TIDMOBD
RNS Number : 9177Z
Oxford BioDynamics PLC
17 January 2024
Oxford Biodynamics Plc
("OBD" or the "Company" and, together with its subsidiaries, the
"Group")
Preliminary results for the year ended 30 September 2023
and
Notice of Annual General Meeting
Oxford, UK - 17 January 2024 - Oxford BioDynamics Plc (AIM:
OBD), a biotechnology company developing precision medicine tests
based on the EpiSwitch(R) 3D genomics platform , today announces
its final results for the year ended 30 September 2023.
Highlights
Corporate and operational highlights
-- 516 EpiSwitch(R) CiRT tests processed in the year, ordered by
57 doctors (FY22: 79 tests, 7 doctors)
-- Second FNIH PACT Award, investigating immuno-oncology-related
Hyper Progressive Disease (HPD), worth $963,000 over one year (May
2023)
-- US clinical laboratory established in Frederick, MD (from
April 2023)
-- Two successful fundraises: raising gross proceeds of GBP9.3m
(October 2022) and GBP6.1m (August 2023)
-- Successful launch of EpiSwitch(R) PSE Prostate Screening Test
in the US and UK, ahead of schedule (September 2023)
Financial highlights
-- Revenue of GBP0.5m (FY22: GBP0.2m)
-- Other operating income of GBP0.8m (FY22: GBP0.4m)
-- Operating loss of GBP10.2m (FY22: GBP8.6m); increase
reflecting investment in team and infrastructure to support CiRT
and PSE
-- Cash and term deposits of GBP5.3m as at 30 September 2023
(FY22: GBP1.0m)
Post-year end highlights
-- Unique CPT-PLA code assigned for PSE, available for use from
1 January 2024, enabling accurate reimbursement in the US from
Medicare, Medicaid or private payors (October 2023)
-- Agreement with leading UK health insurer, Bupa UK, to cover
EpiSwitch CiRT (October 2023)
-- Confidential discussions commenced with third parties
regarding monetizing OBD's two most advanced pipeline assets:
EpiSwitch(R) NST (No Stool Test) for colorectal/bowel cancer and
EpiSwitch(R) SCB (Specific for Canine Blood) blood test for
detection of multiple types of canine cancer
-- Total PSE orders to date of 144, total CiRT orders to date of
770
Commenting on the results, Chief Executive Officer Jon Burrows
said:
"This year we again made significant, rapid progress in the
transformation of OBD that began with our expanded strategy in late
2020.
"We now have two precision medicine tests on the market, each
with a unique CPT-PLA code in the US. We established and are now
running PSE tests from our own CLIA-registered clinical operations
laboratory in Frederick, MD, with a UK lab scheduled to begin
operation by the end of March 2024. We received a second
prestigious PACT Award, a further sign of the growing recognition
of the power of our EpiSwitch(R) technology to address seemingly
intractable problems with non-invasive precision medicine
testing.
"We have achieved all this thanks to the support of our
investors, both longstanding and new to OBD, from whom we raised
over GBP15 million during the year.
"This year, we are dedicated to growing sales of both
EpiSwitch(R) CiRT and EpiSwitch(R) PSE across all our markets and
channels and to pursuing opportunities to monetize assets from
within our pipeline of deployable tests."
-Ends-
Notice of Annual General Meeting
The Company's Annual General Meeting will be held at 3140 Rowan
Place, John Smith Drive, Oxford Business Park South, Oxford, OX4
2WB, UK on 27 March 2024 at 12.00 pm.
The information included in this announcement is extracted from
the Annual Report, which was approved by the Directors on 16
January 2024. Defined terms used in the announcement refer to terms
as defined in the Annual Report unless the context requires
otherwise. This announcement should be read in conjunction with,
and is not a substitute for, the full Annual Report.
The information contained within this announcement is deemed to
constitute inside information as stipulated under the Market Abuse
Regulations (EU) No. 596/2014 which is part of domestic UK law
pursuant to the Market Abuse (Amendment) (EU Exit) Regulations (SI
2019/310) ("UK MAR"). Upon the publication of this announcement,
this inside information (as defined in UK MAR) is now considered to
be in the public domain.
Investor webinar
The Company's management team will conduct a live presentation
via the Yellowstone Advisory webinar platform to investors, at 3pm
GMT on Wednesday 17 January 2024.
The online presentation is open to both existing and potential
shareholders. Questions will be addressed at the end of the
presentation and may be submitted either before the presentation by
emailing info@yellowstoneadvisory.com or during the
presentation.
To register for the presentation, please visit:
https://us02web.zoom.us/webinar/register/7417006471039/WN_ZqpvQz8hQOKMufXOKPwH1A
or alternatively via the Yellowstone events calendar at
www.yellowstoneadvisory.com/events .
For further details please contact:
Oxford BioDynamics Plc Tel: +44 (0)1865 518910
Jon Burrows, CEO
Paul Stockdale, CFO
Shore Capital - Nominated Adviser and Tel: +44 (0)20 7408 4090
Broker
Advisory: Stephane Auton / Iain Sexton
Broking: Fiona Conroy
Instinctif Partners - Financial PR Tel: +44 (0)20 7457 2020
Melanie Toyne-Sewell / Katie Duffell OxfordBioDynamics@instinctif.com
/ Jack Kincade
Chief Executive Officer's review
Introduction
We had two main objectives during the year to September 2023: to
continue to grow orders of our flagship EpiSwitch(R) CiRT
(Checkpoint Inhibitor Response Test) and to accelerate the
development and launch of our next test, the EpiSwitch(R) PSE
Prostate Screening Test. We are pleased with the progress made in
pursuit of both these objectives. CiRT orders grew consistently
over the year and we launched PSE - "the 94% test" - in the US and
UK ahead of schedule on 26 September 2023.
Alongside this work, we received a second FNIH PACT Award to
fund work on immuno-oncology (IO)-related hyper progressive
disease, announced compelling results using OBD's EpiSwitch
platform in the diagnosis and stratification of amyotrophic lateral
sclerosis (ALS, or motor neurone disease) and completed two
successful fundraises, raising a total of GBP15.4m (before costs)
in the year.
