this Indenture with respect to any Security of such
series or in the Securities of such series and such default or breach continues for a period of 30 consecutive days after written notice
to the Company by the Trustee or to the Company and the Trustee by the Holders of 25% or more in aggregate principal amount of the Securities
of all series affected thereby specifying such default or breach and requiring it to be remedied and stating that such notice is a “Notice
of Default” hereunder;
(d) a court having jurisdiction
in the premises shall enter a decree or order for relief in respect of the Company in an involuntary case under any applicable bankruptcy,
insolvency or other similar law now or hereafter in effect, or appointing a receiver, liquidator, assignee, custodian, trustee, sequestrator
(or similar official) of the Company or for any substantial part of its property or ordering the winding up or liquidation of its affairs,
and such decree or order shall remain unstayed and in effect for a period of 60 consecutive days;
(e) the Company (i) commences
a voluntary case under any applicable bankruptcy, insolvency or other similar law now or hereafter in effect, or consents to the entry
of an order for relief in an involuntary case under any such law, (ii) consents to the appointment of or taking possession by a receiver,
liquidator, assignee, custodian, trustee, sequestrator or similar official of the Company or for all or substantially all of the property
and assets of the Company or (iii) effects any general assignment for the benefit of creditors; or
(f) any other Event of Default
established pursuant to Section 2.03 with respect to the Securities of such series occurs.
Section 6.02. Acceleration.
(a) If an Event of Default other than as described in clauses (d) or (e) of Section 6.01 with respect to the Securities of any series
then outstanding occurs and is continuing, then, and in each and every such case, except for any series of Securities the principal of
which shall have already become due and payable, either the Trustee or the Holders of not less than 25% in aggregate principal amount
of the Securities of any such series then outstanding hereunder (each such series treated as a separate class) by notice in writing to
the Company (and to the Trustee if given by Securityholders), may declare the entire principal (or, if the Securities of any such series
are Original Issue Discount Securities, such portion of the principal amount as may be specified in the terms of such series established
pursuant to Section 2.03) of all Securities of such series, and the interest accrued thereon, if any, to be due and payable immediately,
and upon any such declaration the same shall become immediately due and payable.
(b) If an Event of Default
described in clause (d) or (e) of Section 6.01 occurs and is continuing, then the principal amount (or, if any Securities are Original
Issue Discount Securities, such portion of the principal as may be specified in the terms thereof established pursuant to Section 2.03)
of all the Securities then outstanding and interest accrued thereon, if any, shall be and become immediately due and payable, without
any notice or other action by any Holder or the Trustee, to the full extent permitted by applicable law.
The foregoing provisions,
however, are subject to the condition that if, at any time after the principal (or, if the Securities are Original Issue Discount Securities,
such portion of the principal as may be specified in the terms thereof established pursuant to Section 2.03) of the Securities of any
series (or of all the Securities, as the case may be) shall have been so declared or become due and payable, and before any judgment or
decree for the payment of the moneys due shall have been obtained or entered as hereinafter provided, the Company shall pay or shall deposit
with the Trustee a sum sufficient to pay all matured installments of interest upon all the Securities of each such series (or of all the
Securities, as the case may be) and the principal of any and all Securities of each such series (or of all the Securities, as the case
may be) which shall have become due otherwise than by acceleration (with interest upon such principal and, to the extent that payment
of such interest is enforceable under applicable law, on overdue installments of interest, at the same rate as the rate of interest or
Yield to Maturity (in the case of Original Issue Discount Securities) specified in the Securities of each such series to the date of such
payment or deposit) and such amount as shall be sufficient to cover all amounts owing the Trustee under Section 7.07, and if any and all
Events of Default under the Indenture, other than the non-payment of the principal of Securities which shall have become due by acceleration,
shall have been cured, waived or otherwise remedied as provided herein, then and in every such case the Holders of a majority in aggregate
principal amount of all the then outstanding Securities of all such series that have been accelerated (voting as a single class), by written
notice to the Company and to the Trustee, may waive all defaults with respect to all such series (or with respect to all the Securities,
as the case may be) and rescind and annul such declaration and its consequences, but no such waiver or rescission and annulment shall
extend to or shall affect any subsequent default or shall impair any right consequent thereon.
For all purposes under this
Indenture, if a portion of the principal of any Original Issue Discount Securities shall have been accelerated and declared or become
due and payable pursuant to the provisions hereof, then, from and after such declaration, unless such declaration has been rescinded and
annulled, the principal amount of such Original Issue Discount Securities shall be deemed, for all purposes hereunder, to be such portion
of the principal thereof as shall be due and payable as a result of such acceleration, and payment of such portion of the principal thereof
as shall be due and payable as a result of such acceleration, together with interest, if any, thereon and all other amounts owing thereunder,
shall constitute payment in full of such Original Issue Discount Securities.
Section 6.03. Other
Remedies. If a payment default or an Event of Default with respect to the Securities of any series occurs and is continuing, the Trustee
may pursue, in its own name or as trustee of an express trust, any available remedy by proceeding at law or in equity to collect the payment
of Principal of and interest on the Securities of such series or to enforce the performance of any provision of the Securities of such
series or this Indenture.
The Trustee may maintain
a proceeding even if it does not possess any of the Securities or does not produce any of them in the proceeding.
Section 6.04. Waiver
of Past Defaults. Subject to Sections 6.02, 6.07 and 9.02, the Holders of at least a majority in principal amount (or, if the Securities
are Original Issue Discount Securities, such portion of the principal as is then accelerable under Section 6.02) of the outstanding Securities
of all series affected (voting as a single class), by notice to the Trustee, may waive an existing Default or Event of Default with respect
to the Securities of such series and its consequences, except a Default in the payment of Principal of or interest on any Security as
specified in clauses (a) or (b) of Section 6.01 or in respect of a covenant or provision of this Indenture which cannot be modified or
amended without the consent of the Holder of each outstanding Security affected. Upon any such waiver, such Default shall cease to exist,
and any Event of Default with respect to the Securities of such series arising therefrom shall be deemed to have been cured, for every
purpose of this Indenture; but no such waiver shall extend to any subsequent or other Default or Event of Default or impair any right
consequent thereto.
Section 6.05. Control
by Majority. Subject to Sections 7.01 and 7.02(e), the Holders of at least a majority in aggregate principal amount (or, if any Securities
are Original Issue Discount Securities, such portion of the principal as is then accelerable under Section 6.02) of the outstanding Securities
of all series affected (voting as a single class) may direct the time, method and place of conducting any proceeding for any remedy available
to the Trustee or exercising any trust or power conferred on the Trustee with respect to the Securities of such series by this Indenture;
provided, that the Trustee may refuse to follow any direction that conflicts with law or this Indenture, that may involve the Trustee
in personal liability or that the Trustee determines in good faith may be unduly prejudicial to the rights of Holders not joining in the
giving of such direction; and provided further, that the Trustee may take any other action it deems proper that is not inconsistent with
any directions received from Holders of Securities pursuant to this Section 6.05.
Section 6.06. Limitation
on Suits. No Holder of any Security of any series may institute any proceeding, judicial or otherwise, with respect to this Indenture
or the Securities of such series, or for the appointment of a receiver or trustee, or for any other remedy hereunder, unless:
(a) such Holder has previously
given to the Trustee written notice of a continuing Event of Default with respect to the Securities of such series;
(b) the Holders of at least
25% in aggregate principal amount of outstanding Securities of all such series affected shall have made written request to the Trustee
to institute proceedings in respect of such Event of Default in its own name as Trustee hereunder;
(c) such Holder or Holders
have offered to the Trustee indemnity reasonably satisfactory to the Trustee against any costs, liabilities or expenses to be incurred
in compliance with such request;
(d) the Trustee for 60 days
after its receipt of such notice, request and offer of indemnity has failed to institute any such proceeding; and
(e) during such 60-day period,
the Holders of a majority in aggregate principal amount of the outstanding Securities of all such affected series have not given the Trustee
a direction that is inconsistent with such written request.
A Holder may not use this
Indenture to prejudice the rights of another Holder or to obtain a preference or priority over such other Holder.
Section 6.07. Rights
of Holders to Receive Payment. Notwithstanding any other provision of this Indenture, the right of any Holder of a Security to receive
payment of Principal of or interest, if any, on such Holder’s Security on or after the respective due dates expressed on such Security,
or to bring suit for the enforcement of any such payment on or after such respective dates, shall not be impaired or affected without
the consent of such Holder.
Section 6.08. Collection
Suit by Trustee. If an Event of Default with respect to the Securities of any series in payment of Principal or interest specified
in clause (a) or (b) of Section 6.01 occurs and is continuing, the Trustee may recover judgment in its own name and as trustee of an express
trust against the Company for the whole amount (or such portion thereof as specified in the terms established pursuant to Section 2.03
of Original Issue Discount Securities) of Principal of, and accrued interest remaining unpaid on, together with interest on overdue Principal
of, and, to the extent that payment of such interest is lawful, interest on overdue installments of interest on, the Securities of such
series, in each case at the rate or Yield to Maturity (in the case of Original Issue Discount Securities) specified in such Securities,
and such further amount as shall be sufficient to cover all amounts owing the Trustee under Section 7.07.
Section 6.09. Trustee
May File Proofs of Claim. The Trustee may file such proofs of claim and other papers or documents as may be necessary or advisable
in order to have the claims of the Trustee (including any claim for amounts due the Trustee under Section 7.07) and the Holders allowed
in any judicial proceedings relative to the Company (or any other obligor on the Securities), its creditors or its property and shall
be entitled and empowered to collect and receive any moneys, securities or other property payable or deliverable upon conversion or exchange
of the Securities or upon any such claims and to distribute the same, and any custodian, receiver, assignee, trustee, liquidator, sequestrator
or other similar official in any such judicial proceeding is hereby authorized by each Holder to make such payments to the Trustee and,
in the event that the Trustee shall consent to the making of such payments directly to the Holders, to pay to the Trustee any amount due
to it under Section 7.07. Nothing herein contained shall be deemed to empower the Trustee to authorize or consent to, or accept or adopt
on behalf of any Holder, any plan of reorganization, arrangement, adjustment or composition affecting the Securities or the rights of
any Holder thereof, or to authorize the Trustee to vote in respect of the claim of any Holder in any such proceeding.
Section 6.10. Application
of Proceeds. Any moneys collected by the Trustee pursuant to this Article in respect of the Securities of any series shall be applied
in the following order at the date or dates fixed by the Trustee and, in case of the distribution of such moneys on account of Principal
or interest, upon presentation of the several Securities and coupons appertaining to such Securities in respect of which moneys have been
collected and noting thereon the payment, or issuing Securities of such series and tenor in reduced principal amounts in exchange for
the presented Securities of such series and tenor if only partially paid, or upon surrender thereof if fully paid:
FIRST: To
the payment of all amounts due the Trustee under Section 7.07 applicable to the Securities of such series in respect of which moneys have
been collected;
SECOND:
Subject to Article 11, in case the principal of the Securities of such series in respect of which moneys have been collected shall not
have become and be then due and payable, to the payment of interest on the Securities of such series in default in the order of the maturity
of the installments of such interest, with interest (to the extent that such interest has been collected by the Trustee) upon the overdue
installments of interest at the same rate as the rate of interest or Yield to Maturity (in the case of Original Issue Discount Securities)
specified in such Securities, such payments to be made ratably to the persons entitled thereto, without discrimination or preference;
THIRD: Subject
to Article 11, in case the principal of the Securities of such series in respect of which moneys have been collected shall have become
and shall be then due and payable, to the payment of the whole amount then owing and unpaid upon all the Securities of such series for
Principal and interest, with interest upon the overdue Principal, and (to the extent that such interest has been collected by the Trustee)
upon overdue installments of interest at the same rate as the rate of interest or Yield to Maturity (in the case of Original Issue Discount
Securities) specified in the Securities of such series; and in case such moneys shall be insufficient to pay in full the whole amount
so due and unpaid upon the Securities of such series, then to the payment of such Principal and interest or Yield to Maturity, without
preference or priority of Principal over interest or Yield to Maturity, or of interest or Yield to Maturity over Principal, or of any
installment of interest over any other installment of interest, or of any Security of such series over any other Security of such series,
ratably to the aggregate of such Principal and accrued and unpaid interest or Yield to Maturity; and
FOURTH:
To the payment of the remainder, if any, to the Company or any other person lawfully entitled thereto.
Section 6.11. Restoration
of Rights and Remedies. If the Trustee or any Holder has instituted any proceeding to enforce any right or remedy under this Indenture
and such proceeding has been discontinued or abandoned for any reason, or has been determined adversely to the Trustee or to such Holder,
then, and in every such case, subject to any determination in such proceeding, the Company, the Trustee and the Holders shall be restored
to their former positions hereunder and thereafter all rights and remedies of the Company, Trustee and the Holders shall continue as though
no such proceeding had been instituted.
Section 6.12. Undertaking
for Costs. In any suit for the enforcement of any right or remedy under this Indenture or in any suit against the Trustee for any
action taken or omitted by it as Trustee, in either case in respect to the Securities of any series, a court may require any party litigant
in such suit (other than the Trustee) to file an undertaking to pay the costs of the suit, and the court may assess reasonable costs,
including reasonable attorneys’ fees, against any party litigant (other than the Trustee) in the suit having due regard to the merits
and good faith of the claims or defenses made by the party litigant. This Section 6.12 does not apply to a suit by a Holder pursuant to
Section 6.07, a suit instituted by the Trustee or a suit by Holders of more than 10% in principal amount of the outstanding Securities
of such series.
Section 6.13. Rights
and Remedies Cumulative. Except as otherwise provided with respect to the replacement or payment of mutilated, destroyed, lost or
wrongfully taken Securities in Section 2.08, no right or remedy herein conferred upon or reserved to the Trustee or to the Holders is
intended to be exclusive of any other right or remedy, and every right and remedy shall, to the extent permitted by law, be cumulative
and in addition to every other right and remedy given hereunder or now or hereafter existing at law or in equity or otherwise. The assertion
or employment of any right or remedy hereunder, or otherwise, shall not prevent the concurrent assertion or employment of any other appropriate
right or remedy.
Section 6.14. Delay
or Omission not Waiver. No delay or omission of the Trustee or of any Holder to exercise any right or remedy accruing upon any Event
of Default shall impair any such right or remedy or constitute a waiver of any such Event of Default or an acquiescence therein. Every
right and remedy given by this Article 6 or by law to the Trustee or to the Holders may be exercised from time to time, and as often as
may be deemed expedient, by the Trustee or by the Holders, as the case may be.
ARTICLE 7
TRUSTEE
Section 7.01. General. The
duties and responsibilities of the Trustee shall be as provided by the Trust Indenture Act and as set forth herein. Notwithstanding the
foregoing, no provision of this Indenture shall require the Trustee to expend or risk its own funds or otherwise incur any financial liability
in the performance of any of its duties hereunder, or in the exercise of any of its rights or powers, unless it receives indemnity satisfactory
to it against any loss, liability or expense. Whether or not therein expressly so provided, every provision of this Indenture relating
to the conduct or affecting the liability of or affording protection to the Trustee shall be subject to the provisions of this Article
7.
Section 7.02. Certain
Rights of Trustee. Subject to Trust Indenture Act Sections 315(a) through (d):
(a) the Trustee may rely
and shall be protected in acting or refraining from acting upon any resolution, certificate, Officers’ Certificate, Opinion of Counsel
(or both), statement, instrument, opinion, report, notice, request, direction, consent, order, bond, debenture, note, other evidence of
indebtedness or other paper or document believed by it to be genuine and to have been signed or presented by the proper person or persons.
The Trustee need not investigate any fact or matter stated in the document, but the Trustee, in its discretion, may make such further
inquiry or investigation into such facts or matters as it may see fit;
(b) before the Trustee acts
or refrains from acting, it may require an Officers’ Certificate and/or an Opinion of Counsel, which shall conform to Section 10.04
and shall cover such other matters as the Trustee may reasonably request. The Trustee shall not be liable for any action it takes or omits
to take in good faith in reliance on such certificate or opinion. Subject to Sections 7.01 and 7.02, whenever in the administration of
the trusts of this Indenture the Trustee shall deem it necessary or desirable that a matter be proved or established prior to taking or
suffering or omitting any action hereunder, such matter (unless other evidence in respect thereof be herein specifically prescribed) may,
in the absence of negligence or bad faith on the part of the Trustee, be deemed to be conclusively proved and established by an Officers’
Certificate delivered to the Trustee, and such certificate, in the absence of negligence or bad faith on the part of the Trustee, shall
be full warrant to the Trustee for any action taken, suffered or omitted by it under the provisions of this Indenture upon the faith thereof;
(c) the Trustee may act through
its attorneys and agents not regularly in its employ and shall not be responsible for the misconduct or negligence of any agent or attorney
appointed with due care;
(d) any request, direction,
order or demand of the Company mentioned herein shall be sufficiently evidenced by an Officers’ Certificate (unless other evidence
in respect thereof be herein specifically prescribed); and any Board Resolution may be evidenced to the Trustee by a copy thereof certified
by the Secretary or an Assistant Secretary of the Company;
(e) the Trustee shall be
under no obligation to exercise any of the rights or powers vested in it by this Indenture at the request, order or direction of any of
the Holders, unless such Holders shall have offered to the Trustee reasonable security or indemnity against the costs, expenses and liabilities
that might be incurred by it in compliance with such request or direction;
(f) the Trustee shall not
be liable for any action it takes or omits to take in good faith that it believes to be authorized or within its rights or powers or for
any action it takes or omits to take in accordance with the direction of the Holders in accordance with Section 6.05 relating to the time,
method and place of conducting any proceeding for any remedy available to the Trustee, or exercising any trust or power conferred upon
the Trustee, under this Indenture;
(g) the Trustee may consult
with counsel and the written advice of such counsel or any Opinion of Counsel shall be full and complete authorization and protection
in respect of any action taken, suffered or omitted by it hereunder in good faith and in reliance thereon; and
(h) prior to the occurrence
of an Event of Default hereunder and after the curing or waiving of all Events of Default, the Trustee shall not be bound to make any
investigation into the facts or matters stated in any resolution, certificate, Officers’ Certificate, Opinion of Counsel, Board
Resolution, statement, instrument, opinion, report, notice, request, consent, order, approval, appraisal, bond, debenture, note, coupon,
security, or other paper or document unless requested in writing so to do by the Holders of not less than a majority in aggregate principal
amount of the Securities of all series affected then outstanding; provided that, if the payment within a reasonable time to the Trustee
of the costs, expenses or liabilities likely to be incurred by it in the making of such investigation is, in the opinion of the Trustee,
not reasonably assured to the Trustee by the security afforded to it by the terms of this Indenture, the Trustee may require reasonable
indemnity against such expenses or liabilities as a condition to proceeding.
Section 7.03. Individual
Rights of Trustee. The Trustee, in its individual or any other capacity, may become the owner or pledgee of Securities and may otherwise
deal with the Company or its Affiliates with the same rights it would have if it were not the Trustee. Any Agent may do the same with
like rights. However, the Trustee is subject to Trust Indenture Act Sections 310(b) and 311. For purposes of Trust Indenture Act Section
311(b)(4) and (6), the following terms shall mean:
(a) “cash transaction”
means any transaction in which full payment for goods or securities sold is made within seven days after delivery of the goods or securities
in currency or in checks or other orders drawn upon banks or bankers and payable upon demand; and
(b) “self-liquidating
paper” means any draft, bill of exchange, acceptance or obligation which is made, drawn, negotiated or incurred by the Company
for the purpose of financing the purchase, processing, manufacturing, shipment, storage or sale of goods, wares or merchandise and which
is secured by documents evidencing title to, possession of, or a lien upon, the goods, wares or merchandise or the receivables or proceeds
arising from the sale of the goods, wares or merchandise previously constituting the security, provided the security is received by the
Trustee simultaneously with the creation of the creditor relationship with the Company arising from the making, drawing, negotiating or
incurring of the draft, bill of exchange, acceptance or obligation.
Section 7.04. Trustee’s
Disclaimer. The recitals contained herein and in the Securities (except the Trustee’s certificate of authentication) shall be
taken as statements of the Company and not of the Trustee and the Trustee assumes no responsibility for the correctness of the same. Neither
the Trustee nor any of its agents (a) makes any representation as to the validity or adequacy of this Indenture or the Securities and
(b) shall be accountable for the Company’s use or application of the proceeds from the Securities.
Section 7.05. Notice
of Default. If any Default with respect to the Securities of any series occurs and is continuing and if such Default is known to the
actual knowledge of a Responsible Officer with the Corporate Trust Department of the Trustee, the Trustee shall give to each Holder of
Securities of such series notice of such Default within 90 days after it occurs (a) if any Unregistered Securities of such series are
then outstanding, to the Holders thereof, by publication at least once in an Authorized Newspaper in the Borough of Manhattan, The City
of New York and at least once in an Authorized Newspaper in London and (b) to all Holders of Securities of such series in the manner and
to the extent provided in Section 313(c) of the Trust Indenture Act, unless such Default shall have been cured or waived before the mailing
or publication of such notice; provided, however, that, except in the case of a Default in the payment of the Principal of or interest
on any Security, the Trustee shall be protected in withholding such notice if the Trustee in good faith determines that the withholding
of such notice is in the interests of the Holders.
Section 7.06. Reports
by Trustee to Holders. The Trustee shall transmit to Holders such reports concerning the Trustee and its actions under this Indenture
as may be required pursuant to the Trust Indenture Act at the times and in the manner provided pursuant thereto. If required by Section
313(a) of the Trust Indenture Act, the Trustee shall, within 60 days after each May 15 following the date of this Indenture, deliver to
Holders a brief report, dated as of such May 15, which complies with the provisions of such Section 313(a).
A copy of each such report
shall, at the time of such transmission to Holders, be filed by the Trustee with each stock exchange upon which any Securities are listed,
with the Commission and with the Company. The Company will promptly notify the Trustee when any Securities are listed on any stock exchange.
Section 7.07. Compensation
and Indemnity. The Company shall pay to the Trustee such compensation as shall be agreed upon in writing from time to time for its
services. The compensation of the Trustee shall not be limited by any law on compensation of a Trustee of an express trust. The Company
shall reimburse the Trustee and any predecessor Trustee upon request for all reasonable out-of-pocket expenses, disbursements and advances
incurred or made by the Trustee or such predecessor Trustee. Such expenses shall include the reasonable compensation and expenses of the
Trustee’s or such predecessor Trustee’s agents, counsel and other persons not regularly in their employ.
The Company shall indemnify
the Trustee and any predecessor Trustee for, and hold them harmless against, any loss or liability or expense incurred by them without
negligence or bad faith on their part arising out of or in connection with the acceptance or administration of this Indenture and the
Securities or the issuance of the Securities or of series thereof or the trusts hereunder and the performance of duties under this Indenture
and the Securities, including the costs and expenses of defending themselves against or investigating any claim or liability and of complying
with any process served upon them or any of their officers in connection with the exercise or performance of any of their powers or duties
under this Indenture and the Securities.
To secure the Company’s
payment obligations in this Section 7.07, the Trustee shall have a lien prior to the Securities on all money or property held or collected
by the Trustee, in its capacity as Trustee, except money or property held in trust to pay Principal of, and interest on particular Securities.
The obligations of the Company
under this Section to compensate and indemnify the Trustee and each predecessor Trustee and to pay or reimburse the Trustee and each predecessor
Trustee for expenses, disbursements and advances shall constitute additional indebtedness hereunder and shall survive the satisfaction
and discharge of this Indenture or the rejection or termination of this Indenture under bankruptcy law. Such additional indebtedness shall
be a senior claim to that of the Securities upon all property and funds held or collected by the Trustee as such, except funds held in
trust for the benefit of the Holders of particular Securities or coupons, and the Securities are hereby subordinated to such senior claim.
