UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
WASHINGTON,
D.C. 20549
FORM
10-K
(Mark
One)
☒ ANNUAL
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For
the fiscal year ended December 31, 2023
OR
☐
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(D) OF THE SECURITIES EXCHANGE ACT OF 1934
For
the transition period from ____ to ____
Commission
File No. 000-51783
Dror
Ortho-Design, Inc.
(Exact
name of registrant as specified in its charter)
| Delaware | | 85-0461778 |
(State or other jurisdiction of incorporation or organization) | | (I.R.S. Employer Identification Number) |
Shatner Street 3
Jerusalem, Israel | | N/A |
| (Address of principal executive office) | | (Zip Code) |
Registrant’s
telephone number, including area code: +972 (0)74-700-6700
Novint
Technologies, Inc.
100 Merrick Road-Suite 400W, Rockville Center, NY, 11570
| (Former name or former address,
if changed since last report) |
Securities
registered pursuant to Section 12(b) of the Act:
| Title of each class | | Trading Symbol | | Name of each exchange on which registered |
| None | | None | | None |
Securities
registered pursuant to Section 12(b) of the Act: Common Stock, par value $0.0001 per share
Indicate
by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No ☒
Indicate
by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☒
Indicate
by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange
Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports) and (2)
has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate
by check mark whether the registrant has submitted electronically and every Interactive Data File required to be submitted pursuant to
Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit such
files). Yes ☒ No ☐
Indicate
by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting
company or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,”
“smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer | ☐ | Accelerated filer | ☐ |
| Non-accelerated filer | ☒ | Smaller reporting company | ☒ |
| | | Emerging growth company | ☐ |
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate
by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness
of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered
public accounting firm that prepared or issued its audit report. ☐
If
securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant
included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate
by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation
received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate
by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ☐ No ☒
The aggregate market value of the voting and non-voting common equity
held by non-affiliates of the registrant on June 30, 2023, based on the price at which the common equity was last sold of $0.0249 was
$4,310,365.
The
number of shares outstanding of the registrant’s Common Stock as of March 31, 2024 was 495,454,546 shares.
Documents
Incorporated by Reference
None.
DROR
ORTHO-DESIGN, INC.
TABLE
OF CONTENTS
Cautionary
Note Regarding Forward-Looking Statements
Statements
in this Annual Report on Form 10-K (this “Annual Report”) of Dror Ortho-Design, Inc. (f/k/a Novint Technologies, Inc.)
(“we,” “us,” “Dror,” or the “Company”) and other written reports made from time to time
by us that are not historical facts, including statements regarding our strategy, future operations, future financial position, future
revenue, projected costs, prospects, plans, objectives of management and expected market growth, constitute so-called “forward-looking
statements,” all of which are subject to risks and uncertainties. Forward-looking statements can be identified by the use of words
such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,”
“may,” “plan,” “potential,” “predict,” “project,” “should,” “target,”
“will,” “would,” and other words of similar meaning, although not all forward-looking statements contain these
identifying words. Forward-looking statements are likely to address our estimates of our expenses, ongoing losses, future revenue, capital
requirements and our needs for or ability to obtain additional financing; our ability to retain and recruit key personnel;
our financial performance; our ability to become profitable and generate consistent cash flows to remain profitable; our ability
to fund our working capital requirements; developments and projections relating to our competitors or our industry; and our Platform
(as defined below) and any other products, among other things. You should carefully consider any such statement and should understand
that many factors could cause actual results to differ from our forward-looking statements. Such risks and uncertainties include but
are not limited to the following:
| ● | our
operations and financial performance depend on global and regional economic conditions. Inflation, fluctuations in currency exchange
rates, changes in consumer confidence and demand, and weakness in general economic conditions and threats, or actual recessions, could
materially affect our business, results of operations, and financial condition; |
| ● | our
Company is in the development stage, is not generating revenues and has no operating history in the manufacturing and distribution of
orthodontic medical devices or platforms for consumer use; |
| ● | our
products and technologies may not be accepted by the intended commercial consumers of our products, which could harm our future financial
performance; |
| ● | we
expect continued operating losses and cannot be certain of our future profitability; |
| ● | our
net revenues will depend primarily on our Platform and any decline in sales or average selling price of our Platform may adversely affect
net revenues, gross margin and net income; |
| ● | our
Company will face competition from large internationally established aligner companies whose products have been widely accepted; |
| ● | our
growth and future success may depend on our ability to enhance our Platform or to develop, obtain regulatory clearance for, successfully
introduce, and achieve market acceptance of new products and services; |
| ● | we
are subject to operating risks, including excess or constrained capacity and operational inefficiencies, which could adversely affect
our results of operations; |
| ● | our
products and information technology systems are critical to our business. Issues with product development or enhancements, IT system
integration, implementation, updates and upgrades could disrupt our operations and have a material impact on our business and operating
results; |
| ● | complying
with regulations enforced by FDA and other regulatory authorities is expensive and time consuming, and failure to comply could result
in substantial penalties; |
| ● | we
may not receive the necessary authorizations to market our Platform or any future new products, and any failure to timely do so may adversely
affect our ability to grow our business; |
| ● | certain
modifications to our products may require new 510(k) clearance or other marketing authorizations; |
| ● | ongoing
changes in healthcare regulation could negatively affect our revenues, business and financial condition; |
| ● | we
are subject to certain federal, state, and foreign fraud and abuse laws, health information privacy and security laws, and transparency
laws, which, if violated, could subject us to substantial penalties. Additionally, any challenge to or investigation into our practices
under these laws could cause adverse publicity and be costly to respond to, and thus could harm our business; |
| ● | our
success depends in part on our proprietary technology, and if we are unable to successfully enforce our intellectual property rights,
our competitive position may be harmed; |
| ● | the
relative lack of U.S. public company experience of our management team may put us at a competitive disadvantage; |
| ● | our
common stock, par value $0.0001 per share (“Common Stock”), is not listed on any stock exchange and there is a limited market
for shares of our Common Stock. Even if a market for our Common Stock develops, our Common Stock could be subject to wide fluctuations;
and |
| ● | other
risks and uncertainties outlined in section of this Annual Report entitled “1A. Risk Factors” and other risks detailed from
time to time in our filings with the United States Securities and Exchange Commission (the “SEC”) or otherwise. |
These
factors may include inaccurate assumptions and a broad variety of other risks and uncertainties, including some that are known and some
that are not. No forward-looking statement can be guaranteed, and actual future results may vary materially. Information regarding market
and industry statistics contained in this Annual Report is included based on information available to us that we believe is accurate.
It is generally based on industry and other publications that are not produced for purposes of securities offerings or economic analysis.
We have not reviewed or included data from all sources and cannot assure investors of the accuracy or completeness of the data included
in this Annual Report. Forecasts and other forward-looking information obtained from these sources are subject to the same qualifications
and the additional uncertainties accompanying any estimates of future market size, revenue and market acceptance of products and services.
We do not assume any obligation to update any forward-looking statement. As a result, investors should not place undue reliance on these
forward-looking statements.
These
forward-looking statements are based on information available as of the date of this Annual Report and current expectations, forecasts
and assumptions, and involve a number of judgments, risks and uncertainties. Accordingly, forward-looking statements should not be relied
upon as representing our views as of any subsequent date, and we do not undertake any obligation to update forward-looking statements
to reflect events or circumstances after the date they were made, whether as a result of new information, future events or otherwise,
except as may be required under applicable securities laws.
Risk
Factors Summary
Our
business is subject to numerous risks and uncertainties, including those highlighted in the section entitled “Risk Factors”
immediately following this prospectus summary, that represent challenges that we face in connection with the successful implementation
of our strategy and the growth of our business. In particular, the following considerations, among others, may offset our competitive
strengths or have a negative effect on our business strategy, which could cause a decline in the price of shares of our Common Stock
or warrants and result in a loss of all or a portion of your investment:
| ● | Our
financial statements have been prepared on a going concern basis; we must raise additional capital to fund our operations in order to
continue as a going concern. |
| ● | We
conduct our operations in Israel. Conditions in Israel, including the recent attack by Hamas and other terrorist organizations from the
Gaza Strip and Israel’s war against them, may affect our operations. |
| ● | Our
operations and financial performance depend on global and regional economic conditions. Inflation, fluctuations in currency exchange
rates, changes in consumer confidence and demand, and weakness in general economic conditions and threats, or actual recessions, could
materially affect our business, results of operations, and financial condition. |
| ● | Our
Company is in the development stage, is not generating revenues and has no operating history in the manufacturing and distribution of
orthodontic medical devices or platforms for consumer use. |
| ● | Our
products and technologies may not be accepted by the intended commercial consumers of our products, which could harm our future financial
performance. |
| ● | We
expect continued operating losses and cannot be certain of our future profitability. |
| ● | Our
net revenues will depend primarily on our Platform and any decline in sales or average selling price of our Platform may adversely affect
net revenues, gross margin and net income. |
| ● | We
will face competition from large internationally established aligner companies whose products have been widely accepted. |
| ● | Our
growth and future success may depend on our ability to enhance our Platform or to develop, obtain regulatory clearance for, successfully
introduce, and achieve market acceptance of new products and services. |
| ● | We
are subject to operating risks, including excess or constrained capacity and operational inefficiencies, which could adversely affect
our results of operations. |
| ● | Our
products and information technology systems are critical to our business. Issues with product development or enhancements, IT system
integration, implementation, updates and upgrades could disrupt our operations and have a material impact on our business and operating
results. |
| ● | Complying
with regulations enforced by FDA and other regulatory authorities is expensive and time consuming, and failure to comply could result
in substantial penalties. |
| ● | We
may not receive the necessary authorizations to market our Platform or any future new products, and any failure to timely do so may adversely
affect our ability to grow our business. |
| ● | Certain
modifications to our products may require new 510(k) clearance or other marketing authorizations. |
| ● | Ongoing
changes in healthcare regulation could negatively affect our revenues, business and financial condition. |
| ● | We
are subject to certain federal, state, and foreign fraud and abuse laws, health information privacy and security laws, and transparency
laws, which, if violated, could subject us to substantial penalties. Additionally, any challenge to or investigation into our practices
under these laws could cause adverse publicity and be costly to respond to, and thus could harm our business. |
| ● | Our
success depends in part on our proprietary technology, and if we are unable to successfully enforce our intellectual property rights,
our competitive position may be harmed. |
| ● | The
relative lack of U.S. public company experience of our management team may put us at a competitive disadvantage. |
| ● | Our
Common Stock is not listed on any stock exchange and there is a limited market for shares of our Common Stock. Even if a market for our
Common Stock develops, our Common Stock could be subject to wide fluctuations. |
| ● | Other
risks and uncertainties outlined in section of this Annual Report entitled “Risk Factors” and other risks detailed from time
to time in our filings with the SEC or otherwise. |
These
factors may include inaccurate assumptions and a broad variety of other risks and uncertainties, including some that are known and some
that are not. No forward-looking statement can be guaranteed, and actual future results may vary materially.
PART
I
Item
1. Business.
As
used in this Report, all references to “we,” “our” and “us” for periods prior to the closing of the
Share Exchange refer to Dror Ortho-Design Ltd., a company incorporated under the laws of the State of Israel, and for periods subsequent
to the closing of the Share Exchange refer to Dror Ortho-Design, Inc., a Delaware corporation and its direct and indirect subsidiaries.
Overview
We were incorporated as Novint Technologies, Inc. in the State of New
Mexico in April 1999. On February 26, 2002, we changed our state of incorporation to Delaware by merging with Novint Technologies, Inc.,
a Delaware corporation. On July 5, 2023, we entered into a share exchange agreement with the shareholders of Dror Ortho-Design, Ltd. (“Private
Dror”), pursuant to which the shareholders of Private Dror agreed to exchange all of their outstanding ordinary shares Private Dror
for shares of our Common Stock and convertible preferred stock (the “Share Exchange”). On August 14, 2023, the Share
Exchange was consummated and we changed our name from “Novint Technologies, Inc.” to “Dror Ortho-Design, Inc.”
Following the Share Exchange, we succeeded to the business of Private Dror as our sole line of business.
Our
Company
We
have reimagined the way people can correct their smile.
We
plan to disrupt the aligner market by offering millions of people a revolutionary alternative. We believe that people do not need to
change their lifestyle to correct their smile as they are required to do with existing aligner solutions.
Existing
aligner solutions generally share the same treatment principles, which are different from our solution. In most cases, patients seeking
to improve their smile need to undergo a 12-to-15 month process of wearing plastic aligners, which need to be worn the entire day and
should only be removed while eating or drinking. Patients are prescribed a series of 20 to 30 aligners that are intended to forcefully
move teeth progressively closer to their intended final position. This process causes pain every time a new aligner is used and restricts
blood circulation, which counterproductively slows down tooth movement. All-day aligner solutions are also intrusive, as patients need
to conduct their lives at work or school wearing the plastic aligners. In addition, most existing aligner therapies require multiple
visits to an orthodontist to monitor the progress of treatment plans through intraoral scanning, physical examination and patient testimony.
We
believe that recent rapid advancements in technology have made traditional aligner solutions no longer the most effective treatment option
for smile correction. Our Company has developed a proprietary AI-based platform to correct people’s smiles in a discreet and less
painful manner (the “Platform”). The Platform uses only one smart aligner to gently move teeth into their optimum position
with pulsating air while the patient is sleeping or at home. The Company has several patents for the technology used in the Platform
and is currently in the process of preparing the prototype for FDA approval.
Our
predecessor first generation Aerodentis System is a Class II medical device, which was cleared by FDA for commercialization in the U.S.
pursuant to the 510(k) notification process for movement and alignment of teeth during orthodontic treatment of malocclusion in April
2020. The Company is preparing to apply for 510(k) clearance for the Platform as a Class II medical device, which constitutes an updated
version of the currently cleared device. Such updated Platform contains new and/or different components than the original device, which
is why a new 510(k) clearance is required prior to marketing the Platform in the U.S. We have not yet filed a 510(k) submission for the
Platform, and it has, thus, not been found by the FDA to be substantially equivalent to the first generation Aerodentis System.
The
Company currently does not generate revenues to fund operations and anticipates that it will continue to incur significant losses as
it continues to develop the Platform. Please refer to “Risk Factors - We are in the development stage, are not generating revenues
and have no operating history in the manufacturing and distribution of orthodontic medical devices or platforms for consumer use.”
for additional information. The Company intends to spend approximately $2.5 million over the next 18 months on software and hardware
development as well as the accompanying regulatory approvals and IP protection associated with such software and hardware projects.
Our
Product
The
First Generation Aerodentis System
Our
Company was founded in 2005 with the goal of offering millions of people a chance to correct their smile in a more discreet and less
painful manner. The first generation of our product underwent ten years of development by a team of twelve orthodontists, engineers,
industrial designers and dental technicians. This team developed a new clinically-proven method for correcting Class 1 and Class 2 malocclusion
using pulsating air. The team discovered that using pulsating air improved blood circulation in the gums, which is essential to tooth
movement. This first-generation product (the “Aerodentis System”) was composed of a base control unit that contained a pump
and motor that would deliver pulses of air to a micro balloon that was part of a mouthpiece to be used by the patient to deliver the
treatment. The use of pulsating air is the base patented technology that distinguishes our Aerodentis System from clear aligner therapies,
which are designed to move teeth using continuous resistant force delivered by the aligner, which impairs blood flow.

Pictured:
Base control unit, containing micro-pump and controls, attached to the smart aligner. The smart aligner is composed of an outer mouthpiece
structure, which is shaped based on the final position of the teeth for a perfect smile. Behind the outer mouthpiece structure is a micro
balloon that is attached to the base control unit with a fine and flexible microtube. The balloon delivers pulsating air by inflating
and deflating. Behind the balloon is the “push structure,” which provides the balloon with a surface to push against as it
gently moves the teeth.
In
January 2013, the Aerodentis System composed of the base control unit and custom mouthpiece received the European CE Mark. In 2020, it
received FDA clearance via the 510(k) process as a Class II medical device, with broad indication for use “in movement and alignment
of teeth during orthodontic treatment of malocclusion.” Clinical trials demonstrated that Aerodentis System was suitable for adults
and pediatric patients with Class 1 and Class 2 malocclusion, including crowding, proclination and retroclination. Further, clinical
trials have demonstrated that the effectiveness of Aerodentis System was consistent with the results achieved by the Invisalign clear
aligners solution provided by Align Technology, Inc.

Pictured:
Close up of smart aligner with (1) outer structure formed based on the final tooth position desired for a perfect smile and (2) micro
balloon inserted between the outer structure and the inner structure to support the balloon’s expansion.
The
Platform
Building
on the Aerodentis System, we have developed a prototype of the Platform, our next generation, comprehensive enhanced solution to Class
1 and Class 2 malocclusion for which we intend to submit a 510(k) application for marketing in the U.S., as the Platform is beyond the
scope of our current FDA clearance. The prototype of the Platform was developed over the course of eighteen months and is intended to
advance the proven clinical features of the Aerodentis System while incorporating recent developments in artificial intelligence utilized
in our Aerodentis AI Cloud (as defined below) component of the Platform, secure wireless and Internet communications with Internet of
Things (“IoT”) devices used in our Smart Aligner System component of the Platform and advanced imaging and 3D printing technologies.
IoT devices revers to pieces of hardware, such as sensors, actuators, gadgets, appliances, or machines, that are programmed for certain
applications and can transmit data over the internet or other networks.
Our Platform
is comprised of three primary components:
| ● | the
Aerodentis smartphone application; |
| ● | our
AI-based cloud service (“Aerodentis AI Cloud”), which is used to perform analytics and manage patient treatment plans; and |
| ● | the
smart aligner system used by the patient, which consists of: (i) a base control unit containing the pump and the IoT components and (ii)
a smart aligner containing the micro-balloon that gently pushes teeth into their intended final position using pulsating air (the “Smart
Aligner System”). |

The following
provides a more detailed description of each of the components of our Platform:
Aerodentis
Smartphone Application
Our
freely downloadable Aerodentis smartphone application will allow potential patients to make a video of their smile and teeth and upload
the video to the Aerodentis AI Cloud. This 2D video will be converted into a 3D model using our proprietary patent-pending AI based image
analysis technology. The underlying algorithms will then perform an initial analysis to determine if the patient can potentially benefit
from our solution. This complex analysis will be performed in minutes and will deliver a “Go/No Go” response. Once a patient
begins treatment, they will use the smartphone application to provide their dental professional with ongoing remote monitoring of their
treatment progress. The smartphone application can be used to upload additional teeth videos showing progress and to transmit data from
the Smart Aligner System (described below), including the amount of time the patient used the Smart Aligner System and the pressure and
pulse levels administered.
Aerodentis
AI Cloud
The
Aerodentis AI Cloud will be used to analyze data uploaded by patients and to facilitate communication between patients and dental professionals.
If the analysis performed on the initial video upload from the Aerodentis smartphone application delivers a “Go” result,
the patient will be invited to have an intraoral scan performed by a dental professional from our network of participating providers.
The results of this intraoral scan will be uploaded to the Aerodentis AI Cloud by the dental professional, and the Aerodentis AI Cloud
will use a machine learning algorithm to compare the scan with the initial model generated from the patient’s initial video upload.
The machine learning algorithm is designed to learn with every scan how to improve the accuracy of the 3D images it generates from smartphone
videos. We believe that the Platform’s image analysis of smartphone videos will eventually approach the level of accuracy observed
in intraoral scans. If we achieve this, we will be in a position to be able provide highly accurate image analysis of teeth that can
be used throughout the dental industry since it would allow for smartphones to essentially replace the need for intraoral scans for certain
cases. This would dramatically increase the efficiency and treatment delivery cycle in the dental industry and result in a potentially
material economic benefit to our Company in the future.
The
Aerodentis AI Cloud will also be used for ongoing analysis of patient data and management of a patient’s treatment plan throughout
the treatment. A dental professional will use our Platform to develop a customized treatment plan, including any interproximal reduction
necessary before treatment begins, based on the Smart Aligner System. As a patient uploads progress videos from their smartphone, the
Platform will compare tooth positions in previous videos to current positions. A dental professional will be able to use this data to
remotely monitor the treatment progress and modify the treatment plan remotely as needed.
Smart
Aligner System
The
Platform’s Smart Aligner System features a newer, more advanced version of our first generation Aerodentis System, featuring completely
redesigned micropump and motor mechanisms. The redesign has significantly increased the pump’s pressure capacity, efficiency, and
durability. In addition, the base control unit of Smart Aligner System is now IoT-enabled to allow external secure communication with
the device using Wi-Fi and Bluetooth. The device will thus be able to communicate with the patient’s smartphone as well as the
Aerodentis AI Cloud and the designated dental professional, subject to FDA clearance.
The
clear aligner of a patient’s Smart Aligner System will be created using 3D printing based on various 3D images of the patient’s
teeth that are collected and analyzed in the Aerodentis AI Cloud. This will represent a significant development in our industry since,
today, aligners are not printed but produced using a thermoforming process. Other companies have implemented 3D printing to produce the
aligner models but not the actual aligners. Although using 3D printing is a superior method for production due to its level of precision
and customizability, it has not been implemented in the production of aligners in the traditional aligner market because it would be
financially prohibitive to do so, since traditional aligner solutions would need to print multiple aligners for each patient. Since our
solution requires only one smart aligner to be produced for each patient, we will be able to take advantage of this cost-effective production
method that will also have economies of scale.
Market
Opportunity
Malocclusion
is one of the most prevalent clinical dental conditions in the world, affecting approximately 60% to 75% of the global population.1
It is estimated that there are approximately 500 million people globally with malocclusion who could benefit from straightening
their teeth.2 However, most people afflicted by malocclusion do not seek orthodontic treatment due to a number of reasons,
including negative perceptions of metal braces, affordability of treatment, and accessibility to doctors in certain markets and geographies.
Annually, only approximately 21 million or 4.2% of the affected individuals elect treatment by orthodontists.3 Today, most
orthodontic patients continue to have their malocclusions treated with the use of traditional corrective methods such as metal arch wires
and brackets, referred to as braces, augmented with elastics, metal expanders, headgear or functional appliances, and other ancillary
devices as needed. Upon completion of a patient’s treatment, their dental professional may recommend the patient use a retainer
appliance to preserve the benefits of their treatments.
According
to a 2022 study conducted by Precedent Research (“Precedence Research 2022 Study”), the global clear aligners market size
was estimated at $6.29 billion in 2022 and is expected to surpass around $46.3 billion by 2030, expanding at a compound annual growth
rate (CAGR) of 28.34% during the period 2022 to 2030.4
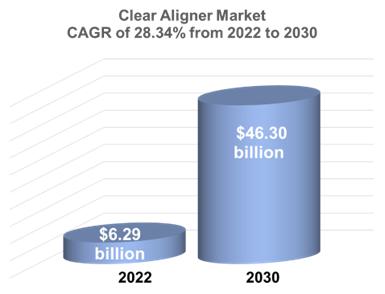
Source:
Precedence Research Study, 2022
Our
Platform seeks to address this large and underserved global market by offering a discreet, less intrusive and less painful treatment
alternative to available clear aligners and traditional orthodontic treatments. Our Platform is optimized to correct malocclusions that
relate to the “social six,” which are the front upper six and lower six teeth. We believe that at least 30% of those who
currently seek treatment, or 6.6 million people, could benefit from using Aerodentis to correct their smiles. According to the Precedence
Research 2022 Study, by 2028, the market for clear aligners will surpass 22 million people, which is our total addressable market.
| 1 | See
Alhammadi, Maged Sultan, et al. “Global distribution of malocclusion traits: A systematic review.” Dental press journal of
orthodontics 23 (2018): 40-e1. |
| 2 | Fortune
Business Insight. The global clear aligners market is projected to grow from $3.80 billion in 2023 to $17.27 billion by 2030, at a CAGR
of 24.2% during the forecast period, 2023-2030 (June 2023), available at https://www.fortunebusinessinsights.com/industry-reports/clear-aligners-market-101377. |
| 3 | Medi-Tech
Insights. Global Orthodontic Supplies Market Report 2027 – Improving Oral Health Care, available at https://meditechinsights.com/global-orthodontic-supplies-market/. |
| 4 | Precedence
Research. Clear Aligners Market (By Age: Adults, Teenagers; By Type: At-home aligners/Direct-to-consumer (DTC) Aligners, In-office Aligners;
By Product: Hard Type, Medium Type, Soft Type; By Material Type: Polyurethane, Plastic Polyethylene Terephthalate Glycol, Poly-vinyl
Chloride; By Distribution Channel: Direct Sales, Laboratories, Others; By End-User: Hospitals, Standalone Practices, Group Practices,
Others) - Global Industry Analysis, Size, Share, Growth, Trends, Regional Outlook, and Forecast 2022-2030 (October 2022), available at
https://www.precedenceresearch.com/clear-aligners-market. |

Source:
Precedence Research Study, 2022
| ● | Ourtotal
addressable market also stands to benefit from the recent trend toward dentists, rather than orthodontists, delivering orthodontic care
through clear aligners. In order for a smile correction solution to work properly, a treatment plan and monitoring needs to be executed
by a dental professional, such as dentists and orthodontists. Since the Aerodentis Platform provides the necessary information to develop
and administer a treatment plan using our solution, it may be used by dentists as well as orthodontists, which expands our target distribution
channel to cover both orthodontists and dentists. According to a 2019 Journal of Family Medicine and Primary Care article, approximately
36% of dentists were already performing orthodontic procedures such as the malocclusion corrections.5 This is an indication
of an ongoing trend of dentists assuming more orthodontic treatment offerings in their practices. We believe that the ease of use of
our Platform will also facilitate eventually selling our solution directly to the consumer in qualified cases with remote dental professional
involvement. |
Business
Model
Our
business model is focused on engaging the customer throughout their smile correction journey and beyond. Our solution provides an innovative,
proprietary end-to-end platform that spans all stages of customer engagement, from initial acquisition to treatment and ongoing maintenance—all
with minimal need for office visits and lifestyle inconvenience.

Customer
Initiated Dentist-Controlled Treatment
Unlike
other solutions in the market, such as traditional clear aligners, we believe our Platform will provide greater access and interaction
with the customers and allow customers to feel more involved in their own treatment process. We hope to engage the power of social media
and other digital outlets to initiate initial demand for our Platform by the customers.
| 5 | See Jayaprakash, Poonam K., et al. “A survey on orthodontic
services provided by general dental practitioners.” Journal of family medicine and primary care 8.7 (2019): 2490-2495. |
Customer
Engagement – Value Creation
Our
Platform is designed to have a high level of engagement with customers, if cleared for marketing in the U.S., as users will be able to
scan their teeth with any smartphone and see how our solution can improve their smile. We intend to engage the customer from their first
interest in correcting their smile and guide them throughout our convenient process.
Network
of Dental Professionals
If
cleared by FDA, the Platform will generally function via the following process: if the customer can benefit from our Platform, based
on the severity of their tooth alignment and malocclusions, they will be referred to a dental professional in our network for an intraoral
scan. Once the results of the scan are uploaded to our Aerodentis AI Cloud, a remote dental professional will develop a treatment plan
for that patient using our Platform. If the patient requires any tooth preparation before initiating treatment with the smart aligner,
the patient will again be referred to a dental professional in our network. The Company currently does not have any written agreements
or arrangements with any dental professionals governing provision of orthodontic services using our Platform.
Monetization
– Value Capture
We
intend to generate revenues by:
| ● | reselling
our solution through a professional dental network; |
| ● | providing
ongoing monitoring and treatment plans for those who have completed their smile correction and may require smile maintenance throughout
their life; and |
| ● | eventually
selling directly to the consumer in qualified cases with remote dental professional involvement |
Sales
and Marketing
We
intend to market our Platform in Israel, the European Union (“E.U.”), United Kingdom, United States, and Canada, subject
to each country’s requisite regulatory authorization. We intend to utilize social media to promote our Platform to our targeted
audience. The Platform has a potentially viral social media message that we hope will drive demand by placing user-generated content
on all major social medial platforms. Our marketing strategy themes and promotional messages will emphasize the ease and convenience
offered by our Platform as compared to other available treatments.
Research
and Development
We
have a research and development team with software development, medical device development, dental/orthodontic, data science and other
innovation focused backgrounds. Our current research and development efforts are primarily focused on enhancing the Platform and developing
software and processes to enable the manufacture of our smart aligner systems in volume as well as productizing the prototype through
the development of UI/UX and system integration with existing patent systems.
As
of March 2023, our outsourced software development team is composed of eight professionals with years of experience in artificial intelligence
development, data science, application and software engineering. Members of the team come from the elite intelligence units of the Israeli
Defense Force and have a breadth of experience in computer vision, imaging and targeting systems development. Our software development
team is headed by Yossi Avni, who has 25 years of experience in developing advanced artificial intelligence applications, behavioral
biometrics, behavioral profiling and advanced security systems and holds over 100 patents in these areas.
Our
hardware and systems development team is composed of six professionals with years of experience in FDA-compliant medical device development.
They are a part of Aran Research Development Prototypes Ltd. (“Aran”), a leading Israeli product design and development firm
and our third-party hardware development partner. Aran is ISO 13485 certified and maintains a ISO 7 cleanroom for testing and assembly.
Aran also has manufacturing facilities and a full suite of 3D printing capabilities, which are compliant with FDA guidelines. Our hardware
and systems development team is headed by Avi Kayton, a skilled development manager and systems engineer with 16 years of experience,
including extensive experience in medical device companies.
Intellectual
Property
We
have three issued U.S. patents, four pending U.S. patents and numerous global patent applications. These patents and applications cover
critical aspects of our Platform, including the movement of teeth using pulsating air, our diagnostic process, Platform technology, and
3D printing. Our issued U.S. patents 7819661, 10806376, and 10820965 expire in 2030, 2040, and 2040 respectively. We currently do not
own any trademarks.
We
intend to continue to pursue further intellectual property protection through U.S. and non-U.S. patent applications, trademark applications,
and non-disclosure and non-compete agreements. We also intend to seek to protect our software, documentation and other written materials
under trade secret and copyright laws. There can be no assurance that patents will be issued as a result of any patent application or
that patents that have been issued to us or may issue in the future will be found to be valid and enforceable and sufficient to protect
our technology or products.
Seasonality
Our
business is generally not seasonal. However, we may experience moderate sales fluctuations, at certain periods of the year, such as January,
due to renewed consumer focus on health improvement and aesthetics.
Competition
The
dental industry is in a period of immense and rapid digital transformation involving products, technologies, distribution channels and
business models. We face competition in the market for our Platform from the clear aligners market and we expect competition from existing
competitors and new companies that may enter the market or introduce new technologies in the future. We compete with several well-established
companies both in the traditional orthodontic industry and the direct-to-consumer clear aligner industry, including Align Technologies,
Smile Direct Club, Dentsply Sirona (Byte), 3M Clarity Aligners, and Straumann Group. Although these companies offer clear aligner solutions,
and thus do not use technologies similar to the Platform, we expect that potential patients will view clear aligner products as alternatives
to the Platform. For this reason, we view any company in the clear aligners market as a potential competitor.
We believe
that the principal competitive factors in the market for orthodontic appliances include:
| ● | price
and financing options; |
| ● | aesthetic
appeal of the treatment method; |
| ● | comfort
associated with the treatment method; |
| ● | duration
and effectiveness of treatment; |
| ● | orthodontist
chair time. |
We
believe that our Platform will compare favorably with respect to each of these factors.
Government
Regulation
Our
products (including the currently cleared version, as well as the next generation Platform for which we have not yet submitted the requisite
510(k) application to FDA) are considered medical devices, and, accordingly, are subject to rigorous regulation by government agencies
in the United States and other countries in which we intend to sell our products. These regulations vary from country to country but
cover, among other things, the following activities with respect to medical devices:
| ● | design,
development and manufacturing; |
| ● | testing,
labeling, content and language of instructions for use and storage; |
| ● | product
storage and safety; |
| ● | marketing,
sales and distribution; |
| ● | pre-market
clearance and approval; |
| ● | record
keeping procedures; |
| ● | advertising
and promotion; |
| ● | recalls
and field safety corrective actions; |
| ● | post-market
surveillance; |
| ● | post-market
approval studies; and |
| ● | product
import and export |
FDA
Regulation
In
the U.S., numerous laws and regulations govern the processes by which medical devices are developed, manufactured, brought to market
and marketed. These include the Federal Food, Drug, and Cosmetic Act (“FD&C Act”) and its implementing regulations issued
by FDA, among others. Unless an exemption applies, each medical device commercially distributed in the United States requires FDA clearance
of a 510(k) premarket notification (“510(k) clearance”), granting of a de novo request, or approval of an application
for premarket approval (“PMA”). In general, under the FD&C Act, medical devices are classified in one of three classes
on the basis of the controls necessary to reasonably assure their safety and effectiveness. A medical device’s classification determines
the level of FDA review and approval to which the device is subject before it can be marketed to consumers:
| ● | Class
I devices, the lowest-risk FDA device classification, include devices with the lowest risk to the patient and are those for which safety
and effectiveness can be assured by adherence to FDA’s medical device general controls, including labeling, establishment registration,
device product listing, adverse event reporting, and, for some products, adherence to good manufacturing practices through FDA’s
Quality System Regulations. |
| ● | Class
II devices, moderate-risk devices, also require compliance with general controls and in some cases, special controls as deemed necessary
by FDA to ensure the safety and effectiveness of the device. These special controls may include performance standards, particular labeling
requirements, or post-market surveillance obligations. While most Class I devices are exempt from the 510(k) premarket notification requirement,
typically a Class II device also requires pre-market review and 510(k) clearance as well as adherence to the Quality System Regulations/good
manufacturing practices for devices. |
| ● | Class
III devices, high-risk devices that are often implantable or life-sustaining, also require compliance with the medical device general
controls and Quality System Regulations, and generally must be approved by FDA before entering the market through a PMA application.
Approved PMAs can include post-approval conditions and post-market surveillance requirements, analogous to some of the special controls
that may be imposed on Class II devices. |
Our
manufacturing quality system is required to be in compliance with the Quality System Regulations enforced by FDA and similar regulations
enforced by other worldwide regulatory authorities. FDA’s Quality System Regulations require manufacturers to follow stringent
design, testing, process control, documentation, and other quality assurance procedures.
Our
first generation Aerodentis System is a Class II medical device, which was cleared by FDA for commercialization in the U.S. pursuant
to the 510(k) notification process for movement and alignment of teeth during orthodontic treatment of malocclusion in April 2020. We
are preparing to apply for 510(k) clearance for the updated version of the currently cleared device. Such updated Platform contains new
and/or different components than the original device, which is why a new 510(k) clearance is required prior to marketing the Platform
in the U.S. We have not yet filed a 510(k) submission for the Platform, and it has, thus, not been found by the FDA to be substantially
equivalent to the first generation Aerodentis System. The manufacture, marketing and distribution of the Aerodentis System, as well as
our next-generation Platform once cleared by FDA, if ever, is subject to continuing regulation and enforcement by FDA and other government
authorities, which includes routine FDA inspections of our facilities to determine compliance with facility registration requirements,
product listing requirements, medical device reporting regulations, and Quality System Regulations, among others. If FDA finds that we
have failed to comply with Quality System Regulations or other legal or regulatory requirements, it or other government agencies may
institute a wide variety of enforcement actions against us, ranging from Warning Letters to more severe sanctions, including but not
limited to financial penalties, withdrawal of 510(k) clearances already granted, and criminal prosecution. We have passed our International
Organization for Standardization (“ISO”) and Medical Device Single Audit Program (“MDSAP”) certification process
and have added the U.S. to our ISO/MDSAP certification in 2019
The
510(k) Process
Under
the 510(k) process, the manufacturer must submit to FDA a premarket notification demonstrating that the device is “substantially
equivalent” to either a device that was legally marketed prior to May 28, 1976, the date upon which the Medical Device Amendments
of 1976 were enacted, and for which a PMA is not required, a device that has been reclassified from Class III to Class II or Class I,
or another commercially available device that was cleared through the 510(k) process. To be “substantially equivalent,” the
proposed device must have the same intended use as the predicate device, and either have the same technological characteristics as the
predicate device or have different technological characteristics and not raise different questions of safety or effectiveness than the
predicate device. Clinical data is sometimes required to support substantial equivalence.
After
a 510(k) premarket notification is submitted, FDA determines whether to accept it for substantive review. If it lacks necessary information
for substantive review, FDA will refuse to accept the 510(k) notification. If it is accepted for filing, FDA begins a substantive review.
By statute, FDA is required to complete its review of a 510(k) notification within 90 days of receiving the 510(k) notification. As a
practical matter, clearance often takes longer, and clearance is never assured. FDA may require further information, including clinical
data, to make a determination regarding substantial equivalence, which may significantly prolong the review process. If FDA agrees that
the device is substantially equivalent to a predicate device currently on the market, it will grant 510(k) clearance to commercially
market the device.