The last year has been a successful one for the Company on
several fronts. CiRT is becoming established in the US market and
PSE is also now launched after we expedited its final development
in response to unprecedented interest. OBD's EpiSwitch technology,
through which all of our commercial products are developed, is
increasingly recognised as able to address the clinical challenges
of personalized medicine, cancer treatment, and immune health.
Our financial results reflect the Company's ongoing commercial
development. Revenues of GBP0.51m (2022: GBP0.15m) included the
first significant amounts from the Group's proprietary tests as we
began to receive reimbursements from US payors for EpiSwitch CiRT,
alongside revenues from projects for pharma and other customers.
Other operating income, from our two PACT Awards and our
involvement in the EU-funded HIPPOCRATES Consortium also increased,
to GBP0.83m (2022: GBP0.35m). Our investment in supporting CiRT and
bringing PSE to market led to cost increases, the largest of which
were in staff and general and administrative costs. Overall, the
operating loss for the year was 18% higher than last year, at
GBP10.17m (2022: GBP8.60m). More detail is provided in the
financial review that follows.
We begin 2024 with continued focus on our products - seeking to
continue to increase adoption of both PSE and CiRT - and on our
promising product pipeline and R&D work. We have already
commenced discussions with third parties, to explore the potential
monetization of the two most advanced assets in our pipeline
(EpiSwitch(R) NST for colorectal/bowel cancer and EpiSwitch(R) SCB
for canine cancer), which we believe could lead to significant
non-dilutive funding for the Company.
EpiSwitch(R) CiRT (Checkpoint Inhibitor Response Test)
Launched in February 2022, EpiSwitch(R) CiRT is OBD's flagship
product, a first-of-its-kind predictive test of a patient's likely
response to immune checkpoint inhibitors (ICIs), which work to
stimulate a patient's immune system to find and fight cancer. 516
CiRT tests were ordered in the year to 30 September 2023, by a
total of 57 doctors (FY22: 79 tests, ordered by 7 doctors). Up to
the date of this report, a total of 770 CiRT tests have now been
ordered.
Building on early progress in a single territory in the prior
year, we expanded our sales and market access team to two more
territories to introduce the test to more doctors. A unique CPT-PLA
code, allowing reimbursement for CiRT tests from US insurers, has
been available throughout the period. We have achieved strong
reimbursements from US payors under the unique code, against a list
price of $4,950. As previously outlined, the Group has worked with
its partner laboratory, NEXT Molecular Diagnostics, to manage the
claim reimbursement process. The extensive experience and
specialist knowledge of members of both the OBD and NEXT teams
continues to be invaluable in optimising claims reimbursement for
the test. With NEXT, we have maintained an excellent level of
service, with an average turn-around time for CiRT (measured from
sample receipt to provision of a final test report) for the period
of just over four days.
Over the year, OBD's US sales and market access professionals
have spent time learning from oncologists how the test has aided
them in determining treatment pathways for their patients. Later in
the year, we began a series of peer group advisory sessions, at
which doctors who routinely order CiRT tests shared their
experience of the test with colleagues. We expect to continue with
this peer-to-peer approach to growing demand for CiRT through the
current financial year, as part of the comprehensive strategy for
the test outlined below.
After particularly strong growth into June 2023, we expected and
saw lower orders through the summer months. Post-year end, in the
final quarter of the calendar year, orders per day have remained at
similar levels to the summer: this reflected a combination of
continued growth in some territories but at the same time, the
impact of an unexpected staff leave of absence that affected orders
in our prime territory and the operation of some of our recently
introduced clinical advisory boards. It is positive to note that we
had continued orders from a core group of oncologists in this
territory, but this has demonstrated the current extent of our
reliance on what is still a relatively small team.
In the meantime, to enhance the leadership of the CiRT sales
team, we brought Ryan Mathis, MD onboard at the beginning of
December 2023. Dr Mathis is a physician who, along with clinical
expertise, has an impressive background in business development and
running sales teams for innovative healthcare products. He also
brings an added level of gravitas to our peer-to-peer approach with
doctors. To date, CiRT has been sold primarily to innovator and
early adopter oncologists, who are specialists in providing expert
care to cancer patients. Ryan has started to analyze our progress
and success in selling bottom-up into this segment of oncologists,
to understand how these doctors are applying CiRT with respect to
the algorithms they have been trained to use to treat their
patients. He intends to refine our speaker programs and clinical
advisory boards to continue to take advantage of and grow our
peer-to-peer sales strategy. He will also implement a rigorous
clinical sales training program, along with a national conference
strategy.
HEOR (Health Economics and Outcomes Research) data is critical
for payors seeking to use their resources as effectively and
efficiently as possible and informs their decisions on coverage and
payment / reimbursement for the test and IO treatments. Dr Mathis
will use the clinical data from the 750-plus real-world cases we
have gathered so far from oncologists to present the test's usage
and clinical utility, engaging with an expert third party
consulting group to set out the compelling HEOR story of CiRT.
Building the HEOR case for CiRT with this real-world evidence,
we also expect to prepare data to support an assertive campaign for
CiRT to be added to the National Comprehensive Cancer Network
(NCCN) Guidelines(R) and physician compendia, published resources
from independent professional organizations which are the
recognized standard for clinical direction and policy in cancer
care and which drive physician behaviour. The NCCN alliance
includes 33 cancer centers across the US with its own peer-reviewed
journal. The intent of the NCCN Guidelines is to assist in the
decision-making process of individuals involved in cancer care -
including physicians, nurses, pharmacists, payers, patients and
their families-with the ultimate goal of improving patient care and
outcomes. Inclusion in the NCCN Guidelines is vital for bringing
the test into the orbit of as many oncologists as possible. Tests
that are included in the guidelines are regularly added by default
to the databases of the electronic medical record (EMR) systems
used in doctors' practices and healthcare networks. This would
therefore rapidly bring CiRT to the attention of more doctors, make
ordering the test easier for oncologists and likely lead to more
patients benefiting from the test. Inclusion in the Guidelines can
also be relevant to some payors' coverage decisions.