Without prejudice to any other rights available to the Trustee under applicable law, if the Trustee renders services and incurs expenses
following an Event of Default under Section 6.01(d) or Section 6.01(e) hereof, the parties hereto and the holders by their acceptance
of the Securities hereby agree that such expenses are intended to constitute expenses of administration under any bankruptcy law.
Section 7.08. Replacement
of Trustee. A resignation or removal of the Trustee as Trustee with respect to the Securities of any series and appointment of a successor
Trustee as Trustee with respect to the Securities of any series shall become effective only upon the successor Trustee’s acceptance
of appointment as provided in this Section 7.08.
The Trustee may resign as
Trustee with respect to the Securities of any series at any time by so notifying the Company in writing. The Holders of a majority in
principal amount of the outstanding Securities of any series may remove the Trustee as Trustee with respect to the Securities of such
series by so notifying the Trustee in writing and may appoint a successor Trustee with respect thereto with the consent of the Company.
The Company may remove the Trustee as Trustee with respect to the Securities of any series if: (i) the Trustee is no longer eligible under
Section 7.11 of this Indenture; (ii) the Trustee is adjudged a bankrupt or insolvent; (iii) a receiver or other public officer takes charge
of the Trustee or its property; or (iv) the Trustee becomes incapable of acting.
If the Trustee resigns or
is removed as Trustee with respect to the Securities of any series, or if a vacancy exists in the office of Trustee with respect to the
Securities of any series for any reason, the Company shall promptly appoint a successor Trustee with respect thereto. Within one year
after the successor Trustee takes office, the Holders of a majority in principal amount of the outstanding Securities of such series may
appoint a successor Trustee in respect of such Securities to replace the successor Trustee appointed by the Company. If the successor
Trustee with respect to the Securities of any series does not deliver its written acceptance required by Section 7.09 within 30 days after
the retiring Trustee resigns or is removed, the retiring Trustee, the Company or the Holders of a majority in principal amount of the
outstanding Securities of such series may petition any court of competent jurisdiction for the appointment of a successor Trustee with
respect thereto.
The Company shall give notice
of any resignation and any removal of the Trustee with respect to the Securities of any series and each appointment of a successor Trustee
in respect of the Securities of such series to all Holders of Securities of such series. Each notice shall include the name of the successor
Trustee and the address of its Corporate Trust Office.
Notwithstanding replacement
of the Trustee with respect to the Securities of any series pursuant to this Section 7.08 and Section 7.09, the Company’s obligations
under Section 7.07 shall continue for the benefit of the retiring Trustee.
Section 7.09. Acceptance
of Appointment by Successor. In case of the appointment hereunder of a successor Trustee with respect to all Securities, every such
successor Trustee so appointed shall execute, acknowledge and deliver to the Company and to the retiring Trustee an instrument accepting
such appointment, and thereupon the resignation or removal of the retiring Trustee shall become effective and such successor Trustee,
without any further act, deed or conveyance, shall become vested with all the rights, powers, trusts and duties of the retiring Trustee;
but, on the request of the Company or the successor Trustee, such retiring Trustee shall, upon payment of its charges and subject to the
lien provided for in Section 7.07, execute and deliver an instrument transferring to such successor Trustee all the rights, powers and
trusts of the retiring Trustee and shall duly assign, transfer and deliver to such successor Trustee all property and money held by such
retiring Trustee hereunder.
In case of the appointment
hereunder of a successor Trustee with respect to the Securities of one or more (but not all) series, the Company, the retiring Trustee
and each successor Trustee with respect to the Securities of one or more series shall execute and deliver an indenture supplemental hereto
wherein each successor Trustee shall accept such appointment and which (1) shall contain such provisions as shall be necessary or desirable
to transfer and confirm to, and to vest in, each successor Trustee all the rights, powers, trusts and duties of the retiring Trustee with
respect to the Securities of that or those series to which the appointment of such successor Trustee relates, (2) if the retiring Trustee
is not retiring with respect to all Securities, shall contain such provisions as shall be deemed necessary or desirable to confirm that
all the rights, powers, trusts and duties of the retiring Trustee with respect to the Securities of that or those series as to which the
retiring Trustee is not retiring shall continue to be vested in the retiring Trustee, and (3) shall add to or change any of the provisions
of this Indenture as shall be necessary to provide for or facilitate the administration of the trusts hereunder by more than one Trustee,
it being understood that nothing herein or in such supplemental indenture shall constitute such Trustees co-trustees of the same trust
and that each such Trustee shall be trustee of a trust or trusts hereunder separate and apart from any trust or trusts hereunder administered
by any other such Trustee; and upon the execution and delivery of such supplemental indenture the resignation or removal of the retiring
Trustee shall become effective to the extent provided therein and each such successor Trustee, without any further act, deed or conveyance,
shall become vested with all the rights, powers, trusts and duties of the retiring Trustee with respect to the Securities of that or those
series to which the appointment of such successor Trustee relates; but, on request of the Company or any successor Trustee, such retiring
Trustee shall duly assign, transfer and deliver to such successor Trustee all property and money held by such retiring Trustee hereunder
with respect to the Securities of that or those series to which the appointment of such successor Trustee relates.
Upon request of any such
successor Trustee, the Company shall execute any and all instruments for more fully and certainly vesting in and confirming to such successor
Trustee all such rights, powers and trusts referred to in the first or second preceding paragraph, as the case may be.
No successor Trustee shall
accept its appointment unless at the time of such acceptance such successor Trustee shall be eligible under this Article and qualified
under Section 310(b) of the Trust Indenture Act.
Section 7.10. Successor
Trustee By Merger, Etc. If the Trustee consolidates with, merges or converts into, or transfers all or substantially all of its corporate
trust business to, another corporation or national banking association, the resulting, surviving or transferee corporation or national
banking association without any further act shall be the successor Trustee with the same effect as if the successor Trustee had been named
as the Trustee herein.
Section 7.11. Eligibility.
This Indenture shall always have a Trustee who satisfies the requirements of Trust Indenture Act Section 310(a). The Trustee shall have
a combined capital and surplus of at least $25,000,000 as set forth in its most recent published annual report of condition.
Section 7.12. Money
Held in Trust. The Trustee shall not be liable for interest on any money received by it except as the Trustee may agree in writing
with the Company. Money held in trust by the Trustee need not be segregated from other funds except to the extent required by law and
except for money held in trust under Article 8 of this Indenture.
ARTICLE 8
SATISFACTION
AND DISCHARGE OF INDENTURE; UNCLAIMED MONEYS
Section 8.01. Satisfaction
and Discharge of Indenture. If at any time (a) the Company shall have paid or caused to be paid the Principal of and interest
on all the Securities of any series outstanding hereunder (other than Securities of such series which have been destroyed, lost or stolen
and which have been replaced or paid as provided in Section 2.08) as and when the same shall have become due and payable, or (b) the Company
shall have delivered to the Trustee for cancellation all Securities of any series theretofore authenticated (other than any Securities
of such series which shall have been destroyed, lost or stolen and which shall have been replaced or paid as provided in Section 2.08)
or (c) (i) all the securities of such series not theretofore delivered to the Trustee for cancellation shall have become due and payable,
or are by their terms to become due and payable within one year or are to be called for redemption within one year under arrangements
satisfactory to the Trustee for the giving of notice of redemption, and (ii) the Company shall have irrevocably deposited or caused to
be deposited with the Trustee as trust funds the entire amount in cash (other than moneys repaid by the Trustee or any paying agent to
the Company in accordance with Section 8.04) or U.S. Government Obligations, maturing as to principal and interest in such amounts and
at such times as will insure (without consideration of the reinvestment of such interest) the availability of cash, or a combination thereof,
sufficient to pay at maturity or upon redemption all Securities of such series (other than any Securities of such series which shall have
been destroyed, lost or stolen and which shall have been replaced or paid as provided in Section 2.08) not theretofore delivered to the
Trustee for cancellation, including principal and interest due or to become due on or prior to such date of maturity or redemption as
the case may be, and if, in any such case, the Company is not prohibited from making payments in respect of the Securities by Article
11 hereof and shall also pay or cause to be paid all other sums payable hereunder by the Company with respect to Securities of such series,
then this Indenture shall cease to be of further effect with respect to Securities of such series (except as to (i) rights of registration
of transfer and exchange of securities of such series, and the Company’s right of optional redemption, if any, (ii) substitution
of mutilated, defaced, destroyed, lost or stolen Securities, (iii) rights of holders to receive payments of principal thereof and interest
thereon, upon the original stated due dates therefor (but not upon acceleration) and remaining rights of the holders to receive mandatory
sinking fund payments, if any, (iv) the rights, obligations and immunities of the Trustee hereunder and (v) the rights of the Securityholders
of such series as beneficiaries hereof with respect to the property so deposited with the Trustee payable to all or any of them), and
the Trustee, on demand of the Company accompanied by an Officers’ Certificate and an Opinion of Counsel and at the cost and expense
of the Company, shall execute proper instruments acknowledging such satisfaction of and discharging this Indenture with respect to such
series; provided, that the rights of Holders of the Securities to receive amounts in respect of Principal of and interest on the Securities
held by them shall not be delayed longer than required by then-applicable mandatory rules or policies of any securities exchange upon
which the Securities are listed. The Company agrees to reimburse the Trustee for any costs or expenses thereafter reasonably and properly
incurred and to compensate the Trustee for any services thereafter reasonably and properly rendered by the Trustee in connection with
this Indenture or the Securities of such series.
Section 8.02. Application
by Trustee of Funds Deposited for Payment of Securities. Subject to Section 8.04, all moneys (including U.S. Government Obligations
and the proceeds thereof) deposited with the Trustee pursuant to Section 8.01, Section 8.05 or Section 8.06 shall be held in trust and
applied by it to the payment, either directly or through any paying agent to the Holders of the particular Securities of such series for
the payment or redemption of which such moneys have been deposited with the Trustee, of all sums due and to become due thereon for Principal
and interest; but such money need not be segregated from other funds except to the extent required by law. Funds and U.S. Government Obligations
held in trust under Section 8.01, 8.05 or 8.06 shall not be subject to the claims of the holders of Senior Indebtedness under Article
11.
Section 8.03. Repayment
of Moneys Held by Paying Agent. In connection with the satisfaction and discharge of this Indenture with respect to Securities
of any series, all moneys then held by any paying agent under the provisions of this Indenture with respect to such series of Securities
shall, upon demand of the Company, be repaid to it or paid to the Trustee and thereupon such paying agent shall be released from all further
liability with respect to such moneys.
Section 8.04. Return
of Moneys Held by Trustee and Paying Agent Unclaimed for Two Years. Any moneys deposited with or paid to the Trustee or any paying
agent for the payment of the Principal of or interest on any Security of any series and not applied but remaining unclaimed for two years
after the date upon which such Principal or interest shall have become due and payable, shall, upon the written request of the Company
and unless otherwise required by mandatory provisions of applicable escheat or abandoned or unclaimed property law, be repaid to the Company
by the Trustee for such series or such paying agent, and the Holder of the Security of such series shall, unless otherwise required by
mandatory provisions of applicable escheat or abandoned or unclaimed property laws, thereafter look only to the Company for any payment
which such Holder may be entitled to collect, and all liability of the Trustee or any paying agent with respect to such moneys shall thereupon
cease.
Section 8.05. Defeasance
and Discharge of Indenture. The Company shall be deemed to have paid and shall be discharged from any and all obligations in
respect of the Securities of any series, on the 123rd day after the deposit referred to in clause (i) hereof has been made, and the provisions
of this Indenture shall no longer be in effect with respect to the Securities of such series (and the Trustee, at the expense of the Company,
shall execute proper instruments acknowledging the same), except as to: (a) rights of registration of transfer and exchange, and the Company’s
right of optional redemption, (b) substitution of apparently mutilated, defaced, destroyed, lost or stolen Securities, (c) rights of holders
to receive payments of principal thereof and interest thereon, upon the original stated due dates therefor (but not upon acceleration),
(d) the rights, obligations and immunities of the Trustee hereunder and (e) the rights of the Securityholders of such series as beneficiaries
hereof with respect to the property so deposited with the Trustee payable to all or any of them; provided that the following conditions
shall have been satisfied:
(i) with reference
to this provision the Company has deposited or caused to be irrevocably deposited with the Trustee (or another qualifying trustee satisfying
the requirements of Section 7.11) as trust funds in trust, specifically pledged as security for, and dedicated solely to, the benefit
of the Holders of the Securities of such series, (A) money in an amount, or (B) U.S. Government Obligations which through the payment
of interest and principal in respect thereof in accordance with their terms will provide not later than one day before the due date of
any payment referred to in subclause (x) or (y) of this clause (i) money in an amount, or (C) a combination thereof, sufficient, in the
opinion of a nationally recognized firm of independent public accountants expressed in a written certification thereof delivered to the
Trustee, to pay and discharge without consideration of the reinvestment of such interest and after payment of all federal, state and local
taxes or other charges and assessments in respect thereof payable by the Trustee (x) the principal of, premium, if any, and each installment
of interest on the outstanding Securities of such series on the due dates thereof and (y) any mandatory sinking fund payments or analogous
payments applicable to the Securities of such series on the day on which such payments are due and payable in accordance with the terms
of Securities of such series and the Indenture with respect to the Securities of such series;
(ii) the Company
has delivered to the Trustee (A) either (x) an Opinion of Counsel to the effect that Holders of Securities of such series will not recognize
income, gain or loss for federal income tax purposes as a result of the Company’s exercise of its option under this Section 8.05
and will be subject to federal income tax on the same amount and in the same manner and at the same times as would have been the case
if such deposit, defeasance and discharge had not occurred, which Opinion of Counsel must be based upon a ruling of the Internal Revenue
Service to the same effect or a change in applicable federal income tax law or related treasury regulations after the date of this Indenture
or (y) a ruling directed to the Trustee received from the Internal Revenue Service to the same effect as the aforementioned Opinion of
Counsel and (B) an Opinion of Counsel to the effect that the creation of the defeasance trust does not violate the Investment Company
Act of 1940, as amended, and after the passage of 123 days following the deposit, the trust fund will not be subject to the effect of
Section 547 of the U.S. Bankruptcy Code or Section 15 of the New York Debtor and Creditor Law;
(iii) immediately
after giving effect to such deposit on a pro forma basis, no Event of Default, or event that after the giving of notice or lapse of time
or both would become an Event of Default, shall have occurred and be continuing on the date of such deposit or during the period ending
on the 123rd day after the date of such deposit, and such deposit shall not result in a breach or violation of, or constitute a default
under, any other agreement or instrument to which the Company is a party or by which the Company is bound;
(iv) if at such
time the Securities of such series are listed on a national securities exchange, the Company has delivered to the Trustee an Opinion of
Counsel to the effect that the Securities of such series will not be delisted as a result of such deposit, defeasance and discharge;
(v) the Company
shall have delivered to the Trustee an Officers’ Certificate and an Opinion of Counsel, each stating that all conditions precedent
to the defeasance and discharge under this Section have been complied with; and
(vi) if the Securities
of such series are to be redeemed prior to the final maturity thereof (other than from mandatory sinking fund payments or analogous payments),
notice of such redemption shall have been duly given pursuant to this Indenture or provision therefor satisfactory to the Trustee shall
have been made.
Section 8.06. Defeasance
of Certain Obligations. The Company may omit to comply with any term, provision or condition set forth in, and this Indenture
will no longer be in effect with respect to, any covenant established pursuant to Section 2.03(r) and clause (c) (with respect to any
covenants established pursuant to Section 2.03(r)) and clause (f) of Section 6.01 shall be deemed not to be an Event of Default, if
(a) with reference to this
Section 8.06, the Company has deposited or caused to be irrevocably deposited with the Trustee (or another qualifying trustee satisfying
the requirements of Section 7.11) as trust funds in trust, specifically pledged as security for, and dedicated solely to, the benefit
of the Holders of the Securities of such series and the Indenture with respect to the Securities of such series, (i) money in an amount
or (ii) U.S. Government Obligations which through the payment of interest and principal in respect thereof in accordance with their terms
will provide not later than one day before the due dates thereof or earlier redemption (irrevocably provided for under agreements satisfactory
to the Trustee), as the case may be, of any payment referred to in subclause (x) or (y) of this clause (a) money in an amount, or (iii)
a combination thereof, sufficient, in the opinion of a nationally recognized firm of independent public accountants expressed in a written
certification thereof delivered to the Trustee, to pay and discharge without consideration of the reinvestment of such interest and after
payment of all federal, state and local taxes or other charges and assessments in respect thereof payable by the Trustee (x) the principal
of, premium, if any, and each installment of interest on the outstanding Securities on the due date thereof or earlier redemption (irrevocably
provided for under arrangements satisfactory to the Trustee), as the case may be, and (y) any mandatory sinking fund payments or analogous
payments applicable to the Securities of such series and the Indenture with respect to the Securities of such series on the day on which
such payments are due and payable in accordance with the terms of the Indenture and of Securities of such series and the Indenture with
respect to the Securities of such series;
(b) the Company has delivered
to the Trustee (i) an Opinion of Counsel to the effect that Holders of Securities of such series will not recognize income, gain or loss
for federal income tax purposes as a result of the Company’s exercise of its option under this Section 8.06 and will be subject
to federal income tax on the same amount and in the same manner and at the same times as would have been the case if such deposit and
defeasance had not occurred and (ii) an Opinion of Counsel to the effect that the creation of the defeasance trust does not violate the
Investment Company Act of 1940, as amended, and after the passage of 123 days following the deposit, the trust fund will not be subject
to the effect of Section 547 of the U.S. Bankruptcy Code or Section 15 of the New York Debtor and Creditor Law;
(c) immediately after giving
effect to such deposit on a pro forma basis, no Event of Default, or event that after the giving of notice or lapse of time or both would
become an Event of Default, shall have occurred and be continuing on the date of such deposit or during the period ending on the 123rd
day after the date of such deposit, and such deposit shall not result in a breach or violation of, or constitute a default under, any
other agreement or instrument to which the Company is a party or by which the Company is bound;
(d) if at such time the Securities
of such series are listed on a national securities exchange, the Company has delivered to the Trustee an Opinion of Counsel to the effect
that the Securities of such series will not be delisted as a result of such deposit, defeasance and discharge; and
(e) the Company shall have
delivered to the Trustee an Officers’ Certificate and an Opinion of Counsel, each stating that all conditions precedent to the defeasance
under this Section have been complied with.
Section 8.07. Reinstatement. If
the Trustee or paying agent is unable to apply any monies or U.S. Government Obligations in accordance with Article 8 by reason of any
legal proceeding or by reason of any order or judgment of any court or governmental authority enjoining, restraining or otherwise prohibiting
such application, the Company’s obligations under this Indenture and the Securities shall be revived and reinstated as though no
deposit had occurred pursuant to this Article until such time as the Trustee or paying agent is permitted to apply all such monies or
U.S. Government Obligations in accordance with Article 8; provided, however, that if the Company has made any
payment of Principal of or interest on any Securities because of the reinstatement of its obligations, the Company shall be subrogated
to the rights of the Holders of such Securities to receive such payment from the monies or U.S. Government Obligations held by the Trustee
or paying agent.
Section 8.08. Indemnity. The
Company shall pay and indemnify the Trustee (or other qualifying trustee, collectively for purposes of this Section 8.08 and Section 8.02,
the “Trustee”) against any tax, fee or other charge, imposed on or assessed against the U.S. Government Obligations deposited
pursuant to Section 8.01, 8.05 or 8.06 or the principal or interest received in respect thereof other than any such tax, fee or other
charge which by law is for the account of the Holders of the Securities and any coupons appertaining thereto.
Section 8.09. Excess
Funds. Anything in this Article 8 to the contrary notwithstanding, the Trustee shall deliver or pay to the Company from time
to time upon request of the Company, any money or U.S. Government Obligations (or other property and any proceeds therefrom) held by it
as provided in Section 8.01, 8.05 or 8.06 which, in the opinion of a nationally recognized firm of independent public accountants expressed
in a written certification thereof delivered to the Trustee, are in excess of the amount thereof which would then be required to be deposited
to effect a discharge or defeasance, as applicable, in accordance with this Article 8.
Section 8.10. Qualifying
Trustee. Any trustee appointed pursuant to Section 8.05 or 8.06 for the purpose of holding money or U.S. Government Obligations
deposited pursuant to such Sections shall be appointed under an agreement in form acceptable to the Trustee and shall provide to the Trustee
a certificate, upon which certificate the Trustee shall be entitled to conclusively rely, that all conditions precedent provided for herein
to the related defeasance have been complied with. In no event shall the Trustee be liable for any acts or omissions of said trustee.
ARTICLE 9
AMENDMENTS,
SUPPLEMENTS AND WAIVERS
Section 9.01. Without
Consent of Holders. The Company and the Trustee may amend or supplement this Indenture or the Securities of any series without
notice to or the consent of any Holder:
(a) to cure any ambiguity,
defect or inconsistency in this Indenture; provided that such amendments or supplements shall not materially and adversely
affect the interests of the Holders;
(b) to comply with Article
5;
(c) to comply with any requirements
of the Commission in connection with the qualification of this Indenture under the Trust Indenture Act;
(d) to evidence and provide
for the acceptance of appointment hereunder with respect to the Securities of any or all series by a successor Trustee and to add to or
change any of the provisions of this Indenture as shall be necessary to provide for or facilitate the administration of the trusts hereunder
by more than one Trustee, pursuant to the requirements of Section 7.09;
(e) to establish the form
or forms or terms of Securities of any series or of the coupons appertaining to such Securities as permitted by Section 2.03;
(f) to provide for uncertificated
or Unregistered Securities and to make all appropriate changes for such purpose; and
(g) to make any change that
does not materially and adversely affect the rights of any Holder.
Section 9.02. With
Consent of Holders. Subject to Sections 6.04 and 6.07, without prior notice to any Holders, the Company and the Trustee may amend
this Indenture and the Securities of any series with the written consent of the Holders of a majority in principal amount of the outstanding
Securities of all series affected by such amendment (all such series voting as a separate class), and the Holders of a majority in principal
amount of the outstanding Securities of all series affected thereby (all such series voting as a separate class) by written notice to
the Trustee may waive future compliance by the Company with any provision of this Indenture or the Securities of such series.
Notwithstanding the provisions
of this Section 9.02, without the consent of each Holder affected thereby, an amendment or waiver, including a waiver pursuant to Section
6.04, may not:
(a) change the stated maturity
of the Principal of, or any sinking fund obligation or any installment of interest on, such Holder’s Security;
(b) reduce the Principal
amount thereof or the rate of interest thereon (including any amount in respect of original issue discount);
(c) reduce the above stated
percentage of outstanding Securities the consent of whose holders is necessary to modify or amend the Indenture with respect to the Securities
of the relevant series; and
(d) reduce the percentage
in principal amount of outstanding Securities of the relevant series the consent of whose Holders is required for any supplemental indenture
or for any waiver of compliance with certain provisions of this Indenture or certain Defaults and their consequences provided for in this
Indenture.
A supplemental indenture
which changes or eliminates any covenant or other provision of this Indenture which has expressly been included solely for the benefit
of one or more particular series of Securities, or which modifies the rights of Holders of Securities of such series with respect to such
covenant or provision, shall be deemed not to affect the rights under this Indenture of the Holders of Securities of any other series
or of the coupons appertaining to such Securities.
It shall not be necessary
for the consent of any Holder under this Section 9.02 to approve the particular form of any proposed amendment, supplement or waiver,
but it shall be sufficient if such consent approves the substance thereof.
After an amendment, supplement
or waiver under this Section 9.02 becomes effective, the Company shall give to the Holders affected thereby a notice briefly describing
the amendment, supplement or waiver. The Company will mail supplemental indentures to Holders upon request. Any failure of the Company
to mail such notice, or any defect therein, shall not, however, in any way impair or affect the validity of any such supplemental indenture
or waiver.