Post-Market
Regulation
After
a device is cleared or approved for marketing, numerous and extensive regulatory requirements may continue to apply. These include but
are not limited to:
| ● | annual
and updated establishment registration and device listing with FDA; |
| ● | Quality
System Regulation requirements, which require manufacturers to follow stringent quality assurance procedures during all aspects of the
design and manufacturing process; |
| ● | restrictions
on sale, distribution, or use of a device; |
| ● | labeling,
advertising, promotion, and marketing regulations, which require that promotion is truthful,
not misleading, and provide adequate directions for use and that all claims are substantiated,
and also prohibit the promotion of products for unapproved or “off-label” uses
(i.e., indications that are inconsistent with or beyond the scope of the applicable FDA approval
or clearance) and impose other restrictions on labeling; |
| ● | clearance
or approval of product modifications to legally marketed devices that could significantly
affect safety or effectiveness or that would constitute a major change in intended use; |
| ● | medical
device reporting regulations, which require that a manufacturer report to FDA if a device
it markets may have caused or contributed to a death or serious injury, or has malfunctioned
and the device or a similar device that it markets would be likely to cause or contribute
to a death or serious injury if the malfunction were to recur; |
| ● | correction,
removal, and recall reporting regulations, and FDA’s recall authority; |
| ● | complying
with the federal law and regulations requiring Unique Device Identifiers on devices; and |
| ● | post-market
surveillance activities and regulations, which apply when deemed by FDA to be necessary to
protect the public health or to provide additional safety and effectiveness data for the
device. |
FDA
has broad regulatory compliance and enforcement powers. If FDA determines that we failed to comply with applicable regulatory requirements,
it can take a variety of compliance or enforcement actions, which may result in any of the following sanctions:
| ● | warning
letters, untitled letters, fines, injunctions, consent decrees, and civil penalties; |
| ● | recalls,
withdrawals, or administrative detention, or seizure of our products; |
| ● | operating
restrictions or partial suspension or total shutdown of production; |
| ● | refusing
or delaying requests for 510(k) marketing clearance or PMA approvals of new products or modified
products; |
| ● | withdrawing
510(k) clearances or PMA approvals that have already been granted; |
| ● | refusal
to grant export or import approvals for our products; or |
International
Regulation
Many
countries throughout the world have established regulatory frameworks for marketing and commercialization of medical devices. As a designer,
manufacturer, and marketer of medical devices, we are obligated to comply with the respective frameworks of these countries to obtain
and maintain access to these global markets. The frameworks often define requirements for marketing authorizations which vary by country.
Failure to obtain appropriate marketing authorization and to meet all local requirements, including specific quality and safety standards
in any country in which we currently market our products, could cause commercial disruption and/or subject us to sanctions and fines.
Delays in receipt of, or a failure to receive, such marketing authorizations, or the loss of any previously received authorizations,
could have a material adverse effect on our business, financial condition and results of operations.
There
is currently no premarket government review of medical devices in the European Economic Area (“EEA”). However, all medical
devices placed on the market in the EEA must meet the relevant essential requirements laid down in Annex I of Directive 93/42/EEC concerning
medical devices, or the Medical Devices Directive. The most fundamental essential requirement is that a medical device must be designed
and manufactured in such a way that it will not compromise the clinical condition or safety of patients, or the safety and health of
users and others. In addition, the device must achieve the performances intended by the manufacturer and be designed, manufactured, and
packaged in a suitable manner. The European Commission has adopted various standards applicable to medical devices. These include standards
governing common requirements, such as sterilization and safety of medical electrical equipment, and product standards for certain types
of medical devices. There are also harmonized standards relating to design and manufacture. While not mandatory, compliance with these
standards is viewed as the easiest way to satisfy the essential requirements as a practical matter. Compliance with a standard developed
to implement an essential requirement also creates a rebuttable presumption that the device satisfies that essential requirement.
On
April 5, 2017, the European Parliament passed the Medical Devices Regulation (Regulation 2017/745), which repeals and replaces the E.U.
Medical Device Directive and became effective on May 26, 2021. The Medical Devices Regulation, among other things, is intended to establish
a uniform, transparent, predictable, and sustainable regulatory framework across the EEA for medical devices and ensure a high level
of safety and health while supporting innovation. The new regulations, among other things:
| ● | strengthen
the rules on placing devices on the market and reinforce surveillance once they are available; |
| ● | establish
explicit provisions on manufacturers’ responsibilities for the follow-up of the quality, performance, and safety of devices placed
on the market; |
| ● | improve
the traceability of medical devices throughout the supply chain to the end-user or patient through a unique identification number; |
| ● | set
up a central database to provide patients, healthcare professionals, and the public with comprehensive information on products available
in the E.U.; and |
| ● | strengthen
rules for the assessment of certain high-risk devices, such as implants, which may have to undergo an additional check by experts before
they are placed on the market. |
We
received our European CE mark and ISO/MDSAP certification in 2019. In light of our ISO/MDSAP certification, we believe that we are in
substantial compliance with applicable E.U. regulations and do not anticipate having to make any material expenditures as a result of
E.U. or other currently applicable regulatory requirements. Under Medical Devices Regulation, manufacturing facilities are subject to
periodic inspections by regulatory authorities and must comply with device safety and effectiveness requirements as set forth therein.
To that end, we have implemented controls and procedures intended to ensure that our Access Dental Lab Quality System meets FDA’s
and ISO requirements. We passed our audit to renew our ISO/MDSAP certification in April 2023.
Quality
System Regulations
Our
manufacturing quality system is required to be in compliance with the Quality System Regulations enforced by FDA and similar regulations
enforced by other worldwide regulatory authorities. FDA’s Quality System Regulations require manufacturers to follow stringent
design, testing, process control, documentation, and other quality assurance procedures. If FDA finds that we have failed to comply with
Quality System Regulations or other legal or regulatory requirements, it or other government agencies may institute a wide variety of
enforcement actions against us, ranging from Warning Letters to more severe sanctions, including but not limited to financial penalties,
withdrawal of 510(k) clearances already granted, and criminal prosecution. In addition, under Canadian regulation, manufacturing facilities
are subject to periodic inspections by regulatory authorities and must comply with device safety and effectiveness requirements as required
by the Medical Devices Regulation.
State
Professional Regulation
Our
ability to conduct business in each state is dependent in part upon that particular state’s treatment of remote healthcare delivery
under such state’s laws, rules and policies governing the practice of dentistry, which are subject to changing political, regulatory
and other influences. Orthodontists and dentists who provide professional services to a patient via teledentistry must, in most instances,
hold a valid license to practice or to provide treatment in the state in which the patient is located. In addition, certain states require
an orthodontist or dentist providing telehealth services to be physically located in the same state as the patient. Failure to comply
with these laws and regulations can give rise to civil or criminal penalties.
Other
U.S. Federal and State Laws
We
are also subject to various laws inside and outside the U.S. concerning our relationships with healthcare professionals and government
officials, price reporting and regulation, the promotion, sales and marketing of our products and services, the importation and exportation
of our products, reimbursement for our products and services, the operation of our facilities, and the distribution of our products.
Initiatives sponsored by government agencies, legislative bodies, and the private sector regarding these matters, including efforts to
limit the growth of healthcare expenses generally, are ongoing in markets where we do business. It is not possible to predict at this
time the long-term impact of such cost containment and other measures on our future business.
We
intend to enter into contracts with orthodontists, dentists, or professional corporations to deliver our products and services to their
patients. Such contractual relationships will be subject to various state laws that prohibit the practice of dentistry by lay entities
or persons and are intended to prevent unlicensed persons from interfering with or influencing the orthodontist’s or dentist’s
professional judgment. In addition, laws in various states also generally prohibit the sharing of professional services income with nonprofessional
or business interests. Activities other than those directly related to the delivery of healthcare may be considered an element of the
practice of dentistry in many states. Under the corporate practice of dentistry restrictions of certain states, non-clinical decisions
and activities may implicate the restrictions on the corporate practice of dentistry. We will continually monitor state requirements
as to what constitutes the practice of dentistry and take steps to ensure that the orthodontists and dentists who utilize our services
and teledentistry platform handle all clinical aspects of their patients’ care to ensure we do not violate those laws and regulations.
As
a participant in the health care industry we are subject to extensive and frequently changing regulation under many other laws administered
by governmental entities at the federal, state, and local levels, some of which are, and others of which may be, applicable to our business.
Laws regulating medical device manufacturers and health care providers cover a broad array of subjects.
Several
states have fraud and abuse and consumer protection laws that apply to healthcare items or services reimbursed by any third-party payor,
including commercial insurers, not just those reimbursed by a federally funded healthcare program, or apply regardless of payor. The
scope of these laws and the interpretations of them vary from state to state and are enforced by state courts and regulatory authorities,
each with broad discretion. A determination of liability under such laws could result in fines and penalties and restrictions on our
ability to operate in these jurisdictions.
Health
Information Privacy and Security Laws
There
are numerous U.S. federal and state laws and regulations related to the privacy and security of PII, including health information. Among
others, the federal Health Insurance Portability and Accountability Act of 1996, as amended by HITECH, and their implementing regulations,
which we collectively refer to as HIPAA, establish privacy and security standards that limit the use and disclosure of PHI and require
covered entities and business associates to implement administrative, physical, and technical safeguards to ensure the confidentiality,
integrity, and availability of individually identifiable health information in electronic form, among other requirements.
Violations
of HIPAA may result in civil and criminal penalties. We must also comply with HIPAA’s breach notification rule which requires notification
to affected individuals and HHS, and in certain cases to media outlets, in the case of a breach of unsecured PHI. The regulations also
require business associates of covered entities to notify the covered entity of breaches by the business associate.
State
attorneys general also have the right to prosecute HIPAA violations committed against residents of their states, and HIPAA standards
have been used as the basis for the duty of care in state civil suits, such as those for negligence or recklessness in misusing personal
information. In addition, HIPAA mandates that HHS conduct periodic compliance audits of HIPAA covered entities and their business associates
for compliance.
Many
states also have laws that protect the privacy and security of sensitive and personal information, including health information. These
laws may be similar to or even more protective than HIPAA and other federal privacy laws. For example, the laws of the State of California,
are more restrictive than HIPAA. Where state laws are more protective than HIPAA, we must comply with the state laws we are subject to,
in addition to HIPAA. California passed the California Consumer Privacy Act or CCPA on June 28, 2018, which went into effect January
1, 2020. On November 3, 2020, the California Privacy Rights Act of 2020 (“CPRA”), which amends the CCPA and adds new privacy
protections that became effective on January 1, 2023, was enacted through a ballot initiative. While information we maintain that is
covered by HIPAA may be exempt from the CCPA, other records and information we maintain on our patients may be subject to the CCPA. In
certain cases, it may be necessary to modify our planned operations and procedures to comply with these more stringent state laws. Not
only may some of these state laws impose fines and penalties upon violators, but also some, unlike HIPAA, may afford private rights of
action to individuals who believe their personal information has been misused. In addition, state and federal privacy laws subject to
frequent change.
In
addition to HIPAA and state health information privacy laws, we may be subject to other state and federal privacy laws, including laws
that prohibit unfair privacy and security practices and deceptive statements about privacy and security, laws that place specific requirements
on certain types of activities, such as data security and texting, and laws requiring holders of personal information to maintain safeguards
and to take certain actions in response to a data breach.
Foreign
data protection, privacy, and other laws and regulations are often more restrictive than those in the U.S. The E.U., for example, traditionally
has imposed stricter obligations under its laws and regulations relating to privacy, data protection and consumer protection than the
U.S. In May 2018, the General Data Protection Regulation (the “GDPR”), which governs data practices and privacy in the E.U.,
became effective and replaced the data protection laws of the individual member states. GDPR requires companies to meet stringent requirements
regarding the handling of personal data of individuals in the E.U. These more stringent requirements include expanded disclosures to
inform members about how we may use their personal data, increased controls on profiling members, and increased rights for members to
access, control and delete their personal data. In addition, there are mandatory data breach notification requirements. The law also
includes significant penalties for non-compliance, which may result in monetary penalties of up to 20 million Euros or 4% of a company’s
worldwide turnover, whichever is higher. GDPR and other similar regulations require companies to give specific types of notice and informed
consent is required for the placement of a cookie or similar technologies on a user’s device for online tracking for behavioral
advertising and other purposes and for direct electronic marketing, and the GDPR also imposes additional conditions in order to satisfy
such consent, such as a prohibition on pre-checked consents. It remains unclear how the U.K. data protection laws or regulations will
develop in the medium to longer term and how data transfer to the U.K. from the E.U. will be regulated. Outside of the E.U., there are
many other countries with data protection laws, and new countries are adopting data protection legislation with increasing frequency.
Many of these laws may require consent from individuals for the use of data for various purposes, including marketing, which may reduce
our ability to market our products.
There
is no harmonized approach to these laws and regulations globally. Consequently, we increase our risk of non-compliance with applicable
foreign data protection laws and regulations when we expand internationally. We may need to change and limit the way we use personal
information in operating our business and may have difficulty maintaining a single operating model that is compliant. Compliance with
such laws and regulations will result in additional costs and may necessitate changes to our business practices and divergent operating
models, limit the effectiveness of our marketing activities, adversely affect our business, results of operations, and financial condition,
and subject us to additional liabilities.
Environmental
Matters
We
have no material expenditures for compliance with Federal, State or local provisions regulating the discharge of materials into the environment,
or otherwise relating to the protection of the environment.
Employees
As
of August 14, 2023, we had 3 full-time employees and 1 part-time employee.
Company
Information
Our
principal executive offices are located at Shatner Street 3, Jerusalem, Israel, and our telephone number is +972 (0)74-700-6700. Our
web page address is www.aerodentis.com. References to our website address do not constitute incorporation by reference of the information
contained on the website, and the information contained on the website is not part of this document or any other document that we file
with or furnish to the SEC.
Item 1A.
Risk Factors.
Our
business and an investment in our securities are subject to a variety of risks. The following risk factors describe the most significant
events, facts or circumstances that we believe could have a material adverse effect upon our business, financial condition, results of
operations, ability to implement our business plan, and the price at which our common stock is quoted on the OTC Pink Market. Many of
these events are outside of our control. The risks described below are not the only ones facing our Company. Additional risks not presently
known to us or that we currently believe are immaterial may also impair our business operations. If any of these risks actually occur,
our business, financial condition or results of operation may be materially adversely affected. In such case investors in our securities
could lose all or part of their investment.
Risks
Related to Our Capital Requirements and Financing
Our
financial statements have been prepared on a going concern basis; we must raise additional capital to fund our operations in order to
continue as a going concern.
In
its report dated April 1, 2024, Barzily & Co., our independent registered public accounting firm, expressed substantial doubt
about our ability to continue as a going concern as we have suffered recurring losses from operations and have insufficient liquidity
to fund our future operations. If we are unable to improve our liquidity position, we may not be able to continue as a going concern.
The accompanying financial statements do not include any adjustments that might result if we are unable to continue as a going concern
and, therefore, be required to realize our assets and discharge our liabilities other than in the normal course of business which could
cause investors to suffer the loss of all or a substantial portion of their investment. As of December 31, 2023, we had approximately
$3.3 million of cash. In order to have sufficient cash to fund our operations in the future, we will need to raise additional equity
or debt capital and cannot provide any assurance that we will be successful in doing so. If are unable to raise sufficient capital to
fund our operations, we may need to delay, reduce or eliminate certain research and development programs or other operations, sell some
or all of our assets or merge with another entity.
Macroeconomic
and External Risks
We
conduct our operations in Israel. Conditions in Israel, including the recent attack by Hamas and other terrorist organizations from the
Gaza Strip and Israel’s war against them, may affect our operations.
Because
our wholly-owned subsidiary is incorporated under the laws of the state of Israel, all of our operations are conducted in Israel, and
all of our employees and management personnel are located in Israel, our business and operations are directly affected by economic, political,
geopolitical and military conditions in Israel. Since the establishment of the State of Israel in 1948, a number of armed conflicts have
occurred between Israel and its neighboring countries and terrorist organizations active in the region. These conflicts have involved
missile strikes, hostile infiltrations and terrorism against civilian targets in various parts of Israel, which have negatively affected
business conditions in Israel.
In
October 2023, Hamas terrorists infiltrated Israel’s southern border from the Gaza Strip and conducted a series of attacks on civilian
and military targets. Hamas also launched extensive rocket attacks on Israeli population and industrial centers located along Israel’s
border with the Gaza Strip and in other areas within the State of Israel. Following the attack, Israel’s security cabinet declared
war against Hamas and a military campaign against these terrorist organizations commenced in parallel to their continued rocket and terror
attacks. Moreover, the clash between Israel and Hezbollah in Lebanon may escalate in the future into a greater regional conflict.
Any
hostilities involving Israel, or the interruption or curtailment of trade within Israel or between Israel and its trading partners could
adversely affect our operations and results of operations and could make it more difficult for us to raise capital. Parties with whom
we may do business have sometimes declined to travel to Israel during periods of heightened unrest or tension, forcing us to make alternative
arrangements when necessary. The conflict situation in Israel could cause situations where medical product certifying or auditing bodies
could not be able to visit manufacturing facilities of our subcontractors in Israel in order to review our certifications or clearances,
thus possibly leading to temporary suspensions or even cancellations of our product clearances or certifications. The conflict situation
in Israel could also result in parties with whom we have agreements involving performance in Israel claiming that they are not obligated
to perform their commitments under those agreements pursuant to force majeure provisions in such agreements.
There
have been travel advisories imposed as related to travel to Israel, and restriction on travel, or delays and disruptions as related to
imports and exports may be imposed in the future. An inability to receive supplies and materials, shortages of materials or difficulties
in procuring our materials, among others, may adversely impact our ability to commercialize and manufacture our product candidates and
products in a timely manner. This could cause a number of delays and/or issues for our operations, including delay of the review of our
product candidates by regulatory agencies, which in turn would have a material adverse impact on our ability to commercialize our product
candidates.
The
Israel Defense Force (the “IDF”), the national military of Israel, is a conscripted military service, subject to certain
exceptions. Several employees of our vendors are subject to military service in the IDF and have been and may be called to serve. It
is possible that there will be further military reserve duty call-ups in the future, which may affect our business due to a shortage
of skilled labor and loss of institutional knowledge, and necessary mitigation measures we may take to respond to a decrease in labor
availability, such as overtime and third-party outsourcing, for example, which may have unintended negative effects and adversely impact
our results of operations, liquidity or cash flows.
It
is currently not possible to predict the duration or severity of the ongoing conflict or its effects on our business, operations and
financial conditions. The ongoing conflict is rapidly evolving and developing, and could disrupt our business and operations, interrupt
our sources and availability of supply and hamper our ability to raise additional funds or sell our securities, among others.
Our
operations and financial performance depend on global and regional economic conditions. Inflation, fluctuations in currency exchange
rates, changes in consumer confidence and demand, and weakness in general economic conditions and threats, or actual recessions, could
materially affect our business, results of operations, and financial condition.
Macroeconomic
conditions impact consumer confidence and discretionary spending, which could adversely affect demand for any products we bring to market.
Consumer spending habits are affected by, among other things, inflation, fluctuations in currency exchange rates, weakness in general
economic conditions, threats or actual recessions, pandemics, wars and military actions, levels of employment, wages, debt obligations,
discretionary income, interest rates, volatility in capital, and consumer confidence and perceptions of current and future economic conditions.
Changes and uncertainty can, among other things, reduce or shift spending away from elective treatments and procedures, drive patients
to purchase orthodontic treatments that may cost less than our treatment options, result in a decrease in the number of overall orthodontic
and dental case starts, reduce patient traffic in dentists’ offices or reduce demand for dental services generally. Further, decreased
demand for dental services can cause dentists and labs to postpone investments in capital equipment, such as intraoral scanners and CAD/CAM
equipment and software. The recent declines in, or uncertain economic outlooks for, the U.S., European and certain other international
economies has and may continue to adversely affect consumer and dental practice spending. The increase in the cost of fuel and energy,
food and other essential items along with climbing interest rates could reduce consumers’ disposable income, resulting in less
discretionary spending for products like ours. Decreases in disposable income and discretionary spending or change in consumer confidence
and spending habits may adversely affect our revenues and operating results.
Inflation
continues to adversely impact spending and trade activities and we are unable to predict the impacts of higher inflation on global and
regional economies. Higher inflation has also increased domestic and international shipping costs, raw material prices, and labor rates,
which could adversely impact the costs of producing, procuring and shipping any products we bring to market. If similar trends continue
once we begin marketing our Platform, our ability to recover these cost increases through price increases may have limited effectiveness,
resulting in downward pressure on our operating results. Attempts to offset cost increases with price increases could reduce sales, increase
customer dissatisfaction or otherwise harm our reputation. Further, we are unable to predict the impact of efforts by central banks and
federal, state and local governments to combat elevated levels of inflation. If their efforts to reduce inflation are too aggressive,
they may lead to a recession. Alternatively, if they are insufficient or are not sustained long enough to lower inflation to more acceptable
levels, consumer spending may be adversely impacted for a prolonged period of time. Any of these events could materially affect our business
and operating results.
Our
business could be impacted by major public health issues, including pandemics such as the spread of COVID-19.
Major
public health issues, including pandemics such as the spread of COVID-19, could in the future materially affect our business due to their
impact on the global economy and regional economies, demand for consumer products, the imposition or removal of public safety measures.
Public health concerns may also limit the movement of products between regions, disrupt or delay supply chains and sales and distribution
channels, resulting in interruptions of the supply of products.
COVID-19
has created significant, widespread and unprecedented volatility, uncertainty, and economic instability, disrupting broad aspects of
global and regional economies. Many of these effects continue to varying degree as variants of COVID-19 and outbreaks globally or regionally
continue to harm recovering consumer confidence. As a result of outbreaks of COVID-19 and its variants, consumer demand and doctor availability
has been inconsistent and difficult to predict. The effects of the pandemic continue to linger and evolve and we cannot predict future
direct and ancillary impacts on our business or results of operations, although they may be material to our business as well as economic
activity generally.
Our
business could be impacted by political events, trade and other international disputes, war, and terrorism, including the military conflict
between Russia and Ukraine.
Political
events, trade and other international disputes, war, and terrorism could harm or disrupt international commerce and the global economy
and could have a material effect on our business as well as our potential customers, suppliers, contract manufacturers, distributors,
and other business partners.
Political
events, trade and other international disputes, wars, and terrorism can lead to unexpected tariffs or trade restrictions, which could
adversely impact our business. Tariffs could increase the cost of our products and the components and raw materials to make them. Once
we begin marketing our products, these increased costs could adversely impact our gross margin and make our products less competitive
or reduce demand. Countries could also adopt other measures, such as controls on imports or exports of goods, technology or data, that
could adversely impact our operations and supply chain and limit our ability to offer products and services. These measures could require
us to take various actions, including changing suppliers or restructuring business relationships. Complying with new or changed trade
restrictions is expensive, time-consuming and disruptive to our operations. Such restrictions can be announced with little or no advance
notice and we may be unable to effectively mitigate the adverse impacts of such measures. If disputes and conflicts escalate in the future,
actions by governments in response could be significantly more severe and restrictive and could materially affect our business.
Political
unrest, threats, tensions, actions and responses to any social, economic, business, geopolitical, military, terrorism, or acts of war
involving key commercial, development or manufacturing markets such as China, Mexico, Israel, Europe, or other countries could materially
impact any international operations we undertake. For example, our employees in Israel could be obligated to perform annual reserve duty
in the Israeli military and be called for additional active duty under emergency circumstances. If any of these events or conditions
occur, the impact on us, our employees and potential customers is uncertain, particularly if emergency circumstances, armed conflicts
or an escalation in political instability or violence disrupts our product development, data or information exchange, payroll or banking
operations, product or materials shipping by us or our suppliers and other unanticipated business disruptions, interruptions and limitations
in telecommunication services or critical systems or applications reliant on a stable and uninterrupted communications infrastructure.
U.S.
and global markets are experiencing volatility and disruption following the escalation of geopolitical tensions and the start of the
military conflict between Russia and Ukraine. On February 24, 2022, a full-scale military invasion of Ukraine by Russian troops was reported.
In response to the military conflict, the United States and other North Atlantic Treaty Organization member states, as well as non-member
states, announced targeted economic sanctions on Russia, including certain Russian citizens and enterprises, and the continuation of
the conflict may trigger additional economic and other sanctions. The potential impacts of the conflict and related sanctions could include
supply chain and logistics disruptions, macro financial impacts resulting from the exclusion of Russian financial institutions from the
global banking system, volatility in foreign exchange rates and interest rates, inflationary pressures on raw materials and energy and
heightened cybersecurity threats. We have no way to predict the progress or outcome of the conflict in Ukraine or the reactions by governments,
businesses or consumers. A prolonged conflict, intensified military activities or more extensive sanctions impacting the region and the
resulting economic impact could have a material effect on our business, results of operations, financial condition, liquidity, growth
prospects and business outlook.
Our
operations may be impacted by natural disasters, which may become more frequent or severe as a result of climate change and may adversely
impact our business and operating results as well as those of our potential customers and suppliers.
Natural
disasters can impact us and our potential customers, as well as suppliers critical to our operations. Natural disasters include earthquakes,
tsunamis, floods, droughts, hurricanes, wildfires, and other extreme weather conditions that can cause deaths, injuries, and critical
health crises, power outages, restrictions and shortages of food, water, shelter, and medical supplies, telecommunications failures,
materials scarcity, price volatility and other ramifications. Climate change is likely to increase both the frequency and severity of
natural disasters and, consequently, risks to our business and operations.
We
anticipate that our digital dental modeling and certain of our customer-facing operations will primarily be processed in our facilities
located in Israel. Similarly, a significant portion of our research and development activities is located in Israel. If there is a natural
disaster in the region, our employees could be impacted, our research could be lost, and our ability to create treatment plans, respond
to customer inquiries or manufacture and ship our aligners or intraoral scanners could be compromised, which could result in our future
customers experiencing significant product and services delays.
The
effects of climate change on regional and global economies could change the supply, demand or availability of sources of energy or other
resources material to our products and operations and affect the availability or cost of natural resources and goods and services on
which we and our suppliers rely.
Business
and Industry Risks
We
are in the development stage, are not generating revenues and have no operating history as a manufacturer and distributor of orthodontic
medical devices or platforms for consumer use.
We
are in the development stage and face all of the risks and uncertainties associated with a new and unproven business. Our future is based
on an unproven business plan with no historical facts to support projections and assumptions. We were founded in 2005 and have no operating
history as a manufacturer and distributor of orthodontic medical devices or platforms to the consumer public. We are not currently generating
revenues and do not expect to generate revenue until we have successfully completed the development and testing of our Platform. Investors
should understand that an investment in a start-up business is significantly riskier than an investment in a business with any significant
operating history. There can be no assurance that we will ever achieve revenues or profitability. Our operations are subject to all of
the risks inherent in the establishment of a new business enterprise. The likelihood of our success must be considered in light of the
problems, expenses, difficulties, complications and delays frequently encountered in connection with the formation of a pre-revenue business.
Our lack of a significant and relevant operating history makes it difficult to manage operations and predict future operating results.
Our
products and technologies may not be accepted by the intended commercial consumers of our products, which could harm our future financial
performance.
There
can be no assurance that our Platform will achieve wide acceptance by intended consumers and/or market acceptance generally. The degree
of market acceptance for our Platform will also depend upon a number of factors, including the receipt and timing of regulatory approvals,
if any, and the establishment and demonstration of the ability of our proposed device to provide the level of confidence and independence
in an efficient manner and at a reasonable cost. Our failure to develop a commercial product to compete successfully with existing orthodontic
treatments could delay, limit, or prevent market acceptance. There can be no assurance that the public will believe that our Platform
is necessary or that the dental industry will actively pursue our product. Long-term market acceptance of our Platform will depend, in
part, on the capabilities, operating features and price of our products and technologies as compared to those of other available products
and services. As a result, there can be no assurance that our Platform will be able to achieve market penetration, revenue growth or
profitability.
We
expect continued operating losses and cannot be certain of our future profitability.
We
have incurred net operating losses since inception. For the years ended December 31, 2023 and 2022, we incurred net losses of $3.5 million
and $1.7 million, respectively. From inception through the present, we have spent significant funds in organizational and start up activities,
to recruit key managers and employees, to develop our Platform, and for research and development.
We
expect to continue incurring net operating losses in the foreseeable future as we increase expenditures for the development and marketing
of the Platform. The time required for us to become profitable is uncertain, and there can be no assurance that we will achieve profitability
on a sustained basis, if at all. As a result of our limited operating history, we have neither internal nor industry-based historical
financial data for any significant period of time upon which to project revenues or base planned operating expenses. We expect that our
results of operations may also fluctuate significantly in the future as a result of a variety of factors, including: the ability to enter
into resale agreements with dental professionals, the ability to effectively market to the public, the ease of use of the Platform by
consumers and dental professionals, intense competition from existing and new companies, retain and motivate qualified personnel, specific
economic conditions in the aligner/consumer orthodontic market, general economic conditions; and other factors.
We
may be unable to raise additional capital, which could harm our ability to compete.
We
expect to expend significant capital to establish our brand, build manufacturing infrastructure, and develop both product and process
technology. These initiatives may require us to raise additional capital over the next few years. We may consume available resources
more rapidly than anticipated and we may not be able to raise additional funds when needed or on acceptable terms. If we raise additional
funds through further issuances of equity or convertible debt securities, our existing stockholders could suffer significant dilution,
and any new equity securities we issue could have rights, preferences, and privileges superior to those of holders of our common stock.
In
connection with the Private Placement, we granted the Private Placement Investors a right to participate in future financings, until
the second anniversary of the closing of the Private Placement, that involve the issuance of our common stock or common stock equivalents
for cash consideration. Further, the Securities Purchase Agreement entered into in connection with the Private Placement (the “Securities
Purchase Agreement”) contains “most favored nation” provisions, which may require future amendments to the terms of
the Private Placement to give Private Placement Investors the benefit of more favorable terms governing certain future issuances of our
common stock or common stock equivalents. Such participation right and “most favored nation” provisions may restrict our
ability to secure future financings unless the Private Placement Investors waive their right to participate, the persons providing such
financing accept the participation of the Private Placement Investors or the Private Placement Investors waive their rights under “most
favored nation” provisions, respectively. If we are unable to obtain adequate financing or financing on terms satisfactory to us,
when we require it, our ability to continue to pursue our business objectives and to respond to business opportunities, challenges, or
unforeseen circumstances could be significantly limited, and our business, operating results, financial condition, and prospects could
be materially adversely affected.
We
will depend on the acceptance of teledentistry and a demand for correcting tooth alignment.
Continued
and widespread market acceptance of teledentistry by consumers is critical to our future success. Delivery of our Platform via a teledentistry
model will represent a change from traditional orthodontic treatment, which requires in person visits, and consumers may be reluctant
to accept this model or may not find it preferable to traditional treatment. In addition, consumers may not respond to our direct marketing
campaigns, or we may be unsuccessful in reaching our target audience, particularly in foreign jurisdictions where our advertising may
be more heavily regulated. If consumers prove unwilling to adopt our teledentistry model as rapidly or in the numbers that we anticipate,
our operating results could be materially harmed.
Consumer
spending habits are affected by, among other things, prevailing economic conditions, inflationary factors, levels of employment, salaries
and wage rates, consumer confidence, and consumer perception of economic conditions. In many markets, dental and orthodontic reimbursement
is largely out of pocket for the consumer and, as result, utilization rates can vary significantly depending on economic growth. A general
slowdown in the U.S. economy and certain international economies may result in, among other things, a decrease in the number of overall
orthodontic case starts, a reduction in consumer spending on elective or higher value procedures, or a reduction in demand for dental
and orthodontic services generally, each of which would have an adverse effect on our sales, if any, and operating results. Inflation
and weakness in the global economy result in a challenging environment for selling dental and orthodontic technologies. If there is a
reduction in consumer demand for orthodontic treatment generally, or if consumers choose to use a competitive product rather than our
Platform for any reason, our business, results of operations, and financial condition could be materially harmed.
Adverse
changes in, or interpretations of, laws, rules, and regulations governing remote healthcare and the practice of dentistry could have
a material adverse effect on our business.
Our
current business model is dependent, in part, on current laws, rules, and regulations governing remote healthcare and the practice of
dentistry. If changes in laws, rules, regulations, or their interpretations are inconsistent with our current business model, we would
need to adapt our business model accordingly, and our operations in certain jurisdictions may be disrupted, which could have a material
adverse effect on our business, results of operations, and financial condition.
Our
net revenues will depend primarily on our Platform and any decline in sales or average selling price of our Platform may adversely affect
net revenues, gross margin and net income.
Our
net revenues will be largely dependent on sales of our Platform, making widespread acceptance of our Platform by dental professionals
and consumers critical to our future success. Our operating results could be harmed if:
| ● | dental
professionals experience a reduction in consumer demand for orthodontic services; |
| ● | consumers
are unwilling to adopt system treatment offered by our Platform as rapidly or in the volumes we anticipate and at the prices offered; |
| ● | dental
professionals choose to continue using wires and brackets or competitive products rather than our Platform or the rates at which they
utilize our Platform fail to increase or increase as rapidly as anticipated after we commence sales; or |
| ● | if
the average selling price of our products declines after we commence sales. |
The
average selling prices of our Platform could be influenced by numerous factors, including the type and timing of products sold and foreign
exchange rates.
Our
average selling prices for our Platform may be adversely affected in the future after we commence sales if:
| ● | we
introduce new or change existing promotions, general or volume-based discount programs, product or services bundles, or consumer rebate
programs; |
| ● | participation
in any promotions or programs unexpectedly increases or decreases or drives demand in unexpected and material ways; |
| ● | our
geographic, channel, or product mix shifts to lower priced products or to products that have a higher percentage of deferred revenue; |
| ● | we
decrease prices on one or more products or services in response to increasing competitive pricing pressures; |
| ● | we
introduce new or change existing products or services, or modify how we market or sell any of our new or existing products or services;
or |
| ● | estimates
used in the calculation of deferred revenue differ from actual average selling prices. |
If
our average selling prices decline after we commence sales, our net revenues, gross margin and net income may be adversely affected.
We
will face competition from large internationally established aligner companies whose products have been widely accepted.
The
dental industry is in a period of immense and rapid digital transformation involving products, technologies, distribution channels and
business models. Once we commence marketing our Platform, we will face competition in the market for our Platform from the clear aligners
market, and we expect competition from existing competitors and new companies that may enter the market or introduce new technologies
in the future.
We
expect to compete with a handful of large aligner companies including Align Technologies, SmileDirectClub, Dentsply Sirona, 3M™
Clarity™ Aligners, and Straumann Group. We expect some additional competition from other teledentistry solutions, and from new
entrants into the orthodontic supply or clear aligner markets. Some of these competitors may have greater resources as well as the ability
to leverage existing channels in the dental market to compete directly with us. In addition, we may also face future competition from
companies that introduce new technologies. We may be unable to compete with these competitors, and one or more of these competitors may
render our technology obsolete or economically unattractive.
Our
business model depends on being able to reach consumers to raise brand awareness and encourage downloading our smartphone application,
which may not prove successful or may become less effective or more costly to maintain in the long term.
There
is no assurance our campaigns will achieve the returns on advertising spend desired, increase brand or product awareness sufficiently
or generate goodwill and positive reputational goals. Moreover, should any entity or individual endorsing us or our products take actions,
make or publish statements in support of, or lend support to events or causes which may be perceived by a portion of society negatively,
our sponsorships or support of these entities or individuals may be questioned, boycotts of our products announced, and our reputation
may be harmed, any of which could have a material effect on our gross margin and business overall.
In
addition, various countries prohibit certain types of marketing activities. For example, some countries restrict direct to consumer advertising
of medical devices. We could run afoul of restrictions and be ordered to stop certain marketing activities. Moreover, competitors do
not always follow these restrictions, creating an unfair advantage and making it more difficult and costly for us to compete.
Future
sales of our Platform may depend on our customers’ ability to obtain reimbursement from third-party payors, such as insurance carriers.
Future
sales of our Platform may depend on our customers’ ability to obtain reimbursement from third-party payors, such as insurance carriers.
Where such insurance or third-party reimbursement becomes available in the future, any reduction in insurance or other third-party payor
reimbursement for our Platform may cause negative price pressure, which would reduce our revenues. Without a corresponding reduction
in the cost to produce such products, the result would be a reduction in our overall gross profit. Similarly, any increase in the cost
of such products would reduce our overall gross profit unless there was a corresponding increase in third-party payor reimbursement.
We face additional risks associated with obtaining and maintaining coverage and securing reimbursement from foreign health care payment
systems on a timely basis or at all. Failure by our patients to obtain or maintain coverage or to secure adequate reimbursement for our
treatment by third-party payors could have an adverse effect on our business, results of operations, and financial condition.
Our
growth and future success may depend on our ability to enhance our Platform or to develop, obtain regulatory clearance for, successfully
introduce, and achieve market acceptance of new products and services.
We
intend to continually improve and enhance our Platform and/or develop and introduce new products and services in order to maintain or
increase our sales. The success of new or enhanced products and services may depend on a number of factors, including anticipating and
effectively addressing consumer preferences and demand, the success of our sales and marketing efforts, innovation and timely and successful
research and development, obtaining necessary regulatory clearances, anticipating and responding to competing products and technological
innovations, adequately protecting our intellectual property rights, effective forecasting and management of product demand, effective
management of manufacturing and supply costs, and the quality of our products. There can be no assurance that we will be able to successfully
develop and introduce new or enhanced products and services. Even if new or enhanced products and services are successfully introduced,
they may not rapidly gain market share and acceptance.
The
development of new products and services in the dental and orthodontic industry can be complex and costly. We could experience delays
in the development and introduction of new and enhanced products and services, including delays in obtaining any necessary regulatory
clearances. Unanticipated problems in developing products and services could also divert substantial research and development resources,
which may impair our ability to develop new products and services and enhancements of existing products and services, and could substantially
increase our costs. If new or enhanced product and service introductions are delayed or not successful, we may not be able to achieve
an acceptable return, if any, on our research and development efforts, and our business may be adversely affected. Even if we successfully
innovate and develop new or enhanced products and services, we may incur substantial costs in doing so and our profitability may suffer.
Any
failure in our ability to successfully develop, introduce, or achieve market acceptance of new or enhanced products and services, or
any problems in the design or quality of any products or services we develop, could have a material adverse effect on our business, results
of operations, and financial condition.
Operational
Risks
Business
disruptions could seriously harm our financial condition.
