Baxter Gets FDA Approval for Myxredlin Insulin
22 July 2019 - 11:49PM
Dow Jones News
By Chris Wack
Baxter International Inc. (BAX) said Monday the U.S. Food and
Drug Administration approved Myxredlin insulin for IV infusion.
The medication company said Myxredlin is indicated for use as a
short-acting human insulin to improve glycemic control in adults
and pediatric patients with diabetes mellitus.
Baxter said it expects Myxredlin to be available in the U.S.
before the end of 2019.
Write to Chris Wack at chris.wack@wsj.com
(END) Dow Jones Newswires
July 22, 2019 09:34 ET (13:34 GMT)
Copyright (c) 2019 Dow Jones & Company, Inc.
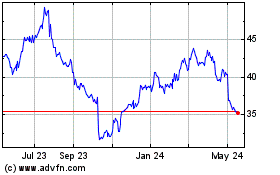
Baxter (NYSE:BAX)
Historical Stock Chart
From Apr 2024 to May 2024
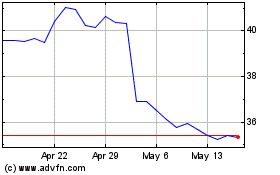
Baxter (NYSE:BAX)
Historical Stock Chart
From May 2023 to May 2024