Section 9.03. Revocation
and Effect of Consent. Until an amendment or waiver becomes effective, a consent to it by a Holder is a continuing consent by
the Holder and every subsequent Holder of a Security or portion of a Security that evidences the same debt as the Security of the consenting
Holder, even if notation of the consent is not made on any Security. However, any such Holder or subsequent Holder may revoke the consent
as to its Security or portion of its Security. Such revocation shall be effective only if the Trustee receives the notice of revocation
before the date the amendment, supplement or waiver becomes effective. An amendment, supplement or waiver shall become effective with
respect to any Securities affected thereby on receipt by the Trustee of written consents from the requisite Holders of outstanding Securities
affected thereby.
The Company may, but shall
not be obligated to, fix a record date (which may be not less than five nor more than 60 days prior to the solicitation of consents) for
the purpose of determining the Holders of the Securities of any series affected entitled to consent to any amendment, supplement or waiver.
If a record date is fixed, then, notwithstanding the immediately preceding paragraph, those Persons who were such Holders at such record
date (or their duly designated proxies) and only those Persons shall be entitled to consent to such amendment, supplement or waiver or
to revoke any consent previously given, whether or not such Persons continue to be such Holders after such record date. No such consent
shall be valid or effective for more than 90 days after such record date.
After an amendment, supplement
or waiver becomes effective with respect to the Securities of any series affected thereby, it shall bind every Holder of such Securities
unless it is of the type described in any of clauses (a) through (d) of Section 9.02. In case of an amendment or waiver of the type described
in clauses (a) through (d) of Section 9.02, the amendment or waiver shall bind each such Holder who has consented to it and every subsequent
Holder of a Security that evidences the same indebtedness as the Security of the consenting Holder.
Section 9.04. Notation
on or Exchange of Securities. If an amendment, supplement or waiver changes the terms of any Security, the Trustee may require
the Holder thereof to deliver it to the Trustee. The Trustee may place an appropriate notation on the Security about the changed terms
and return it to the Holder and the Trustee may place an appropriate notation on any Security of such series thereafter authenticated.
Alternatively, if the Company or the Trustee so determines, the Company in exchange for the Security shall issue and the Trustee shall
authenticate a new Security of the same series and tenor that reflects the changed terms.
Section 9.05. Trustee
to Sign Amendments, Etc. The Trustee shall be entitled to receive, and shall be fully protected in relying upon, an Opinion of
Counsel stating that the execution of any amendment, supplement or waiver authorized pursuant to this Article 9 is authorized or permitted
by this Indenture, stating that all requisite consents have been obtained or that no consents are required and stating that such supplemental
indenture constitutes the legal, valid and binding obligation of the Company, enforceable against the Company in accordance with its terms,
subject to customary exceptions. The Trustee may, but shall not be obligated to, execute any such amendment, supplement or waiver that
affects the Trustee’s own rights, duties or immunities under this Indenture or otherwise.
Section 9.06. Conformity
with Trust Indenture Act. Every supplemental indenture executed pursuant to this Article 9 shall conform to the requirements
of the Trust Indenture Act as then in effect.
ARTICLE 10
MISCELLANEOUS
Section 10.01. Trust
Indenture Act of 1939. This Indenture shall incorporate and be governed by the provisions of the Trust Indenture Act that are required
to be part of and to govern indentures qualified under the Trust Indenture Act.
Section 10.02. Notices. Any
notice or communication by the Company or the Trustee to the other, or by a Holder to the Company or the Trustee, is duly given if in
writing and delivered in person or mailed by first-class mail (registered or certified, return receipt requested), email or overnight
air courier guaranteeing next day delivery, to the others’ address:
if to the Company:
|
60 Degrees Pharmaceuticals, Inc.
1025 Connecticut Ave. NW, Suite 1000
Washington, DC 20036 |
| Phone: (202) 327-5422 |
| Attention: Chief Executive Officer and President |
if to the Trustee:
| |
[Name of Trustee] |
| |
[Address] |
| |
Phone: |
| |
Attention: |
The Company or the Trustee
by written notice to the other may designate additional or different addresses for subsequent notices or communications.
Any notice or communication
shall be sufficiently given to Holders of any Unregistered Securities, by publication at least once in an Authorized Newspaper in The
City of New York, or with respect to any Security the interest on which is based on the offered quotations in the interbank Eurodollar
market for dollar deposits at least once in an Authorized Newspaper in London, and by mailing to the Holders thereof who have filed their
names and addresses with the Trustee pursuant to Section 313(c)(2) of the Trust Indenture Act at such addresses as were so furnished to
the Trustee and to Holders of Registered Securities by mailing to such Holders at their addresses as they shall appear on the Security
Register. Notice mailed shall be sufficiently given if so mailed within the time prescribed. Copies of any such communication or notice
to a Holder shall also be mailed to the Trustee and each Agent at the same time.
Failure to mail a notice
or communication to a Holder or any defect in it shall not affect its sufficiency with respect to other Holders. Except as otherwise provided
in this Indenture, if a notice or communication is mailed in the manner provided in this Section 10.02, it is duly given, whether or not
the addressee receives it.
Where this Indenture provides
for notice in any manner, such notice may be waived in writing by the Person entitled to receive such notice, either before or after the
event, and such waiver shall be the equivalent of such notice. Waivers of notice by Holders shall be filed with the Trustee, but such
filing shall not be a condition precedent to the validity of any action taken in reliance upon such waiver.
In case it shall be impracticable
to give notice as herein contemplated, then such notification as shall be made with the approval of the Trustee shall constitute a sufficient
notification for every purpose hereunder.
Section 10.03. Certificate
and Opinion as to Conditions Precedent. Upon any request or application by the Company to the Trustee to take any action under
this Indenture, the Company shall furnish to the Trustee:
(a) an Officers’ Certificate
stating that, in the opinion of the signers, all conditions precedent, if any, provided for in this Indenture relating to the proposed
action have been complied with; and
(b) an Opinion of Counsel
stating that, in the opinion of such counsel, all such conditions precedent have been complied with.
Section 10.04. Statements
Required in Certificate or Opinion. Each certificate or opinion with respect to compliance with a condition or covenant provided
for in this Indenture (other than the certificate required by Section 4.04) shall include:
(a) a statement that each
person signing such certificate or opinion has read such covenant or condition and the definitions herein relating thereto;
(b) a brief statement as
to the nature and scope of the examination or investigation upon which the statement or opinion contained in such certificate or opinion
is based;
(c) a statement that, in
the opinion of each such person, he has made such examination or investigation as is necessary to enable him to express an informed opinion
as to whether or not such covenant or condition has been complied with; and
(d) a statement as to whether
or not, in the opinion of each such person, such condition or covenant has been complied with; provided, however, that, with
respect to matters of fact, an Opinion of Counsel may rely on an Officers’ Certificate or certificates of public officials.
Section 10.05. Evidence
of Ownership. The Company, the Trustee and any agent of the Company or the Trustee may deem and treat the Holder of any Unregistered
Security and the Holder of any coupon as the absolute owner of such Unregistered Security or coupon (whether or not such Unregistered
Security or coupon shall be overdue) for the purpose of receiving payment thereof or on account thereof and for all other purposes, and
neither the Company, the Trustee, nor any agent of the Company or the Trustee shall be affected by any notice to the contrary. The fact
of the holding by any Holder of an Unregistered Security, and the identifying number of such Security and the date of his holding the
same, may be proved by the production of such Security or by a certificate executed by any trust company, bank, banker or recognized securities
dealer wherever situated satisfactory to the Trustee, if such certificate shall be deemed by the Trustee to be satisfactory. Each such
certificate shall be dated and shall state that on the date thereof a Security bearing a specified identifying number was deposited with
or exhibited to such trust company, bank, banker or recognized securities dealer by the person named in such certificate. Any such certificate
may be issued in respect of one or more Unregistered Securities specified therein. The holding by the person named in any such certificate
of any Unregistered Securities specified therein shall be presumed to continue for a period of one year from the date of such certificate
unless at the time of any determination of such holding (1) another certificate bearing a later date issued in respect of the same Securities
shall be produced or (2) the Security specified in such certificate shall be produced by some other Person, or (3) the Security specified
in such certificate shall have ceased to be outstanding. Subject to Article 7, the fact and date of the execution of any such instrument
and the amount and numbers of Securities held by the Person so executing such instrument may also be proven in accordance with such reasonable
rules and regulations as may be prescribed by the Trustee or in any other manner which the Trustee may deem sufficient.
The Company, the Trustee
and any agent of the Company or the Trustee may deem and treat the person in whose name any Registered Security shall be registered upon
the Security Register for such series as the absolute owner of such Registered Security (whether or not such Registered Security shall
be overdue and notwithstanding any notation of ownership or other writing thereon) for the purpose of receiving payment of or on account
of the Principal of and, subject to the provisions of this Indenture, interest on such Registered Security and for all other purposes;
and neither the Company nor the Trustee nor any agent of the Company or the Trustee shall be affected by any notice to the contrary.
Section 10.06. Rules
by Trustee, Paying Agent or Registrar. The Trustee may make reasonable rules for action by or at a meeting of Holders. The Paying
Agent or Registrar may make reasonable rules for its functions.
Section 10.07. Payment
Date Other Than a Business Day. Except as otherwise provided with respect to a series of Securities, if any date for payment
of Principal or interest on any Security shall not be a Business Day at any place of payment, then payment of Principal of or interest
on such Security, as the case may be, need not be made on such date, but may be made on the next succeeding Business Day at any place
of payment with the same force and effect as if made on such date and no interest shall accrue in respect of such payment for the period
from and after such date.
Section 10.08. Governing
Law. The laws of the State of New York shall govern this Indenture and the Securities.
Section 10.09. No
Adverse Interpretation of Other Agreements. This Indenture may not be used to interpret another indenture or loan or debt agreement
of the Company or any Subsidiary of the Company. Any such indenture or agreement may not be used to interpret this Indenture.
Section 10.10. Successors. All
agreements of the Company in this Indenture and the Securities shall bind its successors. All agreements of the Trustee in this Indenture
shall bind its successors.
Section 10.11. Duplicate
Originals. The parties may sign any number of copies of this Indenture. Each signed copy shall be an original, but all of them
together represent the same agreement.
Section 10.12. Separability. In
case any provision in this Indenture or in the Securities shall be invalid, illegal or unenforceable, the validity, legality and enforceability
of the remaining provisions shall not in any way be affected or impaired thereby.
Section 10.13. Table
of Contents, Headings, Etc. The Table of Contents and headings of the Articles and Sections of this Indenture have been inserted
for convenience of reference only, are not to be considered a part hereof and shall in no way modify or restrict any of the terms and
provisions hereof.
Section 10.14. Incorporators,
Stockholders, Officers and Directors of Company Exempt from Individual Liability. No recourse under or upon any obligation, covenant
or agreement contained in this Indenture or any indenture supplemental hereto, or in any Security or any coupons appertaining thereto,
or because of any indebtedness evidenced thereby, shall be had against any incorporator, as such or against any past, present or future
stockholder, officer, director or employee, as such, of the Company or of any successor, either directly or through the Company or any
successor, under any rule of law, statute or constitutional provision or by the enforcement of any assessment or by any legal or equitable
proceeding or otherwise, all such liability being expressly waived and released by the acceptance of the Securities and the coupons appertaining
thereto by the holders thereof and as part of the consideration for the issue of the Securities and the coupons appertaining thereto.
Section 10.15. Judgment
Currency. The Company agrees, to the fullest extent that it may effectively do so under applicable law, that (a) if for the purpose
of obtaining judgment in any court it is necessary to convert the sum due in respect of the Principal of or interest on the Securities
of any series (the “Required Currency”) into a currency in which a judgment will be rendered (the “Judgment
Currency”), the rate of exchange used shall be the rate at which in accordance with normal banking procedures the Trustee could
purchase in The City of New York the Required Currency with the Judgment Currency on the day on which final unappealable judgment is entered,
unless such day is not a Business Day, then, to the extent permitted by applicable law, the rate of exchange used shall be the rate at
which in accordance with normal banking procedures the Trustee could purchase in The City of New York the Required Currency with the Judgment
Currency on the Business Day preceding the day on which final unappealable judgment is entered and (b) its obligations under this Indenture
to make payments in the Required Currency (i) shall not be discharged or satisfied by any tender, or any recovery pursuant to any judgment
(whether or not entered in accordance with subsection (a)), in any currency other than the Required Currency, except to the extent that
such tender or recovery shall result in the actual receipt, by the payee, of the full amount of the Required Currency expressed to be
payable in respect of such payments, (ii) shall be enforceable as an alternative or additional cause of action for the purpose of recovering
in the Required Currency the amount, if any, by which such actual receipt shall fall short of the full amount of the Required Currency
so expressed to be payable and (iii) shall not be affected by judgment being obtained for any other sum due under this Indenture.
ARTICLE 11
SUBORDINATION OF SECURITIES
Section 11.01. Agreement
to Subordinate. The Company covenants and agrees, and each Holder of Securities issued hereunder by his acceptance thereof likewise
covenants and agrees, that all Securities shall be issued subject to the provisions of this Article; and each person holding any Security,
whether upon original issue or upon transfer, assignment or exchange thereof accepts and agrees that the Principal of and interest on
all Securities issued hereunder shall, to the extent and in the manner herein set forth, be subordinated and subject in right of payment
to the prior payment in full of all Senior Indebtedness.
Section 11.02. Payments
to Securityholders. No payments on account of Principal of or interest on the Securities shall be made if at the time of such
payment or immediately after giving effect thereto there shall exist a default in any payment with respect to any Senior Indebtedness,
and such default shall not have been cured or waived or shall not have ceased to exist.
Upon any payment or distribution
of assets of the Company of any kind or character, whether in cash, property or securities, to creditors upon any liquidation, dissolution,
winding up, receivership, reorganization, assignment for the benefit of creditors, marshalling of assets and liabilities or any bankruptcy,
insolvency or similar proceedings of the Company, all amounts due or to become due upon all Senior Indebtedness shall first be paid in
full, in cash or cash equivalents, or payment thereof provided for in accordance with its terms, before any payment is made on account
of the Principal of, or interest on the indebtedness evidenced by the Securities, and upon any such liquidation, dissolution, winding
up, receivership, reorganization, assignment, marshalling or proceeding, any payment or distribution of assets of the Company of any kind
or character, whether in cash, property or securities, to which the Holders of the Securities or the Trustee under this Indenture would
be entitled, except for the provisions hereof, shall be paid by the Company or by any receiver, trustee in bankruptcy, liquidating trustee,
agent or other Person making such payment or distribution, directly to the holders of Senior Indebtedness (pro rata to such holders on
the basis of the respective amounts of Senior Indebtedness held by such holders) or their respective representatives, or to the trustee
or trustees under any indenture pursuant to which any instruments evidencing any of such Senior Indebtedness may have been issued, as
their respective interests may appear, to the extent necessary to pay all Senior Indebtedness in full (including, without limitation,
except to the extent, if any, prohibited by mandatory provisions of law, post-petition interest, in any such proceedings), after giving
effect to any concurrent payment or distribution to or for the holders of Senior Indebtedness, before any payment or distribution is made
to the holders of the indebtedness evidenced by the Securities or to the Trustee under this Indenture.
In the event that, notwithstanding
the foregoing, any payment or distribution of assets of the Company of any kind or character, whether in cash, property or securities,
prohibited by the foregoing, shall be received by the Trustee under this Indenture or the Holders of the Securities before all Senior
Indebtedness is paid in full or provision is made for such payment in accordance with its terms, such payment or distribution shall be
held in trust for the benefit of and shall be paid over or delivered to the holders of such Senior Indebtedness or their respective representatives,
or to the trustee or trustees under any indenture pursuant to which any instruments evidencing any of such Senior Indebtedness may have
been issued, as their respective interests may appear, for application to the payment of all Senior Indebtedness remaining unpaid until
all such Senior Indebtedness shall have been paid in full in accordance with its terms, after giving effect to any concurrent payment
or distribution to or for the holders of such Senior Indebtedness.
For purposes of this Article,
the words, “cash, property or securities” shall not be deemed to include shares of stock of the Company as reorganized or
readjusted, or securities of the Company or any other corporation provided for by a plan of arrangement, reorganization or readjustment,
the payment of which is subordinated (at least to the extent provided in this Article with respect to the Securities) to the payment of
all Senior Indebtedness which may at the time be outstanding; provided, that (i) the Senior Indebtedness is assumed by the
new corporation, if any, resulting from any such arrangement, reorganization or readjustment, and (ii) the rights of the holders of the
Senior Indebtedness are not, without the consent of such holders, altered by such arrangement, reorganization or readjustment. The consolidation
of the Company with, or the merger of the Company into, another corporation or the liquidation or dissolution of the Company following
the sale, conveyance or transfer of all or substantially all of its property and assets to another corporation upon the terms and conditions
provided in Article 5 shall not be deemed a dissolution, winding-up, liquidation or reorganization for the purposes of this Section if
such other corporation shall, as a part of such consolidation, merger, sale, conveyance or transfer, comply with the conditions stated
in Article 5. Nothing in this Section shall apply to claims of, or payments to, the Trustee under or pursuant to Article 7. This Section
shall be subject to the further provisions of Section 11.05.
Section 11.03. Subrogation
of Securities. Subject to the payment in full of all Senior Indebtedness, the Holders of the Securities shall be subrogated to
the rights of the holders of Senior Indebtedness to receive payments or distributions of cash, property or securities of the Company applicable
to the Senior Indebtedness until the principal of and interest on the Securities shall be paid in full; and, for the purposes of such
subrogation, no payments or distributions to the holders of the Senior Indebtedness of any cash, property or securities to which the Holders
of the Securities or the Trustee on their behalf would be entitled except for the provisions of this Article, and no payment over pursuant
to the provisions of this Article to the holders of Senior Indebtedness by Holders of the Securities or the Trustee on their behalf shall,
as between the Company, its creditors other than holders of Senior Indebtedness and the Holders of the Securities, be deemed to be a payment
by the Company to or on account of the Senior Indebtedness; and no payments or distributions of cash, property or securities to or for
the benefit of the Securityholders pursuant to the subrogation provision of this Article, which would otherwise have been paid to the
holders of Senior Indebtedness shall be deemed to be a payment by the Company to or for the account of the Securities. It is understood
that the provisions of this Article are and are intended solely for the purpose of defining the relative rights of the holders of the
Securities, on the one hand, and the Holders of the Senior Indebtedness, on the other hand.
Nothing contained in this
Article or elsewhere in this Indenture or in the Securities is intended to or shall impair, as between the Company, its creditors other
than the holders of Senior Indebtedness, and the Holders of the Securities, the obligation of the Company, which is absolute and unconditional,
to pay to the Holders of the Securities the Principal of and interest on the Securities as and when the same shall become due and payable
in accordance with their terms, or is intended to or shall affect the relative rights of the Holders of the Securities and creditors of
the Company other than the holders of the Senior Indebtedness, nor shall anything herein or therein prevent the Holder of any Security
or the Trustee on his behalf from exercising all remedies otherwise permitted by applicable law upon default under this Indenture, subject
to the rights, if any, under this Article of the holders of Senior Indebtedness in respect of cash, property or securities of the Company
received upon the exercise of any such remedy.
Upon any payment or distribution
of assets of the Company referred to in this Article, the Trustee, subject to the provisions of Sections 7.01 and 7.02, and the holders
of the Securities shall be entitled to rely upon any order or decree made by any court of competent jurisdiction in which such liquidation,
dissolution, winding up, receivership, reorganization, assignment or marshalling proceedings are pending, or a certificate of the receiver,
trustee in bankruptcy, liquidating trustee, agent or other person making such payment or distribution, delivered to the Trustee or to
the Holders of the Securities, for the purpose of ascertaining the Persons entitled to participate in such distribution, the holders of
the Senior Indebtedness and other indebtedness of the Company, the amount thereof or payable thereon, the amount or amounts paid or distributed
thereon and all other facts pertinent thereto or to this Article.
Section 11.04. Authorization
by Securityholders. Each Holder of a Security by his acceptance thereof authorizes the Trustee in his behalf to take such action
as may be necessary or appropriate to effectuate the subordination provided in this Article and appoints the Trustee his attorney-in-fact
for any and all such purposes.
Section 11.05. Notice
to Trustee. The Company shall give prompt written notice to the Trustee and to any paying agent of any fact known to the Company
which would prohibit the making of any payment of moneys to or by the Trustee or any paying agent in respect of the Securities pursuant
to the provisions of this Article or would end such prohibition. Regardless of anything to the contrary contained in this Article or elsewhere
in this Indenture, the Trustee shall not be charged with knowledge of the existence of any Senior Indebtedness or of any default or event
of default with respect to any Senior Indebtedness or of any other facts which would prohibit the making of any payment of moneys to or
by the Trustee or which would end such prohibition, unless and until the Trustee shall have received notice in writing at its principal
Corporate Trust Office to that effect signed by an officer of the Company, or by a holder or agent of a holder of Senior Indebtedness
or by the trustee under any indenture pursuant to which Senior Indebtedness shall be outstanding, who shall have been certified by the
Company or otherwise established to the reasonable satisfaction of the Trustee to be such holder or agent or trustee, and, prior to the
receipt of any such written notice, the Trustee shall, subject to Sections 7.01 and 7.02, be entitled to assume that no such facts exist;
provided that if on a date at least three Business Days prior to the date upon which by the terms hereof any such moneys shall become
payable for any purpose (including, without limitation, the payment of the Principal of, or interest on any Security) the Trustee shall
not have received with respect to such moneys the notice of prohibition provided for in this Section, then, regardless of anything herein
to the contrary, the Trustee shall have full power and authority to receive such moneys and to apply the same to the purpose for which
they were received, and shall not be affected by any notice to the contrary which may be received by it on or after such prior date.
Regardless of anything to
the contrary herein, nothing shall prevent (a) any payment by the Company or the Trustee to the Securityholders of amounts in connection
with a redemption of Securities if (i) notice of such redemption has been given pursuant to Article 3 prior to the receipt by the Trustee
of written notice of prohibition as aforesaid, and (ii) such notice of redemption is given not earlier than 60 days before the redemption
date, or (b) any payment by the Trustee to the Securityholders of amounts deposited with it pursuant to Sections 8.01, 8.05 or 8.06.
The Trustee shall be entitled
to rely on the delivery to it of a written notice by a Person representing himself to be a holder of Senior Indebtedness (or a trustee
or agent on behalf of such holder) to establish that such notice has been given by a holder of Senior Indebtedness or a trustee or agent
on behalf of any such holder. In the event that the Trustee determines in good faith that further evidence is required with respect to
the right of any Person as a holder of Senior Indebtedness to participate in any payment or distribution pursuant to this Article, the
Trustee may request such Person to furnish evidence to the reasonable satisfaction of the Trustee as to the amount of Senior Indebtedness
held by such Person, the extent to which such Person is entitled to participate in such payment or distribution and any other facts pertinent
to the rights of such Person under this Article, and if such evidence is not furnished the Trustee may defer any payment to such Person
pending judicial determination as to the right of such Person to receive such payment.
Section 11.06. Trustee’s
Relation to Senior Indebtedness. The Trustee and any agent of the Company or the Trustee shall be entitled to all the rights
set forth in this Article with respect to any Senior Indebtedness which may at any time be held by it in its individual or any other capacity
to the same extent as any other holder of Senior Indebtedness and nothing in this Indenture shall deprive the Trustee or any such agent,
of any of its rights as such holder. Nothing in this Article shall apply to claims of, or payments to, the Trustee under or pursuant to
7.07.