The
occurrence of any material or prolonged business disruptions, whether internal or at key suppliers, could harm our business and results
of operations, result in material losses, seriously harm our development efforts and future revenues, profitability and financial condition,
adversely affect our competitive position, increase our costs and expenses, and require substantial expenditures and recovery time in
order to fully resume operations.
When
business disruptions occur, they may, individually or in the aggregate, affect our ability to continue critical research and development
and could cause production delays or limitations, create adverse effects on distributors, disrupt supply chains, result in shipping and
distribution disruptions and reduce the availability of or access to one or more facilities.
We
are subject to operating risks, including excess or constrained capacity and operational inefficiencies, which could adversely affect
our results of operations.
We
are subject to operating risks, including excess or constrained capacity and pressure on our internal systems, personnel and suppliers.
In order to manage current and anticipated future operations effectively, we must continually implement and improve our operational,
financial and management information systems, hire, train, motivate, manage and retain employees, and ensure our suppliers remain diverse
and capable of meeting growing demand for the systems, raw materials, parts and components essential to the manufacture and delivery
of our products. We may be unable to balance near-term efforts to meet existing demand with future customer demand, including adding
personnel, creating scalable, secure and robust systems and operations, and automating processes needed for long term efficiencies. Any
such failure could have a material impact on our business, operations and prospects.
Our
products and information technology systems are critical to our business. Issues with product development or enhancements, IT system
integration, implementation, updates and upgrades could disrupt our operations and have a material impact on our business and operating
results.
We
rely on the efficient, uninterrupted and secure operation of our IT systems and are dependent on key third-party software embedded in
our products and IT systems as well as third-party hosted IT systems to support our operations. All software and IT systems are vulnerable
to damage, cyber attacks or interruption from a variety of sources. To effectively manage and improve our operations, our IT systems
and applications require an ongoing commitment of significant expenditures and resources to maintain, protect, upgrade, enhance and restore
existing systems and develop new systems to keep pace with continuing changes in information processing technology, evolving industry
and regulatory standards, increasingly sophisticated cyber threats, and changing consumer preferences. Failure to adequately protect
and maintain the integrity of our products and IT systems may result in a material effect on our financial position, results of operations
and cash flows.
We
plan to continuously upgrade and issue new releases of our products and customer-facing software applications, upon which customer-facing,
manufacturing and treatment planning operations depend. Software applications and products containing software frequently contain errors
or defects, especially when first introduced or when new versions are released. Additionally, the third-party software integrated into
or interoperable with our products and services will routinely reach end of life, and as a consequence, may be exposed to additional
vulnerabilities, including increased security risks, errors and malfunctions that may be irreparable or difficult to repair. The discovery
of a defect, error or security vulnerability in our products, software applications or IT systems, incompatibility with future customers’
computer operating systems and hardware configurations with a new release or upgraded version or the failure of our products or primary
IT systems may cause adverse consequences, including: delay or loss of revenues, significant remediation costs, delay in market acceptance,
loss of data, disclosure of financial, health or other personal information of any customers or patients, product recalls, damage to
our reputation, or increased service costs, any of which could have a material effect on our business, financial condition or results
of our operations and the operations of our potential customers or our business partners.
Our
success depends on key executive personnel, vendors, and relationships with key dental professionals and organizations.
Our
success depends on the expertise and experience of our key personnel, including our CEO, CTO and top management. If we lose the services
of any of these key personnel, our business and prospects could be materially and adversely affected. In addition, since the research
and development of the Platform is mainly performed by outsourced third party vendors, although we could transfer the materials to other
vendors, an interruption of service could materially and adversely affect us.
Our
success depends largely on the talents and efforts of our personnel, and if we are unable to attract, motivate, train or retain our personnel,
it may be more difficult to grow effectively and pursue our strategic priorities, and could materially effect on our results of operations.
In addition, our market acceptance and success are dependent on attracting key orthodontists, dentists and dental organization to work
in conjunction with us to educate the consumer market on our Platform.
There
is no assurance that we will be able to attract and retain relationships with these key dental professionals to validate our Platform.
The orthodontics industry is inundated with new products and services which demand the attention of practitioners, who do not have adequate
time or motivation to explore new treatments for their patients or business opportunities of their practices.
Additionally,
facilitating seamless leadership transitions for key positions is a critical factor in sustaining the culture and maintaining the success
of our organization. If our succession planning efforts are not effective, it could adversely impact our business. We continue to assess
the key personnel that we believe are essential to our long-term success, as future organizational changes could also cause our employee
attrition rate to increase. If we fail to effectively manage any organizational or strategic changes, our financial condition, results
of operations, and reputation, as well as our ability to successfully attract, motivate and retain key employees, could be harmed.
Legal,
Regulatory and Compliance Risks
Complying
with regulations enforced by FDA and other regulatory authorities is expensive and time consuming, and failure to comply could result
in substantial penalties.
Our
products (including the currently cleared version, as well as the next generation Platform for which we have not yet submitted the requisite
510(k) application to FDA) are considered medical devices and, accordingly, are subject to rigorous regulation by government agencies
in the U.S. and other countries in which we intend to sell our products. Compliance with these rigorous regulations will affect capital
expenditures, earnings and our competitive position. These regulations vary from country to country but cover, among other things, the
following activities with respect to medical devices:
| ● | design,
development and manufacturing; |
| ● | testing,
labeling, content and language of instructions for use and storage; |
| ● | product
storage and safety; |
| ● | marketing,
sales and distribution; |
| ● | pre-market
clearance and approval; |
| ● | record
keeping procedures; |
| ● | advertising
and promotion; |
| ● | recalls
and field safety corrective actions; |
| ● | post-market
surveillance; |
| ● | post-market
approval studies; and |
| ● | product
import and export. |
The
regulations to which we are subject are complex. Regulatory changes could result in restrictions on our ability to carry on or expand
our operations, higher than anticipated costs, or lower than anticipated sales. Our failure to comply with applicable regulatory requirements
could result in enforcement action by FDA or state agencies, which may include any of the following sanctions:
| ● | warning
letters, fines, injunctions, consent decrees, and civil penalties; |
| ● | repair,
replacement, refunds, recall, or seizure of our products; |
| ● | operating
restrictions or partial suspension or total shutdown of production; |
| ● | refusing
our requests for 510(k) clearance or pre-market approval of new products, new intended uses, or modifications to existing products; |
| ● | withdrawing
clearance or pre-market approvals that have already been granted; and |
If
any of these events were to occur, they could harm our business.
We
may not receive the necessary authorizations to market our Platform or any future new products, and any failure to timely do so may adversely
affect our ability to grow our business.
Before
we can sell a new medical device in the U.S., or market a new use of, new claim for, or significant modification to a legally marketed
device, we must first obtain either FDA 510(k) clearance or approval, unless an exemption applies. In the 510(k) clearance process, before
a device may be marketed, the applicant must submit a premarket notification to FDA under Section 510(k) of the FD&C Act, and FDA
must determine that a proposed device is “substantially equivalent” to a legally-marketed “predicate” device.
To be “substantially equivalent,” the proposed device must have the same intended use as the predicate device, and either
have the same technological characteristics as the predicate device or have different technological characteristics, not raise different
questions of safety or effectiveness than the predicate device, and be as safe and as effective as the predicate device. The 510(k) clearance
process can be expensive and uncertain and can take from three to 12 months, but may last significantly longer. Clinical data may be
required in connection with an application for 510(k) clearance. Furthermore, even if we are granted regulatory clearances or approvals,
they may include limitations on the indications for use or intended uses of the device, which may limit the market for the device.
Our
first generation Aerodentis System is a Class II medical device, which was cleared by FDA for commercialization in the U.S. pursuant
to the 510(k) notification process for movement and alignment of teeth during orthodontic treatment of malocclusion in April 2020. We
are preparing to apply for 510(k) clearance for the updated version of the currently cleared device. Such updated Platform contains new
and/or different components than the original device, which is why a new 510(k) clearance is required prior to marketing the Platform
in the U.S. We have not yet filed a 510(k) submission for the Platform, and it has, thus, not been found by the FDA to be substantially
equivalent to the first generation Aerodentis System.
FDA
can delay, limit, or deny 510(k) clearance, or other approval or reclassification, of a device for many reasons, including:
| ● | we
may be unable to demonstrate to FDA’s satisfaction that the products or modifications are substantially equivalent to a proposed
predicate device or safe and effective for their intended uses; |
| ● | we
may be unable to demonstrate that the clinical and other benefits of the device outweigh the risks; and |
| ● | the
applicable regulatory authority may identify deficiencies in our submissions or in the facilities or processes of our third party contract
manufacturers. |
Any
delay or failure to obtain necessary regulatory clearances or approvals could harm our business. Once cleared for marketing in the U.S.,
if ever, to the extent we decide to market the Platform for any additional indications for use and/or make any material modifications
to any element of the device and/or the manufacturing or distribution thereof in the future, an additional 510(k) submission, and FDA
clearance thereof, will be required.
In
addition, FDA may change its policies, adopt additional regulations, revise existing regulations, or take other actions, or Congress
may enact different or additional statutory requirements, which may prevent or delay clearance of our future products under development
or impact our ability to modify our currently marketed products on a timely basis. Such policy, statutory, or regulatory changes could
impose additional requirements upon us that could delay our ability to obtain new 510(k) clearances, increase the costs of compliance,
or restrict our ability to maintain our current marketing authorizations.
We
received our European CE mark and ISO/MDSAP certification in 2019. In light of our ISO/MDSAP certification, we believe that we are in
substantial compliance with applicable E.U. regulations. We will also need to obtain regulatory approval in other foreign jurisdictions
in which we plan to market and sell our products. The time required to obtain registrations or approvals, if required by other countries,
may be longer than that required for FDA clearance, and requirements for such registrations, clearances, or approvals may significantly
differ from FDA requirements. If we modify our products, we may need to apply for additional regulatory approvals before we are permitted
to sell the modified product. In addition, we may not continue to meet the quality and safety standards required to maintain the authorizations
that we have received. If we are unable to maintain our authorizations in a particular country, we will no longer be able to sell the
applicable product in that country.
Failure
to comply with these rules, regulations, self-regulatory codes, circulars, and orders could result in significant civil and criminal
penalties and costs and could have a material adverse impact on our business. Also, these regulations may be interpreted or applied by
a prosecutorial, regulatory, or judicial authority in a manner that could require us to make changes in our operations or incur substantial
defense and settlement expenses. Even unsuccessful challenges by regulatory authorities or private relators could result in reputational
harm and the incurring of substantial costs. In addition, many of these laws are vague or indefinite and have not been interpreted by
the courts and have been subject to frequent modification and varied interpretation by prosecutorial and regulatory authorities, increasing
compliance risks.
Certain
modifications to our products may require new 510(k) clearance or other marketing authorizations.
Once
a medical device is permitted to be legally marketed in the U.S. pursuant to a 510(k) clearance, a manufacturer may be required to notify
FDA of certain modifications to the device. Manufacturers determine in the first instance whether a change to a product requires a new
premarket submission, but FDA may review any manufacturer’s decision.
While
our first generation Aerodentis System has received 510(k) clearance in 2020, we are preparing to apply for 510(k) clearance for the
updated components of our Platform, which must, then, be found by the FDA to be substantially equivalent to the Aerodentis System and,
thus, may not be lawfully marketed in the U.S. until FDA make a substantial equivalence determination and issues the requisite 510(k)
clearance for the updated Platform. Although the development of our Platform has been carefully monitored and documented by professionals
who are experienced in the FDA clearance process, there is no assurance that the FDA will agree that our Platform is substantially equivalent
to the Aerodentis System and allow our Platform to be marketed in the United States. The FDA may determine that the device is not substantially
equivalent and require a PMA or, more likely, a de novo reclassification, and/or require further information, such as additional
test data, including data from clinical studies, before it is able to make a determination regarding substantial equivalence. By requesting
additional information, the FDA can delay market introduction of our Platform. Delays in receipt of or failure to receive any necessary
510(k) clearance, de novo classification, or PMA, or the imposition of stringent restrictions for our Platform could have a material
adverse effect on our business, results of operations and financial condition.
In
the future, we may make other modifications to our products, including our Platform, and determine, based on our review of the applicable
FDA regulations and guidance, that in certain instances new 510(k) clearances or other premarket submissions are not required. If FDA
disagrees with our determinations, we may be subject to a wide range of enforcement actions, including, for example, a warning letter,
among other consequences, after which we will likely have to cease marketing the applicable modified product and/or to recall distributed
units of such modified product until we obtain the requisite clearance or approval.
Our
products must be manufactured in accordance with federal, state, and international regulations, and we could be forced to recall our
products or terminate production and/or face other regulatory enforcement actions if we fail to comply with these regulations.
The
methods used in, and the facilities used for, the manufacture of our products must comply with FDA’s Quality System Regulation
which is a complex regulatory scheme that covers the procedures and documentation of, among other requirements, the design, testing,
validation, verification, complaint handling, production, process controls, quality assurance, labeling, supplier evaluation, packaging,
handling, storage, distribution, installation, servicing, and shipping of medical devices. Furthermore, we are required to verify that
our suppliers maintain facilities, procedures, and operations that comply with our quality standards and applicable regulatory requirements.
FDA enforces the Quality System Regulation through, among other oversight methods, periodic announced or unannounced inspections of medical
device manufacturing facilities, which may include the facilities of contractors, suppliers, or contract manufacturing organizations.
Our products are also subject to similar state regulations as well as similar laws and regulations of foreign countries. Our failure
to comply with the Quality System Regulation or similar requirements could result in enforcement actions, sanctions, recalls, detentions,
seizures, or similar market actions with respect to our products, among other potential consequences. If any of these or other events
occur, there could be a negative impact on the supply of our products, our reputation could be harmed, we could be exposed to product
liability claims, and we could lose customers and suffer reduced revenue and increased costs.
Ongoing
changes in healthcare regulation could negatively affect our revenues, business and financial condition.
There
have been several proposed changes in the United States at the federal and state level for comprehensive reforms regarding the payment
for, the availability of and reimbursement for healthcare services. These proposals have ranged from fundamentally changing federal and
state healthcare reimbursement programs, including providing comprehensive healthcare coverage to the public under government-funded
programs, to minor modifications to existing programs. One example, among countless others, is the Patient Protection and Affordable
Care (the “Affordable Care Act”) which was the most significant Federal healthcare reform law enacted in the U.S. in recent
history. The Affordable Care Act has undergone substantial challenges and changes since its enactment in 2010, and numerous other federal
healthcare reform legislation, executive orders, and judicial rulings have been implemented in the years since, most of which have been
or are aimed at lowering healthcare costs in the U.S. To the extent any such reform measures or any future initiatives reduce reimbursement
or coverage eligibility or amount(s) for our Platform and/or any future products we may market in the U.S. (if any), our business may
be adversely affected.
Healthcare
reform initiatives will continue to be proposed and may reduce healthcare related funding in an effort. It is impossible to predict the
ultimate content and timing of any healthcare reform legislation and its resulting impact on us. If significant reforms are made to the
healthcare system in the United States, or in other jurisdictions, those reforms may increase our costs or otherwise negatively effect
on our business, results of operations, and financial condition.
On
April 5, 2017, the European Parliament passed the Medical Devices Regulation (Regulation 2017/745), which repeals and replaces the E.U.
Medical Device Directive and became effective on May 26, 2021. The Medical Devices Regulation, among other things, is intended to establish
a uniform, transparent, predictable, and sustainable regulatory framework across the EEA for medical devices and ensure a high level
of safety and health while supporting innovation. The new regulations, among other things:
| ● | strengthen
the rules on placing devices on the market and reinforce surveillance once they are available; |
| ● | establish
explicit provisions on manufacturers’ responsibilities for the follow-up of the quality, performance and safety of devices placed
on the market; |
| ● | improve
the traceability of medical devices throughout the supply chain to the end-user or patient through a unique identification number; |
| ● | set
up a central database to provide patients, healthcare professionals and the public with comprehensive information on products available
in the European Union; and |
| ● | strengthen
rules for the assessment of certain high-risk devices, such as implants, which may have to undergo an additional check by experts before
they are placed on the market. |
These
modifications may have an effect on the way we conduct our business in the EEA.
Any
change in the laws or regulations that govern the clearance and approval processes relating to our current, planned and future products
could make it more difficult and costly to obtain clearance or approval for new products or to produce, market and distribute existing
products. Significant delays in receiving clearance or approval or the failure to receive clearance or approval for our new products
would have an adverse effect on our ability to expand our business.
Our
products may cause or contribute to adverse medical events that we are required to report to FDA and other governmental authorities,
and if we fail to do so, we would be subject to sanctions that could harm our reputation, business, results of operations, and financial
condition. The discovery of serious safety issues with our products, or a recall of our products either voluntarily or at the direction
of FDA or another governmental authority, could have a negative impact on us.
We
are required to timely file various reports with FDA, including reports required by the medical device reporting regulations which require
us to report to FDA when we receive or become aware of information that reasonably suggests that one of our products may have caused
or contributed to a death or serious injury or malfunctioned in a way that, if the malfunction were to recur to the device or a similar
device that we market, could cause or contribute to a death or serious injury. If we fail to comply with our reporting obligations, FDA
or other governmental authorities could take action, including warning letters, untitled letters, administrative actions, criminal prosecution,
imposition of civil monetary penalties, revocation of our device clearance, seizure of our products, or delay in clearance of future
products. FDA and certain foreign regulatory bodies have the authority to require the recall of commercialized products under certain
circumstances.
A
government-mandated or voluntary recall by us could occur as a result of an unacceptable risk to health, component failures, malfunctions,
manufacturing defects, labeling or design deficiencies, packaging defects, or other deficiencies, or failures to comply with applicable
regulations. If we do not adequately address problems associated with our devices, we may face additional regulatory requirements or
enforcement action, including required new marketing authorizations, FDA warning letters, product seizure, injunctions, administrative
penalties, or civil or criminal proceedings.
We
may initiate voluntary withdrawals, removals, or corrections for our products in the future that we determine do not require notification
of FDA. If FDA disagrees with our determinations, it could require us to report those actions and we may be subject to enforcement action.
A future recall announcement or other corrective action could harm our financial results and reputation, potentially lead to product
liability claims against us, require the dedication of our time and capital, and negatively affect our sales.
In
addition, FDA’s and other regulatory authorities’ policies may change, and additional government regulations may be enacted
that could prevent, limit, or delay regulatory approval of our product candidates. For example, in November 2018, FDA announced that
it plans to develop proposals to drive manufacturers utilizing the 510(k) pathway toward the use of newer predicates. It is unclear the
extent to which any proposals, if adopted, could impose additional regulatory requirements on us that could delay our ability to obtain
new 510(k) clearances, increase the costs of compliance, or restrict our ability to maintain our current clearances.
We
also cannot predict the likelihood, nature, or extent of government regulation that may arise from future legislation or administrative
or executive action, either in the U.S. or abroad. For example, the Trump Administration previously enacted several executive actions
that could impose significant burdens on, or otherwise materially delay, FDA’s ability to engage in routine regulatory and oversight
activities. It is difficult to predict how these executive actions and executive actions that may be taken under the Biden Administration
may affect FDA’s ability to exercise its regulatory authority. If these executive actions impose constraints on FDA’s ability
to engage in oversight and implementation activities in the normal course, our business may be negatively impacted.
Changes
in internet regulations could adversely affect our business.
Laws,
rules, and regulations governing internet communications, advertising, and e-commerce are dynamic, and the extent of future government
regulation is uncertain. Federal and state regulations govern various aspects of our online business, including intellectual property
ownership and infringement, trade secrets, the distribution of electronic communications, marketing and advertising, user privacy and
data security, search engines, and internet tracking technologies. Future taxation on the use of the internet or e-commerce transactions
could also be imposed. Existing or future regulation or taxation could increase our operating expenses and expose us to significant liabilities.
Disruptions
at the FDA, other agencies or notified bodies caused by funding shortages or global health concerns could hinder their ability to hire,
retain, or deploy key leadership and other personnel, or otherwise prevent new or modified products from being developed, cleared or
approved, or commercialized in a timely manner, or at all, which could negatively impact our business.
The
ability of the FDA, other agencies and notified bodies to review and authorize or certify for marketing new products can be affected
by a variety of factors, including government budget and funding levels, statutory, regulatory and policy changes, agency’s or
notified body’s ability to hire and retain key personnel and accept the payment of user fees, and other events that may otherwise affect
the agency’s or notified body’s ability to perform routine functions. Average review times at the FDA and other agencies and notified
bodies have fluctuated in recent years as a result. In addition, government funding of other government agencies that fund research and
development activities is subject to the political process, which is inherently fluid and unpredictable. Disruptions at the FDA, other
agencies and notified bodies may also slow the time necessary for new medical devices or modifications to be reviewed and/or cleared,
approved or certified by necessary agencies or notified bodies, which would adversely affect our business. For example, over the last
several years, the U.S. government has shut down several times and certain regulatory agencies, such as the FDA, have had to furlough
critical FDA employees and stop critical activities.
Separately, in response to
the global COVID-19 pandemic, the FDA postponed most inspections of domestic and foreign manufacturing facilities at various points. Even
though the FDA has since resumed standard inspection operations of domestic facilities where feasible, the FDA has continued to monitor
and implement changes to its inspectional activities to ensure the safety of its employees and those of the firms it regulates as it adapts
to the evolving COVID-19 pandemic, and any resurgence of the virus or emergence of new variants may lead to further inspectional delays.
Regulatory authorities outside the United States may adopt similar policy measures in response to the COVID-19 pandemic. If a prolonged
government shutdown occurs, or if global health concerns continue to prevent the FDA or other regulatory authorities from conducting their
regular inspections, reviews, or other regulatory activities, it could significantly impact the ability of the FDA or other regulatory
authorities to timely review and process our regulatory submissions, which could have a material adverse effect on our business.
In the E.U., notified bodies
must be officially designated to certify products and services in accordance with the MDR. While several notified bodies have been designated
the COVID-19 pandemic has significantly slowed down their designation process and the current designated notified bodies are facing a
large amount of requests with the new regulation as a consequence of which review times have lengthened although a new regulation amending
the E.U. MDR was recently adopted in March 2023, extending existing transitional provisions. This situation could significantly impact
the ability of notified bodies to timely review and process our regulatory submissions, which could have a material adverse effect on
our business in the E.U. and the EEA (which consists of the 27 E.U. member states plus Norway, Liechtenstein and Iceland).
The misuse or off-label use of our Platform
may harm our reputation in the marketplace, result in injuries that lead to product liability suits or result in costly investigations,
fines or sanctions by regulatory bodies, particularly if we are deemed to have engaged in the promotion of these uses, any of which could
be costly to our business.
Our first generation Aerodentis
System is a Class II medical device was cleared by FDA for commercialization in the U.S. pursuant to the 510(k) notification process for
movement and alignment of teeth during orthodontic treatment of malocclusion in April 2020. We are preparing to apply for 510(k) clearance
for the Platform. If and when our Platform receives 510(k) clearance, it will be cleared for marketing by the FDA only for movement and
alignment of teeth during orthodontic treatment of malocclusion. We, thus, will not be able to promote it for any other indications for
use or make any promotional claims that are inconsistent with, or outside the scope of, such FDA clearance (often referred to as “off-label
uses”). However, the assessment of whether a given claim is or is not consistent with a given FDA clearance or approval can often
be subjective, and we cannot guarantee that FDA will always agree with our position regarding a particular claim or that all of our employees,
representatives, and agents will abide by our marketing policies. If FDA determines that we have promoted any product without the requisite
clearance or approval and/or for an off-label or unapproved use, it could take any number of enforcement actions against us, including
(among others), issuing untitled or warning letters and/or pursuing an injunction, seizure, civil fine and/or criminal penalties. It is
also possible that other federal, state or foreign enforcement authorities might take action under other regulatory authority, such as
laws prohibiting false claims for reimbursement, any of which would have a material adverse effect on our business, financial condition,
and/or business as a whole.
Additionally, we must have
competent and reliable scientific evidence or, where applicable, other adequate substantiation for each reasonable interpretation of every
promotional claim we make. In particular, comparative or superiority claims generally require adequate, well controlled, head-to-head
clinical studies, comparing the product to the applicable competing products. To the extent we make any claims, or are otherwise held
responsible for third-party claims about any product we may market in the United States, without the requisite clinical substantiation,
we could be subject to enforcement action by FDA and/or the Federal Trade Commission (the “FTC”), as well as a competitor
challenge via the National Advertising Division (the “NAD”) of the Better Business Bureau. Our plans to utilize social media
as a primary promotional tool for our device(s) increases the applicable enforcement risk, as it makes it easier for our employees, affiliates,
and any third parties with which we may have a relationship and/or arrangement under which we are deemed responsible for such party’s
claims about our product(s) to disseminate promotional claims about our product(s) that may be inconsistent with applicable regulations
governing device promotions. Further, consumers can bring private false-advertising lawsuits, including class actions, against us for
any material misrepresentations and/or deceptive or unsubstantiated claims (among other similar causes of action) in our promotional materials
or other advertising. Any of the foregoing could have a material adverse effect on our business.
Laws and Regulations Governing Healthcare,
including Health Information Privacy and Security Laws
We are subject to certain federal, state,
and foreign fraud and abuse laws, health information privacy and security laws, and transparency laws, which, if violated, could subject
us to substantial penalties. Additionally, any challenge to or investigation into our practices under these laws could cause adverse publicity
and be costly to respond to, and thus could harm our business.
There are numerous U.S. federal
and state, as well as foreign, laws pertaining to healthcare fraud and abuse, including anti-kickback, false claims, and physician transparency
laws. Efforts to ensure that our business arrangements with third parties will comply with applicable healthcare laws and regulations
involve substantial costs. Our business practices and relationships with providers and patients are subject to scrutiny under these laws.
We may also be subject to patient information privacy and security regulation by both the federal government and the states and foreign
jurisdictions in which we conduct our business. The healthcare laws and regulations that may affect our ability to operate include:
| ● | the federal healthcare Medicare and Medicaid Patient Protection
Act of 1987 (the “Anti-Kickback Statute”), which prohibits, among other things, persons, and entities from knowingly and
willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce or reward
either the referral of an individual for, or the purchase, lease, order, or arrange for or recommend a good or service, for which payment
may be made, in whole or in part, under federal healthcare programs, such as Medicare and Medicaid. The term “remuneration”
has been broadly interpreted to include anything of value. The government can establish a violation of the Anti-Kickback Statute without
proving that a person or entity had actual knowledge of the law or a specific intent to violate. Moreover, the government may assert
that a claim including items or services resulting from a violation of the federal healthcare Anti-Kickback Statute constitutes a false
or fraudulent claim for purposes of the federal civil False Claims Act. Although there are a number of statutory exceptions and regulatory
safe harbors to the federal healthcare Anti-Kickback Statute protecting certain common business arrangements and activities from prosecution
or regulatory sanctions, the exceptions and safe harbors are drawn narrowly. Practices that involve remuneration to those who prescribe,
purchase, or recommend medical device products, including discounts, or engaging individuals as speakers, consultants, or advisors, may
be subject to scrutiny if they do not fit squarely within an exception or safe harbor. Our practices may not in all cases meet all of
the criteria for safe harbor protection from anti- kickback liability. Moreover, there are no safe harbors for many common practices,
such as reimbursement support programs, educational or research grants, or charitable donations; |
| ● | the federal civil False Claims Act, which prohibits, among
other things, individuals or entities from knowingly presenting, or causing to be presented, false or fraudulent claims for payment of
federal government funds, and knowingly making, using or causing to be made or used a false record or statement material to a false or
fraudulent claim to avoid, decrease or conceal an obligation to pay money to the federal government. Private individuals, commonly known
as “whistleblowers,” can bring civil False Claims Act qui tam actions, on behalf of the government and such individuals and
may share in amounts paid by the entity to the government in recovery or settlement. False Claims Act liability is potentially significant
in the healthcare industry because the statute provides for treble damages and serious mandatory penalties for each false or fraudulent
claim or statement. The government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback
Statute constitutes a false or fraudulent claim under the federal civil False Claims Act. Many pharmaceutical and medical device manufacturers
have been investigated and have reached substantial settlements under the federal civil False Claims Act in connection with alleged off-label
promotion of their products and allegedly providing free products to customers with the expectation that the customers would bill federal
health care programs for the product. In addition, manufacturers can be held liable under the federal civil False Claims Act even when
they do not submit claims directly to government payers if they are deemed to “cause” the submission of false or fraudulent
claims. There are also criminal penalties, including imprisonment and criminal fines, for making or presenting false, fictitious or fraudulent
claims to the federal government; |
| ● | Health Insurance Portability and Accountability Act of 1996
(“HIPAA”), which created additional federal criminal statutes that prohibit, among other things, knowingly and willfully
executing or attempting to execute a scheme to defraud any healthcare benefit program, including private third-party payers, knowingly
and willfully embezzling or stealing from a healthcare benefit program, willfully obstructing a criminal investigation of a healthcare
offense, and knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious
or fraudulent statements or representations, or making or using any false writing or document knowing the same to contain any materially
false, fictitious or fraudulent statement or entry in connection with the delivery of, or payment for, healthcare benefits, items or
services. Similar to the federal healthcare Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the statute
or specific intent to violate it to have committed a violation; |
| ● | the federal Physician Payments Sunshine Act under the Affordable
Care Act which requires certain manufacturers of drugs, devices, biologics and medical supplies for which payment is available under
Medicare, Medicaid or the Children’s Health Insurance Program (with certain exceptions) to report annually to the United States
Department of Health and Human Services, Centers for Medicare and Medicaid Services, information related to payments and other transfers
of value to physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors) and teaching hospitals, and
applicable manufacturers and group purchasing organizations, as well as ownership and investment interests held by physicians and their
immediate family members. Since January 2022, applicable manufacturers are also required to report information regarding payments and
transfers of value provided to physician assistants, nurse practitioners, clinical nurse specialists, certified nurse anesthetists, and
certified nurse-midwives; |
| ● | HIPAA, as amended by Health Information Technology for Economic
and Clinical Health Act (“HITECH”), and their respective implementing regulations, which imposes privacy, security, and breach
reporting obligations with respect to Protected Health Information (“PHI”), upon entities subject to the law, such as health
plans, healthcare clearinghouses and certain healthcare providers, and their respective business associates that perform services on
their behalf that involve PHI. HITECH also created new tiers of civil monetary penalties, amended HIPAA to make HIPAA compliance as well
as civil and criminal penalties directly applicable to business associates, and gave state attorneys general new authority to file civil
actions for damages or injunctions in federal courts to enforce the HIPAA laws and seek attorneys’ fees and costs associated with
pursuing federal civil actions; and |
| ● | analogous state and foreign law equivalents of each of the
above federal laws, such as anti-kickback and false claims laws which may apply to items or services reimbursed by any third-party payer,
including commercial insurers or patients; state laws that require device companies to comply with the industry’s voluntary compliance
guidelines and the applicable compliance guidance promulgated by the federal government or otherwise restrict payments that may be made
to healthcare providers and other potential referral sources; state and local laws that require the licensure of sales representatives;
state laws that require device manufacturers to report information related to payments and other transfers of value to physicians and
other healthcare providers or marketing expenditures and pricing information; data privacy and security laws and regulations in foreign
jurisdictions that may be more stringent than those in the United States (such as the E.U., which adopted the GDPR, which became effective
in May 2018); state laws governing the privacy and security of health information in certain circumstances, many of which differ from
each other in significant ways and may not have the same effect, thus complicating compliance efforts; and state laws related to insurance
fraud in the case of claims involving private insurers. |
These laws and regulations,
among other things, constrain our business, marketing, and other promotional activities by limiting the kinds of financial arrangements,
including sales programs, we may have with physicians or other potential purchasers of our products. We have also entered into consulting
agreements with physicians, which are subject to these laws. Further, while we do not submit claims and our future customers will make
the ultimate decision on how to submit claims, we may provide reimbursement guidance and support regarding our products. Due to the breadth
of these laws, the narrowness of statutory exceptions and regulatory safe harbors available, and the range of interpretations to which
they are subject, it is possible that some of our current or future practices might be challenged under one or more of these laws.
To enforce compliance with
healthcare regulatory laws, certain enforcement bodies have recently increased their scrutiny of interactions between healthcare companies
and healthcare providers, which has led to a number of investigations, prosecutions, convictions and settlements in the healthcare industry.
For example, U.S. federal and state regulatory and enforcement agencies continue to actively investigate violations of healthcare laws
and regulations, including pursuing novel theories of liability under these laws. These government agencies recently have increased regulatory
scrutiny and enforcement activity with respect to manufacturer reimbursement support activities and patient support programs, including
bringing criminal charges or civil enforcement actions under the federal healthcare Anti-Kickback statute, federal civil False Claims
Act, the health care fraud statute, and HIPAA privacy provisions. Responding to investigations can be time and resource consuming and
can divert management’s attention from the business. Any such investigation or settlement could increase our costs or otherwise
have an adverse effect on our business. Even an unsuccessful challenge or investigation into our practices could cause adverse publicity,
and be costly to respond to.
If our operations are found
to be in violation of any of the healthcare laws or regulations described above or any other healthcare regulations that apply to us,
we may be subject to administrative, civil and criminal penalties, damages, fines, disgorgement, substantial monetary penalties, exclusion
from participation in government healthcare programs, such as Medicare and Medicaid, imprisonment, additional reporting obligations, and
oversight if we become subject to a corporate integrity agreement or other agreement to resolve allegations of non-compliance with these
laws, reputational harm, and the curtailment or restructuring of our operations.
Since our Platform will utilize cloud-based
information systems and the exchange of information between patents and doctors, we will be subject to numerous U.S. federal and state
laws and regulations related to the privacy and security of personally identifiable information, including health information.
Among other data-privacy and/or
confidentiality laws to which we may be subject, HIPAA establishes privacy and security standards that limit the use and disclosure of
PHI and require covered entities and business associates to implement administrative, physical, and technical safeguards to ensure the
confidentiality, integrity, and availability of individually identifiable health information in electronic form, among other requirements.
Violations of HIPAA may result
in civil and criminal penalties. We must also comply with HIPAA’s breach notification rule which requires notification to affected
individuals and the Secretary of Health and Human Services (“HHS”), and in certain cases to media outlets, in the case of
a breach of unsecured PHI. The regulations also require business associates of covered entities to notify the covered entity of breaches
by the business associate.
State attorneys general also
have the right to prosecute HIPAA violations committed against residents of their states, and HIPAA standards have been used as the basis
for the duty of care in state civil suits, such as those for negligence or recklessness in misusing personal information. In addition,
HIPAA mandates that HHS conduct periodic compliance audits of HIPAA covered entities and their business associates for compliance.
Many states also have laws
that protect the privacy and security of sensitive and personal information, including health information. These laws may be similar to
or even more protective than HIPAA and other federal privacy laws. For example, the laws of the State of California are more restrictive
than HIPAA. Where state laws are more protective than HIPAA, we must comply with the state laws we are subject to, in addition to HIPAA.
California passed the California Consumer Privacy Act or CCPA on June 28, 2018, which went into effect January 1, 2020. On November 3,
2020, the California Privacy Rights Act of 2020 (“CPRA”), which amends the CCPA and adds new privacy protections that became
effective on January 1, 2023, was enacted through a ballot initiative. While information we maintain that is covered by HIPAA may be exempt
from the CCPA, other records and information we maintain on our patients may be subject to the CCPA. In certain cases, it may be necessary
to modify our planned operations and procedures to comply with these more stringent state laws. Not only may some of these state laws
impose fines and penalties upon violators, but also some, unlike HIPAA, may afford private rights of action to individuals who believe
their personal information has been misused. In addition, state and federal privacy laws subject to frequent change.
In addition to HIPAA and state
health information privacy laws, we may be subject to other state and federal privacy laws, including laws that prohibit unfair privacy
and security practices and deceptive statements about privacy and security, laws that place specific requirements on certain types of
activities, such as data security and texting, and laws requiring holders of personal information to maintain safeguards and to take certain
actions in response to a data breach.
Foreign data protection, privacy,
and other laws and regulations are often more restrictive than those in the U.S. The E.U., for example, traditionally has imposed stricter
obligations under its laws and regulations relating to privacy, data protection and consumer protection than the U.S. In May 2018, the
GDPR governing data practices and privacy in the E.U., became effective and replaced the data protection laws of the individual member
states. GDPR requires companies to meet stringent requirements regarding the handling of personal data of individuals in the E.U. These
more stringent requirements include expanded disclosures to inform members about how we may use their personal data, increased controls
on profiling members, and increased rights for members to access, control and delete their personal data. In addition, there are mandatory
data breach notification requirements. The law also includes significant penalties for non-compliance, which may result in monetary penalties
of up to 20 million Euros or 4% of a company’s worldwide turnover, whichever is higher. GDPR and other similar regulations require
companies to give specific types of notice and informed consent is required for the placement of a cookie or similar technologies on a
user’s device for online tracking for behavioral advertising and other purposes and for direct electronic marketing, and the GDPR
also imposes additional conditions in order to satisfy such consent, such as a prohibition on pre-checked consents. It remains unclear
how the U.K. data protection laws or regulations will develop in the medium to longer term and how data transfer to the U.K. from the
E.U. will be regulated. Outside of the E.U., there are many other countries with data protection laws, and new countries are adopting
data protection legislation with increasing frequency. Many of these laws may require consent from individuals for the use of data for
various purposes, including marketing, which may reduce our ability to market our products.
There is no harmonized approach
to these laws and regulations globally. Consequently, we increase our risk of non-compliance with applicable foreign data protection laws
and regulations when we expand internationally. We may need to change and limit the way we use personal information in operating our business
and may have difficulty maintaining a single operating model that is compliant. Compliance with such laws and regulations will result
in additional costs and may necessitate changes to our business practices and divergent operating models, limit the effectiveness of our
marketing activities, adversely affect our business, results of operations, and financial condition, and subject us to additional liabilities.