Post-year end, in October 2023, we announced our agreement with
the UK's leading health insurer Bupa UK, to give Bupa patients who
are being considered for or already on ICI therapy access to OBD's
EpiSwitch CiRT. This marked our first direct agreement with a
private medical insurer for the reimbursement of CiRT and the first
agreement with a major customer outside of the US. As well as
agreeing to reimburse EpiSwitch CiRT, the partnership represents
the first time that Bupa will be actively marketing a genomic test
to their network of healthcare providers. Bupa is advocating for
CiRT's adoption by facilitating a series of OBD roadshows in some
of the UK's largest private cancer care clinics throughout the
first half of 2024. Bupa UK provides health and dental insurance to
over 3 million people. We expect that joining forces with a
pioneering healthcare organization like Bupa will significantly
enhance access to EpiSwitch CiRT across the UK. Gaining
reimbursement from the UK's leading health insurer was a milestone
for OBD and we intend to capitalize on this with similar agreements
with other insurers and healthcare networks, in all our markets,
over the coming months.
The market opportunity for CiRT is significant, with unmet need
for patients, doctors and payors alike. Nine anti-PD-(L)1 ICIs are
now approved by the FDA for a variety of cancer indications.
Hundreds of thousands of patients are treated with these
therapeutics each year, but, whilst efficacy is improving and
varies across different types of cancer, on average fewer than one
third of patients show a positive response from treatment and many
experience unwelcome, sometimes serious, side effects. Doctors have
shared several case studies in which the actionable information
provided by CiRT has helped them to determine treatment pathways
for their patients with increased confidence. From the perspective
of payors, it is estimated that in excess of $10 billion is spent
on ineffective ICI therapy every year in the US alone, meaning that
smart testing with CiRT has the potential to offer more efficient,
effective use of patients' and payors' financial resources. CiRT
also offers obvious potential advantages for pharmaceutical
development programs, by helping to stratify and analyse patients
in more targeted clinical trials.
We therefore aim, with Ryan leading the CiRT sales team, to
resume growth in orders of CiRT through 2024 and our build process
is continuing to that end, with the mindful allocation of finite
resources to targeted expansion of the team, with a particular
focus on major accounts, generating the HEOR story for CiRT and
getting the test included in the NCCN Guidelines, alongside
expansion into more territories.
EpiSwitch(R) PSE (Prostate Screening Test)
EpiSwitch PSE ("the 94% test") is a non-invasive blood test that
accurately detects prostate cancer risk, reducing the number of men
referred for an unnecessary biopsy and treatment. The PSE test
measures five epigenetic biomarkers and combines these with a
patient's PSA (prostate-specific antigen) score to accurately
predict the presence or absence of prostate cancer.
PSE has high overall accuracy of 94% (sensitivity 86%,
specificity 97%), representing a huge boost in accuracy compared to
a PSA test alone. Crucially, the positive predictive value (PPV) of
PSE is 93%, compared to just 32% for PSA. This low PPV is one of
the main impediments to using PSA as a population-wide screening
test. Fewer than one third of men with a raised PSA will go on to
be diagnosed with prostate cancer. PSE's PPV of 93%, means that 93
of every 100 men who receive a "high probability" PSE result will
go on to receive a prostate cancer diagnosis.
Our work on EpiSwitch PSE, culminating in its launch in
September 2023, represents a major achievement for the year.
Publication in February 2023 of compelling results involving OBD's
technology in the multi-disciplinary PROSTAGRAM study led to such
significant interest that we decided to expedite the final
development and commercial launch of the test. Indeed, the paper
co-authored by members of the OBD team and investigators from the
University of East Anglia, Imperial College London and King's
College London entitled "Circulating chromosome conformation
signatures significantly enhance PSA positive predicting value and
overall accuracy for prostate cancer detection" and published in
the journal Cancers(1) , was one of the most viewed papers in that
journal on cancer causes, screening and diagnosis in 2023.
Following publication of the groundbreaking results, OBD
completed the development and validation of the commercial test and
leased, staffed and commissioned a CLIA-registered US clinical
laboratory in Frederick, MD, where the test is performed. An
application for a unique CPT-PLA(++) code for PSE was submitted in
early July 2023 and the code, 0433U, was assigned in September 2023
and has been available for use by Medicare, Medicaid and private
payors from 1 January 2024. We also announced plans to develop a UK
clinical laboratory, compliant with the requirements of ISO
15189:2012 (Medical Laboratories), in our existing Oxford HQ. We
will begin validation procedures in January 2024 and expect the UK
lab to begin processing PSE clinical samples by the end of March
2024.
The addressable market for PSE is very large: there are
approximately 47 million men aged between 50 and 74 in the US and
approximately 10 million men in the same age bracket in the UK.
There is no population-wide screening programme for prostate
cancer, although some 25 million PSA tests are performed annually
in the US, which lead to around one million biopsies being carried
out. With a prostate cancer incidence rate of approximately 250,000
new cases in the US each year, there are too many needless,
invasive procedures currently being performed. We believe PSE
represents an opportunity to build an efficient screening regimen
to go from PSA through PSE and onto invasive biopsy then treatment
only when necessary. A screening programme which would be minimally
invasive, fast, accurate and cost-effective could improve early
detection of this terrible disease, thereby improving treatment
outcomes at the same time as minimising unnecessary biopsies.
Our sales and marketing approach for PSE reflects its
applicability to all men in the at-risk age bracket (rather than
those already diagnosed with cancer and being considered for a
particular therapy, as is the case with the CiRT test). OBD's
online advertising in the US therefore addresses men and their
families, educating them, as well as their physicians on the
benefits of "the 94% test". Like CiRT, PSE must be ordered by a
registered doctor.
An example of the growing awareness and recognition of PSE was
the appearance in December 2023 of OBD's Laboratory Medical
Director, Dr Robert Heaton, on the Prostate Health Podcast(2) ,
hosted by Garrett D. Pohlman, MD, a board-certified urologist who
has treated over 4,000 men for various prostate conditions and has
begun using the PSE test for his patients. The podcast, focused on
prostate health education, reached many thousands of viewers and
listeners in 148 countries in 2023. As an experienced
board-certified pathologist, Dr Heaton is eminently qualified to
explain how the test is being used to benefit patients, by
providing physicians with a precise tool to assess whether a
patient should undergo a biopsy or opt for continued
monitoring.