With respect to the holders
of Senior Indebtedness, the Trustee undertakes to perform or to observe only such of its covenants and obligations as are specifically
set forth in this Article, and no implied covenants or obligations with respect to the holders of Senior Indebtedness shall be read into
this Indenture against the Trustee. The Trustee shall not be deemed to owe any fiduciary duty to the holders of Senior Indebtedness and,
subject to the provisions of Sections 7.01 and 7.02, the Trustee shall not be liable to any holder of Senior Indebtedness if it shall
pay over or deliver to Holders of Securities, the Company or any other Person moneys or assets to which any holder of Senior Indebtedness
shall be entitled by virtue of this Article or otherwise.
Section 11.07. No
Impairment of Subordination. No right of any present or future holder of any Senior Indebtedness to enforce subordination as
herein provided shall at any time in any way be prejudiced or impaired by any act or failure to act on the part of the Company or by any
act or failure to act, in good faith, by any such holder, or by any noncompliance by the Company with the terms, provisions and covenants
of this Indenture, regardless of any knowledge thereof which any such holder may have or otherwise be charged with.
SIGNATURES
IN WITNESS WHEREOF, the parties
hereto have caused this Indenture to be duly executed, all as of the date first written above.
| |
60 DEGREES PHARMACEUTICALS, INC.,
as the Company |
| |
|
|
| |
By: |
|
| |
Name: |
|
| |
Title: |
|
| |
_________________, as the Trustee |
Exhibit 5.1

July 12, 2024
60 Degrees Pharmaceuticals, Inc.
1025 Connecticut
Avenue NW Suite 1000
Washington, D.C.
20036
| |
Re: |
Registration Statement on Form S-3 |
Dear Board of Directors:
We have acted as counsel to
60 Degrees Pharmaceuticals, Inc., a Delaware corporation (the “Company”), in connection with the filing of a registration
statement on Form S-3 (the “Registration Statement”) under the Securities Act of 1933, as amended (the “Securities Act”).
The Registration Statement relates to the offer and sale by the Company, from time to time pursuant to Rule 415 under the Securities Act,
of (i) common stock, $0.0001 par value per share, of the Company (the “Common Stock”); (ii) preferred stock, $0.0001 par value
per share, of the Company (the “Preferred Stock” and, collectively with the Common Stock, the “Capital Stock”);
(iii) warrants to purchase Common Stock or Preferred Stock (the “Warrants”); (iv) units consisting of any combination of Common
Stock, Preferred Stock or Warrants (the “Units”); and (v) debt securities (the “Debt Securities”) having an aggregate
initial public offering price not to exceed $15,000,000, in each case on terms to be determined at the time of offering by the Company.
The Common Stock, Preferred Stock, Warrants, Units and Debt Securities are collectively referred to herein as the “Securities.”
The Securities will be offered in amounts, at prices and on terms to be set forth in supplements (each, a “Prospectus Supplement”)
to the prospectus (the “Prospectus”) contained in the Registration Statement.
In connection with the foregoing,
we have examined originals or copies of such corporate records of the Company, certificates and other communications of public officials,
certificates of officers of the Company and such other documents as we have deemed relevant or necessary for the purpose of rendering
the opinions expressed herein. As to questions of fact material to those opinions, we have, to the extent we deemed appropriate, relied
on certificates of officers of the Company and on certificates and other communications of public officials. We have assumed the genuineness
of all signatures on, and the authenticity of, all documents submitted to us as originals, the conformity to authentic original documents
of all documents submitted to us as copies thereof, the due authorization, execution and delivery by the parties thereto other than the
Company of all documents examined by us, and the legal capacity of each individual who signed any of those documents. In addition, we
have assumed that:
| |
(a) |
The Certificate of Incorporation, as corrected (the “Certificate of Incorporation”), and the Amended and Restated Bylaws (the “Bylaws”) of the Company, each as amended through the date hereof, will not have been further amended in any manner that would affect any legal conclusion set forth herein and all Certificates of Designation in respect of any series of Preferred Stock will be in conformity therewith and with applicable law; |
| |
|
|
| |
(b) |
The Company will be validly existing and in good standing under the laws of the State of Delaware as of the date that any Securities are offered or sold; |
| |
|
|
| |
(c) |
The consideration paid for any shares of Capital Stock will comply with the Delaware General Corporation Law (the “DGCL”) or any successor provision; |
| |
(d) |
The Registration Statement, and any amendments thereto (including post-effective amendments), will have become effective under the Securities Act and such effectiveness will not have been terminated or rescinded; |
| |
(e) |
A Prospectus Supplement will have been prepared and filed with the Securities and Exchange Commission describing the Securities offered thereby; |
| |
|
|
| |
(f) |
All Securities will be offered and sold in compliance with applicable federal and state securities or “blue sky” laws and in the manner specified in the Registration Statement and applicable Prospectus Supplement; |
| |
|
|
| |
(g) |
With respect to Securities to be offered through an agent, underwriter or dealer or to or through a market maker, the form, terms and conditions of a definitive purchase, placement, agency, underwriting or similar agreement with respect to such Securities or, with respect to Securities to be sold by the Company directly to investors in privately negotiated transactions, the form, terms and conditions of a definitive purchase agreement with respect to such Securities (such agreement with respect to any offering of Securities, the “Definitive Agreement”), will have been duly authorized and validly executed and delivered by the Company and the other parties thereto; |
| |
|
|
| |
(h) |
In the case of Common Stock, the Board of Directors of the Company (“Board”) will have taken all necessary corporate action to approve the issuance of the Common Stock, and the issuance of such shares of Common Stock will not violate any applicable law or result in a default under or breach of any agreement or instrument binding upon the Company and will comply with any requirements or restrictions imposed by any court or governmental body having jurisdiction over the Company; |
| |
|
|
| |
(i) |
In the case of Preferred Stock of any series, the Board will have taken all necessary corporate action to approve the issuance of the Preferred Stock of such series and to designate and establish the terms of such series, will have caused an appropriate Certificate of Designations, Rights and Preferences or amendment to the Articles of Incorporation (after obtaining all required stockholder approvals) with respect to such series of Preferred Stock to be prepared and filed with the Secretary of State of the State of Delaware, and the issuance of the Preferred Stock and the terms of such series of Preferred Stock will not violate any applicable law or result in a default under or breach of any agreement or instrument binding upon the Company and will comply with any requirements or restrictions imposed by any court or governmental body having jurisdiction over the Company; |
| |
|
|
| |
(j) |
In the case of Warrants, (i) the Board will have taken all necessary corporate action to authorize the creation of and the terms of such Warrants and the issuance of the Securities to be issued upon exercise of such Warrants and to approve any warrant agreement relating thereto (the “Warrant Agreement”); (ii) such Warrant Agreement will have been duly executed and delivered by the Company and the warrant agent thereunder, if any, appointed by the Company; (iii) each person signing the Warrant Agreement will have the legal capacity and authority to do so; (iv) neither such Warrants nor such Warrant Agreement will include any provision that is unenforceable, that violates any applicable law or results in a default under or breach of any agreement or instrument binding upon the Company; (v) such Warrants or certificates representing such Warrants will have been duly executed, countersigned, registered and delivered in accordance with the provisions of such Warrant Agreement; and (vi) the issuance and sale of the Warrants will not violate any applicable law or result in a default under or breach of any agreement or instrument binding upon the Company and will comply with any requirements or restrictions imposed by any court or governmental body having jurisdiction over the Company; |
| |
|
|
| |
(k) |
In the case of Units, (i) the Board will have taken all necessary corporate action to authorize the creation of and the terms of such Units and the issuance of the Capital Stock or Warrants comprising such Units and to approve any unit agreement relating thereto (the “Unit Agreement”); (ii) such Unit Agreement will have been duly executed and delivered by the Company and the unit agent thereunder, if any, appointed by the Company; (iii) each person signing the Unit Agreement will have the legal capacity and authority to do so; (iv) none of the Units, the Capital Stock or Warrants comprising such Units, such Unit Agreement nor any applicable Definitive Agreement will include any provision that is unenforceable, that violates any applicable law or results in a default under or breach of any agreement or instrument binding upon the Company; (v) such Units or certificates representing such Units, if any, will have been duly executed, countersigned, registered and delivered in accordance with the provisions of such Unit Agreement; and (vi) the issuance and sale of the Units will not violate any applicable law or result in a default under or breach of any agreement or instrument binding upon the Company and will comply with any requirements or restrictions imposed by any court or governmental body having jurisdiction over the Company; |
| |
(l) |
In the case of Debt Securities, (i) the Board will have taken all necessary corporate action to authorize the creation of and the terms of such Debt Securities and approve any debt agreement relating thereto (the “Debt Agreement”); (ii) such Debt Agreement will have been duly executed and delivered by the Company and the unit agent thereunder, if any, appointed by the Company; (iii) each person signing the Debt Agreement will have the legal capacity and authority to do so; (iv) neither the Debt Agreement nor any applicable Definitive Agreement will include any provision that is unenforceable, that violates any applicable law or results in a default under or breach of any agreement or instrument binding upon the Company; (v) such Debt Securities will have been duly executed, countersigned, registered and delivered in accordance with the provisions of such Debt Agreement; and (vi) the issuance and sale of the Debt Securities will not violate any applicable law or result in a default under or breach of any agreement or instrument binding upon the Company and will comply with any requirements or restrictions imposed by any court or governmental body having jurisdiction over the Company; |
| |
|
|
| |
(m) |
Certificates representing shares of Capital Stock will have been duly executed, countersigned, registered and delivered, or if uncertificated, valid book-entry notations will have been made in the share register by the Company, in each case in accordance with the provisions of the Articles of Incorporation and Bylaws, each as amended and then in effect; |
| |
|
|
| |
(n) |
There will be sufficient Common Stock or Preferred Stock authorized under the Company’s Certificate of Incorporation as amended and then in effect and not otherwise issued or reserved for issuance; |
| |
|
|
| |
(o) |
The purchase price for Common Stock payable to the Company or, if such shares are issuable upon conversion, exchange, redemption or exercise of other Securities, the consideration payable to the Company for such conversion, exchange, redemption or exercise will not be less than the par value of such shares, and will not be less than the purchase price determined by the Board; |
| |
|
|
| |
(p) |
The purchase price for Preferred Stock payable to the Company or, if such shares are issuable upon conversion, exchange, redemption or exercise of other Securities, the consideration payable to the Company for such conversion, exchange, redemption or exercise will not be less than the par value of such shares, will not be less than the purchase price determined by Board and will not be less than the amount determined by the Board to constitute the stated capital applicable to such shares; and |
| |
|
|
| |
(q) |
Any Securities issuable upon conversion, exchange or exercise of any Securities being offered will have been duly authorized, created and, if appropriate, reserved for issuance upon such conversion, exchange or exercise. |
Based upon the foregoing,
and having due regard for such legal considerations as we deem relevant, we are of the opinion that:
| |
1. |
With respect to the Capital Stock, when the Capital Stock has been issued and delivered either (i) in accordance with the terms of the applicable Definitive Agreement, upon payment of the consideration therefor provided therein or (ii) upon conversion, exchange or exercise of any Security, in accordance with the terms of such Security or the instrument governing such Security providing for such conversion, exchange or exercise, including payment of the consideration therefor provided therein, the Capital Stock will be validly issued, fully paid and nonassessable. |
| |
|
|
| |
2. |
With respect to the Warrants, when the Warrants have been duly executed and countersigned in accordance with the Warrant Agreement and issued and delivered in accordance with the terms of the applicable Definitive Agreement upon payment of the consideration therefor provided therein, such Warrants will constitute valid and binding obligations of the Company, enforceable against the Company in accordance with their terms. |
| |
3. |
With respect to the Units, when the Units have been duly executed and countersigned in accordance with the Unit Agreement and issued and delivered in accordance with the terms of the applicable Definitive Agreement upon payment of the consideration therefor provided therein, such Units will constitute valid and binding obligations of the Company, enforceable against the Company in accordance with their terms. |
| |
4. |
With respect to the Debt Securities, when the Debt Securities have been duly executed and countersigned in accordance with the Debt Agreement and issued and delivered in accordance with the terms of the applicable Definitive Agreement upon payment of the consideration therefor provided therein, such Debt Securities will constitute valid and binding obligations of the Company, enforceable against the Company in accordance with their terms. |
We express no opinion concerning
(a) any provision that relates to severability or separability or purports to require that all amendments, supplements or waivers to be
in writing; or (b) the enforceability of indemnification or exculpation provisions to the extent they purport to relate to liabilities
resulting from or based upon negligence, misconduct or any violation of federal or state securities or blue sky laws. In addition, our
opinions in paragraphs 1, 2 and 3 above are subject to: (a) the effect of bankruptcy, insolvency, reorganization, fraudulent conveyance,
moratorium or other similar laws now or hereafter in effect relating to or affecting the rights and remedies of creditors; (b) constitution
and public policy limitations and the effect of general principles of equity, including without limitation, concepts of materiality, reasonableness,
good faith and fair dealing and the possible unavailability of specific performance or injunctive relief, regardless of whether enforcement
is considered in a proceeding in equity or at law, and the discretion of the court before which any proceeding therefore may be brought;
(c) the unenforceability under certain circumstances under law or court decisions of provisions providing for the indemnification of,
or contribution to, a party with respect to a liability where such indemnification or contribution is contrary to public policy; (d) the
rights or remedies available to any party for violations or breaches of any provisions of the Warrants and Units, as applicable, that
are immaterial or the enforcement of which would be unreasonable under the then existing circumstances, (e) the rights or remedies available
to any party for material violations or breaches that are the proximate result of actions taken by any party to the Warrants and Units,
as applicable, other than the party against whom enforcement is sought, which actions such other party is not entitled to take pursuant
to the Warrants and Units, as applicable, or that otherwise violate applicable laws, (f) the rights or remedies available to any party
that takes discretionary action that is arbitrary, unreasonable or capricious, or is not taken in good faith or in a commercially reasonable
manner, whether or not the Warrants and Units, as applicable, permit such action or (g) the effect of the exercise of judicial discretion,
whether in a proceeding in equity or at law. In addition, we express no opinion as to any right to collect any payment to the extent that
such payment constitutes a penalty, premium or forfeiture; whether the exercise of a remedy limits or precludes the exercise of another
remedy; the enforceability of any governing law and forum selection provisions contained in any Warrant Agreement, Unit Agreement or Definitive
Agreement; the effectiveness and enforceability of waivers of counterclaims, defenses, setoff, or statutory, regulatory or constitutional
rights; any modification or waiver of rules of evidence, or any provision that purports to specify which party bears the burden of proof,
for litigation or similar proceedings; waivers of broad or vague rights; waivers of commercial reasonableness or good faith and fair dealing;
or the availability of the appointment of a receiver.
The opinions expressed herein
are limited exclusively to the laws of the State of New York, and applicable provisions of the DGCL, in each case as currently in effect,
and we are expressing no opinion as to the effect of the laws of any other jurisdiction.
This opinion letter has been
prepared in accordance with the customary practice of lawyers who regularly give, and lawyers who regularly advise opinion recipients
concerning, opinions of the type contained herein.
We hereby consent to the filing
of this opinion as an exhibit to the Registration Statement and the reference to this firm under the caption “Legal Matters”
in the Prospectus. In giving such consent, we do not hereby admit that we are within the category of persons whose consent is required
under Section 7 of the Securities Act, and the rules and regulations thereunder.
This opinion is rendered to
you as of the date hereof and we assume no obligation to advise you or any person hereafter with regard to any change after the date hereof
in the circumstances or the law that may bear on the matters set forth herein even though the change may affect the legal analysis or
legal conclusion or other matters of law.
Very truly yours,
| /s/ Sichenzia Ross Ference Carmel LLP |
|
| |
|
| Sichenzia Ross Ference Carmel LLP |
|
4
Exhibit 5.2

July 12, 2024
60 Degrees Pharmaceuticals, Inc.
1025 Connecticut
Avenue NW Suite 1000
Washington, D.C.
20036
| |
Re: |
Registration Statement on Form S-3 |
Dear Board of Directors:
We have acted as counsel to
60 Degrees Pharmaceuticals, Inc., a Delaware corporation (the “Company”), in connection with the filing of a registration
statement on Form S-3 (the “Registration Statement”) under the Securities Act of 1933, as amended (the “Securities Act”).
The Registration Statement relates to the offer and sale by the Company, from time to time pursuant to Rule 415 under the Securities Act,
of (i) common stock, $0.0001 par value per share, of the Company (the “Common Stock”); (ii) preferred stock, $0.0001 par value
per share, of the Company (the “Preferred Stock” and, collectively with the Common Stock, the “Capital Stock”);
(iii) warrants to purchase Common Stock or Preferred Stock (the “Warrants”); (iv) units consisting of any combination of Common
Stock, Preferred Stock or Warrants (the “Units”); and (v) debt securities (the “Debt Securities”) having an aggregate
initial public offering price not to exceed $15,000,000, in each case on terms to be determined at the time of offering by the Company.
The Common Stock, Preferred Stock, Warrants, Units and Debt Securities are collectively referred to herein as the “Securities.”
The Securities will be offered in amounts, at prices and on terms to be set forth in supplements (each, a “Prospectus Supplement”)
to the prospectus (the “Prospectus”) contained in the Registration Statement.
A separate opinion has been
rendered in connection with certain matters relating to the Registration Statement, which is being filed as Exhibit 5.1 thereto.
With respect to certain matters
in connection with the proposed offer and sale by the Company of an aggregate of up to $1,253,603, of shares (the “Shares”)
of the Common Stock, pursuant to the Registration Statement, the Prospectus and a Prospectus Supplement relating to the offer and sale
of the Shares (the Prospectus and the Prospectus Supplement shall collectively be referred to as the “Sales Prospectus”).
We understand that the Shares are proposed to be offered and sold by the Company through WallachBeth Capital LLC, as sales agent (the
“Agent”), pursuant to that certain At the Market Offering Sales Agreement, dated as of July [*], 2024, by and between the
Sales Agent and the Company (the “Sales Agreement”).
In our capacity as your counsel
in connection with such registration, we are familiar with the proceedings taken and proposed to be taken by the Company in connection
with the preparation and filing of the Registration Statement, the preparation and filing of the Sales Prospectus, the negotiation and
execution of the Sales Agreement, and the authorization, issuance and sale of the Shares.
In connection with the preparation
of this supplemental opinion, we have examined such documents and considered such questions of law as we have deemed necessary or appropriate.
We have assumed the authenticity of all documents submitted to us as originals, the conformity to originals of all documents submitted
to us as copies thereof and the genuineness of all signatures.
Based
on the foregoing, we are of the opinion that the Shares have been duly authorized and, when issued and sold in the manner described in
the Registration Statement, the Sales Prospectus and the Sales Agreement, will be validly issued, fully paid and non-assessable.
The opinions expressed herein
are limited exclusively to the laws of the State of New York, and applicable provisions of the Delaware General Corporation Law in each
case as currently in effect, and we are expressing no opinion as to the effect of the laws of any other jurisdiction.
This supplemental opinion
is for your benefit in connection with the offer and sale of the Shares pursuant to the Registration Statement and may be relied upon
by you and by persons entitled by law to rely upon it pursuant to the applicable provisions of the U.S. federal securities laws. We hereby
consent to the use of this opinion as Exhibit 5.2 to the Registration Statement, and further consent to the use of our name under the
caption “Legal Matters” in the Sales Prospectus which is part of the Registration Statement. In giving such consent, we do
not hereby admit that we are within the category of persons whose consent is required under Section 7 of the Securities Act or the rules
and regulations thereunder.
This opinion letter has been
prepared in accordance with the customary practice of lawyers who regularly give, and lawyers who regularly advise opinion recipients
concerning, opinions of the type contained herein.
This opinion is rendered as
of the date hereof and based solely on our understanding of facts in existence as of such date after the examination described in this
supplemental opinion. We assume no obligation to advise you of any fact, circumstance, event or change in the law or the facts that may
hereafter be brought to our attention whether or not such occurrence would affect or modify the opinions expressed herein.
Very truly yours,
| /s/ Sichenzia Ross Ference Carmel LLP |
|
| |
|
| Sichenzia Ross Ference Carmel LLP |
|
Exhibit 10.1
SPONSORED PROJECT AGREEMENT
Sponsor Agreement Number (if any): N/A
NC State RED
Number: PAM-P24-003709
This Sponsored Project Agreement is
entered into by and between North Carolina State University, Raleigh North Carolina (hereinafter called “University”), and
60 Degrees Pharmaceuticals INC with a principal place of business at 1025 Connecticut Avenue NW Suite 1000, Washington DC, 20036 (hereinafter
called “Sponsor”) (Individually referred to as a “Party” and collectively hereinafter called “Parties”).
RECITALS
| A. | The project contemplated by this Agreement is of mutual interest and benefit to University and Sponsor
and will further the instruction, research and public service objectives of University in a manner consistent with its status as a public
educational institution; and |
| B. | Sponsor desires to provide non-federal funding in support of this project in exchange for receiving certain
rights in the results. |
NOW, THEREFORE, the Parties
agree as follows:
| 1. | Project Field of Research |
University agrees to use reasonable
effort to conduct the project as described in the proposal entitled “Prevalence and characterization of Babesia species in blood
samples from people with chronic illness, severe fatigue (CFS or Chronic Fatigue Syndrome), and neurological symptoms” (hereinafter
called “Project”) that is hereby incorporated into this Agreement as Appendix “A.”
############# will serve as Principal
Investigator and will supervise the Project. If for any reason the Principal Investigator is unable to continue to serve and a successor
acceptable to both Parties is not available, this Agreement may be terminated as hereinafter provided.
The Project will begin on June 1, 2024.