Our business could be adversely affected
by professional and legal challenges to our business model or by new state actions restricting our ability to provide our products and
services in certain states.
Since the success of our business
will be dependent on the widespread adaptation of our Platform as a valid method for smile correction, many patients across multiple geographies
will be needed to use our Platform and provide positive feedback and results. This will expose us to legal risk of patients or dental
practitioners who may have a negative experience with our Platform to file lawsuits claiming damages or other claims. Although the we
will seek insurance coverage for such legal actions, there is no assurance that the amount of coverage will be sufficient to cover these
claims. In addition, such legal actions from consumers and dental professionals may result in material and adverse effects on our ability
to continue to conduct business due to negative press.
A number of dental and orthodontic
professionals believe that aligners are appropriate for only a limited percentage of their patients and may believe that our Platform
is even less appropriate than traditional aligners. National and state dental associations have issued statements discouraging use of
orthodontics using a teledentistry platform. Increased market acceptance of remote treatment may depend, in part, upon the recommendations
of dental and orthodontic professionals and associations, as well as other factors including effectiveness, safety, ease of use, reliability,
aesthetics, and price compared to competing products. Furthermore, our ability to conduct business in each state is dependent, in part,
upon that particular state’s treatment of remote healthcare and that state dental board’s regulation of the practice of dentistry.
Security breaches, data breaches, cyber
attacks, other cybersecurity incidents or the failure to comply with privacy, security and data protection laws could materially impact
our operations, patient care could suffer, we could be liable for damages, and our business, operations and reputation could be harmed.
We expect to retain confidential
customer personal and financial, patient health information and our own proprietary information and data essential to our business operations.
We will rely upon the effective operation of our IT systems, and those of our service providers, vendors, and other third parties to safeguard
the information and data. Additionally, our success may be dependent on the success of healthcare providers, many of whom are comprised
of individual or small operations with limited IT experience and inadequate or untested security protocols, in managing data privacy and
data security requirements. It is critical that the facilities, infrastructure and IT systems on which we depend to run our business and
the products we develop remain secure and be perceived by the marketplace and our potential customers to be secure. Despite the implementation
of security features in our products and security measures in our IT systems, we and our service providers, vendors, and other third parties
may become subject to physical break-ins, computer viruses or other malicious code, unauthorized or fraudulent access, programming errors
or other technical malfunctions, hacking or phishing attacks, malware, ransomware, employee error or malfeasance, cyber attacks, and other
breaches of IT systems or similar disruptive actions, including by organized groups and nation-state actors. For example, we may experience
cybersecurity incidents and unauthorized internal employee exfiltration of company information.
Further, the frequency of
third-party cyber-attacks has increased over the last several years. The military conflict in Ukraine may cause nation-state actors
or hackers sympathetic to either side of the conflict to carry out cyber-attacks to achieve their goals, which may include espionage,
information gathering operations, monetary gain, ransomware, disruption, and destruction. Significant service disruptions, breaches in
our infrastructure and IT systems or other cybersecurity incidents could expose us to litigation or regulatory investigations, impair
our reputation and competitive position, be distracting to our management, and require significant time and resources to address. Affected
parties or regulatory agencies could initiate legal or regulatory action against us, which could prevent us from resolving the issues
quickly or force us to resolve them in unanticipated ways, cause us to incur significant expense and liability, or result in judicial
or governmental orders forcing us to cease operations or modify our business practices in ways that could materially limit or restrict
the products and services we provide. Concerns over our privacy practices could adversely affect others’ perception of us and deter
potential customers, patients and partners from using our products. In addition, patient care could suffer, and we could be liable if
our products or IT systems fail to deliver accurate and complete information in a timely manner. We have internal monitoring and detection
systems as well as cybersecurity and other forms of insurance coverage related to a breach event covering expenses for notification, credit
monitoring, investigation, crisis management, public relations and legal advice. However, damages and claims arising from such incidents
may not be covered or may exceed the amount of any coverage and do not cover the time and effort we may incur investigating and responding
to any incidents, which may be material. The costs to eliminate, mitigate or recover from security problems and cyber attacks and
incidents could be material and depending on the nature and extent of the problem and the networks or products impacted, may result in
network or systems interruptions, decreased product sales, or data loss that may have a material impact on our operations, net revenues
and operating results.
Our business will expose us to potential
liability for the quality and safety of our products and services, how we advertise and market those products and services and how and
to whom we sell them, and we may incur substantial expenses or be found liable for substantial damages or penalties if we are subject
to claims or litigation.
Our products and services
involve an inherent risk of claims concerning their design, manufacture, safety and performance, how they are marketed and advertised
in a complex framework of highly regulated domestic and international laws and regulations, how we package, bundle or sell them to potential
customers, who may be private individuals or companies or public entities such as hospitals and clinics, and how we train and support
doctors, their staffs and patients who administer or use our products. Moreover, consumer products and services are routinely subject
to claims of false, deceptive or misleading advertising, consumer fraud and unfair business practices. Additionally, we may be held liable
if any product we develop or manufacture or services we offer or perform causes injury or is otherwise found unhealthy. If our products
are safe but they are promoted for off-label usage, we may be investigated, fined or have our products or services enjoined or approvals
rescinded or we may be required to defend ourselves in litigation. Although we maintain insurance for product liability, business practices
and other types of activities we make or offer, coverage may not be available on acceptable terms, if at all, and may be insufficient
for actual liabilities. Any claim for product liability, sales, advertising and business practices, regardless of its merit or eventual
outcome, could result in material legal defense costs and damage our reputation, increase our expenses and divert management’s attention.
Increased focus on current and anticipated
environmental, social and governance (“ESG”) laws and increased scrutiny of our ESG policies and practices may materially
increase our costs, expose us to potential liability, adversely impact our reputation, employee retention, willingness of potential customers
and suppliers to do business with us and willingness of investors to invest in us.
Our operations are subject
to a variety of existing local, regional and global ESG laws and regulations, and we will likely be required to comply with new, broader,
more complex and more costly laws and regulations that focus on ESG matters. Our compliance obligations will likely span all aspects of
our business and operations, including product design and development, materials sourcing and other procurement activities, product packaging,
product safety, energy and natural resources usage, facilities design and utilization, recycling and collection, transportation, disposal
activities and workers’ rights.
Environmental regulations
related to greenhouse gases are expected to have an increasingly larger impact on our or our suppliers’ energy sources. Many U.S.
and foreign regulators have enacted or are considering enacting new or additional disclosure requirements or limits on the emissions of
greenhouse gases, including, but not limited to, carbon dioxide and methane, from power generation units using fossil fuels. The effects
of greenhouse gas emission limits on power generation are subject to significant uncertainties, including the timing of any new requirements,
levels of emissions reductions and the scope and types of emissions regulated. These limits may have the effect of increasing our costs
and those of our suppliers and could result in manufacturing, transportation and supply chain disruptions and delays if clean energy alternatives
are not readily available in adequate amounts when required. Moreover, alternative energy sources, coupled with reduced investments in
traditional energy sources and infrastructure, may fail to provide the predictable, reliable, and consistent energy that we, our suppliers
and other businesses need for operations.
Meeting our obligations under
existing ESG laws, rules, or regulations is already costly to us and our suppliers, and we expect those costs to increase as new laws
are enacted, possibly materially. Additionally, we expect regulators to perform investigations, inspections and periodically audit our
compliance with these laws and regulations, and we cannot provide assurance that our efforts or operations will be compliant. If we fail
to comply with any requirements, we could be subject to significant penalties or liabilities and we may be required to implement new and
materially more costly processes and procedures to come into compliance. Further these laws are subject to unpredictable changes. Even
if we successfully comply with these laws and regulations, our suppliers may fail to comply. We may also suffer financial and reputational
harm if future customers require, and we are unable to deliver, certification that our products are conflict free. In all of these situations,
our future customers may stop purchasing products from us, and may take legal action against us, which could harm our reputation, revenues
and results of operations.
Investor advocacy groups,
institutional investors, investment funds, proxy advisory services, stockholders, and consumers are also increasingly focused on corporate
ESG practices. Additionally, public interest and legislative pressure related to public companies’ ESG practices continues to grow.
If our ESG practices fail to meet investor or other industry stakeholders’ evolving expectations and standards, including environmental
stewardship, support for local communities, board of director and employee diversity, human capital management, employee health and safety
practices, product quality, supply chain management, corporate governance and transparency and employing ESG strategies in our operations,
our brand, reputation and employee retention may be negatively impacted, potential customers and suppliers may be unwilling to do business
with us and investors may be unwilling to invest in us. In addition, as we work to align our ESG practices with industry standards, we
have expanded and will likely continue to expand our disclosures in these areas. We also expect to incur additional costs and require
additional resources to monitor, report, and comply with our various ESG practices. If we fail to adopt ESG standards or practices as
quickly as stakeholders desire, report on our ESG efforts or practices accurately, or satisfy the disclosure and other expectations of
stakeholders, our reputation, business, financial performance, growth, and stock price may be adversely impacted.
We are subject to consumer protection laws
that regulate our marketing practices and prohibit unfair or deceptive acts or practices. Our actual or perceived failure to comply with
such obligations could harm our business, and changes in such regulations or laws could require us to modify our products, marketing or
advertising efforts.
In connection with the marketing
or advertisement of our products and services, we could be the target of claims relating to false, misleading, deceptive, or otherwise
noncompliant advertising or marketing practices, including under the auspices of the FTC and state consumer protection statutes. If we
rely on third parties to provide any marketing and advertising of our products and services, we could be liable for, or face reputational
harm as a result of, their marketing practices if, for example, they fail to comply with applicable statutory and regulatory requirements.
If we are found to have breached
any consumer protection, advertising, unfair competition, or other laws or regulations, we may be subject to enforcement actions that
require us to change our marketing and business practices in a manner which may negatively impact us. This could also result in litigation,
fines, penalties, and adverse publicity that could cause reputational harm and loss of patient trust, which could have an adverse effect
on our business.
We will be subject to a number of risks
related to the credit card and debit card payments we plan to accept.
We plan to accept payments
through credit and debit card transactions. For credit and debit card payments, we will be required to pay interchange and other fees,
which may increase over time. An increase in those fees may require us to increase the prices we charge and would increase our operating
expenses, either of which could harm our business, results of operations, and financial condition.
If we or our future processing
vendors fail to maintain adequate systems for the authorization and processing of credit and debit card transactions, it could cause one
or more of the major credit card companies to disallow our continued use of their payment products. In addition, if these systems fail
to work properly and, as a result, we do not charge our patients’ credit or debit cards on a timely basis or at all, our business,
revenue, results of operations, and financial condition could be harmed.
The payment methods that we
will offer can also subject us to potential fraud and theft by criminals, who are becoming increasingly more sophisticated in exploiting
weaknesses that may exist in the payment systems. If we fail to comply with applicable rules or requirements for the payment methods we
will accept, or if payment-related data is compromised due to a breach, we may be liable for significant costs incurred by payment card
issuing banks and other third parties or subject to fines and higher transaction fees, or our ability to accept or facilitate certain
types of payments may be impaired. In addition, our patients could lose confidence in certain payment types, which may result in a shift
to other payment types or potential changes to our payment systems that may result in higher costs. If we fail to adequately control fraudulent
credit card transactions, we may face civil liability, diminished public perception of our security measures, and significantly higher
card-related costs, each of which could harm our business, results of operations, and financial condition.
We will also be subject to
payment card association operating rules, certification requirements, and rules governing electronic funds transfers, which could change
or be reinterpreted to make it more difficult for us to comply. We will be required to comply with payment card industry security standards.
Failing to comply with those standards may violate payment card association operating rules, federal and state laws and regulations, and
the terms of our contracts with payment processors. Any failure to comply fully also may subject us to fines, penalties, damages, and
civil liability, and may result in the loss of our ability to accept credit and debit card payments. Further, there is no guarantee that
such compliance will prevent illegal or improper use of our payment systems or the theft, loss, or misuse of data pertaining to credit
and debit cards, card holders, and transactions.
If we are unable to maintain
our chargeback rate or refund rates at acceptable levels, our future processing vendor may increase our transaction fees or terminate
its relationship with us. Any increases in our credit and debit card fees could harm our results of operations, particularly if we elect
not to raise our rates for our products and services to offset the increase. The termination of our ability to process payments on any
major credit or debit card would significantly impair our ability to operate our business.
We face risks related to our future international
sales, including the need to obtain necessary foreign regulatory clearance or approvals.
Sales of our products outside
the U.S. will subject us to foreign regulatory requirements that vary widely from country to country. We received our European CE mark
and ISO/MDSAP certification in 2019. In light of our ISO/MDSAP certification, we believe that we are in substantial compliance with applicable
E.U. regulations.
We will also need to obtain
regulatory approval in other foreign jurisdictions in which we plan to market and sell our products. The time required to obtain clearances
or approvals required by other countries may be longer than that required for FDA clearance or approval, and requirements for such approvals
may differ from FDA requirements. We may be unable to obtain regulatory approvals and may also incur significant costs in attempting to
obtain foreign regulatory approvals or maintain those we already have. If we experience delays in receipt of approvals to market our products
in new jurisdictions, or if we fail to receive these approvals, we may be unable to market our products in international markets in a
timely manner, if at all, which could materially impact our international expansion and adversely affect our business as a whole. In addition,
we anticipate that regulations in certain foreign countries may challenge our teledentistry model. Some international regulations may
also limit the availability of our Platform to patients in certain jurisdictions without our first obtaining a license or engaging a third
party to provide such financing, or limit the financing options we can offer our patients. If any of these risks were to materialize,
they could limit our expected international growth and profitability.
Intellectual Property Risks
Our success depends in part on our proprietary
technology, and if we are unable to successfully enforce our intellectual property rights, our competitive position may be harmed.
Our success will depend in
part on our ability to maintain existing intellectual property and to obtain and maintain further intellectual property protection for
our products and services, both in the U.S. and in other countries. We intend to protect our intellectual property rights through a combination
of patent, trademark, copyright, and trade secret laws, as well as third-party confidentiality and assignment agreements. Our inability
to do so could harm our competitive position.
We rely on our portfolio of
issued and pending patent applications in the U.S. and other countries to protect a large part of our intellectual property and our competitive
position; however, our currently pending or future patent filings may not result in the issuance of patents. While we generally apply
for patents in those countries where we intend to make, have made, use, or sell patented products, we may not accurately predict all of
the countries where patent protection will ultimately be desirable. If we fail to timely file for a patent, we may be precluded from doing
so at a later date.
Patent rights are territorial,
and patent protection extends only to those countries where we have issued patents. Filing, prosecuting and defending patents on our products
and product candidates in all countries and jurisdictions throughout the world would be prohibitively expensive, and our intellectual
property rights in some countries outside the United States could be less extensive than those in the United States. Many countries do
not protect intellectual property to the same extent as the U.S. or Europe, and their litigation processes differ. Competitors may successfully
challenge or avoid our patents, or manufacture products in countries where we have not applied for patent protection. Changes in the patent
laws in the U.S. or other countries may diminish the value of our patent rights. As a result of these and other factors, the scope, validity,
enforceability, and commercial value of our patent rights are uncertain and unpredictable.
Furthermore, the issuance
of a patent, while presumed valid and enforceable, is not conclusive as to its validity or its enforceability and it may not provide us
with adequate proprietary protection or competitive advantages against competitors with similar products. Any patents issued to us may
be challenged, invalidated, held unenforceable, circumvented, or may not be sufficiently broad to prevent third parties from producing
competing products similar in design to our products. In addition, any protection afforded by foreign patents may be more limited than
that provided under U.S. patent and intellectual property laws. There can be no assurance that any of our patents, any patents licensed
to us, or any patents which we may be issued in the future, will provide us with a competitive advantage or afford us protection against
infringement by others, or that the patents will not be successfully challenged or circumvented by third parties, including our competitors.
Further, there can be no assurance that we will have adequate resources to enforce our patents. Competitors may also be able to design
around our patents. Other parties may develop and obtain patent protection for more effective technologies, designs or methods.
Our ability to enforce our
patent rights depends on our ability to detect infringement. It is difficult to detect infringers who do not advertise the components
that are used in their products. Moreover, it may be difficult or impossible to obtain evidence of infringement in a competitor’s
or potential competitor’s product, particularly in litigation in countries other than the U.S. that do not provide an extensive
discovery procedure. Any litigation to enforce or defend our patent rights, if any, even if we were to prevail, could be costly and time-consuming
and would divert the attention of our management and key personnel from our business operations. We may not prevail in any lawsuits that
we initiate and the damages or other remedies awarded if we were to prevail may not be commercially meaningful.
We also may seek to rely on
protection of copyright, trade secrets, know how, and confidential and proprietary information. We generally enter into confidentiality
and non-compete agreements with our employees, consultants, and collaborative partners upon their commencement of a relationship with
us. However, these agreements may not provide meaningful protection against the unauthorized use or disclosure of our trade secrets or
other confidential information, and adequate remedies may not exist if unauthorized use or disclosure were to occur. The exposure of our
trade secrets and other proprietary information would impair our competitive advantages and could have a material adverse effect on our
operating results, financial condition, and future growth prospects. In particular, a failure to protect our proprietary rights might
allow competitors to copy our technology, which could adversely affect our pricing and market share. We may not be able to prevent the
unauthorized disclosure or use of our technical knowledge or trade secrets by consultants, vendors, former employees and current employees.
Further, other parties may independently develop substantially equivalent know-how and technology.
While we currently do not
own any registered trademarks, we intend to rely on both registered and common law rights for our trademarks in the future. There can
be no assurance that our future trademark applications will be approved. Third parties may also oppose our trademark applications, or
otherwise challenge our use of the trademarks. In the event that our trademarks are successfully challenged, we could be forced to rebrand
our products and services, which could result in loss of brand recognition, and could require us to devote resources to advertising and
marketing new brands. Further, there can be no assurance that competitors will not infringe our trademarks, or that we will have adequate
resources to enforce our trademarks.
Litigation, interferences,
oppositions, re-exams, inter partes reviews, post grant reviews, or other proceedings are, have been, and may in the future be necessary
in some instances to determine the validity and scope of certain of our proprietary rights, and in other instances to determine the validity,
scope, or non-infringement of certain proprietary rights claimed by third parties to be pertinent to the manufacture, use, or sale of
our products or provision of our services. These types of proceedings are unpredictable and may be protracted, expensive, and distracting
to management. The outcome of such proceedings could adversely affect the validity and scope of our patent or other proprietary rights,
hinder our ability to manufacture and market our products and provide our services, require us to seek a license for the infringed product
or technology, or result in the assessment of significant monetary damages. An unfavorable ruling could include monetary damages or, in
cases where injunctive relief is sought, an injunction prohibiting us from selling our products or providing our services. Any of these
results from litigation could adversely affect our business, financial condition, and results of operations.
If we infringe or violate the patents or
proprietary rights of other parties or are subject to an intellectual property infringement or misappropriation claim, our ability to
grow our business may be severely limited.
Our commercial success also
depends upon our ability, and the ability of any third party with which we may partner, to develop, manufacture, market and sell our products,
if approved, and use our patent-protected technologies without infringing the patents of third parties. Extensive litigation over patents
and other intellectual property rights is common in the dental and orthodontic industry.
We may not have identified
all patents, published applications or published literature that affect our business either by blocking our ability to commercialize our
products, by preventing the patentability of one or more aspects of our products, or by covering the same or similar technologies that
may affect our ability to market our products. For example, we may not have conducted a patent clearance search sufficient to identify
potentially obstructing third party patent rights. Moreover, patent applications in the United States are maintained in confidence for
up to 18 months after their filing. In some cases, however, patent applications remain confidential in the U.S. Patent and Trademark Office,
or the USPTO, for the entire time prior to issuance as a U.S. patent. Patent applications filed in countries outside of the United States
are not typically published until at least 18 months from their first filing date. Similarly, publication of discoveries in the scientific
or patent literature often lags behind actual discoveries. We cannot be certain that we were the first to invent, or the first to file,
patent applications covering our products. We also may not know if our competitors filed patent applications for technology covered by
our pending applications or if we were the first to invent the technology that is the subject of our patent applications. Competitors
may have filed patent applications or received patents and may obtain additional patents and proprietary rights that block or compete
with our patents.
We may therefore in the future
be the subject of patent or other litigation. From time to time, we may in the future receive letters from third parties drawing our attention
to their patent rights. While we do not believe that we infringe upon any valid and enforceable rights that have been brought to our attention,
and we take necessary steps to ensure that we do not infringe on the rights of others, there may be other more pertinent rights of which
we are presently unaware. The defense and prosecution of intellectual property suits, interference proceedings, and related legal and
administrative proceedings could result in substantial expense to us and significant diversion of effort by our technical and management
personnel. An adverse determination of any litigation or interference proceeding to which we may become a party could subject us to significant
liabilities. An adverse determination of this nature could also put our patents at risk of being invalidated or interpreted narrowly or
require us to seek licenses from third parties. Licenses may not be available on commercially reasonable terms or at all, in which event,
our business would be materially adversely affected. Intellectual property litigation or claims could force us to cease developing, selling
or otherwise commercializing one or more of our products; to pay substantial damages for past use of the asserted intellectual property;
and redesign, or rename in the case of trademark claims, our product(s) to avoid such third party rights, which may not be possible or
which could be costly and time-consuming. Any of these risks coming to fruition could have a material adverse effect on our business,
results of operations, financial condition and prospects.
Our failure to secure trademark registrations
could adversely affect our ability to market our products and operate our business.
Any future trademark applications
in the United States and any other jurisdictions where we may file may not be allowed registration, and we may not be able to maintain
or enforce our registered trademarks. During trademark registration proceedings, we may receive rejections. Although we are given an opportunity
to respond to those rejections, we may be unable to overcome such rejections. In addition, in the USPTO and in corresponding foreign agencies,
third parties are given an opportunity to oppose pending trademark applications and to seek to cancel registered trademarks. Opposition
or cancellation proceedings may be filed against our applications and/or registrations, and our applications and/or registrations may
not survive such proceedings. Failure to secure such trademark registrations in the United States and in foreign jurisdictions could adversely
affect our ability to market our products and our business.
We may be subject to claims that our employees
have wrongfully used or disclosed alleged trade secrets of their former employers.
As is common in the medical
device industry, we may employ individuals who were previously employed at other companies similar to ours, including our competitors
or potential competitors. We may become subject to claims that these employees or we have inadvertently or otherwise used or disclosed
trade secrets or other proprietary information of their former employers. Litigation may be necessary to defend against these claims.
Even if we are successful in defending against these claims, litigation could result in substantial costs and be a distraction to management.
Obtaining and maintaining patent protection
depends on compliance with various procedures and other requirements, and our patent protection could be reduced or eliminated in case
of non-compliance with these requirements.
Periodic maintenance fees,
renewal fees, annuity fees and various other governmental fees on patents and/or applications will be due to the relevant patent agencies
in several stages over the lifetime of the patents and /or applications. The relevant patent agencies require compliance with a number
of procedural, documentary, fee payment and other provisions during the patent application process. In many cases, an inadvertent lapse
can be cured by payment of a late fee or by other means in accordance with the applicable rules. However, there are situations in which
the failure to comply with the relevant requirements can result in the abandonment or lapse of the patent or patent application, resulting
in partial or complete loss of patent rights in the relevant jurisdiction. In such an event, our competitors might be able to use our
technologies and know-how which could have a material adverse effect on our business, prospects, financial condition and results of operation.
Patent terms may be inadequate to protect
our competitive position on our product candidates for an adequate amount of time.
Patents have a limited lifespan.
In the United States, if all maintenance fees are timely paid, the natural expiration of a patent is generally 20 years from its earliest
U.S. non-provisional filing date. Various extensions may be available, but the life of a patent, and the protection it affords, is limited.
Even if patents covering our products are obtained, once the patent life has expired for a product, we may be open to competition from
competitive products. Given the amount of time required for the development, testing and regulatory review of new products, patents protecting
such products might expire before or shortly after such products are commercialized. As a result, our patent portfolio may not provide
us with sufficient rights to exclude others from commercializing products similar or identical to ours.
Risks Related Our Securities
The relative lack of U.S. public company
experience of our management team may put us at a competitive disadvantage.
Our management team lacks
U.S. public company experience and is generally unfamiliar with the requirements of the U.S. securities laws and U.S. Generally Accepted
Accounting Principles (“GAAP”), which could impair our ability to comply with legal and regulatory requirements such as those
imposed by Sarbanes-Oxley Act of 2002 (the “Sarbanes-Oxley Act”). The individuals who now constitute our senior management
team have never had responsibility for managing a publicly traded company. Such responsibilities include complying with federal securities
laws and making required disclosures on a timely basis. Our senior management may not be able to implement programs and policies in an
effective and timely manner that adequately responds to such increased legal, regulatory compliance and reporting requirements. Our failure
to comply with all applicable requirements could lead to the imposition of fines and penalties and distract our management from attending
to the growth of our business.
Our common stock is not listed on any stock
exchange and there is a limited market for shares of our common stock. Even if a market for our common stock develops, our common stock
could be subject to wide fluctuations.
Our common stock is not listed
on any stock exchange. Although our common stock is quoted on the OTC Pink Market operated by the OTC Markets Group Inc., there is a limited
public market for shares of our common stock, and limited trades of our common stock have taken place on the OTC Pink Market. Even if
the shares of our common stock may in the future trade greater volume on the OTC Pink Market, the liquidity and price of our common stock
is expected to be more limited than if such securities were quoted or listed on a national exchange. No assurances can be given that an
active public trading market for our common stock will develop or be sustained. Trading volume may be limited by the fact that many major
institutional investment funds, including mutual funds, as well as individual investors follow a policy of not investing in over the counter
stocks and certain major brokerage firms restrict their brokers from recommending over the counter stocks because they are considered
speculative, volatile and thinly traded. Lack of liquidity will limit the price at which stockholders may be able to sell our common stock.
Even if our common stock will
in the future trade more actively on the OTC Pink Market, the price of such common stock could be subject to wide fluctuations, in response
to quarterly variations in our operating results, announcements by us or others, developments affecting us, and other events or factors.
In addition, the stock market has experienced extreme price and volume fluctuations in recent years. These fluctuations have had a substantial
effect on the market prices for many companies, often unrelated to the operating performance of such companies, and may adversely affect
the market prices of the securities. Such risks could have an adverse effect on the stock’s future liquidity.
We cannot assure you that our common stock
will become eligible for listing or quotation on any exchange and the failure to do so may adversely affect your ability to dispose of
our common stock in a timely fashion.
In order for our common stock
to become eligible for listing or quotation on any exchange, reverse merger companies must have had their securities traded on an over-the-counter
market for at least one year, maintained a certain minimum closing price for not less than 30 of the most recent 60 days prior to the
filing of an initial listing application and prior to listing, and timely filed with the SEC all required reports since consummation of
the reverse merger, including one annual report containing audited consolidated financial statements for a full fiscal year commencing
after the date of filing of the Current Report on Form 8-K which discloses the reverse merger. We may not be able to meet all of the filing
requirements above and may not be able to satisfy the initial standards for listing or quotation on any exchange in the foreseeable future
or at all. Even if we are able to become listed or quoted on an exchange, we may not be able to maintain a listing of the common stock
on such stock exchange.
As a result of the Share Exchange, we became
a company that is subject to the reporting requirements of federal securities laws, which can be expensive and may divert resources from
other projects, thus impairing our ability to grow.
As a result of the Share Exchange,
we became a public reporting company and, accordingly, subject to the information and reporting requirements of Securities Exchange Act
of 1934, as amended (the “Exchange Act”), and other federal securities laws, including compliance with the Sarbanes-Oxley
Act. The costs of preparing and filing annual and quarterly reports, proxy statements and other information with the SEC (including reporting
of the Share Exchange) and furnishing audited reports to stockholders will cause our expenses to be higher than they would have been if
we remained privately held and did not consummate the Share Exchange.
Public company compliance may make it more
difficult for us to attract and retain officers and directors.
The Sarbanes-Oxley Act and
new rules subsequently implemented by the SEC have required changes in corporate governance practices of public companies. As a public
company, we expect these new rules and regulations to increase our compliance costs and to make certain activities more time consuming
and costly. As a public company, we also expect that these new rules and regulations may make it more difficult and expensive for us to
obtain director and officer liability insurance in the future and we may be required to accept reduced policy limits and coverage or incur
substantially higher costs to obtain the same or similar coverage. As a result, it may be more difficult for us to attract and retain
qualified persons to serve on our board of directors or as executive officers.
Because we became public by means of a reverse
merger, we may not be able to attract the attention of major brokerage firms.
There may be risks associated
with us becoming public through a “reverse merger”. Securities analysts of major brokerage firms may not provide coverage
of us since there is no incentive to brokerage firms to recommend the purchase of our common stock. No assurance can be given that brokerage
firms will, in the future, want to conduct any secondary offerings on our behalf.
Our stock price may be volatile.
The price at which our common
stock is quoted is likely to be highly volatile and could fluctuate widely in price in response to various factors, many of which are
beyond our control, including the following:
| ● | changes in our industry; |
| ● | competitive pricing pressures; |
| ● | our ability to obtain working capital financing; |
| ● | additions or departures of key personnel; |
| ● | limited “public float” in the hands of a small
number of persons whose sales or lack of sales could result in positive or negative pricing pressure on the price at which our common
stock is quoted; |
| ● | sales of our common stock; |
| ● | our ability to execute our business plan; |
| ● | operating results that fall below expectations; |
| ● | loss of any strategic relationship; |
| ● | regulatory developments; |
| ● | economic and other external factors; and |
| ● | period-to-period fluctuations in our financial results. |
In addition, the securities
markets have from time to time experienced significant price and volume fluctuations that are unrelated to the operating performance of
particular companies. These market fluctuations may also materially and adversely affect the price at which our common stock is quoted.
Our securities are restricted securities
with limited transferability.
Our securities should be considered
a long-term, illiquid investment. Our common stock has not been registered under the Securities Act of 1933, as amended (the “Securities
Act”), and cannot be sold without registration under the Securities Act or any exemption from registration. In addition, our common
stock is not registered under any state securities laws that would permit its transfer. Because of these restrictions, a stockholder will
likely find it difficult to liquidate an investment in our common stock.
We are subject to penny stock rules which
will make the shares of our common stock more difficult to sell.
We are subject to the SEC’s
“penny stock” rules since our shares of common stock trade below $5.00 per share. Penny stocks generally are equity securities
with a per share price of less than $5.00. The penny stock rules require broker-dealers to deliver a standardized risk disclosure document
prepared by the SEC that provides information about penny stocks and the nature and level of risks in the penny stock market. The broker-dealer
must also provide the customer with current bid and offer quotations for the penny stock, the compensation of the broker-dealer and its
salesperson, and monthly account statements showing the market value of each penny stock held in the customer’s account. The bid
and offer quotations, and the broker-dealer and salesperson compensation information must be given to the customer orally or in writing
prior to completing the transaction and must be given to the customer in writing before or with the customer’s confirmation.
In addition, the penny stock
rules require that prior to a transaction the broker-dealer must make a special written determination that the penny stock is a suitable
investment for the purchaser and receive the purchaser’s written agreement to the transaction. The penny stock rules are burdensome
and may reduce purchases of any offerings and reduce the trading activity for shares of our common stock. As long as our shares of common
stock are subject to the penny stock rules, the holders of such shares of common stock may find it more difficult to sell their securities.
FINRA sales practice requirements may also
limit a stockholder’s ability to buy and sell our stock.
In addition to the “penny
stock” rules described above, the Financial Industry Regulatory Authority (“FINRA”) has adopted rules that require that
in recommending an investment to a customer, a broker-dealer must have reasonable grounds for believing that the investment is suitable
for that customer. Prior to recommending speculative low priced securities to their non-institutional customers, broker-dealers must make
reasonable efforts to obtain information about the customer’s financial status, tax status, investment objectives and other information.
Under interpretations of these rules, FINRA believes that there is a high probability that speculative low priced securities will not
be suitable for at least some customers. The FINRA requirements make it more difficult for broker-dealers to recommend that their customers
buy our common stock, which may limit your ability to buy and sell our stock and have an adverse effect on the market for our shares.
We do not anticipate paying any cash dividends.
We presently do not anticipate
that we will pay any dividends on any of our capital stock in the foreseeable future. The payment of dividends, if any, would be contingent
upon our revenues and earnings, if any, capital requirements, and general financial condition. The payment of any dividends will be within
the discretion of our board of directors. We presently intend to retain all earnings, if any, to implement our business plan; accordingly,
we do not anticipate the declaration of any dividends in the foreseeable future.
Our shares of common stock are very thinly
traded, and the price may not reflect our value and there can be no assurance that there will be an active market for our shares of common
stock in the future.
Our shares of common stock
are thinly traded. Due to the illiquidity, the price at which our common stock is quoted may not accurately reflect our relative value.
There can be no assurance that there will be an active market for our shares of common stock either now or in the future. Investors may
not be able to liquidate their investment or liquidate it at a price that reflects the value of the business. If a more active market
should develop, the price may be highly volatile. Because there may be a low price for our shares of common stock, many brokerage firms
may not be willing to effect transactions in the securities. Even if an investor finds a broker willing to effect a transaction in the
shares of our common stock, the combination of brokerage commissions, transfer fees, taxes, if any, and any other selling costs may exceed
the selling price. Further, many lending institutions will not permit the use of such shares of common stock as collateral for a loans.
We may apply the proceeds of the Private
Placement to uses that ultimately do not improve our operating results or increase the price of our common stock.
We intend to use the net proceeds
from the Private Placement. However, our management has broad discretion in how we actually use these proceeds. These proceeds could be
applied in ways that do not ultimately improve our operating results or otherwise increase the value of our common stock.
We may need additional financing which may
not be available on acceptable terms, which may in turn dilute your investment in us.
Our future capital requirements
will depend on many factors including but not limited to: market acceptance of our services; competitive pressure on the price of our
products; the extent to which we invest in new locations, develop new relationships with producers of polymers and chemicals as well as
consumers of polymers and chemicals; and the response of competitors to our products. We believe that the existing cash balances, including
the net proceeds from the Private Placement, and funds generated from operations will provide us with sufficient funds to finance our
operations for the foreseeable future. To the extent that our current funds, together with existing resources, are insufficient to fund
our activities over the long-term, we may need to raise additional funds through equity or debt financing or from other sources.
Subject to the lock-up provisions
of the Securities Purchase Agreement and other documents related to the Share Exchange and the Private Placement, –as part of any
future financing, we are generally not restricted from issuing additional securities, including shares of common stock, securities that
are convertible into or exchangeable for, or that represent the right to receive, common stock or substantially similar securities. In
particular, we may conduct one or more additional offerings following the closing of the Private Placement and may seek waiver of the
lock-up provisions of the Securities Purchase Agreement and other documents related to the Share Exchange and the Private Placement –to
conduct such offerings. The sale of additional equity or convertible debt may result in additional dilution to our stockholders and such
securities may have rights, preferences or privileges senior to those of the common stock. To the extent that we rely upon debt financing,
we will incur the obligation to repay the funds borrowed with interest and may become subject to covenants and restrictions that restrict
operating flexibility. No assurance can be given that additional equity or debt financing will be available or that, if available, it
can be obtained on terms favorable to us or our stockholders. Failure to obtain necessary financing could have a material adverse effect
on our business, financial condition and results of operations.
Our board of directors can authorize the
issuance of preferred stock, which could diminish the rights of holders of our common stock, and make a change of control of us more difficult
even if it might benefit our stockholders.
Our board of directors is
authorized to issue shares of preferred stock in one or more series and to fix the voting powers, preferences and other rights and limitations
of the preferred stock. Accordingly, we may issue shares of preferred stock with a preference over our common stock with respect to dividends
or distributions on liquidation or dissolution, or that may otherwise adversely affect the voting or other rights of the holders of common
stock. Issuances of preferred stock, depending upon the rights, preferences and designations of the preferred stock, may have the effect
of delaying, deterring or preventing a change of control, even if that change of control might benefit our stockholders.
Anti-takeover provisions under Delaware
corporate law may make it difficult for our stockholders to replace or remove our board of directors and could deter or delay third parties
from acquiring our Company, which may be beneficial to our stockholders.
We are subject to the anti-takeover
provisions of the Delaware General Corporation Law (“DGCL”), including Section 203 of the DGCL. Under these provisions, if
anyone becomes an “interested stockholder,” we may not enter into a “business combination” with that person for
three (3) years without special approval, which could discourage a third party from making a takeover offer and could delay or prevent
a change of control. For purposes of Section 203 of the DGCL, “interested stockholder” means, generally, someone owning fifteen
percent (15%) or more of our outstanding voting stock or an affiliate of ours that owned fifteen percent (15%) or more of our outstanding
voting stock during the past three (3) years, subject to certain exceptions as described in Section 203 of the DGCL.
Future sales of significant amounts of our
common stock may depress our stock price.
Future issuances of our common
stock or securities convertible into, or exercisable or exchangeable for, our common stock, or the expiration of lock-up provisions that
restrict the issuance of new common stock or the trading of outstanding common stock, could cause the price at which our common stock
is quoted to decline. We cannot predict the effect, if any, of future issuances of our securities, or the future expirations of lock-up
provisions, on the price of our common stock. In all events, future issuances of our common stock would result in the dilution of your
holdings. In addition, the perception that new issuances of our securities could occur, or the perception that locked-up parties will
sell their securities when the lock-ups expire, could adversely affect the price at which our common stock is quoted.
