AC Immune: Semorinemab Meets Co-Primary Endpoint in Alzheimer's Disease
31 August 2021 - 10:33PM
Dow Jones News
By Colin Kellaher
AC Immune SA on Tuesday said a Phase 2 study evaluating the
safety and efficacy of its investigational anti-tau monoclonal
antibody semorinemab in mild-to-moderate Alzheimer's disease met
one of its co-primary endpoints.
The Lausanne, Switzerland, clinical-stage biopharmaceutical
company said semorinemab showed a statistically significant
reduction in cognitive decline from baseline by 43.6% compared with
placebo in the study. The study was being conducted by Roche
Holding AG's Genentech unit, AC Immune's collaboration partner in
Alzheimer's disease since 2006.
AC Immune said there was no effect on the other co-primary
endpoint of reducing the rate of functional decline from baseline,
adding that semorinemab was well tolerated in the study, with an
acceptable safety profile and no unanticipated safety signals.
AC Immune said the study marks the first time that a monoclonal
anti-tau antibody therapy has had a therapeutic impact on cognition
in the mild-to-moderate Alzheimer's patient population.
AC Immune said Genentech plans to continue the open-label
portion of the study and to submit top-line results for
presentation at a conference in November.
Shares of AC Immune, which closed Monday at $6.99, surged nearly
70% in premarket trading Tuesday.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
August 31, 2021 08:18 ET (12:18 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
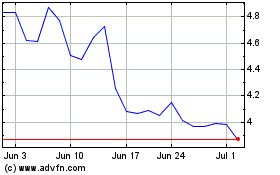
AC Immune (NASDAQ:ACIU)
Historical Stock Chart
From Mar 2024 to Apr 2024
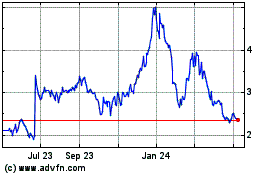
AC Immune (NASDAQ:ACIU)
Historical Stock Chart
From Apr 2023 to Apr 2024