Roche Receives US FDA Emergency Use Authorization for Zika Test
29 August 2016 - 7:24PM
Dow Jones News
By Denise Roland
Roche Holding AG said a Zika virus test developed by its
diagnostics unit has received emergency use authorisation from the
U.S. Food and Drug Administration.
The Swiss drug maker said the test detects the virus in the
blood using its LightMix or cobas z 480 machines, both small and
easy-to-use systems.
It is the latest diagnostic test to receive emergency
authorisation in the U.S. in response to the threat posed by the
Zika virus. The virus can be detected in either the blood or
urine.
The FDA grants this status to unapproved products whose use is
deemed necessary in public health emergencies.
Roche is already providing a test that screens blood donations
for the Zika virus. That test was made available under an
investigative new drug program, another route for unapproved
products to be made available under special circumstances.
Write to Denise Roland at denise.roland@wsj.com
(END) Dow Jones Newswires
August 29, 2016 05:09 ET (09:09 GMT)
Copyright (c) 2016 Dow Jones & Company, Inc.
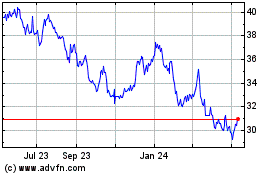
Roche (QX) (USOTC:RHHBY)
Historical Stock Chart
From Apr 2024 to May 2024
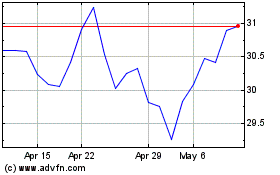
Roche (QX) (USOTC:RHHBY)
Historical Stock Chart
From May 2023 to May 2024