The Securities Purchase Agreement
entered into in connection with the Private Placement contains provisions that prevent us, subject to certain exceptions, from offering
additional shares of capital stock for up to eighteen (18) months after the closing of the Private Placement, subject to the approval
of the Lead Investor. Further, in connection with the Share Exchange, Private Dror shareholders are subject to the lock-up provisions
contained in the Share Exchange Agreement. These lock-up provisions may be waived pursuant to the terms of Securities Purchase Agreement
and the Share Exchange Agreement, as applicable. If these restrictions on future offerings and lock-up restrictions are waived, additional
shares of our common stock may become available for sale or resale, subject to applicable law, including without notice, which could reduce
the price at which our common stock is quoted.
Further, a significant percentage
of our outstanding common stock is currently owned by a small number of stockholders. These stockholders may sell in the future large
amounts of our stock over relatively short periods of time. Sales of substantial amounts of our stock by existing stockholders may adversely
affect the price at which our stock is quoted by creating the perception of difficulties or problems with our business that may depress
our stock price.
Financial, Tax and Accounting Risks
If our goodwill or long-lived assets become
impaired, we may be required to record a material charge to earnings.
Under GAAP, we review our
goodwill and long-lived asset group for impairment when events or changes in circumstances indicate the carrying value may not be recoverable.
Additionally, goodwill must be tested for impairment at least annually. The qualitative and quantitative analysis used to test goodwill
are dependent upon various assumptions and reflect management’s best estimates. Changes in certain assumptions, including revenue
growth rates, discount rates, earnings multiples and future cash flows may cause a change in circumstances indicating that the carrying
value of goodwill or the asset group may be impaired and assessing these assumptions and predicting and forecasting future events can
be difficult. Goodwill and purchased assets require periodic fair value assessments to determine if they have become impaired. Consequently,
we may be required to record a material charge to earnings in the financial statements during the period in which any impairment of goodwill
or long-lived asset group is determined.
Changes in, or interpretations of, accounting
rules and regulations, could result in unfavorable accounting charges.
We prepare our consolidated
financial statements in conformity with GAAP. These principles are subject to interpretation by the SEC and various bodies formed to interpret
and create appropriate accounting policies. A change in these policies or in the way these policies are interpreted by us or regulators
could have a material effect on our reported results and may even retroactively affect previously reported financial statements.
We are required to annually assess our internal
control over financial reporting and any adverse results from such assessment may result in a loss of investor confidence in our financial
reports and adversely affect our stock price.
We are required to furnish
in our Form 10-K a report by our management regarding the effectiveness of our internal control over financial reporting that includes,
among other things, an assessment of the effectiveness of our internal control over financial reporting as of the end of our fiscal year,
including a statement as to whether our internal control over financial reporting is effective. Our internal controls may become inadequate
because of changes in personnel, updates and upgrades to existing software, failure to maintain accurate books and records, changes in
accounting standards or interpretations of existing standards, and, as a result, the degree of compliance of our internal control over
financial reporting with the existing policies or procedures may become ineffective. Establishing, testing and maintaining an effective
system of internal control over financial reporting requires significant resources and time commitments on the part of our management
and our finance staff, may require additional staffing and infrastructure investments and increases our costs of doing business. If we
are unable to assert that our internal control over financial reporting is effective in any future period (or if our auditors are unable
to express an opinion on the effectiveness of our internal controls or conclude that our internal controls are ineffective), the timely
filing of our financial reports could be delayed or we could be required to restate past reports, and cause us to lose investor confidence
in the accuracy and completeness of our financial reports in the future, which could have an adverse effect on our stock price.
Our effective tax rate may vary significantly
from period to period.
We operate globally and are
subject to taxes in the U.S. and foreign countries. Various internal and external factors may affect our future effective tax rate. These
factors include changes in the global economic environment, changes in our legal entity structure or activities performed within our entities,
changes in our business operations, changes in tax laws, regulations and/or rates, new or changes to accounting pronouncements, changing
interpretations of existing tax laws or regulations, changes in relative proportions of revenues and income before taxes in the various
jurisdictions in which we operate that have differing statutory tax rates, changes in overall levels of pretax earnings, the future levels
of tax benefits of stock-based compensation, settlement of income tax audits and non-deductible goodwill impairments.
Our effective tax rate is
also dependent in part on forecasts of full year results which can vary materially. Furthermore, we may continue to experience significant
variation in our effective tax rate related to excess tax benefits on stock-based compensation, particularly in the first quarter of each
year when the majority of our equity awards vest.
New tax laws and practices, changes to existing
tax laws and practices, or disputes regarding the positions we take regarding tax laws, could negatively affect our provision for income
taxes as well as our ongoing operations.
We are subject to tax laws
both within and outside of the U.S. requiring significant judgment in determining our worldwide provision for income taxes. Changes in
tax laws or changes to how those laws are applied to our business in practice, could affect the amount of tax to which we are subject
and the manner in which we operate. Additionally, the Organization for Economic Cooperation and Development’s (“OECD”)
Base Erosion and Profit Shifting (“BEPS”) project has resulted in considerable new reporting obligations worldwide as OECD
member countries have implemented its guidance. The OECD continues to publish guidance pursuant to the BEPS and other projects which,
if adopted by member countries, may affect our tax positions in many of the countries in which we do business.
Moreover, the application
of indirect taxes (such as sales and use tax (“SUT”), value-added tax (“VAT”), goods and services tax (“GST”),
and other indirect taxes) to our operations is complex and evolving. U.S. states, local and foreign taxing jurisdictions have differing
rules and regulations governing differing types of taxes, and these rules and regulations are subject to varying interpretations and exemptions
that may change over time. We collect and remit SUT, VAT, GST and other taxes in many jurisdictions and we are routinely subject to audits.
We are also routinely subject to audits regarding our tax reporting and remissions by local and national government, and we may also be
subject to audits in U.S. states, local and foreign jurisdictions for which we have not accrued tax liabilities. The positions we take
regarding taxes as well as the amounts we collect or remit may be challenged and we may be liable for failing to collect or remit all
or any portion of taxes deemed owed or the taxes could exceed our estimates. One or more U.S. states or countries may seek to impose incremental
or new sales, use, or other tax collection obligations on us or may determine that such taxes should have but have not been paid by us.
If we dispute rulings or positions taken by tax authorities, we may incur expenses and expend significant time and effort to defend our
positions, which may be costly.
On August 16, 2022, the Inflation
Reduction Act of 2022 (“IRA”) was enacted. It contains numerous new U.S. federal tax law provisions, including a corporate
alternative minimum tax on adjusted financial statement income and an excise tax on corporate stock repurchases, both effective after
December 31, 2022. We continue to evaluate the IRA’s impact to our business, which may be material.
The application of existing,
new, or future tax laws, and results of audits, whether in the U.S. or internationally, could harm our business. Furthermore, there have
been and will continue to be substantial ongoing costs associated with complying with the various tax requirements and defending our positions
in the numerous markets in which we conduct or will conduct business.
Item 1B. Unresolved Staff Comments.
None.
Item 1C. Cybersecurity.
We operate in the medical
devices sector, which is subject to various cybersecurity risks that could adversely affect our business, financial condition, and results
of operations, including intellectual property theft; fraud; extortion; harm to employees or customers; violation of privacy laws and
other litigation and legal risk; and reputational risk. We recognize the importance of assessing, identifying, and managing material risks
associated with cybersecurity threats. Both our executive management team and our board of directors are involved in the assessment, identification,
and management of such risks, including prevention, mitigation, detection, and remediation of cybersecurity incidents.
Our executive management team
is responsible for day-to-day assessment, identification and management of material risks from cybersecurity threats, including the prevention,
mitigation, detection, and remediation of cybersecurity incidents. The individuals currently serving in these roles are our Chief Executive
Officer and Chief Technology Officer. The executive management team monitors current events in order to remain aware of current cybersecurity
threats and is informed of cybersecurity incidents as they arise by our frontline personnel.
The executive management team
is also responsible for overseeing and identifying risks from cybersecurity threats associated with our use of any third-party service
providers.
Our board of directors is
responsible for oversight of risks from cybersecurity threats in conjunction with our executive management team. Our board of directors
receives updates from our management team with respect to risks from cybersecurity threats and are notified of any new significant cybersecurity
threats or incidents as they arise. Additionally, our board of directors considers risks from cybersecurity threats as part of its overall
assessment of risk management, including its general oversight of the Company’s business strategy, risk management policies, and
financials.
To date, no cybersecurity incident (or aggregation of incidents) or
cybersecurity threat has materially affected our business strategy, results of operations or financial condition, and we are not aware
of any cybersecurity incidents that are reasonably likely to materially affect the Company, including our business strategy, results of
operations, or financial condition. For further information regarding the risks associated with cybersecurity incidents, see “Risk
Factors—Security breaches, data breaches, cyber-attacks, other cybersecurity incidents or the failure to comply with privacy, security
and data protection laws could materially impact our operations, patient care could suffer, we could be liable for damages, and our business,
operations and reputation could be harmed” in Item 1A of this Annual Report on Form 10 K.
Item 2. Properties.
We lease commercial space
that has a gross area of approximately 860 square feet at Shatner 3, Jerusalem, Israel. This space is used as an office, production, testing,
adjustment, and preparation center for the shipment of orthodontic appliances. This space is approved for our activities by the certification
body MEDCERT. MEDCERT is one of the largest German Notified Bodies that provides ISO and CE certification. The inspection of our facility
is carried out on an annual basis by an auditor on behalf of MEDCERT and includes an inspection of all activities and their adaptation
to ISO 13485:2016 certification. We have successfully passed our most recent annual inspection in April 2023.
We also lease a small, approximately
130-square-foot storage facility at Hartom 7, Jerusalem, Israel.
Item 3. Legal Proceedings.
From time to time, we may
be involved in litigation that arises through the normal course of business. As of the date of this filing, we are not a party to any
material litigation nor are we aware of any such threatened or pending litigation.
There are no proceedings in
which any of our directors, officers or affiliates or any registered or beneficial stockholders is an adverse party or has a material
interest adverse to our interest.
Item 4. Mine Safety Disclosures.
Not applicable.
PART II
Item 5. Market for Registrant’s Common Equity, Related Stockholder
Matters and Issuer Purchases of Equity Securities.
Market for our Common Stock
Our Common Stock is currently
approved for quotation on the OTC Pink Market under the symbol “DROR.” As soon as practicable, and assuming we satisfy all
necessary initial listing requirements, we intend to apply to have our Common Stock listed for trading on The Nasdaq Stock Market, although
we cannot be certain that any such application will be approved.
The following table sets forth
the high and low sale prices for our Common Stock for the periods indicated as reported by OTC. The high and low prices reflect inter-dealer
prices, without retail mark-up, mark-down or commission and may not necessarily represent actual transactions.
Quarter Ended | |
High | | |
Low | |
| December 31, 2023 | |
$ | 0.03 | | |
$ | 0.0093 | |
| September 30, 2023 | |
$ | 0.0474 | | |
$ | 0.01 | |
| June 30, 2023 | |
$ | 0.038 | | |
$ | 0.0102 | |
| March 31, 2023 | |
$ | 0.04475 | | |
$ | 0.0085 | |
| December 31, 2022 | |
$ | 0.0275 | | |
$ | 0.009 | |
| September 30, 2022 | |
$ | 0.054 | | |
$ | 0.0211 | |
| June 30, 2022 | |
$ | 0.074 | | |
$ | 0.036 | |
| March 31, 2022 | |
$ | 0.1326 | | |
$ | 0.035 | |
| December 31, 2021 | |
$ | 0.1349 | | |
$ | 0.0811 | |
| September 30, 2021 | |
$ | 0.15 | | |
$ | 0.0801 | |
| June 30, 2021 | |
$ | 0.34 | | |
$ | 0.107 | |
| March 30, 2021 | |
$ | 0.58 | | |
$ | 0.0475 | |
Shareholders of Record
As
of March 29, 2024, there were 209 stockholders of record holding 495,454,546 shares of common stock.
Recent Sales of Unregistered Securities
During the fiscal year ended
December 31, 2023, all sales of equity securities not registered under the Securities Act were included in our Quarterly Reports on Form
10-Q and in our Current Reports on Form 8-K.
Repurchase of Equity Securities
We have no plans, programs
or other arrangements in regard to repurchases of our common stock.
Dividends
We have not paid any cash
dividends on shares of our Common Stock to date. It is the present intention of our board of directors to retain future earnings for the
development, operation, and expansion of its business, and our board of directors does not anticipate declaring or paying any cash dividends
for the foreseeable future. The payment of dividends is within the discretion of our board of directors and will be contingent upon our
future revenues and earnings, as well as its capital requirements and general financial condition, and we can give no assurances that
we will ever have excess funds available to pay dividends.
Securities Authorized for Issuance under Equity Compensation Plans
The table below sets forth
certain information as of December 31, 2023 regarding the shares of our Common Stock available for grant or granted under stock option
plans and other compensation arrangements that (i) were adopted by our stockholders and (ii) were not adopted by our stockholder.
Plan Category | |
Number of
securities to
be issued
upon exercise
of
outstanding
options,
warrants and
rights | | |
Weighted
average
exercise price
of outstanding
options,
warrants, and
rights | | |
Number of securities
remaining available for
future issuance under equity compensation plans
(excluding securities
reflected in common) | |
| Equity Compensation plans approved by stockholders (1) | |
| 163,142,084 | | |
$ | 0.0038480 | | |
| 245,692,304 | |
| Equity Compensation plans not approved by stockholders | |
| — | | |
| — | | |
| — | |
| Total | |
| 163,142,084 | | |
$ | 0.0038480 | | |
| 245,692,304 | |
| (1) | Represents shares approved for issuance under the 2021 Plan
and the 2023 Plan. All information in this table has been adjusted to give effect to the Share Exchange. |
Use of Proceeds from the Sale of Registered Securities
None.
Item 6. [Reserved]
Item 7. Management’s Discussion and Analysis of Financial
Condition and Results of Operations.
Management’s Discussion
and Analysis of Financial Condition and Results of Operations (“MD&A”) is designed to provide a reader of our financial
statements with a narrative from the perspective of management on our financial condition, results of operations, liquidity and certain
other factors that may affect future results. In certain instances, parenthetical references are made to relevant sections of the Notes
to Financial Statements to direct the reader to a further detailed discussion. This section should be read in conjunction with the Financial
Statements and Supplementary Data included in this Annual Report on Form 10-K. This MD&A contains forward-looking statements
reflecting our current expectations, whose actual outcomes involve risks and uncertainties. Actual results and the timing of events may
differ materially from those stated in or implied by these forward-looking statements due to a number of factors, including those discussed
in the sections entitled “Risk Factors” and “Cautionary Statement Regarding Forward-Looking Statements” contained
in this Annual Report on Form 10-K.
Unless the context otherwise
requires, references in this MD&A to “Dror,” “we”, “us”, “our”, and the “Company”
are intended to refer to (i) following the Share Exchange (as defined below), the business and operations of Dror Ortho-Design, Inc.
and its consolidated subsidiaries, and (ii) prior to the Share Exchange, Dror Ortho-Design Ltd. (the predecessor entity and currently
wholly owned subsidiary of Dror Ortho-Design, Inc.).
All dollar amounts in this
registration statement refer to U.S. dollars unless otherwise indicated.
Overview
We were incorporated as Novint Technologies, Inc. in the State of New
Mexico in April 1999. On February 26, 2002, we changed our state of incorporation to Delaware by merging with Novint Technologies, Inc.,
a Delaware corporation. On July 5, 2023, we entered into a share exchange agreement with the shareholders of Dror Ortho-Design, Ltd. (“Private
Dror”), pursuant to which the shareholders of Private Dror agreed to exchange all of their outstanding ordinary shares Private Dror
for shares of our Common Stock and convertible preferred stock (the “Share Exchange”). On August 14, 2023 the Share Exchange
was consummated and we changed our name to “Dror Ortho-Design, Inc.”
Following the Share Exchange,
we succeeded to the business of Private Dror as its sole line of business. The Share Exchange is being accounted for as a recapitalization,
with Private Dror deemed to be the accounting acquirer and the Company the acquired company. Accordingly, Private Dror’s historical
financial statements for periods prior to the consummation of the Share Exchange have become those of the Company. Operations reported
for periods prior to the Share Exchange are those of Private Dror.
Our Company
We have reimagined the way
people can correct their smile.
We plan to disrupt the aligner
market by offering millions of people a revolutionary alternative. We believe that people do not need to change their lifestyle to correct
their smile as they are required to do with existing aligner solutions.
Existing aligner solutions
generally share the same treatment principles, which are different from our solution. In most cases, patients seeking to improve their
smile need to undergo a 12-to-15 month process of wearing plastic aligners, which need to be worn the entire day and should only be removed
while eating or drinking. Patients are prescribed a series of 20 to 30 aligners that are intended to forcefully move teeth progressively
closer to their intended final position. This process causes pain every time a new aligner is used and restricts blood circulation, which
counterproductively slows down tooth movement. All-day aligner solutions are also intrusive, as patients need to conduct their lives at
work or school wearing the plastic aligners. In addition, most existing aligner therapies require multiple visits to an orthodontist to
monitor the progress of treatment plans through intraoral scanning, physical examination and patient testimony.
We believe that recent rapid
advancements in technology have made traditional aligner solutions no longer the most effective treatment option for smile correction.
Our Company has developed a proprietary AI-based platform to correct people’s smiles in a discreet and less painful manner (the
“Platform”). The Platform uses only one smart aligner to gently move teeth into their optimum position with pulsating air
while the patient is sleeping or at home.
We are involved in the research
and development of an orthodontic alignment platform. We have several patents for the technology used in the Platform and is currently
in the process of preparing the prototype for FDA approval.
Our predecessor first generation
Aerodentis System is a Class II medical device, which was cleared by FDA for commercialization in the U.S. pursuant to the 510(k) notification
process for movement and alignment of teeth during orthodontic treatment of malocclusion in April 2020. The Company is preparing to apply
for 510(k) clearance for the Platform as a Class II medical device, which constitutes an updated version of the currently cleared device.
Such updated Platform contains new and/or different components than the original device, which is why a new 510(k) clearance is required
prior to marketing the Platform in the U.S. We have not yet filed a 510(k) submission for the Platform, and it has, thus, not been found
by the FDA to be substantially equivalent to the first generation Aerodentis System.
The Company currently does
not generate revenues to fund operations and anticipates that it will continue to incur significant losses as it continues to develop
the Platform. Please refer to “Risk Factors - We are in the development stage, are not generating revenues and have no operating
history in the manufacturing and distribution of orthodontic medical devices or platforms for consumer use.” for additional information.
The Company intends to spend approximately $2.5 million over the next 18 months on software and hardware development as well as the accompanying
regulatory approvals and IP protection associated with such software and hardware projects.
Share Exchange
As discussed above, on July
5, 2023, we entered into a Share Exchange Agreement (as amended by that certain Amendment to Share Exchange Agreement, dated August 14,
2023, the “Share Exchange Agreement”) with Private Dror and all shareholders of Private Dror. Pursuant to the Share Exchange
Agreement, on August 14, 2023, the shareholders of Private Dror transferred all of their ordinary shares in Private Dror to us in exchange
for 7,576,999 newly issued shares of our Series A Convertible Preferred Stock, par value $0.0001 per share (the “Series A Preferred
Stock”), and 106,782,187 shares of our Common Stock. As a result of these share exchanges, Private Dror became a wholly owned subsidiary
of the Company.
Pursuant to the terms and
conditions of the Share Exchange Agreement:
| ● | The shareholders of Private Dror transferred 235,088 ordinary
shares of Private Dror to us in exchange for 7,576,999 shares of Series A Convertible Preferred Stock and 106,782,187 shares of Common
Stock (the “Share Exchange”). |
| ● | In connection with the Share Exchange, we assumed all of Private
Dror’s obligations under Private Dror’s outstanding share options. |
| ● | All outstanding Series A-4 Warrants to purchase Private Dror’s
ordinary shares were assumed by the Company and converted into Share Exchange Warrants (as defined below). |
| ● | Simultaneously with the Share Exchange, the board of directors
and certain officers of the Company resigned, and a new board of directors, comprised of Private Dror’s legacy board of directors,
and new officers were appointed for the Company. The Company’s new board of directors consists of Eliyahu (Lee) Haddad, Chaim Hurvitz,
Moshe Shvets, Chaim Ravad and Yehuda Englander. In addition, immediately following the Share Exchange, Mr. Haddad was appointed as the
Company’s chief executive officer, Mr. Shvets as Chief Technology Officer, and Mr. Hurvitz as chairman of the board of directors. |
Private Placement
In connection with the closing
of the Share Exchange, pursuant to the Purchase Agreement, the Company sold (1) the Private Placement Shares and shares of Series A Preferred
Stock, or a combination thereof, at an effective purchase price of $0.011 per Private Placement Share or share of Common Stock underlying
such shares of Series A Preferred Stock and (2) Private Placement Warrants to the Private Placement Investors in connection with the Private
Placement. The Company received aggregate gross proceeds of $5,025,000 in connection with the first closing of the Private Placement on
August 14, 2023 and an additional $200,000 in connection with a second closing of on September 13, 2023.
The Company and the Private
Placement Investors also entered into a Registration Rights Agreement, pursuant to which the Company agreed to register, among other registrable
securities, on Form S-1 (or, if the Company is then eligible, on Form S-3) with the SEC: (i) the Private Placement Shares, (ii) Conversion
Shares issuable in connection with the Purchase Agreement, (iii) the shares of Common Stock underlying the Private Placement Warrants
issued to the Private Placement Investors, and (iv) the shares of Common Stock and Conversion Shares underlying the shares of Series A
Preferred Stock issued to the investors in the December 2021 Transaction in connection with the Share Exchange. The Company filed a registration
statement on Form S-1 covering the aforementioned securities with the SEC on February 9, 2024.
Going Concern
We have experienced net losses
and negative cash flows from operations since our inception. As of December 31, 2023, we had cash of approximately $3.3 million, positive
working capital of $3.2 million, an accumulated deficit of approximately $13.7 million and used cash in operations during the twelve months
ended December 31, 2023 of approximately $2.4 million. The Company does not currently have sufficient available liquidity to fund its
operations for at least the next 12 months. Such factors raise substantial doubt about our ability to sustain operations for at least
one year from the issuance of the audited financial statements included in this Annual Report. The accompanying financial statements do
not include any adjustments related to the recoverability and classification of asset amounts or the classification of liabilities that
might be necessary should we be unable to continue as a going concern.
In response to these conditions
and events, we are evaluating various financing strategies to obtain sufficient additional liquidity to meet our operating and capital
requirements for the next twelve months following the date of this Annual Report. The potential sources of financing that we are evaluating
include one or any combination of secured or unsecured debt, convertible debt and equity in both public and private offerings. We also
plan to finance near-term operations with our cash on hand, as well as by exploring additional ways to raise capital. There is no assurance
we will manage to raise additional capital or otherwise increase cash flows, if required. The sources of financing described above that
could be available to us and the timing and probability of obtaining sufficient capital depend, in part, on our further developing and
commercializing the Platform and on future capital market conditions. If our current assumptions regarding the pace of such development
are incorrect, or if there are any other changes or differences in our current assumptions that negatively impact our financing strategy,
we may have to reduce expenditures or significantly delay, scale back or discontinue the development or commercialization of the Platform.
Results of Operations
Comparison of the Years Ended December 31, 2023 and 2022
The following table sets forth
the results of our operations for the years ended December 31, 2023 and 2022:
| | |
Years Ended December 31, | | |
| | |
| |
| | |
2023 | | |
2022 | | |
Change $ | | |
Change % | |
| Research and development | |
$ | 1,004,443 | | |
$ | 850,680 | | |
$ | 153,763 | | |
| 18 | % |
| General and administrative | |
$ | 1,120,426 | | |
$ | 814,653 | | |
$ | 305,773 | | |
| 38 | % |
| Share-based compensation | |
$ | 2,253,793 | | |
$ | 19,908 | | |
$ | 2,233,885 | | |
| 11221 | % |
| Financial income, net | |
$ | 90,147 | | |
$ | 1,742 | | |
$ | 88,405 | | |
| 5075 | % |
| Gain on retirement of royalty accrual | |
$ | 720,632 | | |
$ | - | | |
$ | 72,632 | | |
| 100 | % |
Research and Development Expenses
Research and development expenses
were $1,004,443 for the year ended December 31, 2023, compared to $850,860 for the year ended December 31, 2022. The increase in research
and development expenses of $153,763 or 18%, was primarily due to increased outsourced consulting activities relating to the development
of our new product and an increase in salaries.
General and Administrative Expenses
General and administrative
expenses were $1,120,426 for the year ended December 31, 2023, compared to $814,653 for the year ended December 31, 2022. The increase
in general and administrative expenses of $305,773 or 38%, was primarily due to an increase in professional fees relating to public company
compliance following the Share Exchange as well as an increase in salaries and related expenses during the year ended December 31, 2023.
Share-based Compensation Expenses
Share-based compensation expenses
were $2,253,793 for the year ended December 31, 2023, compared to $19,908 for the year ended December 31, 2022. The increase in general
and administrative expenses of $2,233,885 or 11221%, was primarily due to the modification of the outstanding stock options as part of
the Share Exchange.
Financial (Income) Expenses, Net
Financial income was $90,147
for the year ended December 31, 2023, compared to $1,742 of income for the year ended December 31, 2022. The increase in financial income,
net of $88,405 or 5075%, was primarily due to exchange rate differences resulting from the translation of NIS based assets and liabilities
to US dollars.
Gain on retirement of royalty accrual
Gain on retirement of royalty
accrual was $720,632 for the year ended December 31, 2023, which resulted from the retirement of outstanding royalty accrual due to the
expiration of the relevant Statute of Limitations. There was not retirement of royalty accrual for the year ended December 31, 2022.
Liquidity and Capital Resources
Sources of Liquidity
We do not have revenues to
fund operations. We anticipate that we will continue to incur significant losses as it continues to develop its product. Historically,
our primary source of cash has been proceeds from the sale of equity instruments. We raised $5.225 million through a private placement
sale of shares to new investors concurrent with the Share Exchange. We intend to spend approximately $2.5 million over the next 18 months
on software and hardware development as well as the accompanying regulatory approvals and IP protection associated with such software
and hardware projects.
We will need to raise additional
capital to fund operating losses and grow our operations. There can be no assurance however that we will be able to raise additional capital
when needed, or at terms deemed acceptable, if at all. Such factors raise substantial doubt about our ability to sustain operations for
at least one year from the issuance of the audited financial statements included in this Annual Report. The accompanying financial statements
do not include any adjustments related to the recoverability and classification of asset amounts or the classification of liabilities
that might be necessary should we be unable to continue as a going concern. For additional information, see the section above titled “MD&A—Going
Concern.”
Private Placement
See the section above titled
“MD&A—Our Company—Private Placement.”
Cash Flows for the Years Ended December 31, 2023 and 2022
| | |
Years ended December 31, | |
| | |
2023 | | |
2022 | |
| Cash provided (used) in | |
| | |
| |
| Operating activities | |
$ | (2,362,386 | ) | |
$ | (1,517,178 | ) |
| Investing activities | |
| 17,966 | | |
| - | |
| Financing activities | |
| 4,653,204 | | |
| - | |
| Net increase (decrease) in cash and cash equivalents | |
$ | 2,308,784 | | |
$ | (1,517,178 | ) |
Cash Used in Operating Activities
Net cash used in operating activities was $2,362,386 for the year ended
December 31, 2023 as compared to $1,517,178 for the year ended December 31, 2022. The amount for the year ended December 31, 2023 primarily
consisted of a net loss of $3,567,883 offset by non-cash charges of $1,533,831 (including: Share-based compensation expense of $2,253,793,
gain on retirement of royalty accrual of $720,632 and depreciation expense of $670), and a decrease in operating assets and liabilities
excluding cash of $328,334. The amount for the year ended December 31, 2022 primarily consisted of a net loss of $1,683,499, partially
offset by non-cash charges of $20,578 (including: Share-based compensation expense of $ 19,908 and depreciation of $670), and a decrease
in operating assets and liabilities excluding cash of $145,743.
Cash Provided by Investing Activities
During the year ended December
31, 2023, net cash provided by investing activities was $17,966 relating to the cash received in the Share Exchange. During the year ended
December 31, 2022, there was no cash provided by or used in investing activities.
Cash Provided by Financing Activities
During the year ended December
31, 2023, net cash provided by financing activities was $4,653,204 relating to the net proceeds from the private placement raise. During
the year ended December 31, 2022, there was no cash provided by or used in financing activities.
Effects of Inflation
Management does not believe
that inflation has had a material impact on our business, sales, or operating results during the periods presented.
Off-Balance Sheet Arrangements
We currently do not have any
off-balance sheet arrangements or financing activities with special-purpose entities.
Critical Accounting Policies and Use of Estimates
The SEC defined a company’s
critical accounting policies as the ones that are most important to the portrayal of our financial condition and results of operations
and which require us to make our most difficult and subjective judgments, often as a result of the need to make estimates of matters that
are inherently uncertain.
Based on this definition,
we have identified the critical accounting policies and judgments addressed below. We also have other key accounting policies that are
significant to understanding our results.
Research and Development
We expense all research and
development costs as they are incurred. Research and development includes expenditures in connection with in-house research and development
salaries and staff costs, consulting fees, as well as proprietary products and technology.
Use of Estimates
The preparation of financial
statements in conformity with U.S. GAAP requires management to make estimates or assumptions that affect the reported amounts of assets
and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of
revenue and expenses during the reporting periods. Actual results could vary from those estimates. Management utilizes various other estimates,
including but not limited to accrued royalties, estimated lives of long-lived assets, the valuation of stock-based compensation, the valuation
allowance for deferred tax assets and other contingencies. The results of any changes in accounting estimates are reflected in the financial
statements in the period in which the changes become evident. Estimates and assumptions are reviewed periodically, and the effects of
revisions are reflected in the period that they are determined to be necessary.
Recent Accounting Pronouncements
In October 2021, the FASB
issued ASU 2021-07-Compensation-Stock Compensation (Topic 718): Determining the Current Price of an Underlying Share for Equity-Classified
Share-Based Awards. The measurement objective in Topic 718 for share-based awards is fair value based, and the current price input is
measured at fair value. This input is used in determining an award’s fair value. The practical expedient in this Update allows a
non-public entity to determine the current price of a share underlying an equity classified share-based award using the reasonable application
of a reasonable valuation method. The practical expedient in this Update is effective prospectively for all qualifying awards granted
or modified during fiscal years beginning after December 15, 2021, and interim periods within fiscal years beginning after December 15,
2022. Early application, including application in an interim period, is permitted for financial statements that have not yet been issued
or made available for issuance as of October 25, 2021. The implementation of this standard did not have a material effect on our financial
statements.
Item 7A. Quantitative and Qualitative Disclosures About Market
Risk.
Not applicable.
Item 8. Financial Statements and Supplementary Data.
Our audited consolidated financial
statements as of and for the years ended December 31, 2023, and December 31, 2022, are included beginning on page F-1 immediately
following the signature page to this Annual Report. See Item 15 for a list of the financial statements included herein.
Item 9. Changes in and Disagreements With Accountants on Accounting
and Financial Disclosure.
Not applicable.
Item 9A. Controls and Procedures.
Disclosure Controls and Procedures
Our principal executive officer
and principal financial officer, after evaluating the effectiveness of our disclosure controls and procedures (as defined in the Exchange
Act) Rule 13a-15(e) and 15d-15(e)) as of the end of the period covered by this Annual Report, have concluded that, based on such evaluation,
our disclosure controls and procedures were not effective to ensure that information required to be disclosed by us in the reports that
we file or submit under the Exchange Act is recorded, processed, summarized and reported, within the time periods specified in the SEC’s
rules and forms, and is accumulated and communicated to our management, including our principal executive officer and principal financial
officer, as appropriate to allow timely decisions regarding required disclosure.
Internal Control over Financial Reporting
Management’s Annual Report on Internal Control over
Financial Reporting
Our management is responsible
for establishing and maintaining adequate internal control over financial reporting, as defined in Rules 13a-15(f) under the Exchange
Act. Internal control over financial reporting refers to the process designed by, or under the supervision of, our principal executive
officer and principal financial officer, and effected by our board of directors, management and other personnel, to provide reasonable
assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance
with GAAP, including those policies and procedures that: (i) pertain to the maintenance of records that, in reasonable detail, accurately
and fairly reflect our transactions and the disposition of our assets, (ii) provide reasonable assurance that transactions are recorded
as necessary to permit preparation of consolidated financial statements in accordance with GAAP and that receipts and expenditures are
being made only in accordance with authorizations of our management and board of directors, and (iii) provide reasonable assurance regarding
prevention or timely detection of unauthorized acquisition, use, or disposition of our assets that could have a material effect on the
consolidated financial statements.
Because of its inherent limitations,
internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness
to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of
compliance with policies and procedures may deteriorate.
Management evaluated the effectiveness of our internal control over
financial reporting based on the 2013 framework in Internal Control — Integrated Framework issued by the Committee of Sponsoring
Organizations of the Treadway Commission. Based on this evaluation management concluded that our internal control over financial reporting
was not effective as of December 31, 2023.
During the year ended December
31, 2023, management identified the following weaknesses, which were deemed to be material weaknesses in internal controls:
| |
1. |
Due to the size of the Company and available resources, there are limited personnel to assist with the accounting and financial reporting function, which results in a lack of segregation of duties. |
| |
2. |
The Company does not have Chief Financial Officer that can oversee day to day operations and the financial reporting function. |
This Annual Report does not
include an attestation report of our registered public accounting firm regarding internal control over financial reporting. Management’s
report was not subject to attestation by our registered public accounting firm pursuant to the Dodd-Frank Wall Street Reform and Consumer
Protection Act, which permits us to provide only management’s report in this Annual Report.
Changes in Internal Controls over Financial Reporting
There were no changes in our
internal control over financial reporting that occurred during our last fiscal quarter ended December 31, 2023 that have materially affected,
or are reasonably likely to affect, our internal control over financial reporting.
Item 9B. Other Information
Not applicable.
Item 9C Disclosure Regarding Foreign Jurisdictions That Prevent
Inspections.
Not applicable.
PART III
Item 10. Directors, Executive Officers and Corporate Governance.
Officers and Directors
The following persons became
our directors and executive officers on August 14, 2023 and hold the positions set forth opposite their respective names as of March 29,
2024:
|
Name |
|
Age |
|
Position |
| Eliyahu (Lee) Haddad |
|
57 |
|
Chief Executive Officer and Director |
| Moshe Shvets |
|
59 |
|
Chief Technology Officer and Director |
| Chaim Hurvitz |
|
63 |
|
Director and Chairman of the Board |
| Chaim Ravad |
|
58 |
|
Director |
| Yehuda Englander |
|
43 |
|
Director |
Directors and Executive Officers
Information concerning our directors and executive officers is set
forth below. The biographical description of each director includes the specific experience, qualifications, attributes and skills that
led the Board to conclude that such person should serve as a director.
Eliyahu (Lee) Haddad
Mr. Haddad has served as our
Chief Executive Officer and director since December 2021. Mr. Haddad is a multi-disciplinary finance and technology expert, with extensive
senior level operational experience in raising capital, growing complex business models, and guiding startups and later stage companies
to successful exits. Prior to his employment at Dror, Mr. Haddad served as Chief Executive Officer of HFT Investments from 2007 through
2021. He also served as a Senior Adviser at Exceed Talent Capital between 2019 and 2023. Over the course of his 30-year career, Mr. Haddad
has structured and managed a number of technology and media transactions valued at an aggregate of over $85 billion, including $250 million
in transactions within the Israeli high-tech space in AI, medical technology, and cybersecurity. Mr. Haddad received a bachelor’s
degree in economics and philosophy from Columbia University, where he was the recipient of the National Science Foundation Award in Theoretical
Physics and started his career in the M&A subgroup of Morgan Stanley’s media and technology group for several years. We believe
that Mr. Haddad’s extensive business experience qualifies him to serve as a member of our Board.
Moshe Shvets
Mr. Shvets has served as a
director and as our Chief Technology Officer since July 20, 2020. Mr. Shvets has also served as a Senior Vice President since December
1, 2021. Mr. Shvets is a seasoned senior executive with 25 years of experience in building companies with over €250M yearly revenues
that involve complex instrumentation & processes, regulation, software, and global infrastructure. Prior to joining Dror, Mr. Shvets
founded and served as a director of BiSec Ltd. from 2015 to 2018. Mr. Shvets has also served as president of OAO Belzan from 2011 to 2013,
and president of OAO DZV from 2011 to 2014. Before joining the management team, Mr. Shvets was one of the investors in our Company. Mr.
Shvets received a bachelor’s degree from Saint Petersburg State University in Aerospace Instrumentation in 1999. We believe that
Mr. Shvets’s extensive experience commercializing new technologies qualifies him to serve as a member of our Board.
Chaim Hurvitz
Mr. Hurvitz has served as
a director and Chairman of our Board since January 17, 2012. Mr. Hurvitz has founded and has served as a chief executive office of C.H.
Health, a healthcare focused venture capital firm since May 2011. His investments through CH Health have included several successful exits
including the NASDAQ IPOs of Galmed Pharmaceuticals Ltd. (NASDAQ: GLMD) (“Galmed”) and UroGen Pharma Ltd. (NASDAQ: URGN) (“UroGen”).
He was previously a member of Teva’s senior management, serving as the President of Teva International Group from 2002 through 2010,
Vice-President of Israeli Pharmaceutical Sales from 1999 through 2002 and President and CEO of Teva Pharmaceuticals Europe from 1992 through
1999. Mr. Hurvitz presently serves the chairman of Univo Pharmaceuticals Ltd., the chairman of Shirat Hachaim Ltd., a director of Celexir,
a director of Genoscience Pharma S.A.S., and has previously served as the chairman CTG Weld Limited, the chairman of PolyPid Ltd. (NASDAQ:
PYPD), as the chairman of Galmed, as a director of UroGen, and as a director of Teva Pharmaceuticals Industries Ltd. (NYSE: TEVA). Mr
Hurvitz is also a member of management of the Manufacturers Association of Israel and Head of its Pharmaceutical branch. Mr. Hurvitz received
a B.A. in political science and economics from Tel Aviv University in 1985. We believe that Mr. Hurvitz’s extensive management experience
in the healthcare industry qualifies him to serve as a member of our Board.
