Gilead & Eisai Enter Into Agreement in Japan for the Co-Promotion of the Investigational Rheumatoid Arthritis Therapy Filgoti...
24 December 2019 - 5:00PM
Business Wire
-- Agreement Extends to Additional Potential
Indications for Filgotinib, Including Ulcerative Colitis, Crohn’s
Disease and Psoriatic Arthritis --
Gilead Sciences, Inc. (Nasdaq: GILD) and Eisai Co., Ltd. (Tokyo,
Japan) announced today that Gilead Sciences K.K. (Tokyo, Japan) and
Eisai have entered into an agreement for the distribution and
co-promotion of filgotinib, an investigational, oral, selective
JAK1 inhibitor, in Japan, pending regulatory approval for the
treatment of rheumatoid arthritis (RA). Through this collaboration,
Gilead Japan will retain responsibility for manufacturing and
marketing approval of filgotinib, while Eisai will be responsible
for product distribution in Japan in RA and other potential future
indications. The companies will jointly commercialize the medicine
if approved.
Approximately 600,000 to 1 million people are living with RA
across Japan, and despite available options, many still do not
experience disease remission. In the global Phase 3 FINCH studies,
filgotinib demonstrated durable efficacy and safety results across
multiple RA patient populations, including in people with prior
inadequate response to methotrexate treatment (MTX), those who were
intolerant to one or more biologic treatments and those who were
MTX treatment-naïve.
“We are very pleased to announce this important new partnership
with Eisai, which brings together our complementary expertise and
commitment in inflammation, to deliver this important new option to
patients living with inflammatory diseases in Japan,” said Luc
Hermans, M.D., President and Representative Director, Gilead
Japan.
“We have extensive clinical development and commercialization
experience spanning more than 20 years in RA and have established a
solid RA franchise in Japan,” said Hidenori Yabune, President of
Eisai Japan, Senior Vice President of Eisai. “With this agreement,
we look forward to contributing more to patients living with RA by
adding filgotinib to our product line-up.”
Global studies investigating filgotinib in additional diseases
are also underway, including the Phase 3 SELECTION trial in
ulcerative colitis, the DIVERSITY Phase 3 trial in Crohn’s disease,
the Phase 3 PENGUIN trials in psoriatic arthritis, as well as Phase
2 studies in uveitis and in small bowel and fistulizing Crohn’s
disease.
Gilead and Galapagos NV (Mechelen, Belgium) have entered into a
global collaboration for the development and commercialization of
filgotinib in inflammatory indications. Filgotinib is an
investigational drug whose efficacy and safety have not been
established. Filgotinib is pending regulatory approval in Japan,
Europe and the United States, based on global Phase 3 trials
evaluating its efficacy and tolerability.
About Gilead Sciences
Gilead Sciences, Inc. is a research-based biopharmaceutical
company that discovers, develops and commercializes innovative
medicines in areas of unmet medical need. The company strives to
transform and simplify care for people with life-threatening
illnesses around the world. Gilead has operations in more than 35
countries worldwide, with headquarters in Foster City,
California.
For more information on Gilead Sciences, please visit the
company’s website at www.gilead.com.
About Eisai Co., Ltd.
Eisai Co., Ltd. is a leading global research and
development-based pharmaceutical company headquartered in Japan. We
define our corporate mission as "giving first thought to patients
and their families and to increasing the benefits health care
provides," which we call our human health care (hhc) philosophy.
With approximately 10,000 employees working across our global
network of R&D facilities, manufacturing sites and marketing
subsidiaries, we strive to realize our hhc philosophy by delivering
innovative products to address unmet medical needs, with a
particular focus in our strategic areas of Neurology and Oncology.
As a global pharmaceutical company, our mission extends to patients
around the world through our investment and participation in
partnership-based initiatives to improve access to medicines in
developing and emerging countries.
For more information about Eisai Co., Ltd., please visit
www.eisai.com/.
Gilead Forward-Looking
Statement
This press release includes forward-looking statements within
the meaning of the Private Securities Litigation Reform Act of 1995
that are subject to risks, uncertainties and other factors,
including the risk that the Japanese Ministry of Health, Labour and
Welfare and other regulatory agencies may not approve filgotinib
for the treatment of RA or other potential indications, and any
marketing approvals, if granted, may have significant limitations
on its use. As a result, filgotinib may never be successfully
commercialized. In addition, Gilead and Eisai may not realize the
potential benefits of this partnership. Further, there is the
possibility of unfavorable results from ongoing and additional
clinical trials involving filgotinib. These risks, uncertainties
and other factors could cause actual results to differ materially
from those referred to in the forward-looking statements. These and
other risks are described in detail in Gilead’s Quarterly Report on
Form 10-Q for the quarter ended September 30, 2019, as filed with
the U.S. Securities and Exchange Commission. All forward-looking
statements are based on information currently available to Gilead,
and Gilead assumes no obligation to update any such forward-looking
statements.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20191223005626/en/
Gilead Sciences, Japan Seiko Noma +81-3-6837-0790
Gilead Sciences, US Sonia Choi +1 650-425-5483
Eisai Co., Ltd. Public Relations Department +81-3-3817-5120
Gilead Sciences (NASDAQ:GILD)
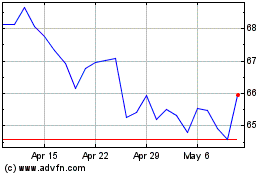
Historical Stock Chart
From Apr 2024 to May 2024
Gilead Sciences (NASDAQ:GILD)
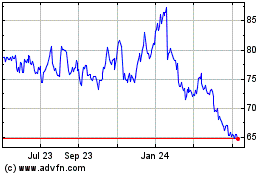
Historical Stock Chart
From May 2023 to May 2024