In addition, post-year end in November 2023, we were pleased to
announce that we had recruited experienced life science business
development executive Dr Steve Arrivo to the OBD team. Steve joined
OBD as SVP, Business and Corporate Development. Dr Arrivo has a big
job, starting with an initial focus on analyzing and evolving our
direct-to-customer marketing approach, engaging partners for
national distribution, and selling access to PSE to larger
accounts. This begins with the large concierge groups and will
expand into the integrated delivery networks (IDNs), other
healthcare systems and the General Purchasing Organizations (GPOs)
that all hospitals work through. During 2024 he will also lead
initiatives to craft and distil the Health Economics and Outcomes
Research (HEOR) story for PSE, drive awareness and utilization of
the test with KOLs, attend and present at strategic conferences,
collaborate with advocacy groups and petition for inclusion of PSE
into the National Comprehensive Cancer Network (NCCN)
Guidelines(R).
Early uptake of the PSE test has been positive, with more than
140 tests processed up to the date of this report, for patients in
the US and UK. Encouragingly, even before Steve's initiatives, the
number of tests ordered per day - a key metric - has increased from
just over one a day in October to just over two a day in December.
US PSE test orders can now be invoiced under our unique CPT-PLA
code (with effect from 1 January 2024). Tests self-paid by
patients, or otherwise reimbursed by non-US insurers, have
accounted for around 15% of orders to date, at a rate of GBP750 (or
equivalent) per test.
Development Pipeline
During the year our R&D and product development teams have
worked on internal, grant-funded and contractual projects in a wide
range of indications and therapy areas. Excellent progress has been
made with our programs in colorectal/bowel cancer, canine oncology
(animal health), amyotrophic lateral sclerosis (ALS, or motor
neurone disease), rheumatoid arthritis, psoriasis/psoriatic
arthritis, immuno-oncology and non-alcoholic steatohepatitis
(NASH).
From OBD's extensive pipeline of deployable molecular tests, two
programs are now ready for commercialization, whether as OBD
proprietary tests, or co-developed or out licensed products. These
two tests are EpiSwitch(R) NST (No Stool Test), a screening blood
test for colorectal/bowel cancer and EpiSwitch(R) SCB (Specific for
Canine Blood), a multi-indication diagnostic test for the most
commonly occurring types of canine cancer.
We recognize that early monetization and commercialization of
each of these two programs is more likely to occur with, and would
benefit from, the involvement of a partner organization with
significant presence in the relevant market. To this end, as noted
above, confidential discussions with third parties have already
commenced to explore possible options for these two most advanced
pipeline assets. As well as expediting the launch and availability
of these high-performing tests, we believe this approach could
potentially lead to significant non-dilutive funding for the
Company.
EpiSwitch(R) Explorer Array Kit
The EpiSwitch Explorer Array Kit (EAK), launched in 2022, allows
members of the life science research community to access OBD's
EpiSwitch 3D genomics technology, using Agilent-manufactured
EpiSwitch whole genome microarrays and OBD's proprietary
biochemical reagents for sample preparation.
The EAK allows whole genome-wide interrogation of just under 1
million of the most critical interactions between 3D anchor sites
(the Company's proprietary "EpiSwitch loci") on the human genome,
offering powerful new discovery information to researchers,
including confirmation or clarification of their hypotheses.
Included in the purchase price of the EAK is access to first tier
analysis software developed in-house by OBD's Data team. For
researchers without access to appropriate microarray equipment, the
Company's scientists can analyse samples of interest using the Kit
as a paid-for service.
Explorer Array Kits have been purchased by scientists from
several prestigious academic research institutions, including The
Francis Crick Institute, the University of Oxford Department of
Biochemistry and the University of the Algarve. Results from
academic life-science research based on EpiSwitch Explorer Arrays
have already been presented at national and international
scientific peer group meetings.
Second PACT Award
In May 2023, the Company was granted a second Partnership for
Accelerating Cancer Therapies ("PACT") Award. PACT is a five-year,
$220 million, public-private research collaboration between the
National Institutes of Health (NIH), the US Food and Drug
Administration (FDA) and 12 leading pharma companies, all managed
by the FNIH.
The second Award to OBD is worth $963,000 over one year (of
which GBP388,000 is recognised as other operating income in the
year ended 30 September 2023) and is funding the reduction to
practice of an EpiSwitch prognostic blood test for cancer patients
most likely to present IO-triggered Hyper-Progressive Disease (HPD)
if given an ICI. HPD is a critical condition observed in a subset
of cancer patients (it has an average prevalence of 12%), who react
adversely to treatment with immune checkpoint inhibitors (ICIs). In
HPD patients, ICI treatment triggers a life-shortening opposite
effect - accelerated tumour growth, with reduced survival. With
increasing adoption of ICI treatments for cancer patients, the lack
of prognostic biomarkers has become an urgent issue for practising
clinicians, drug developers, payors and regulators. The work
enabled by the PACT Award will help to complete the development of
the Hyper-ICI Response Test (HiRT), a blood test to identify
patients at risk of HPD prior to ICI therapy.
Fundraising to support short-term activity
We remain in the early stages of our commercialization of OBD's
technology (we initiated our expanded strategy to include
development of proprietary products only three years ago, in
December 2020). During the year we completed two successful
fundraises, in October 2022 and August 2023, to support the
Company's immediate term plans, raising a total of GBP15.4 million
before costs.
Existing and new investors took part in both fundraises. In the
most recent raise in August 2023, we were pleased to welcome a
number of new institutional investors to our register and to
receive over GBP0.5 million from 194 individual investors who took
part in the fundraise through a PrimaryBid offer. I would again
like to thank all investors in the Company for the support they
have shown throughout the year.
Conclusion and focus for 2024
At the start of the financial year, our primary focus was
expected to be on growing orders of EpiSwitch CiRT. The OBD team
achieved this objective, in addition to meeting the extra challenge
of expediting the development and launch of EpiSwitch PSE. I am
pleased that we set ourselves this stretch goal - as well as the
obvious promising commercial prospects for the Group, there are
clear benefits for patients and their families to having PSE
launched and available as soon as possible.