Chaim Ravad
Mr. Ravad has served as a
director since February 2015. Mr. Ravad has experience in food catering and real estate industries. In his capacity as our director, Mr.
Ravad has served as a major contributor to the development of Dror’s teeth straightening product from its early stages and until
receipt of FDA and CE approval and has in the past successfully assisted in securing private investments in our Company. Mr. Ravad is
a graduate of Hebron Yeshiva.
Yehuda Englander
Mr. Englander has served as
a director since December 6, 2021. Mr. Englander is a co-founder of YYE ALEY SHLECHT ASSETS LTD. and YE RUT Finance Ltd. Prior to that,
Mr. Englander led Yehuda Englander Finance Advisory Ltd. for four years. Mr. Englander received a B.A. in Accounting from Lev Academic
Center at Jerusalem College of Technology. We believe that Mr. Englander’s extensive investment experience qualifies him to serve
as a member of our Board.
Involvement in Certain Legal Proceedings
None of the members of the
Board or our executive officers has, in the last ten years, been involved in any legal proceeding of the type described under Item 103I(2)
or Item 401(f) of Regulation S-K.
Director Independence
Our Common Stock is quoted
on the OTC Pink Market operated by the OTC Markets Group Inc., which does not have director independence requirements. We also have not
established our own definition for determining whether our director and nominees for directors are “independent” nor have
we adopted any other standard of independence employed by any national securities exchange.
We expect our Board, in the
future, to appoint an audit committee, nominating committee and compensation committee, and to adopt charters relative to each such committee.
We intend to appoint such persons to committees of the Board as are expected to be required to meet the corporate governance requirements
imposed by a national securities exchange, although we are not required to comply with such requirements until we elect to seek a listing
on a national securities exchange. In addition, we intend that a majority of our directors will be independent directors, of which at
least one director will qualify as an “audit committee financial expert,” within the meaning of Item 407(d)(5) of Regulation
S-K, as promulgated by the SEC. We do not currently have an “audit committee financial expert” since we currently do not have
an audit committee in place.
Family Relationships
There are no family relationships
among our directors or executive officers.
Delinquent Section 16(a) Reports
Section 16(a) of the Exchange
Act requires our directors and executive officers and each person who owns more than ten percent of a registered class of our equity securities
(collectively, “Reporting Persons”) to file with the SEC initial reports of ownership and reports of changes in ownership
of our Common Stock and our other equity securities. Reporting Persons are required by SEC regulation to furnish us with copies of all
Section 16(a) forms that they file. Based solely on our review of the copies of the forms received by us during the fiscal year ended
December 31, 2023 and written representations that no other reports were required, we believe that each person who, at any time during
such fiscal year, was a director, officer or beneficial owner of more than ten percent of our common stock complied with all Section 16(a)
filing requirements during such fiscal year with the following exceptions: (1) Mr. Haddad, Mr. Englander, Mr. Shvets, and Mr. Ravad
filed Form 3s on September 27, 2023, disclosing their becoming Reporting Persons in connection with the closing of the Share Exchange
on August 14, 2023 and (2) Mr. Haddad, Mr. Hurvitz, Mr. Shvets, and Mr. Ravad filed Form 4s on September 27, 2023, disclosing
the acquisition of shares of Series A Preferred Stock, and warrants to purchase Common Stock on August 14, 2023.
Insider Trading Arrangements and Policies; Code of Ethics
We intend to adopt insider
trading policies and procedures and a code of ethics that will apply to our officers, directors and employees, including our principal
executive officer and principal accounting officer, but have not done so to date due to our relatively small size. We intend to adopt
written insider trading policies and procedures and a written code of ethics in the near future.
Director Nominations by Security Holders
Our Second Amended and Restated Bylaws (the “Bylaws”) contain
provisions that address the process by which a stockholder may nominate an individual to stand for election to our board of directors
(the “Board”). To recommend a nominee for election to the Board, a stockholder must submit his or her recommendation to our
Secretary at our corporate offices at Shatner Street 3, Jerusalem, Israel. Such nomination must satisfy the notice, information and consent
requirements set forth in our Bylaws and must be received by us prior to the date set forth under “Submission of Future Stockholder
Proposals” in our most recent proxy statement. A stockholder’s recommendation must be accompanied by the information with
respect to stockholder nominees as specified in our Bylaws, including among other things, the name, age, address and occupation of the
recommended person, the proposing stockholder’s name and address, the ownership interests of the proposing stockholder and any beneficial
owner on whose behalf the nomination is being made (including the number of shares beneficially owned, any hedging, derivative, short
or other economic interests and any rights to vote any shares) and any material monetary or other relationships between the recommended
person and the proposing stockholder and/or the beneficial owners, if any, on whose behalf the nomination is being made.
Item 11. Executive Compensation.
The following table sets forth
summary compensation information for the respective fiscal years. For the purpose of this prospectus, our “named executive officers”
or “NEOs” are our principal executive officer (“PEO”), Mr. Haddad, and our sole non-PEO executive officer, Mr.
Shvets. We provide a description of the employment arrangements with Mr. Haddad and Mr. Shvets, below under “Employment Agreements.”
The following table includes all compensation earned by our named executive officers for the respective period, regardless of whether
such amounts were actually paid during the period.
This discussion may contain
forward-looking statements that are based on our current plans, considerations, expectations and determinations regarding future compensation
programs.
Summary Compensation Table
The following table sets forth
information concerning the compensation of our named executive officers for the fiscal years indicated below.
| Name and principal position |
|
Year |
|
|
Salary
($)(1) |
|
|
Bonus
($) |
|
|
Stock awards
($) |
|
|
Option awards
($)(2) |
|
|
Nonequity incentive plan
compensation
($) |
|
|
Nonqualified
deferred
compensation
earnings
($) |
|
|
All other
compensation
($) |
|
|
Total
($) |
|
| Eliyahu (Lee) Haddad |
|
|
2023 |
|
|
|
419,962 |
|
|
|
— |
|
|
|
— |
|
|
|
2,506,941 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
2,926,903 |
|
| (Chief Executive Officer and Director) |
|
|
2022 |
|
|
|
343,456 |
|
|
|
20,252 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
363,708 |
|
| Moshe Shvets |
|
|
2023 |
|
|
|
313,770 |
|
|
|
— |
|
|
|
— |
|
|
|
1,504,145 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
1,817,915 |
|
| (Chief Technology Officer) |
|
|
2022 |
|
|
|
228,743 |
|
|
|
20,203 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
248,946 |
|
| (1) | Compensation amounts received in non-U.S. currency have been
converted into U.S. dollars using the average exchange rate for the applicable year. The average exchange rate for 2023 was 3.690 NIS
per dollar and the average exchange rate for 2022 was 3.359 NIS per dollar. |
| (2) | In accordance with SEC rules, this column reflects the aggregate
fair value of the option awards granted during the respective fiscal year computed as of their respective grant dates in accordance with
Financial Accounting Standard Board Accounting Standards Codification Topic 718 for share-based compensation transactions. The assumptions
made in the valuation of the share-based payments are contained in Note 2 to our financial statements included in this prospectus. |
Narrative Disclosure Regarding Summary Compensation Table
Our Board reviews compensation
annually for all employees, including named executive officers. In making compensation determinations, the Board considers compensation
for comparable positions in the market and with peer companies, the historical compensation levels of executives, individual performance
as compared to the board’s expectations and objectives, the board’s desire to motivate employees to achieve short- and long-term
results that are in the best interests of our stockholders and a long-term commitment to our Company.
Annual Base Salaries
Base salaries for the executive
officers are initially established through arm’s-length negotiations at the time of the executive officer’s hiring, taking
into account such executive officer’s qualifications, experience, the scope of his or her responsibilities and competitive market
compensation paid by other companies for similar positions within the industry and geography. Base salaries are reviewed periodically,
typically in connection with our annual performance review process, and adjusted from time to time to realign salaries with market levels
after taking into account individual responsibilities, performance and experience. In making decisions regarding salary increases, we
may also draw upon the experience of members of the Board with executives at other companies.
Bonus Compensation
For 2023, our named executive
officers are not eligible to receive a discretionary annual bonus based on individual and company performance. During fiscal year 2022,
Messrs. Haddad and Shvets earned discretionary bonuses as set forth in the Summary Compensation Table above.
Equity-Based Incentive Awards
Our equity-based incentive
awards are designed to align our interests and those of our stockholders with those of our employees and consultants, including our named
executive officers. We have historically used stock options as incentives for long-term compensation to the named executive officers as
the return on such awards is tied to an increase in our stock price. We may grant equity awards at such times as our Board determines
appropriate in their discretion. Additional grants may occur periodically in order to incentivize executives with respect to achieving
certain corporate goals or to reward them for exceptional performance. See “Outstanding Equity Awards at Fiscal Year-End”
below for additional information regarding outstanding equity awards held by our named executive officers as of December 31, 2023.
Employment Agreements
Eliyahu (Lee) Haddad
On December 6, 2021, Private
Dror entered into an employment agreement (the “Haddad Employment Agreement”) with Mr. Haddad to serve as Private Dror’s
chief executive officer. Pursuant to this employment agreement, Mr. Haddad is entitled to a monthly salary (including all social benefit
payments provided under Israeli law) of $22,256. Mr. Haddad is also entitled to an annual bonus based on achievement of objectives and
Board’s approval. In connection with his employment agreement, Mr. Haddad was granted options to purchase five percent (5%) of our
fully diluted Ordinary Shares issued and issuable on the date of the employment agreement, which options shall vest in three tranches
on the first, second, and third anniversary of the date of the employment agreement. The options are subject to accelerated vesting upon
the achievement by us of certain performance milestones. We cannot terminate Mr. Haddad’s employment not for “cause,”
and in circumstances constituting “cause,” we may terminate the agreement effective immediately. Mr. Haddad can terminate
the agreement for convenience upon 30 days written notice, and may terminate the agreement immediately for “good reason.”
If Mr. Haddad’s employment is terminated without cause, or Mr. Haddad resigns for good reason, he is entitled to twelve month’s
salary.
Following the closing of the
Share Exchange, the Board appointed Mr. Haddad to the office of Chief Executive Officer on the terms of the Haddad Employment Agreement.
Moshe Shvets
On January 26, 2022, Private
Dror entered into an employment agreement (the “Shvets Employment Agreement”) with Mr. Shvets to serve as Private Dror’s
Senior Vice President, effective as of December 1, 2021. Mr. Shvets was named Chief Technology Officer as of July 20, 2020. Pursuant to
his employment agreement, Mr. Shvets is entitled to a monthly gross salary of NIS 32,000. Mr. Shvets is also entitled to certain social
and fringe benefits as set forth in the employment agreement. In connection with his employment agreement, Mr. Shvets was granted options
to purchase three percent (3%) of our fully diluted Ordinary Shares issued and issuable on the date of the employment agreement, which
options shall vest in three tranches on the first, second, and third anniversary of the date of the employment agreement. The options
are subject to accelerated vesting upon the achievement by us of certain performance milestones. Mr. Shvets’ employment can be terminated
by either party for convenience upon 30 days written notice.
Following the closing of the
Share Exchange, the Board appointed Mr. Shvets to the office of Chief Technology Officer on the terms of the Shvets Employment Agreement.
Outstanding Equity Awards at Fiscal Year-End
The following table presents
information regarding outstanding equity awards held by our named executive officers as of December 31, 2023. Information in this table
has been adjusted to give pro forma effect to the Share Exchange.
| |
Option awards |
| Name | |
Number of securities underlying unexercised options (#) exercisable | | |
Number of securities underlying unexercised options (#) unexercisable | | |
Equity incentive plan awards: Number of securities underlying unexercised unearned options (#) | | |
Option exercise price ($) | | |
Option expiration date |
Eliyahu
(Lee) Haddad
(Chief Executive Officer and Director) | |
| 63,977,143 | (1) | |
| 31,988,572 | (1) | |
| — | | |
$ | 0.0038480 | | |
August 14, 2033 |
Moshe Shvets
(Chief Technology Officer and Director) | |
| 38,385,796 | (2) | |
| 19,192,898 | (2) | |
| — | | |
$ | 0.0038480 | | |
August 14, 2033 |
| (1) | On December 6, 2021, Mr. Haddad was granted options to purchase
up to 26,097 ordinary shares of Private Dror at an exercise price of $14.15 per ordinary share. In connection with the Share Exchange,
these options were exchanged for options to purchase up to 95,965,715 shares of Common Stock at an exercise price of approximately $0.0038480
per share. These options vest in three tranches on the first, second, and third anniversary of the employment start date. The options
are subject to accelerated vesting upon the achievement by us of certain performance milestones. |
| (2) | On December 1, 2021, Mr. Shvets was granted options to purchase
up to 15,658 ordinary shares of Private Dror at an exercise price of $14.15 per ordinary share. In connection with the Share Exchange,
these options were exchanged for options to purchase up to 57,578,694 shares of Common Stock at an exercise price of approximately $0.0038480
per share. These options vest in three tranches on the first, second, and third anniversary of the employment start date. The options
are subject to accelerated vesting upon the achievement by us of certain performance milestones. |
Equity Incentive Plans
2021 Share Incentive Plan
Prior to the Share Exchange,
Private Dror adopted the Dror 2021 Share Incentive Plan (the “2021 Plan”), which provides for the granting of stock options,
restricted stock, restricted stock units, and other stock-based awards to employees, directors, officers, consultants, and advisors of
Private Dror or its affiliates. Under the 2021 Plan, 51,482 ordinary shares of Private Dror were initially reserved for issuance as awards,
and stock options covering up to 44,365 ordinary shares of Private Dror (which were exchanged for stock options covering approximately
163,142,084 shares of Common Stock in connection with the Share Exchange) are outstanding as of the date hereof. No other type of equity
award is currently outstanding under the 2021 Plan. As further described below, upon the closing of the Share Exchange, any stock options
outstanding under the 2021 Plan were converted into stock options under the Dror Ortho-Design, Inc. 2023 Long-Term Incentive Plan (the
“2023 Plan”). The 2021 Plan is filed as Exhibit 10.9 to the registration statement on Form S-1 of which this prospectus forms
a part.
2023 Long-Term Incentive Plan
On August 14, 2023, our Board
adopted the 2023 Plan. Under the 2023 Plan, we reserved 235,958,571 shares of our Common Stock for issuance as awards to our key employees,
key contractors, and non-employee directors and those of our subsidiaries, of which 100% may be delivered pursuant to incentive stock
options. A form of the 2023 Plan is filed as Exhibit 10.10 to the registration statement on Form S-1 of which this prospectus forms a
part.
The 2023 Plan currently consists
of the primary plan document that governs all awards granted under the 2023 Plan for eligible U.S. employees, contractors, and non-employee
directors who are subject to U.S. income taxation and a sub-plan annex designated for the purpose of grants of equity awards to eligible
Israeli employees, officers, and contractors of the Company and its affiliates who are subject to Israeli income taxation.
Upon the closing of the Share
Exchange, we became the sponsor of the 2021 Plan, and all outstanding stock option awards previously granted under the 2021 Plan will
be converted into awards under the 2023 Plan. Thus, all outstanding options to purchase ordinary shares of Dror (which are converted into
options to purchase shares of Common Stock of the Company pursuant to the Share Exchange Agreement, as amended) were converted to options
to purchase shares of Common Stock of the Company.
The purpose of the 2023 Plan
is to provide an incentive to attract and retain the services of key employees, key contractors, and non-employee directors of the Company
and its subsidiaries and to provide such persons with a proprietary interest in the Company through the granting of awards. The 2023 Plan
will be administered by our Board or a committee of the Board (the “Committee”) consisting of two or more members. At any
time there is no Committee to administer the 2023 Plan, any reference to the Committee is a reference to the Board. The Committee will
determine the persons to whom awards are to be made, determine the type, size and terms of awards, interpret the 2023 Plan, establish
and revise rules and regulations relating to the 2023 Plan, and make any other determinations that it believes necessary for the administration
of the 2023 Plan. The Committee may delegate certain duties to one or more officers of the Company as provided in the 2023 Plan. Unless
terminated earlier by our Board, the 2023 Plan will expire on August 14, 2033. No awards may be made under the 2023 Plan after its expiration
date, but awards made prior thereto may extend beyond that date.
The 2023 Plan provides for
the granting of incentive stock options, nonqualified stock options, stock appreciation rights, restricted stock, restricted stock units,
performance awards, dividend equivalent rights, and other awards which may be granted singly, in combination, or in tandem, and which
may be paid in cash or shares of the Company’s Common Stock. Awards granted pursuant to the 2023 Plan will be evidenced by a written
award agreement. The Committee will determine the terms of each award at the time of grant, including, without limitation, the number
of shares subject to such award, the term of the award, the exercise price to be paid for the award (if applicable), the vesting and forfeiture
conditions, the methods by or forms in which shares will be delivered to participants, the price to be paid for the award (if any), and
any other terms and conditions applicable to such award.
To date, no awards have been
granted pursuant to the 2023 Plan, other than the awards that were previously granted pursuant to the 2021 Plan and will be converted
into an award under the 2023 Plan, as described above.
The Board may, at any time
and from time to time, without the consent of the participants, alter, amend, revise, suspend or discontinue the 2023 Plan in whole or
in part; provided, however, that (i) no amendment that requires shareholder approval in order for the 2023 Plan and any awards granted
thereunder to continue to comply with Sections 421 and 422 of the Internal Revenue Code of 1986, as amended (the “Code”) (including
any successors to such sections, or other applicable law) or any applicable requirements of any securities exchange or inter-dealer quotation
system on which the Company’s Common Stock is listed or traded, shall be effective unless such amendment is approved by the requisite
vote of the Company’s shareholders entitled to vote on the amendment; and (ii) unless required by law, no action by the Board regarding
amendment or discontinuance of the 2023 Plan may adversely affect any rights of any participant or obligations of the Company to any participant
with respect to any outstanding award under the 2023 Plan without the consent of the affected participant.
Commitments to Grant Stock Options
In addition to the stock option
awards to be granted in substitution of stock options currently outstanding under the 2021 Plan, we currently have a commitment to issue
options to purchase up to 0.5% of the outstanding shares of Common Stock to Mr. Haddad, contingent on the Company achieving certain market
capitalization targets. We anticipate issuing these options pursuant to the 2023 Plan at such time as the Company has a sufficient number
of authorized and unissued shares of Common Stock.
Director Compensation
The following table presents
the total compensation for each person who served as a non-employee member of our Board during the fiscal year ended December 31, 2023.
Other than as set forth in the table and described more follow below, and as set forth in the Summary Compensation Table with respect
to our employee directors, we did not pay any compensation to, reimburse any expense of, make any equity awards or non-equity awards to,
or pay any other compensation to any of the other members of our Board in 2023.
Name | |
Fees earned
or paid
in
cash
($) | | |
Stock
awards
($) | | |
Option
awards
($)(1) | | |
Non-equity
incentive plan
compensation
($) | | |
Nonqualified
deferred
compensation
earnings
($) | | |
All other
compensation
($) | | |
Total
($) | |
| Chaim Hurvitz | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | |
| Chaim Ravad (2) | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | |
| Yehuda Englander (3) | |
| 11,383 | | |
| — | | |
| 250,723 | | |
| — | | |
| — | | |
| — | | |
| 262,106 | |
| (1) | In accordance with SEC rules, this column reflects the aggregate
fair value of option awards granted during the fiscal year ended December 31, 2022, computed as of their respective grant dates in accordance
with Financial Accounting Standard Board Accounting Standards Codification Topic 718 for share-based compensation transactions. The assumptions
made in the valuation of the share-based payments are contained in Note 2 to our financial statements included in this prospectus. |
| (2) | On February 7, 2024, we entered into a consulting agreement (the
“Ravad Consulting Agreement”) with Mr. Ravad, pursuant to which, in consideration for certain services provided as a board
member, Mr. Ravad would receive a cash fee of $5,000 each month. The Ravad Consulting Agreement is terminable by either party upon 30 days
written notice to the other party, and it will terminate automatically once Mr. Ravad has received fees in the aggregate amount of $55,000. |
| (3) | On June 1, 2022, Private Dror entered into a consulting agreement
(the “Englander Consulting Agreement”) with Mr. Englander, pursuant to which, in consideration for certain financial and
strategic consulting services, Mr. Englander receives a cash fee of NIS 3,500 + VAT each month and was also granted with options to purchase
2,610 Ordinary Shares of Private Dror, which options were exchanged for options to purchase 9,597,675 shares of Common Stock in connection
with the Share Exchange and shall vest in three tranches on the first, second, and third anniversary of the date of the consulting agreement.
The options are subject to accelerated vesting upon an exit event. |
Effective as of February 7,
2024, we entered into the First Amendment to the Englander Consulting Agreement with Mr. Englander, which provided that Mr. Engalnder’s
monthly cash fee in respect of the services provided would be equal to $2,500 + VAT.
Item 12. Security Ownership of Certain Beneficial Owners and Management
and Related Stockholder Matters.
The following table sets forth
information regarding the beneficial ownership of Common Stock as of March 29, 2024:
| ● | each person, or group of affiliated persons, known by us to
beneficially own more than 5% of outstanding shares of any class of our voting securities; |
| ● | each of our named executive officers; and |
| ● | all directors and executive officers as a group. |
Unless otherwise indicated
below, beneficial ownership is determined according to the rules of the SEC, which generally provide that a person has beneficial ownership
of a security if he, she, or it possesses sole or shared voting or investment power over that security, including options and warrants
that are currently exercisable or exercisable within 60 days.
The beneficial ownership percentages
set forth in the following table are based on 495,454,546 shares of Common Stock and 10,463,363 shares of Preferred Stock, which are entitled
to cast an aggregate of 749,721,570 votes, outstanding as of March 29, 2024.
Name of Beneficial Owner (1) | |
Number of Shares of Common Stock Beneficially Owned | | |
Percent of Class | | |
Number of Shares of Series A Convertible Preferred Stock Beneficial Owned | | |
Percent of Class | | |
Percent of Voting Power(2) | |
| 5% Stockholders | |
| | |
| | |
| | |
| | |
| |
| Orin Hirschmann/AIGH (3) | |
| 49,588,407 | (4) | |
| 9.99 | % | |
| 3,054,544 | (5) | |
| 29.19 | % | |
| 4.11 | % |
| Congregation Ahavas Tzdokah Vchesed Inc. (6) | |
| 61,722,996 | (7) | |
| 12.46 | % | |
| — | | |
| — | | |
| 5.12 | % |
| Moshe Bodner | |
| 54,989,344 | (8) | |
| 9.99 | % | |
| 1,511,873 | | |
| 14.45 | % | |
| 4.56 | % |
| The Hewlett Fund (9) | |
| 45,453,150 | (10) | |
| 8.65 | % | |
| 301,804 | | |
| 2.88 | % | |
| 3.77 | % |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Directors and Named Executive Officers | |
| | | |
| | | |
| | | |
| | | |
| | |
| Eliyahu (Lee) Haddad | |
| 63,977,143 | (11) | |
| 11.44 | % | |
| 45,455 | | |
| * | | |
| * | |
| Moshe Shvets | |
| 54,989,344 | (12) | |
| 9.99 | % | |
| 691,621 | | |
| 6.61 | % | |
| 1.38 | % |
| Chaim Hurvitz | |
| 54,989,344 | (13) | |
| 9.99 | % | |
| 592,151 | | |
| 5.66 | % | |
| 4.56 | % |
| Chaim Ravad | |
| 54,989,344 | (14) | |
| 9.99 | % | |
| 2,150,946 | | |
| 20.56 | % | |
| 4.56 | % |
| Yehuda Englander | |
| 3,199,225 | (15) | |
| * | | |
| — | | |
| — | | |
| * | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| All Directors and Executive Officers as a Group (5 persons) | |
| 223,144,400 | | |
| 42.05 | % | |
| 3,480,172 | | |
| 33.26 | % | |
| 12.47 | % |
| * | Represents beneficial ownership of less than 1%. |
| (1) | Except as expressly noted in the footnotes below, beneficial
ownership has been determined in accordance with Rule 13d-3 under the Exchange Act. The amounts set forth in this table reflect the application
of various limitations on the exercise of certain warrants and the conversion of shares of Preferred Stock, including beneficial ownership
limitations.
Unless otherwise indicated below, the address for each beneficial owner listed is c/o Dror Ortho-Design, Inc., Shatner 3, Jerusalem,
Israel. |
| (2) | Stockholders are entitled to one vote per each share of Common
Stock owned. Stockholders are entitled to the number of votes per each share of Preferred Stock owned equal to the number of shares of
Common Stock into such share of Preferred Stock is convertible into pursuant to the Certificate of Designations, after giving effect
to beneficial ownership limitations. |
| (3) | Mr. Orin Hirschman has sole voting and dispositive power over
shares held by AIGH Investment Partners, LP (“AIGH LP”), and its affiliated entities, AIGH Investment Partners, LLC (“AIGH
LLC”), WVP Emerging Manager Onshore Fund, LLC – AIGH Series (“WVP-AIGH”), and WVP Emerging Manager Onshore Fund,
LLC – Optimized Equity Series (“WVP-OES”). The principal business address of Mr. Hirschman and each such entity is
6006 Berkeley Avenue, Baltimore, MD 21209. |
| (4) | Represents (1) 30,000,000 shares of Common Stock held by AIGH
LP, (2) 8,662,500 shares of Common Stock held by AIGH LLC, (3) 7,000,000 shares of Common Stock held by WVP-AIGH, (4) 3,000,000 shares
of Common Stock held by WVP-OES, and (5) 925,907 shares of Common Stock issuable upon the conversion of shares of Preferred Stock held
by such entities that are convertible within 60 days of March 29, 2024. |
| (5) | Represents (1) 1,600,000 shares of Preferred Stock held by AIGH
LP, (2) 954,543.85 shares of Preferred Stock held by AIGH LLC, (3) 400,000 shares of Preferred Stock held by WVP-AIGH, and (4) 100,000
shares of Preferred Stock held by WVP-OES. |
| (6) | Rabbi Nusyn Pinches Erlich has sole voting and dispositive power
over these shares. The address for Congregation Ahavas Tzdokah Vchesed Inc. is 1655 E 24th St, Brooklyn, NY 11229. |
| (7) | Represents shares of Common Stock. |
| (8) | Represents 54,989,344 shares of Common Stock issuable upon the
conversion of shares of Preferred Stock held by Mr. Bodner that are convertible within 60 days of March 29, 2024. |
| (9) | Martin Chopp has voting and dispositive power over the securities
held by The Hewlett Fund LP (“Hewlett”). Hewlett’s address is 100 Merrick Road, Suite 400W, Rockville Centre, NY 11570. |
| (10) | Represents (1) 15,272,727 shares of Common Stock held by Hewlett
and (2) 30,180,423 shares of Common Stock issuable upon the conversion of shares of Preferred Stock held by Hewlett that are convertible
within 60 days of March 29, 2024 |
| (11) | Represents 63,977,143 shares of Common Stock issuable upon the
exercise of options upon that are exercisable within 60 days of March 29, 2024. |
| (12) | Represents (1) 38,385,796 shares of Common Stock issuable upon
the exercise of options and (2) 26,141,712 shares of Common Stock issuable upon the conversion of shares of Preferred Stock held by Mr.
Shvets that are exercisable or convertible within 60 days of March 29, 2024. |
| (13) | Represents 54,989,344 shares of Common Stock issuable upon the
conversion of shares of Preferred Stock held by Shirat Hachaim Ltd. (“Shirat Hachaim”) that are convertible within 60 days
of March 29, 2024. Mr. Hurvitz is the sole owner of Shirat Hachaim and has sole voting and dispositive power over shares held by Shirat
Hachaim. |
| (14) | Represents 54,989,344 shares of Common Stock issuable upon the
conversion of shares of Preferred Stock held by Mr. Ravad that are convertible within 60 days of March 29, 2024. |
| (15) | Represents 3,199,225 shares of Common Stock issuable upon the
exercise of options held by Mr. Englander that are exercisable within 60 days of March 29, 2024. |
Item 13. Certain Relationships and Related Transactions, and Director
Independence.
In addition to the compensation
arrangements discussed under “Executive Compensation,” the following is a description of transactions since January 1, 2022
to which we have been a party, in which the amount involved exceeds or will exceed the lesser of $120,000 or one percent of the average
of the Company’s total assets at year-end for the last two completed fiscal years, and in which any of our directors, executive
officers or beneficial owners of more than 5% of our capital stock, or an affiliate or immediate family member thereof, had or will have
a direct or indirect material interest. We also describe below certain other transactions with our directors, executive officers and stockholders.
We believe that we have executed
all of the transactions set forth below on terms no less favorable to us than we could have obtained from unaffiliated third parties.
It is our intention to ensure that all future transactions between us and our officers, directors and principal stockholders and their
affiliates are approved by our audit committee, once it has been formed and its members appointed, and a majority of the members of our
Board, including a majority of the independent and disinterested members of our Board, and are on terms no less favorable to us than those
that we could obtain from unaffiliated third parties.
Indemnification Agreements and Directors’ and Officers’
Liability Insurance
We have entered into separate
indemnification agreements with our directors and executive officers, in addition to indemnification provided for in our Amended Charter
and our Bylaws. Each indemnification agreement provides for indemnification and advancement by the Company of certain expenses and costs
relating to claims, suits, or proceedings arising from service to the Company or, at its request, service to other entities to the fullest
extent permitted by applicable law. We also maintain directors’ and officers’ liability insurance.
Item 14. Principal Accountant Fees and Services.
Accounting Fees
Our independent registered public
accounting firm is Barzily and Co., CPA’s (PCAOB Firm ID No.: 2015) (“Barzily”) located in Jerusalem, Israel. From 2017
until October 18, 2023, our independent accountant was Sadler, Gibb & Associates, LLC (“Sadler”). The following table
presents fees for professional audit services rendered (i) by Barzily for the audit of our annual financial statements for the year ended
December 31, 2023 and the review of our quarterly financial statements for the third quarter of 2023, and (ii) by Sadler for the audit
of our annual financial statements for the year ended December 31, 2022 and the review of our quarterly financial statements for the first
and second quarters of 2023, and fees billed for other services rendered by Barzily and Sadler during those periods.
| | |
For the year ended
December 31, | |
| | |
2023 | | |
2022 | |
| Audit fees(1) | |
$ | 59,782 | | |
$ | 24,000 | |
| Audit-related fees(2) | |
$ | 7,939 | | |
$ | 19,000 | |
| Tax-related fees(3) | |
$ | 0 | | |
$ | 0 | |
| All other fees(4) | |
$ | 0 | | |
$ | 0 | |
| Total fees | |
$ | 67,721 | | |
$ | 43,000 | |
| (1) | Audit fees for 2023 primarily related to the audit of our annual consolidated
financial statements for the 2023 fiscal year, and the reviews of the financial statements included in our Quarterly Reports on Form 10-Q
or included in a Form 8-K for the 2023 fiscal year. Audit fees for 2022 primarily related to the audit of our annual consolidated financial
statements for the 2022 fiscal year, and the reviews of the financial statements included in our Quarterly Reports on Form 10-Q for the
2022 fiscal year (including direct engagement expenses). |
| (2) | Audit-related fees billed in 2023 included services performed relating
to the Share Exchange. Audit-related fees billed in 2022 included fees, if any, for assurance and related services by Sadler that were
reasonably related to the performance of the audit or review of our financial statements and were not reported under “audit fees.” |
| (3) | There were no tax-related fees billed in 2023 or 2022. |
| (4) | There were no other fees billed in 2023 or 2022. |
Audit Committee Pre-Approval Policy and Procedures
Our Board does not presently
have a separately designated standing audit committee. As such, the percentage of services set forth above in the categories audit-related
fees, tax-related fees, and all other fees that were approved by the Audit Committee pursuant to Rule 2-01(c)(7)(i)(C) (relating to the
approval of a de minimis amount of non-audit services after the fact but before completion of the audit) was 0%. The functions of an audit
committee are undertaken by our Board.
PART IV
Item 15. Exhibit and Financial Statement Schedules.
The following documents are
filed as part of this report:
(1) Financial Statements
(2) Financial Statement Schedules:
None.
(3) Exhibits:
See “Index to Exhibits”
for a description of our exhibits.
Item 16. Form 10–K Summary.
None.
Index
to Exhibits
| Exhibit
No. |
|
Description |
| 2.1 |
|
Share Exchange Agreement, dated July 5, 2023, by and among Dror Ortho-Design, Inc., Dror Ortho-Design Ltd., and certain shareholders of Dror Ortho-Design Ltd. (incorporated by reference to Exhibit 2.1 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 2.2 |
|
Amendment to the Share Exchange Agreement, dated August 14, 2023, by and among Dror Ortho-Design, Inc., Dror Ortho-Design Ltd., and certain shareholders of Dror Ortho-Design Ltd. (incorporated by reference to Exhibit 2.2 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 3.1 |
|
Amended and Restated Certificate of Incorporation of Dror Ortho-Design, Inc. (incorporated by reference to Exhibit 3.1 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 3.2 |
|
Certificate of Designations of Preferences, Rights and Limitations of Series A Convertible Preferred Stock (incorporated by reference to Exhibit 3.2 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 3.3 |
|
Certificate of Correction to the Certificate of Designation of Preferences, Rights and Limitations of Series A Convertible Preferred Stock of Dror Ortho-Design, Inc. (incorporated by reference to Exhibit 3.1 to the Current Report on Form 8-K, filed with the Commission on November 14, 2023) |
| 3.4 |
|
Amended and Restated Certificate of Incorporation of Dror Ortho-Design, Inc. (incorporated by reference to Exhibit 3.1 to the Current Report on Form 8-K, filed with the Securities and Exchange Commission on January 4, 2024) |
| 3.5 |
|
Amended and Restated Bylaws (incorporated by reference to Exhibit 3.5 to the Current Report on Form 8-K, filed with the Commission on March 1, 2007) |
| 3.6 |
|
Second Amended and Restated Bylaws (incorporated by reference to Exhibit 3.1 to the Current Report on Form 8-K/A, filed with the Commission on November 14, 2023) |
| 4.1 |
|
Form of Class A Common Stock Purchase Warrant (incorporated by reference to Exhibit 4.1 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 4.2* |
|
Description of Securities |
| 10.1+ |
|
Employment Agreement, dated December 6, 2021, between Dror Ortho-Design Ltd. and Eliyahu (Lee) Haddad (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 10.2+ |
|
Employment Agreement, dated January 26, 2022, between Dror Ortho-Design Ltd. and Moshe Shvets (incorporated by reference to Exhibit 10.2 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 10.3+ |
|
Indemnification Agreement, dated December 6, 2021, between Dror Ortho-Design Ltd. and Eliyahu (Lee) Haddad (incorporated by reference to Exhibit 10.3 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 10.4+ |
|
Indemnification Agreement, dated December 6, 2021, between Dror Ortho-Design Ltd. and Moshe Shvets (incorporated by reference to Exhibit 10.4 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 10.5+ |
|
Indemnification Agreement, dated December 6, 2021, between Dror Ortho-Design Ltd. and Chaim Hurvitz (incorporated by reference to Exhibit 10.5 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 10.6+ |
|
Indemnification Agreement, dated December 6, 2021, between Dror Ortho-Design Ltd. and Chaim Ravad (incorporated by reference to Exhibit 10.6 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 10.7+ |
|
Indemnification Agreement, dated December 6, 2021, between Dror Ortho-Design Ltd. and Yehuda Englander (incorporated by reference to Exhibit 10.7 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 10.8+ |
|
Consulting Agreement, dated December 6, 2021, between Dror Ortho-Design Ltd. and Yaacov Bodner (incorporated by reference to Exhibit 10.8 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 10.9+ |
|
2021 Share Incentive Plan (incorporated by reference to Exhibit 10.9 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 10.10+ |
|
2023 Long-Term Incentive Plan (incorporated by reference to Exhibit 10.10 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 10.11 |
|
Securities Purchase Agreement, dated August 14, 2023, between Dror Ortho-Design, Inc. and certain purchasers identified therein (incorporated by reference to Exhibit 10.11 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 10.12 |
|
Registration Rights Agreement, dated August 14, 2023, between Dror Ortho-Design, Inc. and certain purchasers identified therein (incorporated by reference to Exhibit 10.12 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 10.13 |
|
Form of Lock-Up Agreement (incorporated by reference to Exhibit 10.13 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on August 14, 2023) |
| 10.14+ |
|
Services Agreement, dated June 1, 2022, between Dror Ortho-Design Ltd. and Yehuda Englander (incorporated by reference to Exhibit 10.4 to the Current Report on Form 8-K/A filed with the Securities and Exchange Commission on August 18, 2023) |
| 10.15+* |
|
First Amendment to Services Agreement, dated February 7, 2023, between Dror Ortho-Design, Inc. and Yehuda Englander |
| 10.16+* |
|
Services Agreement, dated February 7, 2023, between Dror Ortho-Design, Inc. and Chaim Ravad |
| 16.1 |
|
Letter from Sadler, Gibb & Associates, LLC to the Securities and Exchange Commission dated October 20, 2023 (incorporated by reference to Exhibit 16.1 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on October 24, 2023) |
| 21.1 |
|
List of Subsidiaries (incorporated by reference to Exhibit 21.1 to the Registration Statement on Form S-1 filed with the Securities and Exchange Commission on February 9, 2024) |
| 31.1* |
|
Certification of Chief Executive Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 |
| 32.1** |
|
Certification of Chief Executive Officer pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
| |
|
|
| 101 INS* |
|
Inline XBRL Instance Document |
| 101 SCH* |
|
Inline XBRL Taxonomy Extension Schema Document |
| 101 CAL* |
|
Inline XBRL Taxonomy Calculation Linkbase Document |
| 101 DEF* |
|
Inline XBRL Taxonomy Extension Definition Linkbase Document |
| 101 LAB* |
|
Inline XBRL Taxonomy Labels Linkbase Document |
| 101 PRE* |
|
Inline XBRL Taxonomy Presentation Linkbase Document |
| 104* |
|
Cover Page Interactive Data File (embedded within the Inline XBRL document) |
| + | Management contract or compensatory plan or arrangement. |
SIGNATURES
Pursuant to the requirements
of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto
duly authorized.
| |
DROR-ORTHO DESIGN, INC. |
| |
|
|
| Date: April 1, 2024 |
By: |
/s/ Eliyahu (Lee) Haddad |
| |
Name: |
Eliyahu (Lee) Haddad |
| |
Title: |
Chief Executive Officer |
| |
|
(Principal Executive Officer and
Principal Financial and Accounting Officer) |
Pursuant to the requirements
of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in
the capacities and on the dates indicated.
| Signature |
|
Title |
|
Date |
| /s/ Eliyahu (Lee) Haddad |
|
Chief Executive Officer and Director
(Principal Executive Officer and |
|
April 1, 2024 |
| Eliyahu (Lee) Haddad |
|
Principal Financial and Accounting Officer) |
|
|
| |
|
|
|
|
| /s/ Chaim Hurvitz |
|
Director and Chairman of the Board |
|
April 1, 2024 |
| Chaim Hurvitz |
|
|
|
|
| |
|
|
|
|
| /s/ Moshe Shvets |
|
Chief Technology Officer and
Director |
|
April 1, 2024 |
| Moshe Shvets |
|
|
|
|
| |
|
|
|
|
| /s/ Chaim Ravad |
|
Director |
|
April 1, 2024 |
| Chaim Ravad |
|
|
|
|
| |
|
|
|
|
| /s/ Yehuda Englander |
|
Director |
|
April 1, 2024 |
| Yehuda Englander |
|
|
|
|
DROR ORTHO-DESIGN, INC.
