FDA Approves Genentech's Evrysdi for Spinal Muscular Atrophy
08 August 2020 - 5:17AM
Dow Jones News
By Stephen Nakrosis
The U.S. Food and Drug Administration on Friday said it gave
approval to Genentech Inc.'s Evrysdi as a treatment for spinal
muscular atrophy.
The FDA said Evrysdi, or risdiplam, was approved to "treat
patients two months of age and older with spinal muscular atrophy,
a rare and often fatal genetic disease affecting muscle strength
and movement," and added, "This is the second drug and the first
oral drug approved to treat this disease."
The FDA also said "The efficacy of Evrysdi for the treatment of
patients with infantile-onset and later-onset SMA was evaluated in
two clinical studies."
--Write to Stephen Nakrosis at stephen.nakrosis@wsj.com
(END) Dow Jones Newswires
August 07, 2020 15:02 ET (19:02 GMT)
Copyright (c) 2020 Dow Jones & Company, Inc.
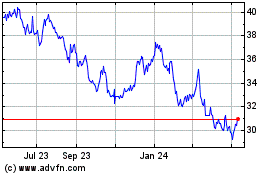
Roche (QX) (USOTC:RHHBY)
Historical Stock Chart
From Apr 2024 to May 2024
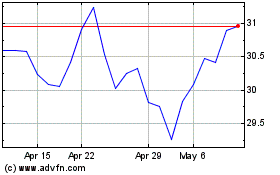
Roche (QX) (USOTC:RHHBY)
Historical Stock Chart
From May 2023 to May 2024