We remain committed to working with commercial and other
partners to provide unique and critical insight with our 3D
genomics technology. At the same time, we see the unpredictable
pace at which such projects are often agreed as validation of OBD's
determination to develop our own products, directly building the
market for 3D genomics ourselves.
Looking forward to 2024, my team and I will be focused on four
main areas:
-- Following the initial introduction of EpiSwitch PSE into the
market last year, with the leadership of Dr Arrivo, we aim to drive
significant awareness and adoption by targeting large organization
accounts and partnering to generate nationwide access and
distribution of the test. This will involve extensive business
development and sales and marketing activity, within our available
resources. We will also bring PSE online in our UK clinical
laboratory by the end of March 2024.
-- We will continue to drive adoption and increase orders of
EpiSwitch CiRT. Dr Mathis's approach will allow us to capitalize on
the foundation of the 750 tests used to date. We will focus our
efforts by identifying insights from the data such as usage niches,
algorithm alignment and key accounts. Distilling the HEOR story and
petitioning for adoption into NCCN Guidelines and compendia will
put the test in the hands of a greater number of oncologists, not
just the early adopters. We expect this will also help us to enter
into further direct agreements with insurers and healthcare
delivery networks (IDNs, GPOs and hospitals).
-- We will continue the recently initiated confidential
discussions with third parties regarding our two most advanced
pipeline assets, EpiSwitch NST for colorectal/bowel cancer and
EpiSwitch SCB for canine cancer and will assess and explore
opportunities for monetizing these and other programs from our
extensive portfolio of deployable 3D genomic tests.
-- Finally, we will continue to work on internal and grant- and
award-funded research and development and on projects for
commercial partners.
We are already accelerating on all of these fronts and look
forward to reporting back to shareholders later in the year.
Dr Jon Burrows
Chief Executive Officer
Oxford BioDynamics Plc
16 January 2024
CAP-CLIA regulated laboratories are accredited by the College of
American Pathologists as being compliant with the Clinical
Laboratory Improvement Amendments, 1988 (42 CFR, Part 493).
++ A Current Procedural Terminology - Proprietary Laboratory
Analysis (CPT-PLA) code is used in the US to report medical and
diagnostic services to entities such as health care professionals
and payors.
Sources :
(1) Pchejetski, D., et al. (2023). Circulating Chromosome
Conformation Signatures Significantly Enhance PSA Positive
Predicting Value and Overall Accuracy for Prostate Cancer
Detection. Cancers, 15(3), 821.
http://dx.doi.org/10.3390/cancers15030821
(2)
https://www.prostatehealthpodcast.com/96-advancing-precision-medicine-episwitch-pse-prostate-cancer-screening-test-with-94-accuracy-robert-heaton-md/
Business performance and position: financial review
Overview
The year ended 30 September 2023 saw increased revenue
(GBP0.51m, FY22: GBP0.15m) and other operating income (GBP0.83m,
FY22: GBP0.35m) compared to the prior year. Revenue included
amounts in respect of sales of proprietary tests and work for
commercial customers. Product revenue predominantly arose from
reimbursement by US payors for CiRT tests, which is recognized only
on receipt of funds and therefore lags performance of the relevant
test.
As noted in the CEO's review, during the year the Company raised
a total of GBP15.4m before costs (FY22: GBP3.6m before costs) in
two equity fundraisings to support the immediate term plans of the
Group.
The tables below present summary explanations of what comprises
the main elements of the Group's financial performance for the year
and its position at the year end, together with the main drivers of
movements compared to the prior year.
Further detail is provided in the financial information and
notes on the following pages, which are extracted from the
Company's annual report and accounts for the year ended 30
September 2023.
Note 2 includes a description of the Board's assessment and
conclusion that it is appropriate to adopt the going concern
assumption in preparing the accounts, but that, as at the previous
three year ends, a number of factors exist that, taken together,
present a material uncertainty which may cast significant doubt on
the Company's ability to continue as a going concern and,
therefore, it may be unable to realize its assets and discharge its
liabilities in the normal course of business.
Financial performance
Element Comprising: 2023 2022 Year-on-year Main drivers of movement
change
GBPm
GBPm GBPm
------------------------- -------- ------- -------------- ---------------------------------
Increase driven by
recognition of first
significant revenue
Revenue from from proprietary tests
test sales and and an increase in
contracts with work performed for
Revenue pharma customers 0.51 0.15 0.36 increase Pharma customers.
------------------------- -------- ------- -------------- ---------------------------------
Amounts payable These costs relate
to the Group's to EpiSwitch(R) CiRT
partner lab and tests ordered and
other costs relating processed during the
to proprietary year, with some additional
tests processed amounts recognized
Cost of and/or reimbursed on receipt of reimbursement
sales in the period. (0.24) (0.04) 0.20 increase for tests from payors.
------------------------- -------- ------- -------------- ---------------------------------
R&D activity was increased
relative to the prior
year. The final stages
of the development
R&D expenditure Lab consumables, of the EpiSwitch(R)
(excluding equipment maintenance PSE test were expedited
staff costs) and similar costs (0.76) (0.53) 0.23 increase during the year.
------------------------- -------- ------- -------------- ---------------------------------
Full year impact of
FY22 recruits, FY23
in-year recruitment.
Average FTEs were
similar to FY22, but
a higher proportion
of the Group's employees
were in senior and/or
US-based roles. Inflationary
pay increases awarded
Staff and directors' in calendar 2023 were
remuneration higher than in recent
Staff costs and benefits (5.40) (4.48) 0.92 increase years.
------------------------- -------- ------- -------------- ---------------------------------
Increases of c.GBP0.3m
in marketing-related
costs to promote EpiSwitch(R)
CiRT and EpiSwitch(R)
PSE tests, c.GBP0.3m
in premises-related
costs, reflecting
increased charges
for utilities and
site service charges
for the UK HQ, and
costs for the new
laboratory in the
US, c.GBP0.2m in travel-related
expenses for sales
Other costs including teams and conference
General marketing, legal attendance, and c.GBP0.1m
and other and other professional in website development
admin costs services (3.41) (2.45) 0.96 increase and other IT services.