CONSOLIDATED FINANCIAL STATEMENTS
Table of Contents
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING
FIRM

To the Board of Directors and Stockholders of
Dror Ortho-Design, Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance
sheets of Dror Ortho-Design, Inc. (the “Company”) as of December 31, 2023 and 2022, the related consolidated statements of
operations, changes in stockholders’ equity and cash flows for the years then ended, and the related notes (collectively referred
to as the “Financial Statements”). In our opinion, the financial statements present fairly, in all material respects, the
financial position of the Company as of December 31, 2023 and 2022, and the results of its operations and its cash flows for the years
then ended, in conformity with accounting principles generally accepted in the United States of America.
Going Concern
The financial statements are presented on a going
concern basis. As described in Note 1 to the financial statements, the Company has not yet generated any material revenues, has suffered
recurring losses from operations with an accumulated deficit of $13,730,705 as of December 31, 2023, and is dependent upon external sources
for financing its operations. There is no assurance that profitable operations, if achieved, could be sustained on a continuing basis.
Further, the Company’s future operations are dependent on the success of the Company’s efforts to raise additional capital,
its research and commercialization efforts, regulatory approvals, and ultimately the market acceptance of the Company’s products.
There is no assurance that the Company will be successful in raising these funds. These financial statements do not include adjustments
that may result from the outcome of these uncertainties. The Company is exploring additional fundraising opportunities.
Basis for Opinion
These financial statements are the responsibility
of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our
audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”)
and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable
rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the
standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the financial
statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged
to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding
of internal control over financial reporting, but not for the purpose of expressing an opinion on the effectiveness of the Company’s
internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess
the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond
to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements.
Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating
the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matters
The critical audit matters communicated below
are matters arising from the current-period audit of the financial statements that were communicated or required to be communicated to
the board of directors and that (1) relate to accounts or disclosures that are material to the financial statements and (2) are especially
challenging, subjective, or complex judgments. The communication of critical audit matters does not alter in any way our opinion on the
financial statements, taken as a whole, and we are not, by communicating the critical audit matters below, providing separate opinions
on the critical audit matters or on the accounts or disclosures to which they relate.
Share Exchange Transaction – Refer to Note 1 of the financial
statements
Description of critical audit matter
As described in Note 1 to the financial statements,
the Company entered into a share exchange agreement with Dror Ortho-Design, Ltd., (“Private Dror”). Pursuant to the agreement,
100% of the outstanding equity capital of Private Dror was exchanged for shares of common and preferred stock of the Company, so that
the Private Dror’s shareholders were issued common and preferred shares in the amount that resulted in them holding 56.1% of the
total voting rights in the Company. In addition, the Company raised $5,225,000 as part of a private placement funding, and warrants and
options exercisable by its terms to Private Dror’s shares were exchanged to the Company. As a result of the transaction, Private
Dror became a wholly-owned subsidiary of the Company.
How the Critical Audit Matter Was Addressed in the Audit
Our audit procedures used to address the accounting
for the share exchange transaction included the following:
| 1. | We obtained and reviewed the share exchange agreement and other related agreements and documents to evaluate
the Company’s application of relevant accounting standards to the transaction. |
| | | |
| 2. | We reviewed the Company’s determination who the legal and accounting acquirer and acquiree were. |
| | | |
| 3. | We reviewed the accounting treatment of the modification of warrants and options. |
| | | |
| 4. | We evaluated the accuracy and completeness of the Company’s presentation of the share exchange agreement
in the financial statements, including evaluating whether disclosures were in accordance with relevant accounting standards. |
As a result of the audit procedures applied, we reached
the conclusion that the Share Exchange Transaction was accounted for correctly in the financial statements as of December 31, 2023.
We have served as the Company’s auditor since 2021.
By: /s/ Barzily and Co.
BARZILY AND CO., CPA’s
Jerusalem, Israel, 2024
April 1, 2024
DROR ORTHO-DESIGN, INC.
CONSOLIDATED BALANCE SHEETS
(U.S. dollars)
| | |
December 31, 2023 | | |
December 31, 2022 | |
| Assets | |
| | |
| |
| Current Assets: | |
| | |
| |
| Cash | |
$ | 3,347,843 | | |
$ | 1,039,059 | |
| Receivables and prepaid expenses | |
| 114,100 | | |
| 101,353 | |
| Total Current Assets | |
| 3,461,943 | | |
| 1,140,412 | |
| | |
| | | |
| | |
| Noncurrent Assets: | |
| | | |
| | |
| Property and equipment at cost, net of accumulated depreciation | |
| 2,328 | | |
| 2,998 | |
| Total Assets | |
| 3,464,271 | | |
| 1,143,410 | |
| | |
| | | |
| | |
| Liabilities And Stockholders’ Equity | |
| | | |
| | |
| | |
| | | |
| | |
| Current Liabilities: | |
| | | |
| | |
| Accounts payable | |
$ | 106,833 | | |
$ | 30,792 | |
| Accrued royalties | |
| — | | |
| — | |
| Founders claim accrual | |
| — | | |
| 240,000 | |
| Accrued expenses and other payables | |
| 190,271 | | |
| 276,126 | |
| Total Current Liabilities | |
| 297,104 | | |
| 546,918 | |
| | |
| | | |
| | |
| Noncurrent Liabilities: | |
| | | |
| | |
| Accrued severance | |
| 5,243 | | |
| 416 | |
| Total Liabilities | |
| 302,347 | | |
| 547,334 | |
| | |
| | | |
| | |
| Commitments and Contingencies (Note 9) | |
| | | |
| | |
| | |
| | | |
| | |
| Stockholders’ Equity* | |
| | | |
| | |
| Preferred A Stock, $0.0001 par value, 12,500,000 shares authorized; 10,463,363 and 7,576,999 shares outstanding at December 31, 2023 and 2022, respectively | |
| 1,047 | | |
| 758 | |
| Common stock, $0.0001 par value; 500,000,000 shares authorized; 495,454,546
and 437,735,093 shares issued and outstanding at December 31, 2023 and 2022, respectively | |
| 49,545 | | |
| 43,774 | |
| Additional paid-in capital | |
| 16,842,037 | | |
| 10,714,366 | |
| Accumulated deficit | |
| (13,730,705 | ) | |
| (10,162,822 | ) |
| Total Stockholders’ Equity | |
| 3,161,924 | | |
| 596,076 | |
| Total Liabilities and Stockholders’ Equity | |
$ | 3,464,271 | | |
$ | 1,143,410 | |
DROR ORTHO-DESIGN INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
(U.S. dollars)
| | |
Year Ended | |
| | |
December 31,
2023 | | |
December 31,
2022 | |
| Operating Expenses | |
| | |
| |
| Research and development | |
$ | 1,004,443 | | |
$ | 850,680 | |
| General and administrative expenses | |
| 1,120,426 | | |
| 814,653 | |
| Share-based compensation | |
| 2,253,793 | | |
| 19,908 | |
| Total Operating Expenses | |
| 4,378,662 | | |
| 1,685,241 | |
| | |
| | | |
| | |
| Loss from operations | |
| (4,378,662 | ) | |
| (1,685,241 | ) |
| | |
| | | |
| | |
| Financial income, net | |
| 90,147 | | |
| 1,742 | |
| Gain on retirement of royalty accrual | |
| 720,632 | | |
| — | |
| Total other income | |
| 810,779 | | |
| 1,742 | |
| | |
| | | |
| | |
| Loss before provision for income taxes | |
| (3,567,883 | ) | |
| (1,683,499 | ) |
| | |
| | | |
| | |
| Provision for income taxes | |
| — | | |
| — | |
| Net loss | |
$ | (3,567,883 | ) | |
$ | (1,683,499 | ) |
| | |
| | | |
| | |
| Net loss per common share | |
| | | |
| | |
Basic and Diluted | |
$ | (0.01 | ) | |
$ | (0.00 | ) |
| | |
| | | |
| | |
| Weighted-average common shares outstanding | |
| | | |
| | |
Basic and Diluted* | |
| 296,664,409 | | |
| 437,735,093 | |
DROR ORTHO-DESIGN INC.
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’
EQUITY
(U.S. dollars)
| | |
Series A
Preferred Stock | | |
Common Stock | | |
Treasury Stock | | |
Additional Paid-In | | |
Accumulated | | |
Total Stockholders’ | |
| | |
Shares* | | |
Amount | | |
Shares* | | |
Amount | | |
Shares | | |
Amount | | |
Capital | | |
Deficit | | |
Equity | |
| Balance at January 1, 2023 | |
| 7,576,999 | | |
$ | 758 | | |
| 437,735,093 | | |
$ | 43,774 | | |
| — | | |
$ | — | | |
$ | 10,714,366 | | |
$ | (10,162,822 | ) | |
$ | 596,076 | |
| Return of founders shares to the Company as part of claim settlement | |
| — | | |
| — | | |
| (330,952,906 | ) | |
| (33,096 | ) | |
| 330,952,906 | | |
| 33,096 | | |
| — | | |
| — | | |
| — | |
| Private Placement Investment, net of issuance costs ($571,796) | |
| 2,886,364 | | |
| 289 | | |
| 186,363,631 | | |
| 18,636 | | |
| — | | |
| — | | |
| 4,634,279 | | |
| — | | |
| 4,653,204 | |
| Settlement of Treasury Stock prior to recapitalization | |
| — | | |
| — | | |
| — | | |
| — | | |
| (330,952,906 | ) | |
| (33,096 | ) | |
| 33,096 | | |
| — | | |
| — | |
| Reverse re-capitalization | |
| — | | |
| — | | |
| 202,308,728 | | |
| 20,231 | | |
| — | | |
| — | | |
| (793,497 | ) | |
| — | | |
| (773,266 | ) |
| Stock-based compensation | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| 2,253,793 | | |
| — | | |
| 2,253,793 | |
| Net loss | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| (3,567,883 | ) | |
| (3,567,883 | ) |
| Balance at December 31, 2023 | |
| 10,463,363 | | |
$ | 1,047 | | |
| 495,454,546 | | |
$ | 49,545 | | |
| — | | |
$ | — | | |
$ | 16,842,037 | | |
$ | (13,730,705 | ) | |
$ | 3,161,924 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Balance at January 1, 2022 | |
| 7,576,999 | | |
$ | 758 | | |
| 437,735,093 | | |
$ | 43,774 | | |
| — | | |
| — | | |
$ | 10,694,458 | | |
$ | (8,479,323 | ) | |
$ | 2,259,667 | |
| Stock-based compensation | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| 19,908 | | |
| | | |
| 19,908 | |
| Net loss | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| (1,683,499 | ) | |
| (1,683,499 | ) |
| Balance at December 31, 2022 | |
| 7,576,999 | | |
$ | 758 | | |
| 437,735,093 | | |
$ | 43,774 | | |
| — | | |
| — | | |
$ | 10,714,366 | | |
$ | (10,162,822 | ) | |
$ | 596,076 | |
DROR ORTHO-DESIGN INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(U.S. dollars)
| | |
For the Year Ended
December 31, | |
| | |
2023 | | |
2022 | |
| Cash flows from operating activities: | |
| | |
| |
| Net loss | |
$ | (3,567,883 | ) | |
$ | (1,683,499 | ) |
| Stock-based compensation expense | |
| 2,253,793 | | |
| 19,908 | |
| Gain on retirement of royalty accrual | |
| (720,632 | ) | |
| — | |
| Depreciation | |
| 670 | | |
| 670 | |
| Changes in operating assets and liabilities: | |
| | | |
| | |
| Receivables and prepaid expenses | |
| (7,264 | ) | |
| (71,734 | ) |
| Accounts payable | |
| 44,111 | | |
| 20,768 | |
| Accrued expenses and other payables | |
| (136,446 | ) | |
| 203,761 | |
| Founders claim accrual | |
| (240,000 | ) | |
| — | |
| Accrued royalties | |
| 6,438 | | |
| — | |
| Accrued severance | |
| 4,827 | | |
| (7,052 | ) |
| Net cash used in operating activities | |
| (2,362,386 | ) | |
| (1,517,178 | ) |
| | |
| | | |
| | |
| Cash flows from investing activities: | |
| | | |
| | |
| Cash acquired in reverse merger | |
| 17,966 | | |
| — | |
| Net cash provided by investing activities | |
| 17,966 | | |
| — | |
| | |
| | | |
| | |
| Cash flows from financing activities: | |
| | | |
| | |
| Proceeds from private placement raise | |
| 5,225,000 | | |
| — | |
| Issuance costs | |
| (571,796 | ) | |
| | |
| Net cash provided in financing activities | |
| 4,653,204 | | |
| — | |
| | |
| | | |
| | |
| Net increase (decrease) in cash | |
| 2,308,784 | | |
| (1,517,178 | ) |
| Cash, beginning of year | |
| 1,039,059 | | |
| 2,556,237 | |
| Cash, end of year | |
$ | 3,347,843 | | |
$ | 1,039,059 | |
| | |
| | | |
| | |
| Supplemental cash flow information: | |
| | | |
| | |
| Cash paid for interest | |
$ | — | | |
$ | — | |
| Cash paid for taxes | |
$ | — | | |
$ | — | |
| | |
| | | |
| | |
| Non-cash activities: | |
| | | |
| | |
| Shares issued at reverse recapitalization | |
$ | 20,231 | | |
$ | — | |
| Net liabilities assumed in merger | |
$ | 791,232 | | |
$ | — | |
| Return of founders shares to the Company as part of claim settlement | |
$ | 33,096 | | |
| — | |
| Settlement of Treasury Stock prior to recapitalization | |
$ | 33,096 | | |
| — | |
DROR ORTHO-DESIGN INC.
NOTES TO FINANCIAL STATEMENTS
Note 1 – Organization and Basis of Presentation
Organization
The Company was incorporated as Novint Technologies,
Inc. in the State of New Mexico in April 1999. On February 26, 2002, the Company changed its state of incorporation to Delaware by merging
with Novint Technologies, Inc., a Delaware corporation. On August 14, 2023, following a share exchange agreement, the Company changed
its name from “Novint Technologies, Inc.” to “Dror Ortho-Design, Inc.”. Following the Share Exchange (as defined
below), the Company succeeded the business of Dror Ortho-Design, Ltd. (“Private Dror”) as its sole line of business. The Company
is involved in the research and development of an orthodontic alignment platform and has not yet reached the sales stage for its product.
The Company’s stock is quoted on the OTC
Pink Market under the symbol “DROR.”
Reverse Recapitalization
On July 5, 2023, Private Dror entered into a share exchange agreement
with the Company and on August 14, 2023 the share exchange was consummated (the “Share Exchange”). As a result of the
Share Exchange, the shareholders of Private Dror exchanged all 235,089 of their outstanding shares of common stock, for 106,782,187 shares
of the Company’s Common Stock and 7,576,999 shares of the Company’s Series A Preferred Stock. Pursuant to the terms of the
Share Exchange, the Company raised $5,225,000 as part of a private placement funding, and the private placement investors received 186,363,631
shares of common stock and 2,886,364 shares of Series A Preferred Stock. As a result, Private Dror became a wholly owned subsidiary of
the Company and the Private Dror shareholders hold 56.1% of the Company’s common stock equivalents based on the common and preferred
shares received in the Share Exchange.
The Share Exchange is being accounted for as a
recapitalization, with Private Dror deemed to be the accounting acquirer, and the Company the accounting acquiree. Accordingly, Private
Dror’s historical financial statements for periods prior to the consummation of the Share Exchange have become those of the registrant.
Assets and liabilities and the historical operations reported for periods prior to the Share Exchange are those of Private Dror other
than equity items. All references to common stock, preferred stock, share and per share amounts have been retroactively restated to reflect
the reverse recapitalization as if the transaction had taken place as of the beginning of the earliest period presented.
Pursuant to the Share Exchange, the Company issued
shares of its common stock and preferred stock to Private Dror’s stockholders, at an exchange ratio of 3,677.27 shares of the Company’s
common stock.
As of August 14, 2023 the fair value of the net
liabilities of the Company was $793,497, which was recorded as Additional Paid-In Capital as part of the Share Exchange.
Going Concern and Management’s Plans
The financial statements are presented on a going
concern basis. The Company has not yet generated any material revenues, has suffered recurring losses from operations with an accumulated
deficit of $13,730,705 as of December 31, 2023, and is dependent upon external sources for financing its operations. There is no assurance
that profitable operations, if achieved, could be sustained on a continuing basis. Further, the Company’s future operations are
dependent on the success of the Company’s efforts to raise additional capital, its research and commercialization efforts, regulatory
approvals, and ultimately the market acceptance of the Company’s products. There is no assurance that the Company will be successful
in raising these funds. These financial statements do not include adjustments that may result from the outcome of these uncertainties.
The Company is exploring additional fundraising opportunities.
NOTE 2 – SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation
The accompanying financial statements for the
years ended December 31, 2023 and 2022 have been prepared in accordance with U.S. generally accepted accounting principles (“U.S.
GAAP”) and applicable rules and regulations of the United States Securities and Exchange Commission (“SEC”).
As the Company completed a reverse recapitalization
on August 14, 2023, the financial information for the periods prior to the reverse recapitalization reflect those of Private Dror. From
August 14, 2023 forward, the financial information presented is the consolidated financial information of the Company and its subsidiary.
Use of Estimates and Assumptions
The preparation of financial statements in conformity
with U.S. GAAP requires management to make estimates or assumptions that affect the reported amounts of assets and liabilities and disclosure
of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenue and expenses during the
reporting periods. Actual results could vary from those estimates. Management utilizes various other estimates, including but not limited
to accrued royalties, accrued expenses, the valuation of stock-based compensation, the valuation allowance for deferred tax assets and
other contingencies. The results of any changes in accounting estimates are reflected in the financial statements in the period in which
the changes become evident. Estimates and assumptions are reviewed periodically, and the effects of revisions are reflected in the period
that they are determined to be necessary.
Functional Currency
The Company accounts for foreign currency transactions
pursuant to ASC 830, “Foreign Currency Matters”. The functional currency of the Company and its subsidiary is the United States
Dollar (“US$”) as the U.S. dollar is the currency of the primary economic environment in which the Company operates. The accompanying
financial statements have been expressed in US$. Transactions denominated in currencies other than the functional currency are translated
into the functional currency at the exchange rates prevailing at the dates of the transaction. Monetary assets and liabilities denominated
in currencies other than the functional currency are translated into the functional currency using the applicable exchange rates at the
balance sheet dates. The resulting exchange differences are recorded in the statements of operations. The exchange rate of the US Dollar
to the Israeli Shekel was 3.627 and 3.519 as of December 31, 2023 and 2022, respectively.
Cash
The Company’s cash is held with financial
institutions in the United States and Israel. Management believes that the financial institutions that hold the Company’s cash are
financially sound and, accordingly, minimal credit risk exists with respect to these investments. Account balances held in the Unites
States may, at times, exceed the Federal Deposit Insurance Corporation (FDIC) insurance limit. As of December 31, 2023 and 2022, the Company
had $145,168 and $643,658, respectively, in excess of the FDIC insurance limit. As of December 31, 2023 and 2022, the Company had $2,935,078
and $144,399, respectively, in Israeli financial institutions, which is uninsured. The Company has not experienced any losses in such
accounts with these financial institutions.
Property and Equipment
Property and equipment are stated at cost less
accumulated depreciation. Depreciation is calculated using the straight–line method on the various asset classes, which currently
consists of office equipment over their estimated useful lives of seven years when placed in service. The cost of repairs and maintenance
is expensed as incurred; major replacements and improvements are capitalized. When assets are retired or disposed of, the cost and accumulated
depreciation are removed from the accounts, and any resulting gains or losses are included in income in the year of disposition.
Research and Development
The Company expenses all research and development costs as they are
incurred. Research and development includes expenditures in connection with in-house research and development as well as proprietary products
and technology, and includes salaries and related costs, consulting fees, and professional services.
Share–based compensation
The Company applies ASC 718-10, “Share-
Based Payment,” which requires the measurement and recognition of compensation expenses for all share-based payment awards made
to employees and directors including employee stock options under the Company’s stock plans and equity awards issued to non-employees
based on estimated fair values.
ASC 718-10 requires companies to estimate the
fair value of equity-based option awards on the date of grant using an option-pricing model. The fair value of the award is recognized
as an expense on a straight-line basis over the requisite service periods in the Company’s statement of operations.
The fair value of an option award is estimated on the date of grant
using the Black–Scholes option valuation model. The Black–Scholes option valuation model requires the development of assumptions
that are inputs into the model. These assumptions are the expected stock volatility, the risk–free interest rate, the expected life
of the option, the dividend yield on the underlying stock and the expected forfeiture rate. Since the Company does not have sufficiant
historical data regarding its volatility of its common stock, the expected volatility used is based on volatility of similar publicly
listed companies in comparable industries. Risk–free interest rates are calculated based on continuously compounded risk–free
rates for the appropriate term.
Determining the appropriate fair value model and
calculating the fair value of equity–based payment awards require the input of the subjective assumptions described above. The assumptions
used in calculating the fair value of equity–based payment awards represent management’s best estimates, which involve inherent
uncertainties and the application of management’s judgment.
Income Taxes
The Company accounts for income taxes using the
asset-and-liability method in accordance with ASC Topic 740, “Income Taxes”. Deferred tax assets and liabilities are recognized
for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities
and their respective tax bases and operating loss and tax credit carry forwards. Deferred tax assets and liabilities are measured using
enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or
settled. The effect on the deferred tax assets and liabilities of a change in tax rate is recognized in the period that includes the enactment
date. A valuation allowance is recorded if it is more-likely-than-not that some portion or all of the deferred tax assets will not be
realized in future periods.
The Company follows the guidance in ASC Topic
740-10 in assessing uncertain tax positions. The standard applies to all tax positions and clarifies the recognition of tax benefits in
the financial statements by providing for a two-step approach of recognition and measurement. The first step involves assessing whether
the tax position is more-likely-than-not to be sustained upon examination based upon its technical merits. The second step involves measurement
of the amount to be recognized. Tax positions that meet the more-likely-than-not threshold are measured at the largest amount of tax benefit
that is greater than 50% likely of being realized upon ultimate finalization with the taxing authority. The Company recognizes the impact
of an uncertain income tax position in the financial statements if it believes that the position is more likely than not to be sustained
by the relevant taxing authority. The Company will recognize interest and penalties related to tax positions in income tax expense. As
of both December 31, 2023 and 2022, there were no unrecognized uncertain income tax positions.
Basic and Diluted Net Loss Per Common Share
The Company computes net loss per share in accordance
with ASC 260, “Earnings per Share” which requires presentation of both basic and diluted earnings per share (EPS) on the face
of the income statement. Basic loss per ordinary share is computed by dividing the loss for the period applicable to common shareholders,
by the weighted average number of shares of common stock outstanding during the period. Diluted net loss per common share is computed
by dividing the net loss by the weighted average number of common shares outstanding for the period and, if dilutive, potential common
shares outstanding during the period. Potentially dilutive securities consist of the incremental common shares issuable upon exercise
of common stock equivalents such as stock options, warrants and convertible debt instruments. Potentially dilutive securities are excluded
from the computation if their effect is anti-dilutive. As a result, the basic and diluted per share amounts for all periods presented
are identical.
For the years ended December 31, 2023 and 2022,
the Company incurred net losses which cannot be diluted; therefore, basic and diluted loss per common share is the same. Each Series A
Preferred Stock is convertible into 100 shares of Common Stock, and is included in the table as if converted. As of December 31, 2023
and 2022, shares issuable which could potentially dilute future earnings were as follows:
| | |
December 31, | |
| | |
2023 | | |
2022 | |
| | |
| | |
| |
| Preferred Shares | |
| 1,046,336,299 | | |
| 757,699,900 | |
| Warrants | |
| 964,834,419 | | |
| 510,794,865 | |
| Stock Options | |
| 163,142,084 | | |
| 163,142,084 | |
| Shares excluded from the calculation of diluted loss per share | |
| 2,174,312,802 | | |
| 1,431,637,849 | |
Reclassification
General and administrative expenses totaling $127,453
and $19,908 for the year ended December 31, 2022 were reclassified to research and development and share-based compensation, respectively,
to conform with current year presentation. The reclassifications had no effect on the net loss for the year ended December 31, 2022.
Recently Issued Accounting Pronouncements
In June 2016, the FASB issued ASU No. 2016-13,
Measurement of Credit Losses on Financial Instruments (“ASU2016-13”), as amended by ASU 2019-10. ASU 2016-13 will change
how companies account for credit losses for most financial assets and certain other instruments. For trade receivables, loans and held-to-maturity
debt securities, companies will be required to recognize an allowance for credit losses rather than reducing the carrying value of the
asset. ASU2016-13 is effective for the Company for the annual reporting period beginning January 1, 2023. The Company adopted this guidance
for the year ended December 31, 2023, however there was no impact to the financial statements.
Note 3 – Prepaid expenses and other current assets:
| | |
December 31, | |
| | |
2023 | | |
2022 | |
| | |
| | |
| |
| VAT receivable | |
$ | 73,784 | | |
| 101,353 | |
| Prepaid expenses | |
| 34,802 | | |
| - | |
| Other assets | |
| 5,514 | | |
| - | |
| | |
$ | 114,100 | | |
| 101,353 | |
Note 4 – Property and Equipment:
| | |
December 31, | |
| | |
2023 | | |
2022 | |
| | |
| | |
| |
| Equipment and furniture | |
$ | 9,567 | | |
| 9,567 | |
| Less accumulated depreciation | |
| (7,239 | ) | |
| (6,569 | ) |
| Property and equipment, net | |
$ | 2,328 | | |
| 2,998 | |
Depreciation expense was $670 for both of the
years ended December 31, 2023 and 2022, respectively.
Note 5 – Accrued expenses:
| | |
December 31, | |
| | |
2023 | | |
2022 | |
| | |
| | |
| |
| Salary and related expenses | |
$ | 95,566 | | |
| 169,297 | |
| Accrued audit fees | |
| 40,000 | | |
| 44,613 | |
| Accrued legal fees | |
| 30,000 | | |
| - | |
| Accrued consulting fees | |
| 24,705 | | |
| 52,811 | |
| Other expenses | |
| - | | |
| 9,405 | |
| | |
$ | 190,271 | | |
| 276,126 | |
Note 6 – Founders claim accrual:
The Company recorded a provision in respect of
a claim made against Private Dror by its founders. The claim related to amounts claimed as a repayment of loan balances and other amounts
including salary and benefit related balances. In January 2023, Private Dror signed an agreement with the founders, settling all-outstanding
claims at $240,000 which included amounts representing the repayment of a loan, reimbursement of expenses and an amount for pain and suffering.
In addition, the agreement stipulated the transfer back of all shares held by the founders to the Private Dror for no additional consideration.
The settlement was paid in the first quarter of 2023. In addition, the agreement stipulated the transfer back of all shares (330,952,906
ordinary shares with par value of NIS 0.0001), held by the founders to the Company.
Note 7 – Accrued royalties
Accrued royalties related to the Company’s licensing agreements
with various parties that provided gaming software to the Company. These licensing agreements contain obligations to pay royalty fees
ranging from 5% to 50% of either gross or net revenue, and a flat fee per end user of $0.50, subject to an obligation to pay minimum annual
royalties of $50,000 as specified in the licensing agreements. As part of the Share Exchange, the Company assumed accrued royalties in
the amount of $714,194, and accrued an additional $6,438 subsequent to the Share Exchange. As the statute of limitations for the collection
of the royalties had passed, the Company retired the royalty accrual amounting to $720,632 during the fourth quarter of 2023 and ceased
to accrue any further amounts.
Note 8 – Accrued severance:
Under Israeli law, companies are required to make
severance payments to terminated Israeli employees. The severance reserve is calculated based on the employee’s last salary and
period of employment. A portion of the severance pay and pension obligation is covered by payment of monthly premiums to insurance companies/
policies under approved plans and to pension funds. The deposits presented in the balance sheet include profits accumulated to the balance
sheet date. The amounts funded as above are not reflected in the balance sheet since they are not under the control and management of
the Company.
A portion of employee severance payments are subject to the terms of section 14 of the Israeli Severance Pay Law, 1963, according to which the Company’s current deposits in pension funds and/or in policies in insurance companies exempt it from any additional undertaking towards employees, for which the aforementioned amounts were deposited.
Note 9 – Commitments and Contingencies
The Company partially financed their research
and development expenditures under grant programs sponsored by the Israel Innovation Authority (“IIA”) of the Ministry of
Economy and Industry (formerly the Office of Chief Scientist) for the support of research and development activities conducted in Israel.
At the time the grants were received from the IIA, successful development of the related projects was not assured. In exchange for participation
in the programs by the IIA, the Company agreed to pay 3% of total sales of products developed within the framework of these programs.
The royalties will be paid up to a maximum amount equaling 100% of the grants provided by the IIA, linked to the dollar, bearing annual
interest at a rate based on LIBOR. Beginning from January 1, 2024 the rate will be adjusted to SOFR (Secured Over Financing Rate). The
obligation to pay these royalties is contingent on actual sales of the products, and in the absence of such sales payment of royalties
is not required. In some cases, the Government of Israel’s participation (through the IIA) is subject to export sales or other conditions.
The maximum amount of royalties is increased in the event of production outside of Israel. The current contingent royalty obligation as
of December 31, 2023 and 2022 is approximately $1.12 and $1.08 million, respectively.
From time to time in the normal course of business,
the Company may be subject to routine litigation incidental to its business. Although there can be no assurances as to the ultimate disposition
of any such matters, it is the opinion of management, based upon the information available at this time, that there are no matters, individually
or in the aggregate, that would have a material adverse effect on the results of operations and financial condition of the Company.
War in Israel
In October 2023, Israel was attacked by a terrorist
organization and entered a state of war. As of the date of these consolidated financial statements, the war in Israel is ongoing and continues
to evolve. The Company’s research and development activities are located in Israel. Currently, such activities in Israel remain
largely unaffected. During the year ended December 31, 2023, the impact of this war on the Company’s results of operations and financial
condition was immaterial. Management will continue to monitor the effect of the war on the Company's financial position and results of
operations.
Note 10 – Stockholders’ Equity
All references to common stock, share and per
share amounts have been retroactively restated to reflect the reverse recapitalization as if the transaction had taken place as of the
beginning of the earliest period presented.
Common Stock
On January 4, 2024, the Company filed its Amended and Restated Certificate of Incorporation, which provided for the number of authorized
shares of the Company’s common stock, par value $0.0001 per share, to be increased from 500,000,000 to 3,254,475,740. All issued
shares of common stock are entitled to vote on a 1 share/1 vote basis. The Company had 495,454,546 and 437,735,093 shares of common stock
issued and outstanding as of December 31, 2023 and 2022, respectively.
Holders of our common stock have no preemptive,
redemption, conversion or subscription rights. No sinking fund provisions are applicable to our common stock. Upon liquidation, dissolution
or winding-up, holders of our common stock are entitled to share in all assets remaining after payment of all liabilities and the liquidation
preferences of any of our outstanding shares of preferred stock. Subject to preferences that may be applicable to any outstanding shares
of preferred stock, holders of our common stock are entitled to receive dividends, if any, as may be declared from time to time by our
board of directors out of our assets which are legally available. Such dividends, if any, are payable in cash, in property or in shares
of capital stock.
As part of the Private Dror founders claim settlement
agreement (see Note 6), 330,952,906 shares of common stock were returned to the Private Dror in February 2023. These shares were initially
classified as Treasury Stock and were retired as part of the Share Exchange Agreement.
Pursuant to the terms of the Share Exchange, the
Company raised $5,225,000 as part of a private placement funding, $5,025,000 from a first closing on August 14, 2023 and an additional
$200,000 from a second closing on September 13, 2023. The private placement investors received 186,363,631 shares of common stock and
2,886,364 shares of Series A Preferred Stock.
Transaction expenses relating to the private placement
funding and for the Share Exchange totaled $571,796, and are offset against the proceeds in Additional Paid-In Capital recorded as part
of the private placement funding and the Share Exchange.
Preferred Stock
The Company is authorized to issue up to 12,500,000 shares
of $0.0001 par value non-redeemable preferred stock. As of December 31, 2022, 7,576,999 shares of Series A Preferred Stock were
outstanding. During the third quarter of 2023, as a result of the private placement funding, 2,886,364 shares of Series A Preferred Stock
were issued to investors.
The following is a summary of the principal terms
of the Series A Preferred Stock as set forth in the Certificate of Designation.
Conversion
The Series A Preferred Stock is convertible into
common stock at any time at a conversion price of $0.011, or 100 shares of Common Stock for each share of Preferred A Stock, subject to
adjustment for certain anti-dilution provisions set forth in the Series A Certificate of Designation. Upon conversion the shares of Series
A Preferred Stock will resume the status of authorized but unissued shares of preferred stock of the Company.
Dividends
The holders of Series A Preferred Stock will be
entitled to dividends, on an as-if converted basis, equal to and in the same form as dividends actually paid on shares of common stock,
when and if actually paid.
Voting Rights
The shareholders of Series A Preferred Stock are
entitled to vote with holders of the Company’s common stock, on all matters that such holders of Common Stock are entitled to vote
upon, in the same manner and with the same effect as the holders of Common Stock, voting together with the holders of Common Stock as
a single class. Each share of Preferred Stock shall entitle the shareholder to cast that number of votes per share of Preferred Stock
equal to the number of shares of Common Stock into which such share of Preferred Stock is convertible (after giving effect to certain
limitations on conversion, as applicable). As long as any shares of Series A Preferred Stock are outstanding, the Company may not, without
the approval of a majority of the then outstanding shares of Series A Preferred Stock (a) alter or change the powers, preferences
or rights given to the Series A Preferred Stock, (b) alter or amend our amended and restated certificate of incorporation, the Series
A Certificate of Designation, or our amended and restated bylaws in such a manner so as to materially adversely affect any rights given
to the Series A Preferred Stock, (c) authorize or create any class of stock ranking as to dividends, redemption or distribution of assets
upon a Liquidation (as defined below) senior to the Series A Preferred Stock, or (d) enter into any agreement to do any of the foregoing.
Liquidation
Upon any liquidation, dissolution or winding-up
of the Company, whether voluntary or involuntary (a “Liquidation”), the then holders of the Series A Preferred Stock are
entitled to receive out of the assets available for distribution to stockholders of the Company the same amount that a holder of common
stock would receive if the Series A Preferred Stock were fully converted (disregarding for such purposes any conversion limitations hereunder)
to common stock which amounts shall be paid pari passu with all holders of common stock..
Warrants
Prior to the Share Exchange, there were 510,794,865 warrants to purchase
Common shares held by Private Dror shareholders. Pursuant to the warrant terms, 20,960,439 warrants expired as a result of the Share Exchange.
On August 14, 2023, the Company issued warrants to purchase up to 489,834,426 shares of Common Stock to Private Dror shareholders
in exchange for their outstanding warrants and warrants to purchase up to 456,818,176 shares of Common Stock to the private placement
investors in respect of their investment, in addition to warrants to purchase up to 18,181,817 shares of Common Stock issued to private
placement investors in a subsequent closing on September 13, 2023. The warrants expire five years from the initial exercise date
and are exercisable at an exercise price of $0.033 per share. The initial exercise date was dependent on the authorization of additional
Common shares which occurred on December 28, 2023. The warrants contain provisions that protect their holders against dilution by adjustment
of the purchase price in certain events such as stock dividends, stock splits and other similar events.