It will be completed by May 31, 2025. This period of performance will be subject to renewal or extension only by written approval of the
Sponsor. Said approval may be granted by an authorized representative of the Sponsor via email correspondence, letter of approval, or
written modification in accordance with Article 18.
| (a) | In consideration of University’s performance hereunder, Sponsor agrees to pay the University the firm
fixed price of $75,240.00. University agrees not to exceed this amount without prior written authorization of Sponsor. |
| (b) | The University will provide invoices to Sponsor in accordance with the schedule below, and invoices should
be paid by Sponsor within thirty (30) days of receipt: |
$37,620 upon
execution of this Agreement
$22,572 six (6) months from the Agreement
start date
$15,048 at the Agreement end date
University shall send Sponsor invoices
to the following individual using the contact information provided below:
Attention: #############
60 Degrees Pharmaceuticals, INC
1025 Connecticut Ave NW, Suite 1000
Washington DC, 20036
Phone: #############
Email: #############@60degreespharma.com
Sponsor shall submit Payments to University
using the address below:
North Carolina State University
Office of Contracts & Grants
Campus Box 7214
2601 Wolf Village Way, Suite 240
Raleigh, NC 27695-7214
Submit financial questions to a Contracts
& Grants Authorized Representative via telephone at 919-515-2153, or via email at cnghelpdesk@ncsu.edu.
| (c) | University’s budget, whether or not incorporated into this Agreement, was provided for a cost estimate
only. University shall have the discretion to alter said budget according to University policies in order to complete the Project. University
is under no obligation to provide the Sponsor with any kind of financial reporting, supporting documentation, or justification of expenditures
made in the performance of the Project as a condition of payment. |
| (d) | Payment not received after thirty (30) days of receipt of an invoice shall be deemed late and may be subject
to collections and attorney’s fees. University reserves the right to terminate this Agreement, pursuant to the provisions of Article
10, described herein, should payment be delayed, without cause and/or mutual agreement by both Parties, by more than sixty (60) days from
the due dates described above. |
| 5. | Export Controls and Confidentiality |
| (a) | The Sponsor does not anticipate exchanging any information, data, materials, equipment, or software that
is export controlled under the Export Administration Regulations (EAR), Title 15, sections 730-774 of the Code of Federal Regulations
(CFR) or the International Traffic in Arms Regulations (ITAR), 22 CFR §§ 120-130. Sponsor agrees that in addition to the requirements
of paragraph (b), Sponsor will give University fifteen (15) days advance written notice of their intention to deliver any information,
data, materials, equipment, software, or technology that is export controlled. University shall have the right to refuse said export-controlled
information, or the right to terminate the Project in accordance with Article 10 should it be unable to meet the necessary compliance
requirements. |
| (b) | In the performance of the Project, it may be necessary for one party to disclose information that is proprietary
and confidential to the disclosing party. All such information must be disclosed in writing and designated as confidential or, if disclosed
orally, must be identified as confidential at the time of disclosure and confirmed in writing and designated as confidential within thirty
(30) days of such disclosure. Except as otherwise provided herein, for a period of three (3) years following the date of such disclosure,
the receiving party agrees to use the confidential information only for purposes of this Agreement and further agrees that it will not
disclose or publish such information except that the restrictions of this §5(b) do not apply to: |
| (i) | information that is or becomes publicly known through no fault of the receiving party; |
| (ii) | information learned from a third party entitled to disclose it; |
| (iii) | information already known to or developed by receiving party before receipt from disclosing party, as
shown by receiving party’s prior written records; |
| (iv) | information for which receiving party obtains the disclosing party’s prior written permission to publish; |
| (v) | information required to be disclosed by court order or operation of law, including, but not limited to,
the North Carolina Public Records Law; or |
| (vi) | information that is independently developed by the receiving party’s personnel who are not privy
to the disclosing party’s confidential information. |
| (c) | The receiving party must use a reasonable degree of care to prevent the inadvertent, accidental, unauthorized
or mistaken disclosure or use by its employees of confidential information disclosed hereunder. |
University must deliver to Sponsor a
final Project report in accordance with Appendix A, within ninety (90) days following conclusion of Project or early termination of this
Agreement.
| (a) | “Intellectual Property” means all forms of intellectual property under the laws of any state
or country, including but not limited to, patentable inventions, patentable plants, copyrightable works, mask works, novel plant varieties,
trademarks, service marks, and trade secrets, that is conceived and actually or constructively reduced to practice by one or more employees
of the University and/or Sponsor, alone or in collaboration with others, in performance of the Project. Inventorship will be determined
in accordance with Title 35 of the United States Code (US Patent Law), and authorship will be determined in accordance with the US Copyright
Act. |
| (b) | University and/or Sponsor own any Intellectual Property conceived and actually or constructively reduced
to practice in the performance of the Project in accordance with the following criteria: |
| (i) | University or Sponsor exclusively owns title to any Intellectual Property conceived and actually or constructively
reduced to practice solely by their respective employees in the performance of the Project (hereinafter called “University Intellectual
Property or “Sponsor Intellectual Property”). |
| (ii) | University and Sponsor jointly own title to any Intellectual Property conceived and actually or constructively
reduced to practice by one or more University employee and one or more Sponsor employee in the performance of the Project (hereinafter
called “Joint Intellectual Property”). |
| (c) | Each party shall promptly disclose to the other party in writing any Intellectual Property of which it
becomes aware during the term of the Project or afterwards. University Principal Investigator must disclose inventions to University Office
of Research Commercialization via eDisclosure portal. University Office of Research Commercialization will disclose invention(s) to Sponsor. |
| (d) | The parties shall agree on the procedure to be used for the protection and administration of Joint Intellectual
Property. University shall have the first right to file a patent application on Joint Intellectual Property in the names of both parties.
All expenses incurred in obtaining and maintaining any patent on such Joint Intellectual Property shall be equally shared except that,
if one Party declines to share in such expenses, the other Party may take over the prosecution and maintenance thereof, at its own expense,
provided that title to the patent remains in the names of both Parties. |
| (e) | Each Party shall have the independent, unrestricted right to license to third parties any such Joint Intellectual
Property without accounting to the other Party, except that the Sponsor shall be entitled to the first right to negotiate for a fee or
royalty-bearing exclusive license or fee bearing option as described in Article 8(b) below. |
| (a) | In consideration for sponsoring the Project, the University shall grant to Sponsor a fully paid-up, non-exclusive,
non-transferable, royalty-free license (without the right to sublicense) to use all Project data and the final report from the Project
owned by University, either solely or jointly, exclusively for Sponsor’s own internal, research and development purposes. |
| (b) | Sponsor also has the first right to negotiate for a fee or royalty-bearing exclusive license or fee bearing
option to any University Intellectual Property and/or University’s interest in Joint Intellectual Property, provided that Sponsor
pays all costs for the preparation, filing, prosecution and maintenance of any patents on such Intellectual Property (“Negotiation
Right”). Sponsor has 90 days following disclosure of an Intellectual Property by the University to exercise its Negotiation Right
(the “Negotiation Period”). The Sponsor must submit a written notice to the University, within the Negotiation Period, in
order to exercise their Negotiation Right. If the Negotiation Period expires before University receives the Sponsor’s written notice
exercising the Negotiation Right or as provided below, the Sponsor shall have no further rights to the Intellectual Property. |
| (c) | If Sponsor has not begun good faith, substantive negotiations with University for an option or license
to such Intellectual Property within 90 days of the date of Sponsor’s exercising its Negotiation Right in accordance paragraph 8(b),
the University may, in its sole and unfettered discretion, terminate Sponsor’s Negotiation Right. |
| (d) | Further, if Sponsor and University have not negotiated a mutually agreeable license or option agreement
on or before twelve months from the disclosure of the Intellectual Property by the University, then University may, in its sole and unfettered
discretion, terminate Sponsor’s Negotiation Right. Sponsor is only obligated to pay expenses incurred and noncancelable obligations
that have accrued up to the date that University terminates Sponsor’s Negotiation Right. |
University has the right to publish
any of the results of the Project. University must furnish Sponsor with a copy of any proposed publication or public disclosure, at least
sixty (60) days in advance of the proposed publication date to allow for the protection of Sponsor’s proprietary, confidential, or information
that if published within sixty (60) days would have an adverse effect on a patent application in which Sponsor owns full or part interest,
or intends to obtain an interest from University pursuant to this Agreement. In the event that Sponsor notifies the University in writing
that the proposed publication or presentation contains Sponsor confidential information as described in Article 5, the University shall
remove the identified Sponsor confidential information from the draft prior to such publication or presentation. In the event Sponsor
requests a delay in publication to file for patent protection, a written request must be submitted to the University’s Office of
Research Commercialization with a copy to the University’s Principal Investigator and the University and the Principal Investigator
shall refrain from making such publication or presentation for a maximum of ninety (90) days from the receipt of such request, and Sponsor
shall indicate with specificity to what manner and degree University may disclose said information during the ninety (90) day period.
Either Party may terminate performance
under this Agreement at any time upon thirty (30) days written notice to the other Party. Upon receipt of notification, University must
proceed in an orderly fashion to limit or terminate any outstanding commitments. Sponsor agrees to reimburse University for all costs
and noncancelable obligations including graduate assistantships, fellowships, and postdoctoral associate appointments incurred in performance
of the Project prior to receipt of termination notice. University agrees to reimburse Sponsor any funds that have been received but remain
unexpended at the time of termination, except for those funds needed to pay for noncancelable obligations.
Neither Party will use the name of the
other in any form of advertising or publicity related to commercial sales without the express written permission of the other party. Sponsor
is advised that this Agreement is subject to the North Carolina Public Records Law and as such, the existence of this Agreement is recorded
in a database accessible to the public.
Any notices required to be given or
which may be given under this Agreement must be in writing delivered by private overnight mail service, first-class mail, facsimile, or
by electronic mail (email) addressed to the Parties as follows:
| |
For University: |
For Sponsor: |
| |
North Carolina State University
Sponsored Programs office
2601 Wolf Village Way
Campus Box 7514
Raleigh, NC 27695-7514
United States of America
Phone: #############
Fax: #############
E-mail: #############@ncsu.edu |
Attention: #############
60 Degrees Pharmaceuticals, INC
1025 Connecticut Ave NW, Suite 1000
Washington DC, 20036, USA
Phone: #############
Email: #############@60degreespharma.com |
For purposes of this Agreement the Parties
are independent contractors and neither may be considered an agent or an employee of the other at any time or for any purpose. No joint
venture, partnership or like relationship is created between the Parties by this Agreement.
This Agreement is binding upon and inures
to the benefit of the Parties and may be assigned only to the successors to substantially the entire business and assets of the respective
Parties. Any other assignment by either party without the prior written consent of the other party is void.
This Agreement is acknowledged to have
been made and must be construed and interpreted in accordance with the laws of the State of North Carolina, without regard for its conflict
of laws provisions, provided that all questions concerning the construction or effect of patent applications and patents shall be decided
in accordance with the laws of the country in which the particular patent application or patent concerned has been filed or granted, as
the case may be.
| (a) | The Sponsor will indemnify, defend, and hold harmless University, its trustees, officers, employees and
agents from and against any liabilities, damages, or claims (including attorneys’ fees) arising out of injuries (including death) or property
damage suffered by any person arising out of Sponsor’s use or possession of the results or inventions produced hereunder or as a result
of Sponsor’s negligence or willful misconduct. |
| (b) | Notwithstanding any other provision of this Agreement, the liability of the University, as an agency of
the State of North Carolina, for any injury or damage arising out of this Agreement or the University’s performance of the Project
is subject to the immunities, procedures and limitations of the North Carolina Tort Claims Act, GS §143-291 et seq. The University
does not waive any rights or defenses under this Act. |
| (c) | All work under this Agreement will be classified as research. University disclaims all warranties, whether
express or implied, including without limitation warranties of merchantability, fitness for a particular purpose, and freedom from infringement,
as to any information, result, design, prototype, product or process deriving directly or indirectly and in whole or part in connection
with work accomplished under this Agreement. |
If any provisions stated in this Agreement,
resulting Sponsor purchase orders, and/or appendices are in conflict, the order of precedence, beginning with the first to last, shall
be (1) this Agreement, and (2) Appendix A. The Parties understand and agree that any purchase order or similar document issued by Sponsor,
even if signed by University, will be for the sole purpose of establishing a mechanism for payment of any sums due and owing hereunder.
Notwithstanding any of the terms and conditions contained in said purchase order, the purchase order will in no way modify or add to the
terms and conditions of this Agreement.
This Agreement, including appendices,
embodies the entire understanding of the Parties for this Project, superseding any prior or contemporaneous representations, either oral
or written regarding this matter. Unless otherwise specified herein, written modifications, signed by both Parties will affect changes
to this Agreement.
| 19. | Counterparts and Electronic Signatures |
This Agreement, agreements ancillary
to this agreement, and related documents entered into in connection with this agreement are signed when a party’s signature is executed
by authorized representatives of the parties via DocuSign, PDF format, or other electronic signature method and delivered by facsimile,
e-mail, or another electronic medium. These signatures must be treated in all respects as having the same force and effect as original
signatures. This Agreement may be executed by the in separate counterparts, each of which when so executed and delivered will be an original,
but all such counterparts will together constitute one and the same instrument.
If any provision of this Agreement is
held to be unenforceable for any reason, that unenforceability shall not affect the enforceability of any other provision of this Agreement,
and the Parties shall negotiate in good faith to substitute an enforceable provision with similar terms.
The rights and obligations of the Parties
that have accrued prior to the expiration or termination of this Agreement shall survive the expiration or termination of this Agreement.
The remedies provided in this Agreement
are not exclusive and the Party suffering from a breach or default of this Agreement may pursue all other remedies, both legal and equitable,
alternatively or cumulatively. No express or implied waiver by a Party of any breach or default will be construed as a waiver of a future
or subsequent breach or default. The failure or delay of any Party in exercising any of its rights under this Agreement will not constitute
a waiver of any such right, and any single or partial exercise of any particular right by any Party will not exhaust the same or constitute
a waiver of any other right provided in this Agreement.
IN WITNESS WHEREOF, the Parties hereto have executed
this Agreement effective as of the date written in Article 3.
| NORTH CAROLINA STATE UNIVERSITY |
|
60 DEGREES PHARMACEUTICALS INC |
| |
|
|
| By: |
/s/
############# |
|
By: |
/s/
############# |
| |
|
|
| Name: |
############# |
|
Name: |
############# |
| |
|
|
| Title: |
############# |
|
Title: |
############# |
| |
|
|
| Date: |
May 10, 2024 |
|
Date: |
May 10, 2024 |
-9-
Exhibit 10.2
CERTAIN APPENDIXES HAVE BEEN OMITTED
PURSUANT TO ITEM 601(a)(5) OF REGULATION S-K. THE COMPANY AGREES TO FILE SUPPLEMENTALLY TO THE COMMISSION A COPY OF ANY OMITTED APPENDIX,
UPON REQUEST. OMITTED APPENDIXES ARE REPRESENTED BY A [*].
VETERINARY TRIAL AGREEMENT
Sponsor Agreement Number (if any): Not applicable
NC State RED
Number: PAM-P24-003176
This Veterinary Trial Agreement (“Agreement”)
is entered into by and between North Carolina State University, Raleigh North Carolina (hereinafter called “University”),
and 60 Degrees Pharmaceuticals LLC with a principal place of business at 1025 Connecticut Avenue NW Suite 1000, Washington DC, 20036 (hereinafter
called “Sponsor”) (Individually referred to as “Party” and collectively hereinafter referred to as “Parties”).
RECITALS
| A. | The project contemplated by this Agreement is of mutual interest and benefit to University and Sponsor
and will further the instruction, research and public service objectives of University in a manner consistent with its status as a public
educational institution. |
| B. | Sponsor desires to conduct a veterinary study (the “Study”) of Arakoda (the “Investigational
Product”) under a protocol entitled Treatment of Canine Babesiosis (the “Protocol”), a copy of which is attached
hereto as Exhibit A; and |
| C. | Sponsor desires to provide non-federal funding in support of this Study in exchange for receiving certain
rights in the results. |
NOW, THEREFORE, the Parties
agree as follows:
1. Study
Field of Research
| (a) | University agrees to use reasonable effort to conduct the Study as described in the Protocol entitled
“Treatment of Canine Babesiosis” and incorporated herein as Appendix A. |
| (b) | University shall conduct the Study strictly in accordance with this Agreement, the Protocol, all reasonable
written instructions of Sponsor’s authorized representative(s) as set forth in this Agreement in order to ensure the safety of the
Study subjects (any such changes to the Protocol must be incorporated in a modification or an amendment pursuant to section (d) below),
and all applicable laws and government regulations (“Applicable Law”), except that the University may deviate from the Protocol
and instructions to ensure safety of Study subjects. |
| (c) | The University shall seek approval of the Study, the Protocol, and a written form of informed consent
mutually acceptable to the University and the Sponsor, from the University’s Animal Care and Use Committee (the “IACUC”).
University shall seek any other approvals from applicable internal safety or veterinary hospital review boards if required. |
| (d) | The Sponsor may amend the Protocol at any time. Any amended Protocol shall be in writing and sent to the
University and will not take effect until approved by the IACUC (when required) and University and incorporated into a duly executed modification
or amendment to this Agreement. The Parties shall negotiate in good faith with respect to any proposed amendment. If the Parties are unable
to agree upon an amendment to this Agreement, either Party may terminate this Agreement pursuant to Article 9. |
2. Principal
Investigator
############# will serve as principal
investigator (“Principal Investigator”) and will supervise the Study. If, for any reason, that person is unable to continue
to serve as Principal Investigator, Sponsor and University shall attempt to find a successor acceptable to both parties. If a successor
acceptable to both Parties is not available, this Agreement may be terminated as hereinafter provided in Article 9.
3. Sponsor
Obligations
| (a) | The Sponsor shall comply with Applicable Law, government regulations, and including, without limitation
the Food and Drug Administration’s (FDA) Good Clinical Practice, regulations governing study investigators and adverse event reporting
requirements, in the performance of its activities relating to the Study. The Sponsor shall conduct such Study-related activities in a
manner consistent with the informed consents and all other applicable consents. |
| (b) | Unless otherwise specified in the Protocol, Sponsor shall supply the University with quantities of the
Investigational Product adequate for the University to conduct the Study. The Investigational Product shall remain the sole property of
the Sponsor. The University shall take reasonable steps to ensure that it has adequate supplies of the Investigational Product to conduct
the Study in accordance with the Protocol. University shall use Investigational Product in accordance with the Protocol guidelines. |
| (c) | The Sponsor shall reimburse the University for actual and reasonable expenses incurred in treating any
injury or illness to a Study subject that is directly related to the administration of the Investigational Product or the proper performance
of any other procedure performed in furtherance of the Study. |
4. Period
of Performance
The Study will begin on February 1,
2024. It will be completed by March 30, 2026. This period of performance will be subject to renewal or extension only by written approval
of the Sponsor. Said approval may be granted by an authorized representative of the Sponsor via email correspondence, letter of approval,
or written modification in accordance with Article 19.
5. Payments: Fixed Price
| (a) | In consideration of University’s performance hereunder, Sponsor agrees to pay the University the firm
fixed price of $23,869.00. University agrees not to exceed this amount without prior written authorization of Sponsor. |
| (b) | The University will provide invoices to Sponsor in accordance with the schedule below, and invoices should
be paid by Sponsor within thirty (30) days of receipt: |
$12,000 upon
execution of this Agreement
$8,000 once 50% of dogs are enrolled in
the study
$3,869 on or by Agreement
end date
University shall send Sponsor invoices
to the following individual using the contact information provided below:
60 Degrees Pharmaceuticals, Inc
ATTN: #############
1025 Connecticut Ave NW, Suite 1000
Washington DC, 20036
Phone: #############
Email: #############@60degreespharma.com
Sponsor shall submit Payments to University
using the address below:
North Carolina State University
Office of Contracts and Grants
Box 7214, 2701 Sullivan Dr.
Raleigh, NC 27695-7214
Submit financial questions to a Contracts
& Grants Authorized Representative via telephone at 919-515-2153, or via email at cnghelpdesk@ncsu.edu.
| (c) | University’s budget, whether or not incorporated into this Agreement, was provided for a cost estimate
only. University shall have the discretion to alter said budget according to University policies in order to complete the Study. University
is under no obligation to provide the Sponsor with any kind of financial reporting, supporting documentation, or justification of expenditures
made in the performance of the Study as a condition of payment. |
| (d) | Payment not received after thirty (30) days of receipt of an invoice shall be deemed late and may be subject
to collections and attorney’s fees. University reserves the right to terminate this Agreement, pursuant to the provisions of Article
9, described herein, should payment be delayed, without cause and/or mutual agreement by both Parties, by more than sixty (60) days from
the due dates described above. |
6. Export
Controls and Confidentiality
| (a) | The Sponsor does not anticipate exchanging any information, data, materials, equipment, or software that
is export controlled under the Export Administration Regulations (EAR), Title 15, sections 730-774 of the Code of Federal Regulations
(CFR) or the International Traffic in Arms Regulations (ITAR), 22 CFR §§ 120-130. Sponsor agrees that in addition to the requirements
of paragraph (b), Sponsor will give University fifteen (15) days advance written notice of their intention to deliver any information,
data, materials, equipment, software, or technology that is export controlled. University shall have the right to refuse said export-controlled
information, or the right to terminate the Study in accordance with Article 8 should it be unable to meet the necessary compliance requirements. |
| (b) | In the performance of the Study, it may be necessary for one party to disclose information that is proprietary
and confidential to the disclosing party. All such information must be disclosed in writing and designated as confidential or, if disclosed
orally, must be identified as confidential at the time of disclosure and confirmed in writing and designated as confidential within thirty
(30) days of such disclosure. Except as otherwise provided herein, for a period of five (5) years following the date of such disclosure,
the receiving party agrees to use the confidential information only for purposes of this Agreement and further agrees that it will not
disclose or publish such information except that the restrictions of this Article 6(b) do not apply to: |
| (i) | information that is or becomes publicly known through no fault of the receiving party; |
| (ii) | information learned from a third party entitled to disclose it; |
| (iii) | information already known to or developed by receiving party before receipt from disclosing party, as
shown by receiving party’s prior written records; |
| (iv) | information for which receiving party obtains the disclosing party’s prior written permission to publish; |
| (v) | information required to be disclosed by court order or operation of law, including, but not limited to,
the North Carolina Public Records Law; or |
| (vi) | information that is independently developed by the receiving party’s personnel who are not privy
to the disclosing party’s confidential information. |
| (a) | The receiving party must use a reasonable degree of care to prevent the inadvertent, accidental, unauthorized
or mistaken disclosure or use by its employees of confidential information disclosed hereunder. |
| (b) | Permitted Disclosures. Notwithstanding the section above, Confidential Information may be disclosed to
third parties to the extent that it: |
| i. | is disclosed to Study subject owners or prospective Study subject owners as reasonably necessary or appropriate
in the course of discussions regarding the Informed Consent; |
| ii. | is disclosed to personnel at other study sites as required for the performance of the Study as approved
by Sponsor; |
| iii. | is required to be disclosed by the University by Applicable Law or regulatory agency. For disclosures
made pursuant to Article 6(d), the University shall use reasonable efforts to disclose the minimum Sponsor Confidential Information necessary
to comply with such requirement. University shall give the Sponsor advance notice of the disclosure when practicable. Reasonable efforts
shall be made to provide this notice in sufficient time to allow the Sponsor to seek an appropriate protective order or modification of
any disclosure. |
7. Records,
Reporting, Audits, Data, and Ownership
| (a) | Study Records. The University shall keep Study data (“Study Data”) and records required by
the Protocol and Applicable Law (the “Study Documents”), including any source documents and Study Deliverables (defined in
this section below) for three (3) years from the Agreement’s early termination or natural expiration. |
| (b) | Periodic Reporting. The University shall provide to the Sponsor the Study Deliverables containing the
data as specified in the Protocol or as otherwise agreed in writing by the Parties. |
| (c) | The University shall make available to the Sponsor (or its agent) the Study site, the study staff, and
all Study Documents for purposes of review and audit upon reasonable advance notice during regular business hours. |
| (d) | The University shall provide the Sponsor prompt, advance notification of any audit directly related to
the Study by a regulatory authority (or, when advance notification is impracticable, prompt notification of any completed audit). To the
extent possible, the University shall permit the Sponsor to review and comment in advance on any written communication from the University
to the regulatory authority in connection with such an audit, if the review does not adversely impact the timeliness of the University’s
response to the regulatory authority. The University shall promptly provide the Sponsor with copies of all communications between the
University and the regulatory authority related to such audit unless prohibited from so doing by the regulatory authority. University
shall promptly take action to correct any deficiencies found by the regulatory authority during the audit. With respect to a pending audit
by the FDA or by any comparable foreign regulatory authority with jurisdiction over drug/device approval directly related to the Study,
the University shall permit the Sponsor’s representatives to be present at such audit unless prohibited from so doing by the regulatory
authority. With respect to any audit by any regulatory authority, which audit is not directly related to the Study, the University shall
promptly notify the Sponsor of any findings of such an audit that would be likely to have an adverse effect on the University’s
ability to conduct the Study. |
| (e) | Without the prior written consent of the Sponsor, University shall not make Study Data available to any
third party in a manner that would reasonably enable such third party to reconstruct the compilation of data contained in the Study Deliverables
(or to construct a substantially similar compilation). However, University may make available Study Data (but may not permit the copying
of such data) to a third party in connection with the peer review of the results of the Study for purposes of Publication in a peer-reviewed
scientific journal subject to Article 8. |
| (f) | All rights, title, and interest in: (i) a Sponsor-written Protocol, (ii) the operations manuals provided
by the Sponsor for use at the Study site, and (iii) any other scientific, technical, business, or other data or information relating to
the Investigational Product and/or this Agreement that is disclosed to the University by the Sponsor (collectively the “Sponsor
Data”) shall be the sole and exclusive property of the Sponsor. Furthermore, all rights (including commercial use), title, and interest
in: (1) the completed case report forms required by the Sponsor-written Protocol (including the data contained in such case report forms),
(2) any electronic databases required to be created under the Protocol (including the data contained in such electronic databases), (3)
any Study reports prepared by the University for the Sponsor as required by the Sponsor-written Protocol (including the data contained
in such Study reports), (4) any compilation of items 1, 2, and 3 thereof (collectively the “Study Deliverables”), and (5)
any invention made by University in performance of the Protocol that relates to the Investigational Product will be owned by the Sponsor. |
All rights, title, and interest in: (a)
other documents generated by University in the course of the Study that are not Study Deliverables (examples of which may include University
required patient or subject forms and corresponding patient or subject records) collectively referred to as “University Documents”,
and (b) inventions made by University outside of the performance of a Sponsor-written Protocol or in performance of a University-written
Protocol (referred to as “University Inventions”) shall be the sole and exclusive property of the University. Sponsor shall
have the right to use the information and data contained in the University Documents for any non-commercial purpose whatsoever, subject
to Applicable Law and the terms of the Informed Consents. University also grants Sponsor the first, time-limited right to negotiate a
license to make, have made, use, and sell any University Inventions, and the terms and conditions of such license will be negotiated diligently
and in good faith (“Sponsor’s Option to Negotiate”). Sponsor’s Option to Negotiate will be valid for six (6) months
after University discloses any such University Invention(s) to Sponsor. The term of Sponsor’s Option to Negotiate may be extended
by mutual agreement of the Parties. University reserves the right to utilize all Study Deliverables, inventions made by University in
performance of the Protocol, and University Documents for its internal research, development, and educational purposes.