------------------------- -------- ------- -------------- ---------------------------------
Options charges are
spread over vesting
periods of up to three
years. More options
were granted than
in the prior year
Non-cash charge and this was offset
spreading fair by reductions in the
value of share amounts recognized
options over in the year in respect
Share option their vesting of options granted
charges period (0.33) (0.39) 0.06 decrease prior to October 2021.
------------------------- -------- ------- -------------- ---------------------------------
Increase results from
higher depreciation
of right-of-use assets
(because of the lease
Depreciation of the Group's US
and amortization clinical laboratory
of intangible in the year) and amortization
assets, property of patents and a smaller
plant and equipment increase in property,
Depreciation and right-of-use plant and equipment
and amortization assets. (1.36) (1.21) 0.15 increase depreciation.
------------------------- -------- ------- -------------- ---------------------------------
Income arises from
the Company's two
PACT Awards and its
Income associated membership of the
Other operating with grants and HIPPOCRATES consortium,
income awards 0.83 0.35 0.48 increase funded by an EU grant.
------------------------- -------- ------- -------------- ---------------------------------
Operating
loss (10.17) (8.60) 1.57 increase As noted above.
-------- ------- -------------- ---------------------------------
The fair value of
Fair value Non-cash movement the warrant liability
(loss) / in fair value increased over the
gain on of liability period, generating
financial recognised in this loss, mainly
liabilities connection with because of the increase
designated warrants issued in the Company's share
as FVTPL in November 2021 (1.25) 1.10 2.35 decrease price.
------------------------- -------- ------- -------------- ---------------------------------
Gain reclassified Non-cash gain 0.11 - 0.11 increase This is a crystallised
to profit arising on the foreign currency translation
or loss deregistration gain, offset by a
on disposal of the Group's reduction in the value
of foreign former Australian of the translation
operation subsidiary entity. reserve, recognised
in other comprehensive
income.
------------------------- -------- ------- -------------- ---------------------------------
Decrease relates to
losses from exchange
rate movements, partly
offset by an increase
in interest receivable
Interest income on invested cash and
Finance and foreign exchange term deposit balances
income gains 0.10 0.13 0.03 decrease during the year.
------------------------- -------- ------- -------------- ---------------------------------
Increase driven by
lease interest costs
on new US laboratory,
partly offset by reducing
Calculated lease interest charges in
Finance interest, foreign respect of the UK
costs exchange losses (0.21) (0.20) 0.01 increase HQ lease.
------------------------- -------- ------- -------------- ---------------------------------
Decrease driven by
higher current tax
charges in subsidiary
entities, and lower
amounts claimable
UK R&D tax credits in respect of R&D
offset by current tax credits following
and deferred legislative changes
Tax taxes in subsidiaries 0.59 0.86 0.27 decrease in the UK.
------------------------- -------- ------- -------------- ---------------------------------
Loss for the Results from the increased
year divided loss and the higher
by weighted average average number of
Loss per number of shares shares in issue for
share in issue (7.3)p (6.7)p 0.6p increase FY23 compared to FY22.
------------------------- -------- ------- -------------- ---------------------------------
Cash flow
Element Comprising: 2023 2022 Year-on-year Main drivers of movement
change
GBPm GBPm GBPm
---------------------- ------- ------- --------------- -----------------------------
Approximately GBP4m
increased loss before
tax and c.GBP0.1m
decrease in tax credits
received. Adjustments
for movements in working
Operating loss, capital, foreign exchange
Net cash adjusted for and non-cash items
used in non-cash items were c.GBP1m higher
operating and movements than in the prior
activities in working capital. (8.29) (5.18) 3.11 increase year.
---------------------- ------- ------- --------------- -----------------------------
Receipts on term deposit
maturities were c.GBP2.1m
lower, offset by c.GBP0.2m
decrease in net expenditure
Net cash Expenditure on on property, plant
(used in) fixed assets, and equipment and
/ generated offset by interest intangible assets
by investing income and maturing and c.GBP0.06m higher
activities term deposits. (0.62) 1.25 1.87 decrease interest receipts.
---------------------- ------- ------- --------------- -----------------------------
Driven by a c.GBP10.58m
increase in net receipts
from equity issues,
a GBP0.04m increase
in rent payments and
Net cash Proceeds from the one-off GBP0.11m
generated equity issues buy-back of minority
by financing offset by lease interest in a subsidiary
activities payments. 13.21 2.56 10.65 increase in FY22.
---------------------- ------- ------- --------------- -----------------------------
Financial position
Element Comprising: 2023 2022 Year-on-year Main drivers of movement
change
GBPm
GBPm GBPm
--------------------- -------- ------- -------------- ----------------------------
Cash and term deposits
increased as a result
of the two fundraisings
during the year, which
provided net funds of
GBP14.14m, offset by
the operating cash outflow
of c.GBP8.29m, capital
expenditure and rent
of c.GBP1.49m and minor
Cash and Cash and term movements in cash held
term deposits deposits 5.25 1.00 4.25 increase in foreign currencies.
--------------------- -------- ------- -------------- ----------------------------
"Right-of-use"
assets associated
with the Group's
leased properties,
tangible and
intangible fixed
assets, deferred
tax assets,
inventories,
debtors and The main component of
prepayments the increase is the
and cash and movement in cash and
Total assets term deposits. 16.13 11.34 4.79 increase term deposits.
--------------------- -------- ------- -------------- ----------------------------
The GBP1.25m increase
Trade creditors, in the estimate of the
accruals, contract fair value of the warrant
liabilities, liability (which is
lease liabilities, mainly driven by the
provisions, increase in the Company's
deferred tax share price over the
liabilities, period) is the main
and warrant driver of the overall
Total liabilities liability. (10.07) (8.76) 1.31 increase increase in liabilities.
--------------------- -------- ------- -------------- ----------------------------
Paul Stockdale
Chief Financial Officer
CONSOLIDATED INCOME STATEMENT
YEARED 30 SEPTEMBER 2023
2023 2022
GBP000 GBP000
Continuing operations Note
Revenue 3 510 154
Cost of sales (244) (38)
--------- --------
Gross profit 266 116
Research & development costs (excluding
staff costs) (758) (526)
Staff costs (5,403) (4,483)
General & other admin costs (3,411) (2,452)
Share option charges (332) (394)
Depreciation and amortization (1,357) (1,213)
Other operating income 4 827 351
--------- --------
Operating loss (10,168) (8,601)
Fair value (loss) / gain on financial liabilities
designated as FVTPL 12 (1,246) 1,095
Gain reclassified to profit or loss on 113 -
disposal of foreign operation
Finance income 103 134
Finance costs (213) (195)
--------- --------
Loss before tax (11,411) (7,567)
Income tax 585 857
--------- --------
Loss for the year from continuing operations 6 (10,826) (6,710)
========= ========
Loss attributable to:
Owners of the Company (10,826) (6,710)
Non-controlling interest - -
--------- --------
(10,826) (6,710)
========= ========
Earnings / (loss) per share
From continuing operations
Basic and diluted (pence per share) 7 (7.3) (6.7)
========= ========
CONSOLIDATED STATEMENT OF COMPREHENSIVE INCOME
YEARED 30 SEPTEMBER 2023
2023 2022
GBP000 GBP000
Note
Loss for the year 6 (10,826) (6,710)
Exchange differences on translation of foreign
operations that may be reclassified to the
income statement (182) (40)
--------- --------
Total comprehensive income for the year (11,008) (6,750)
========= ========
Total comprehensive income attributable
to:
Owners of the Company (11,008) (6,750)
Non-controlling interest - -
--------- --------
(11,008) (6,750)
========= ========
CONSOLIDATED STATEMENT OF FINANCIAL POSITION
AS AT 30 SEPTEMBER 2023
2023 2022
GBP000 GBP000
Assets Note
Non-current assets
Intangible fixed assets 8 1,913 1,601
Property, plant and equipment 9 2,238 2,582
Right-of-use assets 10 4,759 4,396
Deferred tax asset 50 -
Total non-current assets 8,960 8,579
--------- ---------
Current assets
Inventories 274 337
Trade and other receivables 1,643 1,429
Fixed-term deposits - 25
Cash and cash equivalents 5,250 974
--------- ---------
Total current assets 7,167 2,765
--------- ---------
Total assets 16,127 11,344
========= =========
Equity and liabilities
Capital and reserves
Share capital 11 2,023 1,004
Share premium 32,144 19,020
Translation reserves (63) 119
Share option reserve 2,776 3,154
Retained earnings (30,825) (20,709)
--------- ---------
Equity attributable to owners
of the Company 6,055 2,588
Non-controlling interest - -
--------- ---------
Total equity 6,055 2,588
========= =========
Current liabilities
Trade and other payables 1,707 2,000
Warrant liability 12 1,360 114
Lease liabilities 13 818 736
Provisions - -
Current tax liabilities 116 61
--------- ---------
Total current liabilities 4,001 2,911
--------- ---------
Non-current liabilities
Lease liabilities 13 5,621 5,400
Provisions 440 424
Deferred tax 10 21
--------- ---------
Total non-current liabilities 6,071 5,845
--------- ---------
Total liabilities 10,072 8,756
--------- ---------
Total equity and liabilities 16,127 11,344
========= =========
CONSOLIDATED STATEMENT OF CHANGES IN EQUITY
YEARED 30
SEPTEMBER 2023
Attribu-
table
Transla- Share to Non-con-
Share Share tion option Retained share- trolling
capital premium reserve reserve earnings holders interest Total
GBP000 GBP000 GBP000 GBP000 GBP000 GBP000 GBP000 GBP000
At 1 October
2022 1,004 19,020 119 3,154 (20,709) 2,588 - 2,588
--------- --------- --------- --------- --------- --------- --------- ---------
Loss for the
year - - - - (10,826) (10,826) - (10,826)
Other
comprehensive
income for
the period - - (182) - - (182) - (182)
--------- --------- --------- --------- --------- --------- --------- ---------
Total
comprehensive
income for
the period - - (182) - (10,826) (11,008) - (11,008)
--------- --------- --------- --------- --------- --------- --------- ---------
Subscription
for new
shares 1,019 14,368 - - - 15,387 - 15,387
Transaction
costs for new
shares - (1,244) - - - (1,244) - (1,244)
Share option
credit - - - 332 - 332 - 332
Lapse of
vested
share options - - - (710) 710 - - -
--------- --------- --------- --------- --------- --------- --------- ---------
At 30
September
2023 2,023 32,144 (63) 2,776 (30,825) 6,055 - 6,055
========= ========= ========= ========= ========= ========= ========= =========
YEARED 30
SEPTEMBER 2022
Attribu-
table
Transla- Share to Non-con-
Share Share tion option Retained share- trolling
capital premium reserve reserve earnings holders interest Total
GBP000 GBP000 GBP000 GBP000 GBP000 GBP000 GBP000 GBP000
At 1 October
2021 926 16,740 159 3,022 (14,171) 6,676 17 6,693
--------- --------- --------- --------- --------- --------- --------- ---------
Loss for the
year - - - - (6,710) (6,710) - (6,710)
Other
comprehensive
income for
the period - - (40) - - (40) - (40)
--------- --------- --------- --------- --------- --------- --------- ---------
Total
comprehensive
income for
the period - - (40) - (6,710) (6,750) - (6,750)
--------- --------- --------- --------- --------- --------- --------- ---------
Subscription
for new
shares 78 3,545 - - - 3,623 - 3,623
Issue of
warrants
to subscribe
for new
shares - (1,209) - - - (1,209) - (1,209)
Transaction
costs for new
shares - (56) - - - (56) - (56)
Share option
credit - - - 394 - 394 - 394
Lapse of
vested
share options - - - (262) 262 - - -
Buy-back and
cancellation
of minority
interest
shares - - - - (90) (90) (17) (107)
--------- --------- --------- --------- --------- --------- --------- ---------
At 30
September
2022 1,004 19,020 119 3,154 (20,709) 2,588 - 2,588
========= ========= ========= ========= ========= ========= ========= =========
CONSOLIDATED STATEMENT OF CASH FLOWS
YEARED 30 SEPTEMBER 2023
2023 2022
GBP000 GBP000
Note
Loss before tax for the financial year 6 (11,411) (7,567)
Adjustments to reconcile loss for the
year to net operating cash flows:
Net interest 141 184
Loss on disposal of property, plant and
equipment 4 1
Depreciation of property, plant and equipment 9 548 539
Depreciation of right-of-use assets 10 663 574
Amortization of intangible assets 8 146 100
Net foreign exchange movements (122) (278)
Movement in provisions 16 16
Share based payments charge 332 394
Fair value loss / (gain) on financial liabilities 1,246 (1,095)
Working capital adjustments:
(Increase) / decrease in trade and other
receivables (448) 469
Decrease in inventories 63 55
(Decrease) / increase in trade and other