If at the time of the warrant’s exercise
there is no effective registration statement registering, or no current prospectus available for, the resale of the shares of common stock
underlying the warrant, then the holder will have the right to exercise warrant by means of a cashless exercise. In addition, if (i) the
volume-weighted average price of our common stock for 20 consecutive trading days is at least 300% of the exercise price of the warrants,
(ii) the dollar trading volume of our common stock for each trading day within such 20-day trading period equals or exceeds $500,000,
(iii) a registration statement providing for the resale of the private placement shares is effective and such registration statement
has been effective for six (6) months, (iv) the holder of the warrant is not in possession of any information provided by the Company
that constitutes material nonpublic information and (v) the Company has not breached any of the terms of the investment documents
(regardless of if such breach has been cured), then the warrants may be redeemed at a price of $0.001 per warrant up to one-half, in the
aggregate, of the warrants upon not less than 20 days’ prior written notice of redemption to each holder, subject to certain customary
restrictions.
| | |
| | |
| | |
Weighted | | |
| |
| | |
| | |
Weighted | | |
Average | | |
| |
| | |
| | |
Average | | |
Remaining | | |
Aggregate | |
| | |
Number of | | |
Exercise | | |
Contractual | | |
Intrinsic | |
| Warrants | |
Shares | | |
Price | | |
Term | | |
Value | |
| Balance Outstanding, January 1, 2022 | |
| 510,794,865 | | |
$ | 0.02 | | |
| 2.73 | | |
$ | 15,486 | |
| Granted | |
| - | | |
| - | | |
| - | | |
| - | |
| Forfeited | |
| - | | |
| - | | |
| - | | |
| - | |
| Exercised | |
| - | | |
| - | | |
| - | | |
| - | |
| Balance Outstanding, December 31, 2022 | |
| 510,794,865 | | |
$ | 0.02 | | |
| 1.73 | | |
$ | 13,263 | |
| Granted | |
| 474,999,993 | | |
| 0.03 | | |
| - | | |
| - | |
| Forfeited | |
| (20,960,439 | ) | |
| - | | |
| - | | |
| - | |
| Exercised | |
| - | | |
| - | | |
| - | | |
| - | |
| Balance Outstanding, December 31, 2023 | |
| 964,834,419 | | |
$ | 0.03 | | |
| 5.00 | | |
$ | - | |
| | |
| | | |
| | | |
| | | |
| | |
| Exercisable, December 31, 2023 | |
| 964,834,419 | | |
$ | 0.03 | | |
| 5.00 | | |
$ | - | |
The aggregate intrinsic value in the table above represents the total
intrinsic value, based on the Company’s closing common stock price of $2.72, $2.33, and $0.01 as of December 31, 2023, 2022 and
2021, respectively, which would have been received by the warrant holders had all warrant holders exercised their warrants as of that
date.
Equity Incentive Plan
Prior to the Share Exchange, there were 163,142,084 Private Dror employee
stock options that had been granted to two executives and a director. As part of the Share Exchange, the outstanding employee stock options
are to be exchanged and the Company is required to issue new employee stock options under the Company’s 2023 Long-Term Incentive
Plan with the same terms as the previously issued options. As the Company did not have enough available authorized shares underlying the
options to be issued at the time of the merger, the new employee stock options were not issued. In December 2023 the Company authorized
additional shares to cover the employee stock options and is working on the legal filings for the establishment of the 2023 Plan. As the
agreement stipulates that the new options will continue the vesting schedules of the original options, the Company continues to record
the expense over the original vesting period.
The Company treated the exchange of the original options for the new
options as a modification in accordance with ASC 718. The Company calculated the fair value of the original options prior to the Share
Exchange and the fair value of the new options at the time of the Share Exchange. The increase in value due to the modification was $4,261,809
is to be recorded as additional share-based compensation expense. As one third of the options had fully vested prior to the Share Exchange,
the Company recognized one third of the total amount of the increased value, amounting to $1,420,603 at the time of the Share Exchange.
The remaining two thirds of the incremental value relating to the unvested options are going to be recorded over the remaining vesting
period.
The following table summarized the option activity for the years ended
December 31, 2023 and 2022:
| | |
| | |
| | |
Weighted | | |
| |
| | |
| | |
Weighted | | |
Average | | |
| |
| | |
| | |
Average | | |
Remaining | | |
Aggregate | |
| | |
Number of | | |
Exercise | | |
Contractual | | |
Intrinsic | |
| Options | |
Shares | | |
Price | | |
Term (in years) | | |
Value | |
| Balance Outstanding, January 1, 2022 | |
| 174,666,648 | | |
$ | 0.01 | | |
| 8.74 | | |
| - | |
| Granted | |
| 9,597,675 | | |
| 0.00 | | |
| | | |
| - | |
| Forfeited | |
| - | | |
| - | | |
| - | | |
| - | |
| Exercised | |
| - | | |
| - | | |
| - | | |
| - | |
| Expired | |
| (21,122,239 | ) | |
| 0.04 | | |
| - | | |
| - | |
| Balance Outstanding, December 31, 2022 | |
| 163,142,084 | | |
$ | 0.00 | | |
| 8.96 | | |
$ | - | |
| Granted (Share Exchange) | |
| - | | |
| 0.00 | | |
| | | |
| 4,070,727 | |
| Forfeited (Share Exchange) | |
| - | | |
| 0.00 | | |
| | | |
| | |
| Exercised | |
| | | |
| | | |
| | | |
| | |
| Expired | |
| | | |
| | | |
| | | |
| | |
| Balance Outstanding, December 31, 2023 | |
| 163,142,084 | | |
$ | 0.00 | | |
| 9.62 | | |
$ | 1,003,656 | |
| | |
| | | |
| | | |
| | | |
| | |
| Exercisable, December 31, 2023 | |
| 105,562,164 | | |
$ | 0.00 | | |
| 9.62 | | |
$ | 669,104 | |
Share-based compensation expense for the years ended
December 31, 2023 and 2022 amounted to $2,253,793 and $19,908, respectively. Share-based compensation relating to general and administrative
expenses amounted to $1,612,173 and 14,146 for the years ended December 31, 2023 and 2022, respectively. Share-based compensation relating
to research and development expenses amounted to $641,620 and 5,762 for the years ended December 31, 2023 and 2022, respectively. The
fair value of stock options that fully vested during the years ended December 31, 2023 and 2022 was $1,420,603 and $19,225, respectively.
The weighted average grant date fair value for options granted during the years ended December 31, 2023 and 2022 was $0.03 and $1.38,
respectively, using the Black Scholes valuation method.
As of December 31, 2023, there was $2,047,973
of unrecognized compensation cost related to non-vested share-based compensation, which will be amortized over a weighted average period
of 0.96 years.
The aggregate intrinsic value in the table above represents the total
intrinsic value, based on the Company’s closing stock price of $2.72, $2.33, and $0.01 as of December 31, 2023, 2022 and 2021, respectively,
which would have been received by the option holders had all option holders exercised their options as of that date.
Note 11 – Research and development expenses:
The components of research and development expenses are as follows:
| | |
For the Year Ended
December 31, | |
| | |
2023 | | |
2022 | |
| Subcontractors | |
$ | 759,440 | | |
$ | 542,186 | |
| Salaries | |
| 191,825 | | |
| 182,995 | |
| Consultants and others | |
| 53,178 | | |
| 125,499 | |
| Total | |
$ | 1,004,443 | | |
$ | 850,680 | |
Note 12 – General and administrative expenses:
The components of general and administrative expenses are as follows:
| | |
For the Year Ended
December 31, | |
| | |
2023 | | |
2022 | |
| Salaries and related | |
$ | 543,469 | | |
$ | 509,800 | |
| Legal | |
| 206,925 | | |
| 131,922 | |
| Professional fees | |
| 149,126 | | |
| 52,536 | |
| Consulting | |
| 106,264 | | |
| 78,525 | |
| Rent and utilities | |
| 39,157 | | |
| 22,872 | |
| Insurance | |
| 23,119 | | |
| - | |
| Donations | |
| 16,235 | | |
| - | |
| Office expense | |
| 9,557 | | |
| 4,020 | |
| Royalties | |
| 6,438 | | |
| - | |
| Depreciation | |
| 670 | | |
| 670 | |
| Other | |
| 19,466 | | |
| 14,308 | |
| Total | |
$ | 1,120,426 | | |
$ | 814,653 | |
Note 13 – Finance income, net:
The components of finance income, net are as follows:
| | |
For the Year Ended
December 31, | |
| | |
2023 | | |
2022 | |
| Exchange differences | |
$ | 94,020 | | |
$ | 3,410 | |
| Bank fees | |
| (3,873 | ) | |
| (1,668 | ) |
| Total | |
$ | 90,147 | | |
$ | 1,742 | |
Note 14 – Income Taxes:
The Company files corporate income tax returns in the United States
(federal), in New York (state), and in Israel (foreign). The Company is subject to federal, state and local income tax examinations by
tax authorities for the tax years 2020 through 2023. The Israeli subsidiary tax reports through 2017 are considered final assessments
in accordance with the provisions of section 145 of the Income Tax Ordinance.
As of December 31, 2023, the Company had federal net
operating loss carry forwards of $32.8 million. Federal net operating losses generated prior to January 1, 2018, amounting to $32.0 million,
may be offset against future taxable income, subject to limitation under IRC Section 382, which begin to expire in 2024 if not utilized
prior to that date, and fully expire during various years through 2037 for federal purposes. Net operating losses generated after January
1, 2018, amounting to $0.8 million, no longer have an expiration but are limited to 80% of taxable income. Tax loss carryforwards in Israel
amount to approximately USD 9.9 million, (NIS 36.5 million) as of December 31, 2023, and do not expire. There are also Israeli capital
loss carryforwards amounting to $0.3 million (NIS 1.1 million) that can be offset only against capital gains but do not expire.
The valuation allowance overall increased by approximately
$7.9 million and $0.1 million in the years ended 2023 and 2022, respectively, and was approximately $9.9 million and $2.0 million, respectively.
The Company has fully reserved the deferred tax asset resulting from available net operating loss carryforwards.
The reconciliation of income tax expense computed
at the U.S. federal statutory rate to the income tax provision for the years ended December 31, 2023 and 2022 is as follows:
| | |
Year ended December 31, | |
| | |
2023 | | |
2022 | |
| Income before income taxes | |
$ | (3,567,883 | ) | |
$ | (1,683,499 | ) |
| Taxes under statutory US tax rates | |
| (749,255 | ) | |
| (353,535 | ) |
| Foreign Rate Differential | |
| (85,538 | ) | |
| (33,670 | ) |
| Acquisitions | |
| (7,163,604 | ) | |
| - | |
| Expired net operating loss | |
| 118,215 | | |
| - | |
| Other permanent items | |
| (53,837 | ) | |
| 283,891 | |
| Increase (decrease) in valuation allowance | |
| 7,934,019 | | |
| 103,314 | |
| Income tax expense | |
$ | - | | |
$ | - | |
The increase in the Company’s net valuation allowance
was mainly due to the reverse merger and continued net operating losses from ongoing operations.
Deferred income taxes reflect the net tax effects
of temporary differences between the carrying amount of assets and liabilities for financial reporting purposes and amounts used for income
tax purposes. Significant components of the Company’s deferred tax assets and liabilities consist of the following:
| | |
December
31, | |
| | |
2023 | | |
2022 | |
| Deferred tax assets: | |
| | |
| |
| Net loss carryforwards | |
$ | 9,235,425 | | |
$ | 2,026,144 | |
| Capital loss carryforwards | |
| 66,063 | | |
| 72,525 | |
| Stock-based compensation | |
| 518,372 | | |
| | |
| Research
and development | |
| 131,690 | | |
| - | |
| | |
| | | |
| | |
| Deferred asset before valuation
allowance | |
| 9,951,550 | | |
| 2,098,669 | |
| Valuation
allowance | |
| (9,951,550 | ) | |
| (2,098,669 | ) |
| Net
deferred tax asset | |
$ | - | | |
$ | - | |
In assessing the realization of deferred tax assets,
management considers whether it is more likely than not that some portion or all of the deferred tax assets will be realized. The ultimate
realization of deferred tax assets is dependent upon the generation of future taxable income during the periods in which those temporary
differences become deductible. Deferred tax assets consist primarily of the tax effect of NOL carry-forwards. The Company has provided
a full valuation allowance on the deferred tax assets because of the uncertainty regarding its realizability.
The Company’s policy is to record interest
and penalties associated with unrecognized tax benefits as additional income taxes in the statement of operations. As of both December
31, 2023 and 2022 the Company had no unrecognized tax benefits. There were no changes in the Company’s unrecognized tax benefits
during the years ended December 31, 2023 and 2022. The Company did not recognize any interest or penalties during the years ended
December 31, 2023 and 2022 related to unrecognized tax benefits.
NOTE 15 – SUBSEQUENT EVENTS
The Company has evaluated subsequent events through
the date these financial statements were issued. In the opinion of management, there were no subsequent events that would require disclosure
or adjustments to the accompanying financial statements through the date the financial statements were issued other than the following:
On December 28, 2023, the Company’s
stockholders approved the adoption of the Company’s Amended and Restated Certificate of Incorporation (the “Restated Charter”)
and an amendment to the Restated Charter to increase the number of authorized shares of the Company’s common stock, par value $0.0001
per share from 500,000,000 to 3,254,475,740 and to make a corresponding change to the number of authorized shares of capital stock. On
January 4, 2024, the Company filed the Restated Charter, with the provisions of the Authorized Share Increase Amendment incorporated therein,
with the Secretary of State of Delaware.
During the first quarter of 2024, the Company
submitted a request to the Israeli Income Tax Authority, for the approval of a plan for the issuance of employee stock options via a trustee
as defined in section 102 of the Income Tax Ordinance. The Company chose a capital taxation route that would apply to the Company’s employees
and undertook to deduct the full tax applicable to employees before shares are issued to an employee.
On February 1, 2024, we entered into a consulting agreement with a
director, pursuant to which, in consideration for certain services provided as a board member, the director would receive a cash fee of
$5,000 each month. The consulting agreement is terminable by either party upon 30 days written notice to the other party, and it will
terminate automatically once the director has received fees in the aggregate amount of $55,000.
On February 1, 2024, the Company amended an agreement with an additional
director, which increased the monthly cash fee in respect of the services provided to $2,500, plus applicable VAT.
F-16
NONE
0.00
0.01
296664409
437735093
false
FY
0001282980
0001282980
2023-01-01
2023-12-31
0001282980
2023-06-30
0001282980
2024-03-31
0001282980
2023-12-31
0001282980
2022-12-31
0001282980
us-gaap:PreferredClassAMember
2023-12-31
0001282980
us-gaap:PreferredClassAMember
2022-12-31
0001282980
2022-01-01
2022-12-31
0001282980
us-gaap:SeriesAPreferredStockMember
us-gaap:PreferredStockMember
2022-12-31
0001282980
us-gaap:CommonStockMember
2022-12-31
0001282980
dror:TreasuryStocksMember
2022-12-31
0001282980
us-gaap:AdditionalPaidInCapitalMember
2022-12-31
0001282980
us-gaap:RetainedEarningsMember
2022-12-31
0001282980
us-gaap:SeriesAPreferredStockMember
us-gaap:PreferredStockMember
2023-01-01
2023-12-31
0001282980
us-gaap:CommonStockMember
2023-01-01
2023-12-31
0001282980
dror:TreasuryStocksMember
2023-01-01
2023-12-31
0001282980
us-gaap:AdditionalPaidInCapitalMember
2023-01-01
2023-12-31
0001282980
us-gaap:RetainedEarningsMember
2023-01-01
2023-12-31
0001282980
us-gaap:SeriesAPreferredStockMember
us-gaap:PreferredStockMember
2023-12-31
0001282980
us-gaap:CommonStockMember
2023-12-31
0001282980
dror:TreasuryStocksMember
2023-12-31
0001282980
us-gaap:AdditionalPaidInCapitalMember
2023-12-31
0001282980
us-gaap:RetainedEarningsMember
2023-12-31
0001282980
us-gaap:SeriesAPreferredStockMember
us-gaap:PreferredStockMember
2021-12-31
0001282980
us-gaap:CommonStockMember
2021-12-31
0001282980
dror:TreasuryStocksMember
2021-12-31
0001282980
us-gaap:AdditionalPaidInCapitalMember
2021-12-31
0001282980
us-gaap:RetainedEarningsMember
2021-12-31
0001282980
2021-12-31
0001282980
us-gaap:SeriesAPreferredStockMember
us-gaap:PreferredStockMember
2022-01-01
2022-12-31
0001282980
us-gaap:CommonStockMember
2022-01-01
2022-12-31
0001282980
dror:TreasuryStocksMember
2022-01-01
2022-12-31
0001282980
us-gaap:AdditionalPaidInCapitalMember
2022-01-01
2022-12-31
0001282980
us-gaap:RetainedEarningsMember
2022-01-01
2022-12-31
0001282980
2023-10-01
2023-12-31
0001282980
dror:PrivateDrorMember
2023-07-05
2023-07-05
0001282980
2023-07-05
2023-07-05
0001282980
us-gaap:SeriesAPreferredStockMember
2023-07-05
2023-07-05
0001282980
us-gaap:PrivatePlacementMember
2023-07-05
2023-07-05
0001282980
us-gaap:CommonStockMember
2023-07-05
2023-07-05
0001282980
dror:NovintTechnologiesIncMember
dror:PrivateDrorMember
2023-07-05
0001282980
us-gaap:CommonStockMember
2023-01-01
2023-12-31
0001282980
dror:NovintTechnologiesIncMember
2023-08-14
0001282980
us-gaap:AdditionalPaidInCapitalMember
2023-08-14
0001282980
currency:ILS
2023-12-31
0001282980
currency:ILS
2022-12-31
0001282980
country:IL
2023-12-31
0001282980
country:IL
2022-12-31
0001282980
us-gaap:ReclassificationOtherMember
2022-01-01
2022-12-31
0001282980
dror:PrivateDrorMember
2023-01-31
0001282980
2023-01-01
2023-01-31
0001282980
2023-01-31
0001282980
srt:MinimumMember
us-gaap:LicensingAgreementsMember
2023-01-01
2023-12-31
0001282980
srt:MaximumMember
us-gaap:LicensingAgreementsMember
2023-01-01
2023-12-31
0001282980
us-gaap:LicensingAgreementsMember
2023-01-01
2023-12-31
0001282980
us-gaap:LicensingAgreementsMember
2023-12-31
0001282980
us-gaap:CommonStockMember
2024-01-04
0001282980
srt:MinimumMember
us-gaap:CommonStockMember
2024-01-04
0001282980
srt:MaximumMember
us-gaap:CommonStockMember
2024-01-04
0001282980
us-gaap:TreasuryStockCommonMember
2023-02-28
2023-02-28
0001282980
2023-08-14
2023-08-14
0001282980
2023-09-13
2023-09-13
0001282980
us-gaap:CommonStockMember
us-gaap:PrivatePlacementMember
2023-01-01
2023-12-31
0001282980
us-gaap:SeriesAPreferredStockMember
us-gaap:PreferredStockMember
us-gaap:PrivatePlacementMember
2023-01-01
2023-12-31
0001282980
us-gaap:PreferredStockMember
2023-12-31
0001282980
us-gaap:SeriesAPreferredStockMember
2022-12-31
0001282980
us-gaap:SeriesAPreferredStockMember
us-gaap:PreferredStockMember
2023-01-01
2023-09-30
0001282980
us-gaap:SeriesAPreferredStockMember
2023-12-31
0001282980
us-gaap:WarrantMember
2023-01-01
2023-12-31
0001282980
dror:PrivateDrorShareholdersMember
2023-08-14
0001282980
dror:PrivatePlacementCommonStockWarrantsMember
2023-08-14
0001282980
dror:PrivatePlacementCommonStockWarrantsMember
2023-12-31
0001282980
dror:WarrantsMember
2022-12-31
0001282980
dror:WarrantsMember
2023-12-31
0001282980
dror:WarrantsMember
2021-12-31
0001282980
dror:EquityIncentivePlanMember
2023-12-31
0001282980
dror:EquityIncentivePlanMember
2023-01-01
2023-12-31
0001282980
dror:EquityIncentivePlanMember
2022-01-01
2022-12-31
0001282980
dror:EquityIncentivePlanMember
2022-12-31
0001282980
dror:EquityIncentivePlanMember
2021-12-31
0001282980
us-gaap:WarrantMember
2021-12-31
0001282980
us-gaap:WarrantMember
2021-12-31
2021-12-31
0001282980
us-gaap:WarrantMember
2022-01-01
2022-12-31
0001282980
us-gaap:WarrantMember
2022-12-31
0001282980
us-gaap:WarrantMember
2023-12-31
0001282980
us-gaap:OptionMember
2022-01-01
0001282980
us-gaap:OptionMember
2022-01-01
2022-01-01
0001282980
us-gaap:OptionMember
2022-01-01
2022-12-31
0001282980
us-gaap:OptionMember
2022-12-31
0001282980
us-gaap:OptionMember
2023-01-01
2023-12-31
0001282980
us-gaap:OptionMember
2023-12-31
0001282980
dror:SubcontractorsMember
2023-01-01
2023-12-31
0001282980
dror:SubcontractorsMember
2022-01-01
2022-12-31
0001282980
dror:SalariesMember
2023-01-01
2023-12-31
0001282980
dror:SalariesMember
2022-01-01
2022-12-31
0001282980
dror:ConsultingMember
2023-01-01
2023-12-31
0001282980
dror:ConsultingMember
2022-01-01
2022-12-31
0001282980
dror:SalariesAndRelatedMember
2023-01-01
2023-12-31
0001282980
dror:SalariesAndRelatedMember
2022-01-01
2022-12-31
0001282980
dror:LegalMember
2023-01-01
2023-12-31
0001282980
dror:LegalMember
2022-01-01
2022-12-31
0001282980
dror:ProfessionalFeesMember
2023-01-01
2023-12-31
0001282980
dror:ProfessionalFeesMember
2022-01-01
2022-12-31
0001282980
dror:RentAndUtilitiesMember
2023-01-01
2023-12-31
0001282980
dror:RentAndUtilitiesMember
2022-01-01
2022-12-31
0001282980
dror:InsuranceMember
2023-01-01
2023-12-31
0001282980
dror:InsuranceMember
2022-01-01
2022-12-31
0001282980
dror:DonationsMember
2023-01-01
2023-12-31
0001282980
dror:DonationsMember
2022-01-01
2022-12-31
0001282980
dror:OfficeExpenseMember
2023-01-01
2023-12-31
0001282980
dror:OfficeExpenseMember
2022-01-01
2022-12-31
0001282980
dror:RoyaltiesMember
2023-01-01
2023-12-31
0001282980
dror:RoyaltiesMember
2022-01-01
2022-12-31
0001282980
dror:DepreciationMember
2023-01-01
2023-12-31
0001282980
dror:DepreciationMember
2022-01-01
2022-12-31
0001282980
dror:OtherMember
2023-01-01
2023-12-31
0001282980
dror:OtherMember
2022-01-01
2022-12-31
0001282980
dror:ExchangeDifferencesMember
2023-01-01
2023-12-31
0001282980
dror:ExchangeDifferencesMember
2022-01-01
2022-12-31
0001282980
dror:BankFeesMember
2023-01-01
2023-12-31
0001282980
dror:BankFeesMember
2022-01-01
2022-12-31
0001282980
us-gaap:InternalRevenueServiceIRSMember
2023-12-31
0001282980
us-gaap:InternalRevenueServiceIRSMember
dror:PriorTo2018Member
2023-12-31
0001282980
us-gaap:InternalRevenueServiceIRSMember
dror:AfterJanuary12018Member
2018-01-01
0001282980
2018-01-01
2018-01-01
0001282980
us-gaap:IsraelTaxAuthorityMember
2023-12-31
0001282980
us-gaap:InternalRevenueServiceIRSMember
2023-01-01
2023-12-31
0001282980
us-gaap:InternalRevenueServiceIRSMember
2022-01-01
2022-12-31
0001282980
2023-12-28
0001282980
srt:MinimumMember
2023-12-28
0001282980
srt:MaximumMember
2023-12-28
0001282980
us-gaap:SubsequentEventMember
2024-02-01
2024-02-01
0001282980
us-gaap:SubsequentEventMember
2024-02-01
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
xbrli:pure
iso4217:ILS
The following summary of the material terms of the capital stock
of Dror Ortho-Design, Inc. (“we,” “our” and the “Company”) is not intended to be a complete summary
of the rights and preferences of such securities. The full text of the our certificate of incorporation, as amended (the “Amended
Charter”) and our bylaws, as amended (the “Bylaws”) are included as exhibits to our Annual Report on Form 10-K.
You are encouraged to read the applicable provisions of Delaware law, the Amended Charter and Bylaws in their entirety for a complete
description of the rights and preferences of our securities.
We have authorized 3,266,975,740 shares of capital stock, par value
$0.0001 per share, of which 3,254,475,740 are shares of common stock (the “Common Stock”) and 12,500,000 are shares of “blank
check” preferred stock.
As of the date hereof, there are 495,454,546 shares of Common Stock
outstanding.
Pursuant to our Amended Charter, holders of our Common Stock are
entitled to one vote for each share held on all matters submitted to a vote of our stockholders. Holders of our Common Stock have no cumulative
voting rights. All shares of our Common Stock validly authorized and issued, fully paid and nonassessable.
Holders of our Common Stock have no preemptive, redemption, conversion
or subscription rights. No sinking fund provisions are applicable to our Common Stock. Upon liquidation, dissolution or winding-up, holders
of our Common Stock are entitled to share in all assets remaining after payment of all liabilities and the liquidation preferences of
any of our outstanding shares of preferred stock. Subject to preferences that may be applicable to any outstanding shares of preferred
stock, holders of our Common Stock are entitled to receive dividends, if any, as may be declared from time to time by our board of directors
out of our assets which are legally available. Such dividends, if any, are payable in cash, in property or in shares of capital stock.
A holders of one-third of the voting power of the stock issued,
outstanding and entitled to vote thereat, present in person or represented by proxy, will constitute a quorum for the transaction of business
at any meeting of our stockholders. If, however, such quorum is not present or represented at any meeting of the stockholders, then either
(i) the chairperson of the meeting, if there be one, or (ii) the stockholders entitled to vote at the meeting, present in person or represented
by proxy, will have power to adjourn the meeting from time to time, without notice other than announcement at the meeting, until a quorum
is present or represented.
Our board of directors has the authority, without further action
by the stockholders, to issue up to 12,500,000 shares of preferred stock in one or more Series And to fix the designations, powers,
preferences, privileges, and relative participating, optional, or special rights as well as the qualifications, limitations, or restrictions
of the preferred stock, including dividend rights, conversion rights, voting rights, terms of redemption, and liquidation preferences,
any or all of which may be greater than the rights of the Common Stock. Our board of directors, without stockholder approval, can issue
convertible preferred stock with voting, conversion, or other rights that could adversely affect the voting power and other rights of
the holders of Common Stock. Preferred stock could be issued quickly with terms calculated to delay or prevent a change of control or
make removal of management more difficult. Additionally, the issuance of preferred stock may have the effect of decreasing the market
price of our Common Stock, and may adversely affect the voting and other rights of the holders of Common Stock.
On August 11, 2023, we filed a Certificate of Designation of Preferences,
Rights and Limitations with the Secretary of State of the State of Delaware designating 12,500,000 shares out of the authorized but unissued
shares of its preferred stock as Series A Preferred Stock with a stated value of $1.10 per share (the “Certificate of Designation”).
There are 10,463,362.24 shares of Series A Preferred Stock issued and outstanding. The following a summary of the principal terms
of the Series A Preferred Stock as set forth in the Certificate of Designation is qualified in its entirety by reference to our Amendment
Charter, of which the Certificate of Designation forms a part.
The holders of Series A Preferred Stock will be entitled to
dividends, on an as-if converted basis, equal to and in the same form as dividends actually paid on shares of Common Stock, when and if
actually paid.
The shares of Series A Preferred Stock are entitled to vote
with holders of the Common Stock on all matters that such holders of Common Stock are entitled to vote upon, in the same manner and with
the same effect as the holders of Common Stock, voting together with the holders of Common Stock as a single class. Each share of Series A
Preferred Stock shall entitle the Holder thereof to cast that number of votes per share of Series A Preferred Stock equal to the
number of Conversion Shares into which such share of Series A Preferred Stock is convertible into pursuant to the Certificate of
Designation (after giving effect to any applicable limitation on conversion under the Certificate of Designation). As long as any shares
of Series A Preferred Stock are outstanding, we may not, without the approval of a majority of the then outstanding shares of Series A
Preferred Stock (a) alter or change the powers, preferences or rights given to the Series A Preferred Stock, (b) alter
or amend our amended and restated certificate of incorporation, the Certificate of Designation, or our amended and restated bylaws in
such a manner so as to materially adversely affect any rights given to the Series A Preferred Stock, (c) authorize or create
any class of stock ranking as to dividends, redemption or distribution of assets upon a Liquidation (as defined below) senior to the Series A
Preferred Stock, or (d) enter into any agreement to do any of the foregoing.
Upon any liquidation, dissolution or winding-up of the Company,
whether voluntary or involuntary (a “Liquidation”), the then holders of the Series A Preferred Stock are entitled to
receive out of the assets available for distribution to stockholders of the Company the same amount that a holder of Common Stock would
receive if the Series A Preferred Stock were fully converted (disregarding for such purposes any conversion limitations hereunder)
to Common Stock which amounts shall be paid pari passu with all holders of Common Stock.
The Series A Preferred Stock is convertible into Common Stock
at any time at a conversion price of $0.011, subject to adjustment for certain anti-dilution provisions set forth in the Series A
Certificate of Designation (the “Series A Conversion Price”). Upon conversion the shares of Series A Preferred Stock
will resume the status of authorized but unissued shares of preferred stock of the Company.
The Series A Preferred Stock is convertible at the then-effective
Series A Conversion Price at the option of the holder at any time and from time to time.
On the trading day immediately following any day the Company is
able to satisfy some or all of its reservation requirements pursuant to the Certificate of Designation (the “Automatic Conversion
Time”), all, but not less than all, of the outstanding shares of Series A Preferred Stock for which Common Stock has been reserved
will automatically convert, without any action on the part of the holder thereof and without payment of any additional consideration,
into that number of shares of reserved Common Stock, determined by dividing the stated value of such share of Series A Preferred
Stock by the Series A Conversion Price. The Company will provide prompt written notice to the holders of Series A Preferred
Stock of the Automatic Conversion Time on the trading day immediately following the Automatic Conversion Time.
The Series A Preferred Stock cannot be converted to common
stock if the holder and its affiliates would beneficially own more than 4.99% (or 9.99% at the election of the holder) of the outstanding
common stock. However, any holder may increase or decrease such percentage to any other percentage not in excess of 9.99% upon notice
to us, provided that any increase in this limitation will not be effective until 61 days after such notice from the holder to us and such
increase or decrease will apply only to the holder providing such notice.
No holders of Series A Preferred Stock will, as holders of
Series A Preferred Stock, have any preemptive rights to purchase or subscribe for common stock or any of our other securities.
The shares of Series A Preferred Stock are not redeemable by
the Company.
As long as any shares of Preferred Stock are outstanding, unless
the holders of more than 50% in stated value of the then outstanding shares of Preferred Stock shall have otherwise given prior written
consent the Company shall not, subject to certain exceptions, (a) enter into, create, incur, assume, guarantee or suffer to exist any
indebtedness, (b) enter into, create, incur, assume or suffer to exist any liens, (c) amend its organizational documents in any manner
that materially and adversely affects any rights of the holders, (d) repay, repurchase or offer to repay, repurchase or otherwise acquire
any shares of its common stock, common stock equivalents or junior securities, (e) enter into any transaction with any affiliate of the
Company which would be required to be disclosed in any public filing with the SEC, unless such transaction is made on an arm’s-length
basis and expressly approved by a majority of the disinterested directors of the Company, (f) declare or pay a dividend on junior securities
or (g) enter into any agreement with respect to any of the foregoing.
There is no established trading market for any of the Series A
Preferred Stock, and we do not expect a market to develop. We do not intend to apply for a listing for any of the Series A Preferred
Stock on any securities exchange or other nationally recognized trading system. Without an active trading market, the liquidity of the
Series A Preferred Stock will be limited.
In connection with the private placement and the share exchange
that closed on August 14, 2023, we issued to the investors party to such transactions five-year warrants (the “Warrants”)
to purchase up to an aggregate of 964,834,419 shares of Common Stock at an exercise price of $0.033 per share. We are prohibited from
effecting the exercise of any Warrant to the extent that as a result of such exercise the holder of the exercised Warrant beneficially
owns more than 4.99% in the aggregate of the issued and outstanding shares of our Common Stock calculated immediately after giving effect
to the issuance of shares of our Common Stock upon the exercise of the Warrant. The Warrants contain provisions that protect their holders
against dilution by adjustment of the purchase price in certain events such as stock dividends, stock splits and other similar events.
If at the time of a Warrant’s exercise there is no effective
registration statement registering, or no current prospectus available for, the resale of the Warrant Shares, then the holder will have
the right to exercise the Warrant by means of a cashless exercise. In addition, if (i) the volume-weighted average price of our Common
Stock for 20 consecutive trading days is at least 300% of the exercise price of the Warrants, (ii) the dollar trading volume of our Common
Stock for each trading day within such 20-day trading period equals or exceeds $500,000, (iii) a registration statement providing for
the resale of the Warrant Shares is effective and such registration statement has been effective for six (6) months, (iv) the holder of
the Warrant is not in possession of any information provided by the Company that constitutes material nonpublic information and (v) the
Company has not breached any of the terms of the documents governing the warrants or the transaction in which they were issued (regardless
of if such breach has been cured), then we may redeem the Warrants at a price of $0.001 per Warrant up to one-half, in the aggregate,
of the Warrants upon not less than 20 days’ prior written notice of redemption to each holder, subject to certain customary restrictions.
We have shares of Common Stock and preferred stock available for
future issuance without stockholder approval. We may utilize these additional shares for a variety of corporate purposes, including future
public offerings to raise additional capital, to facilitate corporate acquisitions or payment as a dividend on the capital stock.
The existence of unissued and unreserved Common Stock and preferred
stock may enable our board of directors to issue shares to persons friendly to current management or to issue preferred stock with terms
that could render more difficult or discourage a third-party attempt to obtain control of our company by means of a merger, tender offer,
proxy contest or otherwise, thereby protecting the continuity of our company’s management. In addition, our board of directors has
the discretion to determine designations, rights, preferences, privileges and restrictions, including voting rights, dividend rights,
conversion rights, redemption privileges and liquidation preferences of each series of preferred stock, all to the fullest extent permissible
under the Delaware General Corporation Law (the “DGCL”) and subject to any limitations set forth in our Amended Charter. The
purpose of authorizing our board of directors to issue preferred stock and to determine the rights and preferences applicable to such
preferred stock is to eliminate delays associated with a stockholder vote on specific issuances. The issuance of preferred stock, while
providing desirable flexibility in connection with possible financings, acquisitions and other corporate purposes, could have the effect
of making it more difficult for a third-party to acquire, or could discourage a third party from acquiring, a majority of our outstanding
voting stock.
We currently intend to use all available funds to develop our business
and do not anticipate that we will pay dividends in the future. We can give no assurances that we will ever have excess funds available
to pay dividends.
Anti-Takeover Effects of Certain Provisions of our Certificate
of Incorporation, Bylaws and the DGCL
Provisions of our Amended Charter and Bylaws may delay or discourage
transactions involving an actual or potential change in our control or change in our management, including transactions in which stockholders
might otherwise receive a premium for their shares, or transactions that our stockholders might otherwise deem to be in their best interests.
Therefore, these provisions could adversely affect the price of our Common Stock. Among other things, our Amended Charter and Bylaws:
We are subject to Section 203 of the DGCL. Section 203 generally
prohibits a public Delaware corporation from engaging in a “business combination” with an “interested stockholder”
for a period of three years after the date of the transaction in which the person became an interested stockholder, unless:
In general, Section 203 defines an “interested stockholder”
as any entity or person beneficially owning 15% or more of the outstanding voting stock of the corporation and any entity or person affiliated
with, or controlling, or controlled by, the entity or person. The term “owner” is broadly defined to include any person that,
individually, with or through that person’s affiliates or associates, among other things, beneficially owns the stock, or has the
right to acquire the stock, whether or not the right is immediately exercisable, under any agreement or understanding or upon the exercise
of warrants or options or otherwise or has the right to vote the stock under any agreement or understanding, or has an agreement or understanding
with the beneficial owner of the stock for the purpose of acquiring, holding, voting or disposing of the stock.
The restrictions in Section 203 do not apply to corporations that
have elected, in the manner provided in Section 203, not to be subject to Section 203 of the DGCL or, with certain exceptions, which do
not have a class of voting stock that is listed on a national securities exchange or held of record by more than 2,000 stockholders. Our
Amended Charter and Amended Bylaws do not opt out of Section 203.
Section 203 could delay or prohibit mergers or other takeover or
change in control attempts with respect to us and, accordingly, may discourage attempts to acquire us even though such a transaction may
offer our stockholders the opportunity to sell their stock at a price above the prevailing market price.
IN WITNESS WHEREOF, the parties have
executed this Agreement as of the Effective Date.
Service Provider shall receive $US 5,000 per each
full month of services provided (up to $US 55,000).
This certification is furnished solely pursuant
to Section 906 of the Sarbanes-Oxley Act of 2002 (18 U.S.C. 1350) and accompanies the Annual Report on Form 10-K (the “Form 10-K”)
for the year ended December 31, 2023, of Dror-Ortho Design, Inc. (the “Company”). I, Eliyahu (Lee) Haddad, the Chief
Executive Officer of the Company, certify that, based on my knowledge: