Processa Pharmaceuticals Appoints Khoso Baluch and James Neal to its Board of Directors and Justin Yorke to Chairman of the Board
16 August 2022 - 10:30PM

Processa Pharmaceuticals, Inc. (Nasdaq: PCSA), a clinical stage
biopharmaceutical company using its Regulatory Science process to
more effectively develop products to improve the survival and/or
quality of life for patients who have an unmet medical need
condition announced today the appointment of Khoso Baluch and James
Neal to its Board of Directors. In addition to the two new
Independent Directors, current board member Justin Yorke has been
named Chairman of the Board.
Justin Yorke, Processa Chairman stated, “I am
excited and delighted to take on the role of Chair of PCSA’s Board
of Directors as we approach some exciting events on the horizon. I
am stepping into this role to support Dr. Young and the team as
they focus on our three ongoing clinical trials and the development
of all 5 of our pipeline products.
Khoso Baluch comes to Processa with over 36
years of experience across global geographies in the
biopharmaceutical industry. Since 2012, he has served as an
independent director of Poxel SA, a French publicly traded biotech
company where he chairs its compensation committee. He has also
served as the Chairman of the Board for Da Volterra, a French
privately held company, since December 2021. From 2016 to 2021, Mr.
Baluch served as the Chief Executive Officer and a board member of
CorMedix, Inc., a publicly traded pharmaceutical company in the US.
Mr. Baluch has held various senior positions at UCB, SA between
January 2008 to April 2016, including Senior Vice President and
President - Europe, Middle East & Africa. Prior to joining UCB,
Mr. Baluch worked for Eli Lilly for 24 years, holding international
positions spanning Europe, the Middle East, and the United States
in general management, business development, market access, and
product leadership. Mr. Baluch holds a B.S. in Aeronautical
Engineering from City University London and an MBA from Cranfield
School of Management.
Mr. Neal was appointed to the Board in August
2022. Mr. Neal brings more than 25 years of experience in forming
and maximizing business and technology collaborations globally and
in bringing novel products and technologies to market. He currently
serves as the Chief Executive Officer and Chairman of the Board of
XOMA Corp, having been elected to that post in 2016. Prior to XOMA,
Mr. Neal was Acting Chief Executive Officer of Entelos, Inc., a
leading biosimulation company that acquired Iconix Biosciences, a
privately held company where Mr. Neal was Chief Executive Officer.
At Iconix, Mr. Neal established multi-year collaborations with
Bristol-Myers Squibb, Abbott Labs, Eli Lilly and the U.S. Food and
Drug Administration. From, 1999-2002, he was Executive Vice
President of Incyte Genomics, leading the global commercial
activities with pharmaceutical company collaborators and partners
including Pfizer, Aventis and Schering-Plough, as well as sales,
marketing and business development activities for the company.
Earlier, he was associated with Monsanto Company in positions of
increasing responsibility. Mr. Neal earned his B.S. in Biology and
his M.S. in Genetics and Plant Breeding from the University of
Manitoba, Canada, and holds an MBA degree from Washington
University in St. Louis, Missouri.
Dr. David Young, Processa’s Chief Executive
Officer added, “I’m pleased to welcome both Khoso and Jim to our
team. Their comprehensive backgrounds and experience in the biotech
space, including licensing and commercialization, make them ideally
suited to assist Processa at this stage of its evolution. I look
forward to working with them and the rest of our board as we
progress towards unlocking numerous value catalysts in our
pipeline.”
About Processa Pharmaceuticals,
Inc.
The mission of Processa is to develop products
with existing clinical evidence of efficacy for patients with unmet
or underserved medical conditions who need treatment options that
improve survival and/or quality of life. The Company uses these
criteria for selection to further develop its pipeline programs to
achieve high-value milestones effectively and efficiently. Active
clinical pipeline programs include: PCS6422 (metastatic colorectal
cancer and breast cancer), PCS499 (ulcerative necrobiosis
lipoidica) and PCS12852 (GI motility/gastroparesis). Members of the
Processa development team have been involved with more than 30 drug
approvals by the FDA (including drug products targeted to orphan
disease conditions) and more than 100 FDA meetings throughout their
careers. For more information, visit our website
at www.processapharma.com.
Forward-Looking Statements
This release contains forward-looking
statements. The statements in this press release that are not
purely historical are forward-looking statements which involve
risks and uncertainties. Actual future performance outcomes and
results may differ materially from those expressed in
forward-looking statements. Please refer to the documents filed by
Processa Pharmaceuticals with the SEC, specifically the most recent
reports on Forms 10-K and 10-Q, which identify important risk
factors which could cause actual results to differ from those
contained in the forward-looking statements.
For More Information:Michael
Floydmfloyd@processapharma.com(301) 651-4256
Patrick Lin(925)
683-3218plin@processapharma.com
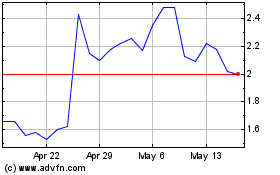
Processa Pharmaceuticals (NASDAQ:PCSA)
Historical Stock Chart
From Jun 2024 to Jul 2024
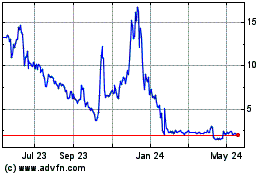
Processa Pharmaceuticals (NASDAQ:PCSA)
Historical Stock Chart
From Jul 2023 to Jul 2024