| (g) | In addition to the terms provided above, University will comply with any required regulatory reporting
regarding adverse events required under Applicable Law including but not limited to any U.S. Department of Agriculture, Environmental
Protection Agency, and Food & Drug Administration regulations subject to animal testing and trials. |
8. Publications
| a) | University has the right to publish any of the results of the Study (including an article, manuscript,
abstract, report, poster, presentation, or other material, a “Publication”). Publications may include any of (i) an analysis
of the results of the Study; (ii) a summary of the Protocol; and (iii) supporting data generated by the Study and identifying information
regarding the Investigational Product, which would be reasonably required for purposes of publication in a peer-reviewed scientific journal. |
| b) | University must furnish Sponsor with a copy of any proposed publication or public disclosure, at least
sixty (60) days in advance of the proposed publication date to allow for the protection of Sponsor’s proprietary, confidential, or information
that if published within sixty (60) days would have an adverse effect on a patent application in which Sponsor owns full or part interest
or intends to obtain an interest from University pursuant to this Agreement. In the event that Sponsor notifies the University in writing
that the proposed publication or presentation contains Sponsor confidential information as described in Article 6, the University shall
remove the identified Sponsor confidential information from the draft prior to such publication or presentation. In the event Sponsor
requests a delay in publication to file for patent protection, a written request must be submitted to the University’s Office of
Research Commercialization with a copy to the University’s Principal Investigator and the University and the Principal Investigator
shall refrain from making such publication or presentation for a maximum of ninety (90) days from the receipt of such request, and Sponsor
shall indicate with specificity to what manner and degree University may disclose said information during the ninety (90) day period. |
9. Termination
Either Party may terminate performance
under this Agreement at any time upon thirty (30) days written notice to the other Party. Upon receipt of notification, University must
proceed in an orderly fashion to limit or terminate any outstanding commitments. Sponsor agrees to reimburse University for all costs
and noncancelable obligations including graduate assistantships, fellowships, and postdoctoral associate appointments incurred in performance
of the Project prior to receipt of termination notice. University agrees to reimburse Sponsor any funds that have been received but remain
unexpended at the time of termination, except for those funds needed to pay for noncancelable obligations.
Within thirty (30) days of termination
or expiration of this Agreement, the University shall return to the Sponsor, at the Sponsor’s expense, any remaining Investigational
Product (except as required by law), any equipment on loan or lease from the Sponsor, and any copies of Confidential Information, except
that University may retain any copies to the extent required by Applicable Law. At the Sponsor’s request and expense, the University
shall dispose of the remaining Investigational Product in accordance with Sponsor’s instructions, subject to Applicable Law.
Additionally, if this Agreement is terminated
before completion of the Study, University shall cease enrolling Study subjects immediately, and shall cease conducting the procedures
set out in the Protocol to the extent that doing so is medically permissible and appropriate. The Sponsor and University shall negotiate
in good faith on the subsequent treatment or transfer of the study subjects. In case of termination of the Study before completion, the
Sponsor shall reimburse University for (i) obligations incurred in accordance with the Study budget that cannot be cancelled or mitigated
by University using reasonable efforts, (ii) reasonable costs incurred in connection with the safe withdrawal of study subjects from the
Study, and (iii) mutually agreed post-termination expenses.
University shall submit a final invoice
and completed case report forms (sent under separate cover) to Sponsor within sixty (60) days post termination or expiration of the Agreement.
10. Use
of Names
Neither party will use the name of the
other in any form of advertising or publicity related to commercial sales without the express written permission of the other party. Sponsor
is advised that this Agreement is subject to the North Carolina Public Records Law and as such, the existence of this Agreement is recorded
in a database accessible to the public. Sponsor has the right to identify the University as the entity that completed the Study and generated
the Study Data. Any such reference must contain the following non-endorsement statement: “Reference to NC State University herein
does not imply endorsement by NC State University for the specified product and/or service.”
11. Notices
Any notices required to be given or
which may be given under this Agreement must be in writing delivered by private overnight mail service, first-class mail, facsimile, or
by electronic mail (email) addressed to the Parties’ authorized representatives as follows:
| |
For University: |
For Sponsor: |
| |
North Carolina State University
Sponsored Programs and Regulatory Compliance
Services
2601 Wolf Village Way, Suite 240
Campus Box 7514
Raleigh, North Carolina 27695-7514
United States of America
Phone: #############
Fax: #############
E-mail: #############@ncsu.edu |
60 Degrees Pharmaceuticals, Inc
1025 Connecticut Ave NW, Suite 1000
Washington DC, 20036
United States of America
Phone: #############
Email: #############@60degreespharma.com |
12. Independent
Parties
For purposes of this Agreement the Parties
are independent contractors and neither may be considered an agent or an employee of the other at any time or for any purpose. No joint
venture, partnership or like relationship is created between the Parties by this Agreement.
13. Assignment
This Agreement is binding upon and inures
to the benefit of the Parties and may be assigned only to the successors to substantially the entire business and assets of the respective
Parties. Any other assignment by either party without the prior written consent of the other party is void.
14. Governing
Law
This Agreement is acknowledged to have
been made and must be construed and interpreted in accordance with the laws of the State of North Carolina, without regard for its conflict
of laws provisions, provided that all questions concerning the construction or effect of patent applications and patents shall be decided
in accordance with the laws of the country in which the particular patent application or patent concerned has been filed or granted, as
the case may be.
15. Representations
Each Party represents that it has and/or
will maintain during the term of this Agreement all regulatory approvals required for the conduct of its respective activities in connection
with the Study, and that all the persons who perform activities under this Agreement on its behalf (including, in the case of the University,
the Study staff) have and will have the necessary expertise, training, qualifications, and certifications.
16. Liability
and Insurance
| (a) | The Sponsor shall indemnify, defend, and hold harmless the University and its officers, directors, employees,
and agents from any loss, liability, damage, or expense (including reasonable attorneys’ fees and costs until such time as the Sponsor
assumes the defense) from any claim of personal injury (including death) or property damage that may arise directly from the Study, Results
of the Study, or inventions produced hereunder. However, to the extent that the claim is a direct result of (a) the failure of the University
or one of its officers, employees, or agents (including the Principal Investigator) to follow the Protocol or the Sponsor’s written
instructions (each when applicable), accepted veterinary practice, or Applicable Law, or (b) any other negligence or willful misconduct
of the University or one of its officers, employees, or agents (including the Principal Investigator), the Sponsor shall have no such
obligation. |
| (b) | Notwithstanding any other provision of this Agreement, the liability of the University, as an agency of
the State of North Carolina, for any injury or damage arising out of this Agreement or the University’s performance of the Project
is subject to the immunities, procedures and limitations of the North Carolina Tort Claims Act, GS §143-291 et seq. The University
does not waive any rights or defenses under this Act. |
| (c) | All work under this Agreement will be classified as research. University disclaims all warranties, whether
express or implied, including without limitation warranties of merchantability, fitness for a particular purpose, and freedom from infringement,
as to any information, result, design, prototype, product or process deriving directly or indirectly and in whole or part in connection
with work accomplished under this Agreement. By signing this Agreement, University is in no way giving its endorsement, approval or authorization
of the Investigational Product. |
| (d) | During the term of this Agreement and for six (6) years thereafter, Sponsor shall carry liability insurance
in the type and amount appropriate and customary for the conduct and sponsorship of clinical trials, respectively (or maintain a comparable
program of self-insurance). Upon request, Sponsor shall provide to University a certificate of such insurance or evidence of such a self-insurance
plan. |
17. Order
of Precedence
If any provisions stated in this Agreement,
resulting Sponsor purchase orders, and/or appendices are in conflict, the order of precedence, beginning with the first to last, shall
be (1) this Agreement, (2) Appendix A, and (3) the Sponsor purchase order. The Parties understand and agree that any purchase order or
similar document issued by Sponsor will be for the sole purpose of establishing a mechanism for payment of any sums due and owing hereunder.
Notwithstanding any of the terms and conditions contained in said purchase order, the purchase order will in no way modify or add to the
terms and conditions of this Agreement.
18. Force Majeure
“Force Majeure” means the
occurrence of an event or circumstance that prevents or impedes a Party from performing one or more of its contractual obligations under
the Agreement, if and to the extent that that Party proves: (a) that such impediment is beyond its reasonable control; and (b) that it
could not reasonably have been foreseen at the time of the conclusion of the Agreement; and (c) that the effects of the impediment could
not reasonably have been avoided or overcome by the affected Party. In the absence of proof to the contrary, the following events affecting
a party shall be presumed to fulfil conditions (a) and (b) of this clause: (i) war (whether declared or not), hostilities, invasion, act
of foreign enemies, extensive military mobilization; (ii) civil war, riot, rebellion and revolution, military or usurped power, insurrection,
act of terrorism, sabotage or piracy; (iii) currency and trade restriction, embargo, sanction; (iv) act of authority whether lawful or
unlawful, compliance with any law or governmental order, expropriation, seizure of works, requisition, nationalization; (v) plague, epidemic,
pandemic, natural disaster or extreme natural event; (vi) explosion, fire, destruction of equipment, prolonged break-down of transport,
telecommunication, information system or energy; (vii) general labor disturbance such as boycott, strike and lock-out, go-slow, occupation
of factories and premises. A Party invoking this clause is relieved from its duty to perform its obligations under the Agreement and from
any liability in damages or from any other contractual remedy for breach of contract, from the time at which the impediment causes inability
to perform, provided that the notice thereof is given without reasonable delay. If notice thereof is not given without reasonable delay,
relief is effective from the time at which notice thereof reaches the other Party. Where the effect of the impediment or event invoked
is temporary, the above consequences shall apply only as long as the impediment invoked impedes performance by the affected Party. Where
the duration of the impediment invoked has the effect of substantially depriving the contracting parties of what they were reasonably
entitled to expect under the contract, either party has the right to terminate the contract by notification within a reasonable period
to the other party. Unless otherwise agreed, the Parties expressly agree that the contract may be terminated in accordance with Article
10 by either Party if the duration of the impediment exceeds 120 days.
19. Entire Agreement
This Agreement, including appendices,
embodies the entire understanding of the Parties for this Study, superseding any prior or contemporaneous representations, either oral
or written regarding this matter. Unless otherwise specified herein, written modifications signed by authorized representatives of both
Parties will affect changes to this Agreement.
20. Counterparts
and Electronic Signatures
This Agreement, agreements ancillary
to this agreement, and related documents entered into in connection with this agreement are signed when a party’s signature is executed
by authorized representatives of the parties via DocuSign, PDF format, or other electronic signature method and delivered by facsimile,
e-mail, or another electronic medium. These signatures must be treated in all respects as having the same force and effect as original
signatures. This Agreement may be executed by the in separate counterparts, each of which when so executed and delivered will be an original,
but all such counterparts will together constitute one and the same instrument.
21. Severability
If any provision of this Agreement is
held to be unenforceable for any reason, that unenforceability shall not affect the enforceability of any other provision of this Agreement,
and the Parties shall negotiate in good faith to substitute an enforceable provision with similar terms.
22. Survival
The rights and obligations of the Parties
that have accrued prior to the expiration or termination of this Agreement shall survive the expiration or termination of this Agreement.
23. Remedies
and Waiver
The remedies provided in this Agreement
are not exclusive and the Party suffering from a breach or default of this Agreement may pursue all other remedies, both legal and equitable,
alternatively or cumulatively. No express or implied waiver by a Party of any breach or default will be construed as a waiver of a future
or subsequent breach or default. The failure or delay of any Party in exercising any of its rights under this Agreement will not constitute
a waiver of any such right, and any single or partial exercise of any particular right by any Party will not exhaust the same or constitute
a waiver of any other right provided in this Agreement.
IN WITNESS WHEREOF, authorized representatives
of the Parties hereto have executed this Agreement effective as of the date written in Article 4.
| NORTH CAROLINA
STATE UNIVERSITY |
|
60 DEGREES
PHARMACEUTICALS, INC |
| |
|
|
| By: |
/s/
############# |
|
By: |
/s/
############# |
| |
|
|
| Name: |
############# |
|
Name: |
############# |
| |
|
|
| Title: |
############# |
|
Title: |
############# |
| |
|
|
| Date: |
3/28/24 |
|
Date: |
3/28/24 |
Appendix A
STUDY OUTLINE
(See attached.)
[*]
-13-
Exhibit 10.3
CERTAIN APPENDIXES HAVE BEEN OMITTED
PURSUANT TO ITEM 601(a)(5) OF REGULATION S-K. THE COMPANY AGREES TO FILE SUPPLEMENTALLY TO THE COMMISSION A COPY OF ANY OMITTED APPENDIX,
UPON REQUEST. OMITTED APPENDIXES ARE REPRESENTED BY A [*].
CLINICAL TRIAL AGREEMENT
PROTOCOL NUMBER: TQ-BA-2024-1 (NCT06207370)
PROTOCOL TITLE: “Double-blind
Placebo-controlled Study to Assess the Safety and Efficacy of Oral Tafenoquine plus Standard of Care versus Placebo plus Standard of Care
in Patients Hospitalized for Babesiosis”
Tufts Medicine, Inc.
800 Washington Street
Boston, MA 02111
United States
(“INSTITUTION”)
and
60 Degrees Pharmaceuticals, Inc
1025 Connecticut Ave NW, Suite 1000
Washington DC, United States, 20036
(“SPONSOR”)
Table
of Contents
Table of Contents
| 1. |
Performance of Study |
3 |
| 2. |
Study Drug, Storage and Return, and Equipment |
7 |
| 3. |
Payments |
8 |
| 4. |
Term and Termination |
9 |
| 5. |
Confidentiality |
10 |
| 6. |
Publications |
12 |
| 7. |
Intellectual Property |
13 |
| 8. |
Warranty |
|
| 9. |
Study Results, Records and Audits |
16 |
| 10. |
Indemnification |
17 |
| 11. |
Insurance |
19 |
| 12. |
Miscellaneous |
19 |
| SPONSOR NAME | Page 2 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
This Clinical Trial Agreement (this “Agreement”)
is effective as of the last date of signature (the “Effective Date”) and sets forth certain agreements by and between 60
DEGREES PHARMACEUTICALS, INC , having its principal place of business at 1025 Connecticut Ave NW, Washington DC, 20036 (“Sponsor”),
and TUFTS MEDICINE, INC., a Massachusetts not-for-profit corporation, on behalf of its affiliates, including but not limited to,
Tufts Medical Center, Inc., Lowell General Hospital, MelroseWakefield Healthcare, and Home Health Foundation, with a principal place of
business at 800 Washington Street, Boston, MA 02111INSTITUTION ADDRESS (hereinafter referred to as “Institution”).
The persons executing this Agreement hereby represent that they are authorized to do so for and on behalf of the above-named companies
and organizations.
WHEREAS, Sponsor and Institution are hereinafter
referred to individually as “Party” and collectively as “Parties;” and
WHEREAS, Sponsor is arranging to use Institution
as a clinical trial site to clinically conduct the study pursuant to the Protocol (as defined in Article 1 below) (the “Study”);
and
WHEREAS, #############
(the “Investigator”) acknowledges having read and understood the Protocol and any documents
appended to the Protocol, as well as this Agreement; and
WHEREAS, Sponsor has contracted with
Fast Track Drugs and Biologics LLC., a Maryland corporation with offices located at 20010 Fisher Avenue Suite G, Poolesville, MD,
20837 (“CRO”) to coordinate and/or perform certain activities as its clinical research organization including, but not limited
to, regulatory activities, monitoring, site initiation and close-out visits, data analysis and audits required for the conduct of the
Study.
NOW, THEREFORE, the Parties, in consideration
of the mutual certifications, representations, and promises contained herein, and for other good and valuable consideration, the sufficiency
of which is hereby acknowledged, have entered into this Agreement and do specifically agree as follows:
1. Performance of Study
1.1 Study Protocol. The scope and nature
of the clinical trial to be performed will be in strict accordance with the Protocol, including Protocol Number TQ-BA-2024-1 (NCT06207370)
entitled, “Double-blind Placebo-controlled Study to Assess the Safety and Efficacy of Oral Tafenoquine plus Standard of Care versus
Placebo plus Standard of Care in Patients Hospitalized for Babesiosis”, and any subsequent amendments thereto, referenced and incorporated
herein (the “Protocol”). The Protocol fully details the clinical research activities and responsibilities to be undertaken,
pursued, and followed with all due diligence by Institution, through the Investigator. The Protocol will be considered final after it
is signed by the Sponsor and approved by the pertinent Institutional Review Board (“IRB”). Thereafter, the Protocol may be
amended only by the Sponsor and subsequent approval by the IRB. A copy of the signed Statement of Investigator (FDA Form 1572) or Investigator
Agreement for medical device studies, the Protocol and any Protocol amendments will be maintained in the Investigator’s Study files.
| SPONSOR NAME | Page 3 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
1.2 Conduct of Study. Subject to approval
of the Study as described herein, Institution, through the Investigator, agrees to conduct the Study in strict compliance with the Protocol,
this Agreement and any amendments thereto, any and all applicable federal, state, and local laws, regulations, good clinical practices,
all reasonable written instructions of the Sponsor or its designee (e.g. CRO) and any other relevant professional standards, with the
prior approval and ongoing review of all appropriate and necessary review authorities, including the IRB. Investigator also agrees specifically
to conduct the Study in accordance with the Statement of Investigator, FDA Form 1572 (or Investigator Agreement for medical device studies),
which Investigator has completed, signed, and delivered to Sponsor prior to the commencement of the Study at the Investigator’s
site. Sponsor shall comply with all-applicable federal, state and local laws and regulations in the performance of its activities relating
to the Study, and shall obtain all approvals and consents required in connection with such activities. Sponsor shall conduct such Study-related
activities in a manner consistent with the informed consents and all other applicable consents. The Institution and Investigator each
further agrees that in the performance of the Study, they shall:
| (a) | Exercise independent medical judgment as to the compatibility
of each Study participant with Protocol requirements (applicable to Investigator only); |
| (b) | Obtain a signed informed consent form (in a form approved
by Sponsor) from each Study participant or his/her legally authorized representative and his/her caregiver in accordance with the Protocol,
which has been approved by the IRB and Sponsor in accordance with 21 CFR §56, et. seq., or any successor thereto; |
| (c) | Properly perform and direct or administer the Study in accordance
with the Protocol, the applicable laws and regulations, the guideline published by the U.S. Food and Drug Administration (“FDA”)
entitled, “Good Clinical Practices, Consolidated Guideline” as amended from time to time, and the other requirements as set
forth therein, including ISO 14155:2020 for medical device studies, if applicable; |
| (d) | Review all Study participant case report forms (hereinafter
“CRFs”) to assure their accuracy and completeness, and assist Sponsor’s representatives and clinical monitors upon
request in promptly resolving any discrepancies or errors on CRFs and in performing random audits of original patient records, laboratory
reports, or other raw data sources underlying data recorded on the CRFs; |
| (e) | Submit all data and information, and undertake all activities,
so that the timeline as defined in the schedule of assessments set forth in the Protocol and this Agreement are strictly met; |
| SPONSOR NAME | Page 4 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
| (f) | Notify Sponsor and the IRB promptly of any failures to comply
with the Protocol; |
| (g) | Maintain records of Study participant identification, clinical
observations, laboratory tests, and drug receipt, storage, return and disposition as specified in the Protocol; |
| (h) | Provide all reasonable cooperation with Sponsor and its designee
in all of their efforts to monitor the Study; |
| (i) | Pursuant to 21 CFR §312.66, assure that the IRB will
comply with the requirements of 21 CFR §56, et. Seq.; |
| (j) | Perform the research in relation to the Study with reasonable
care, diligence and skill and ensure that personnel engaged by it in the provision of such Study services are competent and have appropriate
professional qualifications, training and experience; and |
| (k) | Upon request from Sponsor, Investigator will update his/her
financial disclosure form in a timely matter one (1) year after Study completion. |
1.3 Participating Staff. The
Study shall be conducted under the immediate direction of the Investigator. Institution and Investigator shall ensure that all Staff personally
perform their assigned Study tasks, as indicated in the Protocol and in accordance with this Agreement. Investigator shall qualify such
personnel and oversee their work. Investigator may have one or more sub-investigator(s) work on the Study; provided, however, that Investigator
is responsible for all work conducted by sub-investigator. Any such sub-investigator shall be subject to all of the terms and conditions
of this Agreement, including all obligations of Investigator. No sub-investigator may work on the Study unless he or she is qualified
through experience and training to conduct clinical studies and agrees to be involved through completion of the Study. The sub-investigator’s
name shall appear in the appropriate space on the FDA Form 1572 or Investigator Agreement for medical device studies. Notwithstanding
the foregoing, Sponsor may disapprove any proposed sub-investigator within five (5) days of submission of FDA Form 1572 or Investigator
Agreement for medical device studies. For purposes of this Agreement, “Staff” shall mean all Institution personnel including,
but not limited to, researchers, investigators, sub-investigators, study coordinators, professional staff, and any other person performing
research and/or services related to the Study on behalf of Institution, or to whom Institution provides access to Study materials or study
drug (“Study Drug”).
1.4 Third Parties Staff. Investigator
shall ensure that any third-party personnel working, directly or indirectly, on the Study shall be competent, have appropriate professional
qualifications, training and experience, and are bound by obligations of confidentiality and non-use with respect to Confidential Information
(as defined by this Agreement) that are at least as restrictive as the obligations of confidentiality and non-use imposed by this Agreement.
Investigator shall qualify such personnel and oversee their work.
| SPONSOR NAME | Page 5 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
1.5 Replacement of Investigator.
In addition to any other remedy which may be available to Sponsor, in the event Investigator becomes either unwilling or unable to perform
the duties required by this Agreement, Institution shall promptly notify Sponsor of such circumstance. In addition, upon request by Sponsor,
Institution will cooperate, in good faith and expeditiously, to find a replacement Investigator acceptable to Sponsor. Institution’s
and Investigator’s cooperation in finding an acceptable replacement does not negate their obligations under this Agreement or by
law or regulation.
1.6 Notification of Adverse Events.
Investigator shall promptly notify Sponsor and the Institution’s IRB in writing of any serious adverse drug experience within twenty-four
(24) hours of learning of the event. For serious adverse drug experiences, Institution and Investigator shall assist in the investigation
of the medical circumstances and shall provide Sponsor with all requested information so that Sponsor can submit any required IND Safety
Report to FDA within fifteen (15) days of its initial receipt of the information. For fatal or life-threatening experiences, Institution
and Investigator shall provide Sponsor with all requested information so that Sponsor can submit any required Telephone and Facsimile
Transmittal Safety Report to FDA within seven (7) days of its initial receipt of the information. Investigator shall follow-up with any
Study participant who experienced an adverse drug experience and continue to provide Sponsor with updates.
1.7 Protocol Deviation. Prospective Protocol
waivers or prospective Protocol deviations will not be granted by Sponsor under any circumstance. Deviations from the Protocol which are
medically necessary for a Study participant’s health, welfare, or safety in cases of emergency are not considered failure to comply
with the Protocol. If, during the course of a Study participant’s post-randomization participation in the Study, it is discovered
that the Study participant did not meet all eligibility criteria, s/he will be discontinued, unless the discontinuation presents an unacceptable
medical risk. The justification to allow the Study participant to continue in the Study will be made by the Sponsor, with medical
input from the Investigator, and will be documented. If the Study participant is allowed to remain in the Study, this will
be reported as a major Protocol deviation and not a waiver. All follow-up safety assessments must be completed and documented as outlined
in the Protocol. Other Protocol deviations will be tracked and corrective measures will be put in place to prevent such deviations from
being repeated.
1.8 Data
and Safety Monitoring Plan and Reports Material Subject Information. If Sponsor is responsible for the data and safety monitoring
for the Study, Sponsor will comply with such data and safety monitoring plan for the Study as approved by Institution’s IRB. Such
data and safety monitoring plan will include information about the provision of monitoring reports, including urgent and routine reports,
or other pertinent determinations from the monitoring to the Investigator. Such plan shall specify the respective timeframes for providing
routine and urgent data and safety monitoring reports to the Investigator. Without limiting the foregoing, Sponsor agrees to notify Investigator
in writing promptly (no later than 30 days) of information (such as Study results or findings from a Study monitoring visit), including
results obtained for a period of two (2) years after completion or closure of the Study that could affect the safety or medical care of
subjects who were at any point enrolled in the Study, influence the conduct of the Study, or alter the IRB’s approval. Sponsor and
Institution shall comply with, and nothing herein shall limit, their respective reporting requirements to regulatory authorities, including,
for example, the Food and Drug Administration, the Office for Human Research Protections, and others as required. Institution, through
the Investigator and/or IRB as appropriate, shall be responsible for informing subjects of the above important information they learn
from Sponsor in accordance with the IRB-approved informed consent form and Sponsor shall not contact them. No other provision of
this Agreement shall be construed to override the provisions of this Section 1.8.
| SPONSOR NAME | Page 6 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
2. Study Drug, Storage and Return, and Equipment
2.1 Study Drug. Sponsor shall provide Institution,
at no charge, with such quantities of the Study Drug, comparator products, and materials as may be required for the Study. Investigator
shall use the Study Drug only pursuant to and in accordance with the Protocol and this Agreement, and for no other purpose. Institution
and Investigator shall notify Sponsor and Institution’s IRB regarding any use of the Study Drug without obtaining informed consent
as soon as possible, but no later than five (5) business days after such use. Any use of the Study Drug other than as specified in the
Protocol shall constitute a material breach of this Agreement. Sponsor represents and warrants that it is in compliance with federal,
state, and local legal requirements relating to the manufacture and formulation of any investigational (or unlicensed or unapproved) drug
and related material and other applicable legal requirements.
2.2 Storage. Institution and Investigator
shall keep all Study Drug in a locked, secured area at all times, within the temperatures required in the Protocol for storage of the
Study Drug and maintain complete, up-to-date records showing receipt of shipments of the Study Drug, dispensing and accountability of
the Study Drug and/or Study Device, and returns of the Study Drug as required by the Protocol, applicable federal, state, and local laws,
regulations, and guidelines. Furthermore, Investigator shall store all Study Drug in either a central pharmacy where a qualified pharmacist
supervises dispensing, or in a restricted area and dispensed under the direct supervision of Investigator.
2.3 Equipment. Sponsor may provide, or
arrange for a vendor to provide, certain equipment, if applicable, for use by Investigator and Staff during the conduct of the Study (“Equipment”).
During the term of this Agreement, Investigator and Staff may use the Equipment only for purposes of this Study in accordance with the
Protocol. Until the termination of this Agreement, this Equipment remains the property of Sponsor or the respective vendor that has provided
the Equipment, as applicable. The Equipment shall be returned upon request by Sponsor, to Sponsor or the vendor, promptly upon early termination
of this Agreement or at the completion of the Study. Institution and/or Investigator agree to return the Equipment in the manner directed
by Sponsor or the vendor in substantially the same condition as when received by Institution and/or Investigator, minus reasonable wear
and tear. Institution agrees to be financially responsible to cover any loss or destruction to Equipment while in Institution’s
and Investigator’s care, which exceeds ordinary wear and tear and/or lacks a reasonable causal relationship to proper performance
of the Study. Institution and Investigator further agree that unless otherwise authorized in writing by Sponsor, Institution and Investigator
will not alter the Equipment in any way nor install any components or software, if applicable. Any software provided to Institution and/or
Investigator may not be duplicated. Sponsor shall not have any liability for damages of any sort, including personal injury or property
damage, resulting from the use of Equipment except to the extent that such damages were caused by the gross negligence or willful misconduct
of Sponsor.
| SPONSOR NAME | Page 7 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
2.4 Return of Study Drug and Sponsor Property.
Upon completion of the Study or earlier termination of this Agreement, all unused Study Drug, devices, Equipment, and related materials
and all copies of Confidential Information, including Sponsor Technology (as defined below), that were furnished to Investigator shall
be returned to Sponsor, except for one copy to allow Institution to monitor its obligations under this Agreement, and any record copies
which the Investigator is required to retain by law or regulation. Regarding all unused Study Drug, the Investigator shall, upon the written
direction and approval of Sponsor, either (i) return all such material back to the Sponsor at Sponsor’s expense; (ii) destroy, at
the site where the Study is being conducted, such material in a manner that does not expose humans to risk from the compound or drug and
according to applicable laws and regulations; or (iii) dispose of such materials in another method that that does not expose humans to
risk from the compound or drug. In cases of (ii) or (iii), a certificate of destruction/disposition shall be sent to Sponsor upon such
action.
3. Payments
3.1 In consideration for performance of the Study,
Sponsor will pay the Party identified as “Payee” in accordance with the Payment Schedule attached as Appendix I (“Payment
Schedule”) and the Budget attached as Appendix II (“Budget”), and each made a part hereof. The Parties acknowledge that
the Payee is the appropriate payee under this Agreement and that the Payee is authorized to receive all the payments for the research
performed under this Agreement. If the Investigator is not the Payee, then the Payee’s obligation to reimburse the Investigator will be
determined by a separate agreement between Investigator and Payee, which may involve different payment amounts and different payment intervals
than the payments made by Sponsor to the Payee. Investigator acknowledges that if Investigator is not the Payee, Sponsor will not pay
Investigator even if the Payee fails to reimburse Investigator. The Budget may be modified only upon the prior written consent of the
Parties. Each Institution and Investigator acknowledges and agrees that the Payment Schedule and Budget includes Sponsor’s entire
payment obligation for the performance of the Study pursuant to this Agreement. CRO may process payments to the Payee on behalf of the
Sponsor.
3.2 Institution and Investigator agree that their
judgment with respect to the advice and care of each Study participant will not be affected by the compensation they receive from this
Agreement, that such compensation does not exceed the fair market value of the research they are providing, and that no payments are being
provided to them for the purpose of inducing them to purchase or prescribe any drugs, devices or products. If the Sponsor provides any
free products or items for use in the Study, Institution and Investigator agree that they will not knowingly bill any Study participant,
insurer or governmental agency, or any other third party, for such free products or items. Institution and Investigator agree that they
will not knowingly bill any Study participant, insurer, or governmental agency for any visits, services or expenses incurred during the
Study for which they have received compensation from Sponsor, or which are not part of the ordinary care they would normally provide for
the Study participant.
| SPONSOR NAME | Page 8 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
3.3 Investigator and Institution each acknowledge
that Payee is accepting tax liability for the research performed under this Agreement. The Payee information needs to be completed below
and on an IRS Form W-9 and returned to Sponsor for the Agreement to be effective. This information will be used for preparation of Federal
Form 1099 which will be issued to Payee documenting the payments made to the Payee in each calendar year.
4. Term and Termination
4.1 Term. The term of this Agreement shall
begin upon the Effective Date. Unless terminated earlier, this Agreement will terminate when (a) Institution has submitted all CRFs to
Sponsor, has resolved all data clarification queries, has submitted the closeout report to the IRB and Sponsor, has returned all Equipment
and Study materials to Sponsor, and has met all other close-out obligations; and (b) all payments, reimbursements and refunds have been
made.
4.2 Termination by Sponsor. This Agreement
may be terminated by Sponsor at any time for any reason upon written notice to Institution and Investigator.
4.3 Termination by Institution.
This Agreement may be terminated by the Institution upon prior written notice to Sponsor if the authorization and approval to conduct
the Study in the United States is withdrawn by the FDA. or immediately if necessary to protect the health, welfare or safety of any Study
subject.
4.4 Effect of Termination. In the event
of expiry or termination of this Agreement for any reason, Institution and Investigator shall, and shall procure, the prompt return to
Sponsor of all Confidential Information, Equipment and Inventions (as defined herein) provided to Institution or Investigator or generated
by Institution or Investigator in connection with the provision of the Study research. The Institution and Investigator may retain only
the relevant Study information and data to the extent and for the duration required by applicable law or regulation, and one copy to allow
Institution to monitor its obligations under this Agreement,. Investigator shall (i) use all reasonable efforts to conclude research on
the Study, (ii) stop enrollment of Study participants, (iii) not incur additional expenses, or enter into any further commitments with
regard to the Study after receiving or providing a notice of termination, and (iv) reasonably cooperate with the Sponsor and its designee
to allow for a close-out site visit. In the event of termination, the sum payable under this Agreement shall be limited to prorated fees
based on actual research performed pursuant to the Protocol as determined in accordance with the Budget, and/or obligations incurred in
accordance with the Study budget that cannot be cancelled or mitigated by the Institution using reasonable efforts, and/or reasonable
costs incurred in connection with the safe withdrawal of Study subjects from the Study. Such payment shall be paid upon satisfaction of
all outstanding obligations of Investigator and Institution, including, but not limited to, return of all Equipment and Study materials
to Sponsor. Institution will be entitled to payment for non-cancelable Study participant-related expenses only if properly substantiated,
reasonably incurred in accordance with the Study through the date of termination and within the Budget. Any funds not due to Institution
but already paid to Institution shall be returned to CRO and Sponsor by the earlier of thirty (30) days from the Institution close-out
visit or from the notice of termination.
| SPONSOR NAME | Page 9 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
5. Confidentiality
5.1 All information, including, but not limited
to, documents, descriptions, data, CRFs, photographs, videos and instructions, Study Drug, materials and information derived from patient
specimens, Sponsor Technology (as defined in Article 7 below) and Confidential Information (as defined below), provided to or made available
to Institution or Investigator by Sponsor or its agents (whether verbal, written or electronic) or generated by Institution, Investigator
or their respective Staff, shall be the property of Sponsor.
5.2. “Confidential Information” means
all information, material or data related to the Study, including Sponsor Technology, whether provided by or on behalf of Sponsor (either
before or after execution of this Agreement), or developed for Sponsor, or generated by Institution and/or Investigator in accordance
with this Agreement, whether in writing, electronic, oral or visual transmission, including without limitation, any information and data
concerning the Study Drug and/or Study Device, Study results, reports, Study strategies, the existence or terms of the Protocol or this
Agreement, exclusive of patient medical records. For the avoidance of doubt, Study data / results shall not be deemed Confidential Information
at the time of publication, in accordance with Article 6 herein.
5.3 Investigator and Institution shall maintain
in strict confidence all of the Confidential Information, including the Sponsor Technology, and not disclose or disseminate to any third
party or use for any purpose other than the performance of the Study any of the same. Such Confidential Information, including the Sponsor
Technology, shall remain the confidential and proprietary property of Sponsor, and shall be disclosed only on a need-to-know basis and
only to the Investigator and his/her Staff who have a need to know such Confidential Information for the purposes of performing the Study
according to this Agreement and the Protocol who are bound by confidentiality terms at least as strict as those herein (individually,
a “Required Disclosee”). Investigator and Institution will inform and advise each Required Disclosee of the obligations under
this Agreement with respect to the Confidential Information and each Required Disclosee shall be bound by such obligations in a like fashion.
5.4 Investigator and Institution each shall use
the same degree of care to protect the Confidential Information as it uses to protect its own confidential information, but in no event
less than a reasonable amount of care. Investigator and Institution shall be responsible for any breach of this Agreement by its Staff
to the extent permitted by applicable law. Investigator and Institution each agree to promptly notify Sponsor in the event of any loss
or unauthorized disclosure of any Confidential Information.
| SPONSOR NAME | Page 10 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
5.5 The foregoing obligation of non-disclosure
shall not apply to Confidential Information, including the Sponsor Technology, that Institution or Investigator is able to demonstrate
by written evidence:
| (a) | was generally known to the public prior to the Effective
Date of this Agreement; |
| (b) | becomes generally known to the public through no unlawful
or unauthorized act or omission of Institution or Investigator or any of their Staff, or in violation of this Agreement; |
| (c) | was independently developed by Institution or Investigator,
without reliance upon or reference to Confidential Information as evidence by its written records; or |
| (d) | was disclosed to Institution or Investigator without restriction
by a third party who had the right to make such disclosure. |
5.6 If Institution or Investigator is requested
to produce any of the Confidential Information pursuant to a legal or governmental proceeding, Institution or Investigator shall give
Sponsor adequate prior notice of such requirement and shall use its reasonable efforts to assist Sponsor in objecting to such request.
If Institution or Investigator is compelled to disclose any of the Confidential Information pursuant to such legal or governmental proceeding,
Institution and Investigator, as applicable, shall use its reasonable efforts to assist Sponsor in obtaining confidential treatment for
such disclosed Confidential Information. Any Confidential Information so disclosed shall still be subject to the terms of this Agreement.
5.7 Injunctive Relief. Investigator and
Institution acknowledge and agree that any violation of the terms of this Agreement relating to the disclosure or use of Confidential
Information, including the Sponsor Technology, may result in irreparable injury and damage to Sponsor not adequately compensable in money
damages, and for which Sponsor may have no adequate remedy at law. Investigator and Institution acknowledge and agree, therefore, that
if those disclosure terms are violated, Sponsor may seek injunctions, orders, or decrees in order to protect the Confidential Information
and the Sponsor Technology.
5.8 Privacy Laws. Institution and Investigator
agree to take all reasonable steps to protect the confidentiality of any patient health and medical information that it has access to
and comply with all applicable privacy laws, including, but not limited to the Health Insurance Portability and Accountability Act of
1996 and its implementing regulations. Institution and Investigator consent to the sharing of their personal data, as defined under any
applicable privacy laws, with Sponsor.
5.9 Existence of Agreement. Investigator
and Institution shall not disclose the existence of this Agreement, except to the extent required by academic policies, law or regulation.
| SPONSOR NAME | Page 11 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
6. Publications
6.1 The
Institution and Investigator each understands that if the Study is part of a multi-center study, then any publication by the Institution
or the Investigator of the results of the Study conducted at the Institution shall not be made before the first multi-center publication,
and of the results of the Study shall be made in conjunction with the presentation of a joint multi-center Publication of the Study results
with the investigators from all sites contributing data, analyses, and comments. Sponsor shall also provide the Investigator in any of
these events with a copy of the abstract and manuscript for any multi-center Study publication and with an adequate opportunity to review
and have input into such abstract and manuscript prior to submission for publication. Principal Investigator shall, in all cases, at his/her
sole discretion, have the right to decline to be an author and to have his/her name removed as an author from any multi-center Study publication.
The Study results will be disclosed as soon as possible if there are any significant safety findings. Sponsor further certifies that the
multicenter Study results will be published/disclosed regardless of whether they support the hypothesis being tested or are contrary to
the predicated outcome. Institution may publish the data and Study results individually in accordance with this Article 6 upon first occurrence
of one of the following: (1) at such time as the multi-center results of the Study are submitted for publication or posted on publicly
accessible websites at the time of publication, (2) notification that a submission of the multi-center results is not planned, or (3)
if no multi-center publication is made within eighteen (18) months from completion of the Study for all sites in the Study, then the Institution
or Investigator may publish individually in accordance with the requirements of Section 6.2, below.
6.2 Investigator
may publish the results of Study, relative to his/her own Study participant(s), only after review by Sponsor. At least forty-five (45)
days prior to submitting a manuscript to a publisher or other outside persons (i.e., reviewer(s)) or fifteen (15) days prior to any public
abstract presentation, a copy of the abstract or presentation will be provided to Sponsor by the Investigator for review and comment.
At the expiration of such forty-five (45) day or fifteen (15) day period, Investigator may proceed with submission for publication provided
that any identified Sponsor Confidential Information has been removed; and provided further that if during the review period, Sponsor
notifies Institution that it desires to file patent applications on any Inventions (as defined in Article 7 below) disclosed or contained
in the manuscript or presentation, Institution will defer publication or other disclosure for a period, not to exceed an additional forty-five
(45) days, sufficient to permit Sponsor or its designee to file or have filed any desired patent applications. The Investigator agrees,
following a request from Sponsor, to delete any Confidential Information from any publication or presentation. In no event shall the submission
of such publication of results be delayed for more than ninety (90) days for manuscripts and for more than sixty (60) days for abstracts
from the date such proposed publication was provided to Sponsor; at the end of said ninety (90) or sixty (60) days, the Investigator shall
be free to publish such results as proposed.
6.3 Sponsor may use, refer
to, and disseminate reprints of scientific, medical, and other published articles relating to the Study, which disclose the name of Investigator
and Institution, consistent with U.S. copyright laws.
| SPONSOR NAME | Page 12 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
7. Intellectual Property
7.1 Sponsor Technology. For purposes of
this Agreement, the term “Sponsor Technology” shall mean all data, materials, information, methods or techniques, whether
or not patented or patentable, copyrighted or copyrightable, pertaining to the research, development, manufacture, or use of compounds
obtained or developed by, or on behalf of, Sponsor. In the performance of this Agreement, Sponsor may disclose Sponsor Technology to Investigator.
Sponsor Technology shall remain the confidential and proprietary property of Sponsor. Each of Investigator and Institution agrees that
Sponsor is the sole and exclusive owner of the Sponsor Technology, whether or not patented or subject to copyright or patent protection;
and that the Sponsor Technology is commercially valuable.
7.2 Patent Rights. It is expressly agreed
that neither Sponsor, on the one hand, or Investigator and/or Institution, on the other hand, transfers to the other, by operation of
this Agreement, any patent right, copyright or other proprietary right which any such Party owns as of the Effective Date, except as specifically
set forth herein.
7.3 Inventions. The ownership of any information,
inventions, improvements, new uses, or discoveries (whether patentable, copyrightable, or not), innovations, suggestions, ideas, communications
and reports (collectively “Inventions”), conceived, reduced to practice, made or developed by the Institution or Investigator
and/or all individuals or entities either employed by or subcontracted by Institution or Investigator, as a result of or relating to the
conduct of the Study or Study Drug and/or Study Device, belong to the Sponsor and Institution or Investigator and/or all individuals or
entities either employed by or contracted by Institution or Investigator agrees to assign all of their interests therein without compensation
from Sponsor or its nominee whenever requested to do so by Sponsor. Institution and Investigator and/or all individuals or entities either
employed by or contracted by Investigator or Institution agrees they will execute any and all applications, assignments, or other instruments
and live testimony which Sponsor shall deem necessary to apply for and obtain Letter Patent of the United States or of any foreign country
or to otherwise protect the Sponsor’s interest therein, and Sponsor shall reimburse Investigator for their time devoted to said
activities.
7.4 Further, Investigator and Institution will
disclose to Sponsor any and all Inventions conceived, reduced to practice, made or developed by the Investigator or Institution, as a
result of conducting the Study. However, if the Investigator obtains ownership of any invention arising from research conducted under
the Study, Sponsor shall be granted the first opportunity to acquire an exclusive license for use of the invention based on good faith
negotiations between the Institution and Sponsor.
8. Representations and Certifications
8.1 Study Drug. Sponsor makes no representations
or warranties, expressed or implied, related to the Study Drug, including without limitation, any warranty or merchantability for fitness
for a particular purpose, or non-infringement.
| SPONSOR NAME | Page 13 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
8.2 Appropriate Training and Licenses.
Institution represents that Institution, Investigator and the Staff have, and will maintain throughout the conduct of the Study, all training,
information, business, professional and other licenses, approvals, or certifications that are necessary for safely, adequately and lawfully
performing the Study. Institution further represents that Investigator and Staff are Institution’s employees and are subject to
Institution’s direct control and supervision.
8.3 No Conflicts. Institution and Investigator
represent that to the best of their knowledge they are not a Party to any existing agreements that would prevent Institution and Investigator
from entering into and performing this Agreement. Institution and Investigator will not enter into any other agreement that is in conflict
with their obligations under this Agreement. If Institution is affiliated with a university or other third party, Institution represents
that (i) Institution has complied with any and all applicable policies and procedures of such university or other third party pertaining
to proposed agreements for services, (ii) to the extent necessary or required, received approval from such university or other third party
to enter into this Agreement and be bound by the terms herein, (iii) receipt, generation and use of Confidential Information hereunder
will not conflict with any agreement Institution or Investigator has with any university or third party to which Institution or Investigator
is employed or affiliated, and (iv) no university or third party shall have any interest or rights in Confidential Information or any
Inventions. Institution and Investigator further represent that they are not is currently involved in, nor is aware of, any pending litigation
proceedings relating to, respectively, the Institution’s or the Investigator’s role in the conduct of a human clinical trial.
Investigator will notify Sponsor promptly of any conflicting obligations or legal impediments that may occur during the term of this Agreement.
8.4 Criminal and Civil Liability. INSTITUTION
AND INVESTIGATOR UNDERSTAND AND ACKNOWLEDGE THAT FABRICATION, FALSIFICATION OR ALTERATION BY INSTITUTION, INVESTIGATOR OR ANY STAFF, OF
ANY PATIENT DATA OR OTHER INFORMATION PROVIDED BY INSTITUTION OR INVESTIGATOR PURSUANT TO THIS AGREEMENT CAN RESULT IN CRIMINAL ACTIONS
AND SANCTIONS AGAINST INSTITUTION AND INVESTIGATOR AND IN EACH SUCH PARTY HAVING CIVIL LIABILITY TO SPONSOR.
8.5 Form 1572 or Investigator Agreement for
Medical Device Studies. Investigator certifies and represents that (i) the statements on his/her FDA Form 1572 or Investigator Agreement
for medical device studies are true and accurate in all respects, (ii) he/she is trained and qualified to conduct clinical trials within
the jurisdiction in which the Investigator shall perform the Study, and (iii) Staff shall be appropriately trained in International Conference
on Harmonisation-Good Clinical Practice (“ICH GCP”), the Protocol and the Study procedures.
| SPONSOR NAME | Page 14 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
8.6 Health Care Provider Payment Tracking.
Institution understands and agrees that for purposes of complying with reporting obligations under the Patient Protection and Affordable
Care Act of 2010 (together with any regulations and official guidelines promulgated thereunder) and any applicable state reporting requirements,
Sponsor may collect, aggregate and report any and all payments made pursuant to this Agreement.
8.7 Business and Financial Disclosure.
Investigator and Institution represent and certify that neither they nor any of their Staff are officials, agents, representatives or
employees of any government or political party or any international public organization where they may be in positions of official government
authority able to use that position to help Sponsor obtain or maintain business or obtain a business advantage. Investigator and Institution
further represent and certify that they have not and agree that they shall not make any payment or any offer or promise for payment, either
directly or indirectly, of money or other assets, to government or political party officials, officials of international organizations,
candidates for public office, or representatives of other businesses or persons acting on behalf of any of the foregoing, for the purpose
of influencing decisions or actions or where such payment would constitute violation of any law, including but not limited to applicable
anti-bribery/anti-corruption laws.
8.8 Debarment and Disqualification. Institution
and Investigator each represents and certify that it/he/she, and each of its Staff, has never been and is not currently:
| (i) | an individual who has been debarred by the FDA pursuant to
21 U.S.C. § 335a(a) or (b) from providing services in any capacity to a person that has an approved or pending drug product application
(a “Debarred Individual”), or an employer, employee, or partner of a Debarred Individual; or |
| (ii) | a corporation, partnership, or association that has been
debarred by the FDA pursuant to 21 U.S.C. § 335a(a) or (b) from submitting or assisting in the submission of any abbreviated drug
application (a “Debarred Entity”), or an employee, partner, shareholder, member, subsidiary, or affiliate of a Debarred Entity. |
| (iii) | an individual or corporation, partnership, or association
that has been barred from participation in a “Federal Health Care Program” (as defined in 42 U.S.C. § 1320a(7b(f)),
as amended from time to time or in any other governmental payment program. |
Institution and Investigator further represents
and certify that it/he/she, and each of its Staff, have not engaged in any conduct or activity which could lead to any of the above-mentioned
disqualifications or debarment actions and that it has no notice that FDA or another regulatory authority intends to seek disqualification
or debarment. Institution shall notify Sponsor within ten (10) days of any actual or threatened disqualification, debarment or other ban
or investigation of the Institution, Investigator or Staff that comes to its attention during the course of the Study and for five (5)
years thereafter.
| SPONSOR NAME | Page 15 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
8.9 Compliance with Laws. Institution and
Investigator each represents and warrants that it/he/she, and each of its Staff, will comply with all applicable laws, rules and regulations
and guidelines relating to the conduct of clinical investigations, including, without limitation 21 CFR Parts 50, 54, 56 and 312, the
IHC GCP and other good clinical and medical practice requirements. .
9. Study Results, Records and Audits
9.1 Use of Study Results. Sponsor shall
have the right to use all information resulting from the Study for any and all lawful purposes, subject to Section 9.2. Institution and
Investigator shall have the right to use any information, data or results relating to the Study for non-commercial internal research purposes
and patient care, and/or in a publication subject to Article 6.
9.2 HIPAA. Institution shall comply in
all material respects with all applicable Federal, state and local laws and regulations regarding the privacy of individually identifiable
health information (including its collection, use, storage, and disclosure), including, but not limited to, the Health Insurance Portability
and Accountability Act of 1996 (“HIPAA”) and the regulations promulgated thereunder, as may be amended from time to time.
Sponsor agrees to collect, use and disclose identifiable data collected or produced in the Study (“Patient Data”) only for
the purpose of the Study and for the purpose of complying with applicable law, provided that all such uses are disclosed in the informed
consent form. Sponsor will use all reasonable efforts to protect the privacy and security of Patient Data and will require its business
partners to do so also. Sponsor will not contact any Study subjects unless permitted by the informed consent form or otherwise agreed
to with Institution. No other provision in this Agreement shall be construed to override the provisions of this paragraph.
9.3 Records. Institution shall maintain
all records required to be maintained by all applicable laws and regulations, including, but not limited to, CRFs, safety reports, annual
reports and any other documentation or materials related to the Study and Institution’s Study file, which should include all Study-related
correspondence. Investigator shall sign statements in each Study participant’s CRF (i) attesting to Investigator’s review
of the data; and (ii) that the data constitutes an accurate accounting of the treatment, care, and events surrounding the Study participant’s
involvement in the Study. All Study records shall be complete and up to date. Institution will retain all records for the Study produced
pursuant to this Agreement for the record retention period mandated by all applicable U.S. laws or FDA regulations. Institution further
agrees to permit Sponsor to ensure that the records are retained for a longer period if necessary, at Sponsor expense, under an arrangement
that protects the confidentiality of the records (e.g., secure off-site storage). At Sponsor’s request, Institution will deliver
copies of such records to Sponsor at Sponsor’s cost. Institution shall promptly notify Sponsor of any accidental loss or destruction
of Study records. Notwithstanding anything herein, Institution and Investigator shall not destroy any Study material, information, data
or records without the prior written consent by Sponsor. Institution will contact Sponsor in writing at least sixty (60) days before the
planned destruction of any Study records. If Institution receives no written response within sixty (60) days of sending notice to Sponsor
of Institution’s desire to destroy the records, then Institution may proceed as desired.
| SPONSOR NAME | Page 16 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
9.4 Study Documents. Institution will prepare,
maintain and retain complete, current, accurate, attributable, organized and legible copies of source documents, CRFs, regulatory documents
and other Study documents as required by the Protocol and ICH GCP, to the extent such guidelines have been incorporated into FDA regulations
as binding law.
9.5 Inspection by Sponsor. During the term
of this Agreement and at no cost to Sponsor, Investigator and Institution will permit Sponsor and Sponsor’s representatives to examine
or audit the research performed hereunder, the use of Sponsor’s funds, the facilities systems and equipment at or with which the
research is conducted and records related to such research, upon reasonable advance notice during regular business hours to determine
that the Study is being conducted in accordance with the agreed requirements and that the facilities are adequate. If records pertaining
to the Study are kept electronically, Institution and Investigator shall provide Sponsor’s representatives access to such systems
for monitoring and auditing purposes. Institution shall make Investigator and other appropriate Staff available to Sponsor’s representatives
to discuss such records and reports and to resolve any questions relating to such records and reports. At the request of Sponsor or its
designee, Institution and Investigator shall correct any errors or omissions in such records and reports.
9.6 Inspection by Regulatory Authority.
Investigator and Institution shall notify Sponsor promptly by telephone or facsimile if the FDA or other duly authorized authority requests
permission to or does inspect Investigator’s or Institution’s facilities or research records during the term of this Agreement and
will, to the extent allowed by law, provide in writing to Sponsor copies of all materials, correspondence, statements, forms, and records
which Investigator or Institution receives, obtains or generates pursuant to any such inspection. Institution and Investigator, to the
extent allowed by law, shall allow Sponsor to be present with any such inspection.
10. Indemnification
10.1 Sponsor will defend, indemnify and hold harmless
the Investigator, Institution and its Staff, and Institution’s trustees, officers, employees, and agents and their respective successors,
heirs and assigns (collectively, the “Institution Indemnitees”) from and against all claims, demands, actions, suits and proceedings
(“Claims”) that may be brought or instituted against any Institution Indemnitee by a third party, and all judgments, damages,
losses, liabilities, costs and expenses incurred by any of the Institution Indemnitees resulting therefrom, by reason of (a) any side
effect, adverse reaction, illness, or personal injury (including death) to any person as a result of his or her participation in the Study,
or damage to property which directly result from the performance of the Study in accordance with the Protocol, or (b) Sponsor’s
use of any Study data, or (c) or a breach of security leading to the accidental or unlawful destruction, loss, alteration, unauthorized
disclosure, or access to personal data, each except to the extent that such Claims arise out of or result from (i) the negligence, recklessness,
criminal act or willful misconduct of any Institution Indemnitee, (ii) any failure by an Institution Indemnitee to use the Study Drug
and conduct the Study in accordance with the Protocol, other written information, instructions or warnings furnished by Sponsor, and all
applicable laws, rules or regulations (provided, however that emergency medical care shall not be deemed a violation of the Protocol),
or (iii) any other breach of the Agreement by any Institution Indemnitee.
| SPONSOR NAME | Page 17 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
10.2 Institution shall be responsible for its
negligent acts or omissions and the negligent acts or omissions of its employees, officers, or directors in the performance of the Study,
to the extent allowed by law.
10.3 Indemnification Process. An Institution
Indemnitee shall promptly notify the indemnifying Sponsor in writing upon receipt of notice of any Claim, but in no event later than ten
(10) days after such receipt, and must permit Sponsor’s attorneys and personnel, at Sponsor’s discretion and cost, to handle
and control the defense of such Claim, provided, however, that failure to provide such notice shall not relieve Sponsor of its indemnification
obligations except to the extent that the Sponsor’s ability to defend such Claim is materially, adversely affected by such failure.
Sponsor shall not make any settlement admitting fault or incur any liability on the part of the Institution Indemnitees without Institution
Indemnitees’ prior written consent, such consent not to be unreasonably withheld or delayed. The Institution Indemnitees shall cooperate
with Sponsor in all reasonable respects regarding the defense of any such Claim, at Sponsor’s expense. The Institution Indemnitees
shall be entitled to retain counsel of its choice at its own expense. In the event a Claim falls under this indemnification clause, in
no event shall either party compromise, settle or otherwise admit any liability with respect to any Claim without the prior written consent
of the other party, such consent not to be unreasonably withheld or delayed.
EXCEPT FOR (I) A PARTY’S INTENTIONAL MISCONDUCT,
OR (II) THE OBLIGATIONS TO INDEMNIFY PURSUANT TO THIS AGREEMENT, NEITHER PARTY SHALL BE LIABLE FOR SPECIAL, CONSEQUENTIAL OR INCIDENTAL
DAMAGES ARISING OUT OF OR IN CONNECTION WITH THIS AGREEMENT, EVEN IF ADVISED OF THE POSSIBILITY OF THE SAME.
10.4 Subject Injury. The Sponsor shall
reimburse Institution (or otherwise pay for) for reasonable and necessary medical expenses incurred by Study participants for any medical
care, including hospitalization, in the treatment of side effect, adverse reactions, illness or injury arising directly from procedures
required under the Protocol and/or the Study Drug following administration in connection with the Protocol, to the extent such expenses
(i) are not covered by the Study participant’s medical or hospital insurance coverage or other third party coverage, (ii) are
not the result of the negligence or intentional misconduct of any Staff or Investigator or Institution, (iii) are not the result of a
deviation from the Protocol, or other written instructions consistent with the Protocol provided to Institution, provided, however that
emergency medical care shall not be deemed a violation of the Protocol. The Parties agree that natural progression of an underlying pre-existing
condition does not constitute such an adverse reaction. Alleged lack of efficacy of the Study drug and/or Study Device and/or any consequences
resulting therefrom shall not constitute an adverse reaction. Sponsor will not provide compensation or reimbursement for any other injury-related
costs or expenses, such as lost wages, pain and suffering compensation, or any other consequential or indirect damages.
| SPONSOR NAME | Page 18 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
11. Insurance
11.1 Institution certifies and represents that
it possesses and shall carry at its own expense from a reputable insurance company, comprehensive general liability insurance and/or policies
of self-insurance with limits of not less than $1,000,000 per occurrence and $3,000,000 annual aggregate for each Investigator performing
research under this Agreement, and professional malpractice insurance (or similar errors and omissions insurance) with limits of not less
than $1,000,000 per occurrence and $3,000,000 annual aggregate. Institution’s insurance covers the Study, and specifically covers
the actions of the Investigator, any sub-investigators and other Study personnel, and is not materially encumbered by existing claims.
In addition, Institution shall secure and maintain in full force and effect workers’ compensation insurance in the amount required
by the laws of the state in which Institution is located. Institution shall maintain all such coverage for the duration of this Agreement
and for five (5) years thereafter. Upon request, Institution shall furnish to Sponsor a certificate indicating that such insurance is
in force, which shall indicate any deductible and/or self-insured retention. Institution shall provide Sponsor with at least thirty (30)
days prior written notice of cancellation or any material change in the policy or policies of insurance required.
11.2 Sponsor warrants and represents that it possesses
and shall carry at its own expense from a reputable insurance company, products liability insurance (with clinical trials coverage) with
limits of not less than Three Million Dollars ($3,000,000) per occurrence and Five Million Dollars ($5,000,000) annual aggregate for each
Investigator performing services under this Agreement, and professional malpractice insurance (or similar errors and omissions insurance)
with limits of not less than $1,000,000 per occurrence and $3,000,000 annual aggregate. Institution shall maintain all such coverage for
the duration of this Agreement and for five (5) years thereafter. Upon request, Sponsor shall furnish to Sponsor a certificate indicating
that such insurance is in force, which shall indicate any deductible and/or self-insured retention. Sponsor shall provide Institution
with at least thirty (30) days prior written notice of cancellation or any material change in the policy or policies of insurance required.
12. Miscellaneous
12.1 Publicity. No Party to this Agreement
shall use the name, trade name, service names, trademarks or service marks of any other Party in connection with any press release, advertising
or promotion of any product or service without the prior written permission of such Party except as required by law or regulation.
| SPONSOR NAME | Page 19 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
12.2 Independent Contractors. Each Party
to this Agreement shall act as an independent contractor and shall not be construed for any purpose as the partner, agent, employee, servant,
or representative of the other Parties. Accordingly, the employee(s) of one Party shall not be considered to be employee(s) of any other
Parties, and no Party shall enter into any contract or agreement with a third party which purports to obligate or bind the Party to this
Agreement. Sponsor shall not be responsible for any employee benefits, pensions, workers’ compensation, withholding, or employment-related
taxes as to the Institution or the Investigator or any of their employees or agents.
12.3 Complete Agreement, Amendment. The
Parties agree that this Agreement, including the Appendices hereto, constitutes the sole, full, and complete Agreement by and between
the Parties and supersedes all other written and oral agreements and representations between the Parties with respect to the subject matter
herein. In the event of a conflict between the Protocol and this Agreement, the terms of the Agreement will govern; provided, however,
that in the case of any such conflict relating to clinical matters, the terms of the Protocol shall prevail. No amendments, changes, additions,
deletions, or modifications to or of this Agreement shall be valid unless reduced to writing and signed by the Parties.
12.4 Notices. Any notices or communications
concerning this Agreement should be in writing and shall be deemed to have been given upon receipt, and shall be either by U.S. Mail postage
prepaid, or overnight bonded courier to the following:
| To Sponsor: |
60 Degrees Pharmaceuticals, Inc |
| |
1025 Connecticut Ave NW, Suite 1000 |
| |
Washington DC, 20036 |
| |
Attn: ############# |
| |
Email: #############@60degreespharma.com |
| |
Phone: ############# |
| To CRO: |
############# |
| |
Chief Operating Officer |
| |
Fast-Track Drugs & Biologics, LLC |
| |
20010 Fisher Avenue, Suite G |
| |
Poolesville, MD, 20837 |
| |
Phone: ############# |
| |
Fax: ############# |
| |
Email: #############@fasttrackresearch.com |
| SPONSOR NAME | Page 20 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
| To Institution: |
Tufts Medical Center, Inc. |
| |
Attn: Research Administration |
| |
800 Washington Street, Tufts MC Box 817 |
| |
Boston, MA 02111 |
| |
Phone: 617-636-2892 |
| To Investigator: |
Tufts Medical Center, Inc. |
| |
ATTN: ############# |
| |
800 Washington Street |
| |
Boston, MA 02111 |
| |
Phone: ############# |
12.5 Binding Effect and Survival. This
Agreement shall be binding upon the Parties, their legal representatives, successors, and assigns. The obligations of the Parties contained
in Articles 2, 3, 5, 6, 7, 10, 11 and 12 shall survive the termination or expiration of this Agreement.
12.6 Waiver. Failure to insist upon compliance
with any of the terms and conditions of this Agreement shall not constitute a general waiver or relinquishment of any such terms or conditions,
and the same shall remain at all times in full force and effect.
12.7 Severability. In the event one or
more of the provisions of this Agreement should, for any reason, be held to be invalid, illegal or unenforceable in any respect, the validity,
legality and enforceability of the remaining terms and provisions shall not be affected or impaired thereby and the Parties will attempt
to agree upon a valid, legal and enforceable provision that is a reasonable substitute therefore, and upon so agreeing, shall incorporate
such substitute provision in this Agreement.
12.8 Assignment. It is expressly understood
by the Parties hereto that Investigator and Institution may not assign, delegate, subcontract or transfer any of its rights or obligations
under this Agreement to any Party without the express prior written consent of Sponsor.
| SPONSOR NAME | Page 21 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
12.9 Force Majeure. No Party shall be responsible
to the others for any delay in the performance of, or failure to perform, this Agreement where such delay or failure is caused by circumstances
beyond the reasonable control of the affected Party including, without limitation, strikes, lockouts or any other labor disruptions, war,
civil commotion, natural disaster, pandemics or epidemics, or acts of God. In the event of any such delay or failure in performance, the
affected party shall be granted an extension of time for performance that is equitable in light of the cause of the delay.
12.10 Effective Upon Execution; Authority.
This Agreement shall not be considered accepted, approved, or otherwise effective until signed below by the appropriate Parties.
Each of the Parties hereto represents and warrants that the person signing below on such Party’s behalf has the authority to enter
into this Agreement.
12.11 Counterparts. This Agreement shall
become binding when any one or more counterparts hereof, individually or taken together, shall bear the signatures of the Parties. This
Agreement may be executed by wet ink or authenticated electronic signature and exchanged by facsimile or electronically via PDF copies,
and in two or more counterparts, each of which will be deemed an original document, and all of which, together with this writing, will
be deemed one instrument.
12.12 Heading. Headings used in this Agreement
are for reference purposes only and shall not be used to be duly executed this Agreement as of the Effective Date above.
(Signature Page Follows)
| SPONSOR NAME | Page 22 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
IN TESTIMONY WHEREOF, Sponsor, Institution
and Investigator have caused this Agreement to be executed as of the Effective Date.
| For Sponsor: |
|
For Institution: |
| |
|
|
|
|
| 60 DEGREES PHARMACEUTICALS INC |
|
TUFTS MEDICINE, INC. |
| |
|
|
|
|
| By: |
/s/
############# |
|
By: |
/s/
############# |
| Name: |
############# |
|
Name: |
############# |
| Title: |
############# |
|
Title: |
############# |
| |
|
|
|
|
| Date: |
5/29/2024 |
|
Date: |
5/29/2024 |
| |
READ AND ACKNOWLEDGED: |
| |
|
|
| |
INVESTIGATOR |
| |
|
|
| |
By: |
/s/
############# |
| |
Name: |
############# |
| |
|
|
| |
Date: |
5/29/2024 |
| SPONSOR NAME | Page 23 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
LIST OF APPENDICES
Appendix I: Payment Schedule
Appendix II: Budget
| SPONSOR NAME | Page 24 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
APPENDIX I – PAYMENT SCHEDULE
(See attached.)
[*]
| SPONSOR NAME | Page 25 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
APPENDIX II – BUDGET
(See attached.)
[*]
| SPONSOR NAME | Page 26 of 26 | CONFIDENTIAL |
| < Institution> CTA | | |
Exhibit 23.1
CONSENT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING
FIRM
We consent to the incorporation by reference in
this Registration Statement of 60 Degrees Pharmaceuticals, Inc. on Form S-3 of our report dated April 1, 2024 relating to the financial
statements of 60 Degrees Pharmaceuticals, Inc., as of December 31, 2023 and 2022 and for each of the years in the two-year period ended
December 31, 2023 (which report includes an explanatory paragraph regarding the Company’s ability to continue as a going concern).
We also consent to the reference to us under
the heading “Experts” in the Prospectus, which is part of this Registration Statement.
/s/
RBSM LLP
Las Vegas, Nevada
July 12, 2024
Exhibit 107
Calculation of Filing Fee Table
Form S-3
(Form Type)
60 Degrees Pharmaceuticals, Inc.
(Exact Name of Registrant as Specified in its Charter)
Table 1: Newly Registered Securities
| | |
Security
Type | |
Security
Class
Title | |
Fee
Calculation or
Carry
Forward
Rule | |
Amount
Registered(1) | | |
Proposed
Maximum
Offering
Price Per
Share(2) | | |
Maximum
Aggregate Offering
Price | | |
Fee Rate | | |
Amount of
Registration
Fee | |
| Fees to be Paid | |
Equity | |
Common Stock, $0.0001 par value per share(3) | |
457(o) | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | |
| | |
Equity | |
Preferred Stock, $0.0001 par value per share(4) | |
457(o) | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | |
| | |
Other | |
Warrants(5) | |
457(o) | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | |
| | |
Other | |
Units(6) | |
457(o) | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | |
| | |
Debt | |
Debt Securities(7) | |
457(o) | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | |
| | |
Unallocated (Universal) Shelf | |
(1) | |
457(o) | |
$ | 15,000,000 | | |
$ | — | | |
$ | 15,000,000 | | |
$ | 0.00014760 | | |
$ | 2,214 | |
| Total | |
| |
| |
| |
$ | 15,000,000 | | |
$ | — | | |
$ | 15,000,000 | | |
| 0.00014760 | | |
$ | 2,214 | |
| (1) |
The table lists each class of securities being registered and the aggregate proceeds to be raised in the offering and does not specify by each class information as to the amount to be registered or the proposed maximum offering price per security. Any securities registered hereunder for the offering may be sold separately or together in combination with other securities registered hereunder for the offering. Any securities registered hereunder may be sold separately or as units with any other securities registered hereunder. In no event will the aggregate offering price of all securities issued from time to time in the offering pursuant to the registration statement of which this Exhibit 107 is a part, exceed $15,000,000, inclusive of any exercise price thereof. Pursuant to Rule 416 under the Securities Act of 1933, as amended (the “Securities Act”), the securities being registered hereunder also include such indeterminate number of securities as may be issued from time to time with respect to the securities being registered hereunder as a result of stock splits, stock dividends or similar transactions. |
| (2) |
The proposed maximum offering price per security will be determined from time to time by the registrant in connection with the issuance by the registrant of the securities registered hereunder and is not specified as to each class of security pursuant to Instruction 2.A(iii)(b) of Item 16(b) of Form S-3 under the Securities Act. |
| (3) |
Including such indeterminate amount of common stock as may be issued from time to time at indeterminate prices or upon conversion of debt securities, preferred stock registered hereby or upon exercise of warrants registered hereby, as the case may be. In the event of a stock split, stock dividend or recapitalization involving the common stock, the number of shares registered shall automatically be adjusted to cover the additional shares of common stock issuable pursuant to Rule 416 under the Securities Act. |
| (4) |
Including such indeterminate amount of preferred stock as may be issued from time to time at indeterminate prices or upon conversion of debt securities, preferred stock registered hereby or upon exercise of warrants registered hereby, as the case may be. |
| (5) |
Warrants may be sold separately or together with any of the securities registered hereby and may be exercisable for shares of common stock, preferred stock, debt securities or units registered hereby. Because the warrants will provide a right only to purchase such securities offered hereunder, no additional registration fee is required. |
| (6) |
Because the units will provide a right only to purchase such securities offered hereunder, no additional registration fee is required. |
| (7) |
Including such indeterminate principal amount of debt securities as may be issued from time to time at indeterminate prices or upon exercise of warrants registered hereby, as the case may be. |
60 Degrees Pharmaceuticals (NASDAQ:SXTP)
Historical Stock Chart
From Jun 2024 to Jul 2024
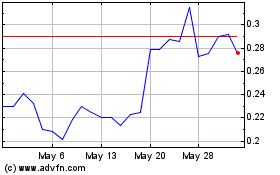
60 Degrees Pharmaceuticals (NASDAQ:SXTP)
Historical Stock Chart
From Jul 2023 to Jul 2024