payables (286) 475
--------- --------
Operating cash flows before interest and
tax paid (9,108) (6,133)
R&D tax credits received 896 969
Tax paid (82) (13)
--------- --------
Net cash used in operating activities (8,294) (5,177)
--------- --------
Investing activities
Interest received 71 14
Lease incentive received - -
Purchases of property, plant and equipment (250) (363)
Purchases of intangible assets (466) (538)
Decrease in term deposits 25 2,138
--------- --------
Net cash (used in) / generated by investing
activities (620) 1,251
--------- --------
Financing activities
Interest paid (213) (195)
Repayment of lease liabilities (723) (703)
Acquisition of minority interest shares
in subsidiary entity - (107)
Issue of equity shares and warrants 15,387 3,623
Transaction costs relating to issue of
equity shares (1,244) (56)
Net cash generated by financing activities 13,207 2,562
--------- --------
Net increase / (decrease) in cash and
cash equivalents 4,293 (1,364)
Foreign exchange movement on cash and cash
equivalents (17) 163
Cash and cash equivalents at beginning
of year 974 2,175
--------- --------
Cash and cash equivalents at end of year 5,250 974
========= ========
1. Corporate information
Oxford Biodynamics plc is a public limited company incorporated
in the United Kingdom, whose shares were admitted to trading on the
AIM market of the London Stock Exchange on 6 December 2016. The
Company is domiciled in the United Kingdom and its registered
office is 3140 Rowan Place, John Smith Drive, Oxford Business Park
South, Oxford, OX4 2WB. The registered company number is 06227084
(England & Wales).
The Group is primarily engaged in the commercialization of
proprietary molecular diagnostics products and biomarker research
and development.
2. Basis of the announcement
Basis of preparation
The final results for the year ended 30 September 2023 were
approved by the Board of Directors on 16 January 2024. The final
results do not constitute full accounts within the meaning of
section 434 of the Companies Act 2006 but are derived from audited
accounts for the year ended 30 September 2023 and the year ended 30
September 2022.
This announcement is prepared on the same basis as set out in
the audited statutory accounts for the year ended 30 September
2023. The accounts for the years ended 30 September 2023 and 30
September 2022, upon which the auditors issued unqualified
opinions, also had no statement under section 498(2) or (3) of the
Companies Act 2006. The auditors' report includes reference to the
material uncertainty relating to going concern. See below for more
details of the going concern assessment performed by the Board of
Directors.
While the financial information included in this results
announcement has been prepared in accordance with the recognition
and measurement criteria of International Financial Reporting
Standards in conformity with the Companies Act 2006 (IFRS), this
announcement does not in itself contain sufficient information to
comply with IFRS.
Reporting currency
The consolidated financial statements are presented in pounds
sterling (GBP), which is also the Company's functional
currency.
Going concern
In assessing the appropriateness of adopting the going concern
assumption, the Group and Parent Company has prepared a detailed
budget ("the budget") for the two-year period ending 30 September
2025. The budget includes:
-- estimates of likely revenue arising from EpiSwitch(R) CiRT
and EpiSwitch(R) PSE (based on the Group's own assessments of
market opportunities);
-- anticipated revenues from contracts with pharmaceutical partners;
-- expected income from existing grants and awards;
-- operating costs reflecting the current cost base (plus
inflationary increases), with some increases in activity to support
the commercial tests already launched; and
-- capital expenditure, primarily to maintain and extend the Group's patent estate.
Combined revenue and other operating income during the year
ended 30 September 2023 was increased compared to the previous
year, but the Group remained lossmaking with income significantly
exceeded by operating costs, which included spending necessary to
expedite the development and launch of the PSE test during the
year. The Group was able to maintain its cash reserves during the
year, including through the raising of GBP9.3m (before costs)
through a placing, subscription and open offer in October 2022 and
GBP6.1m (before costs) through a placing, subscription and
PrimaryBid offer in August 2023.
The Board considers that the budget represents a reasonable best
estimate of the Group's performance over the period to 30 September
2025 and the Directors are satisfied that in the scenario modelled
in the budget, the Group and Parent Company would be able to
continue as a going concern. The Directors note, however, that the
budget includes estimates of product and contract revenue
reflecting significant increases in the volume of CiRT tests to be
ordered in FY24 compared to FY23, significant increases,
post-launch, in orders of PSE tests and expectations of a number of
new contracts with pharma customers. Forecast cash balances in the
budget, whilst positive throughout the period covered, are expected
to be reduced to a low level relative to the Group's cost base
through much of 2024.
The Directors also draw attention to several significant
uncertainties inherent in the preparation of the budget, primarily
relating to balances associated with the revenue / income cycle,
since most of the Group's costs are reasonably predictable and
controllable. These uncertainties include volumes of orders of the
Group's tests, particularly PSE which was launched just before the
end of FY23; reimbursement rates and timing of the reimbursement
cycle (and consequent impact on the Group's working capital); and
the number and value of new pharma/biotech agreements.
Cash resources as predicted in the budget are very sensitive to
changes in the assumptions related to these uncertainties: this was
noted in two alternative scenarios considered by the Directors: a
"possible" scenario that reflects significantly reduced test
volumes compared to the budget and a "downside" scenario with still
lower test volumes and no new pharma projects assumed. Without any
remedial action to reduce costs or delay expenditure, in these
scenarios the Group and Company would need to obtain additional
funds during the second quarter of 2024 in order to continue as a
going concern.
The Group successfully raised a total of GBP15.4m (before costs)
from new and existing shareholders in two fundraises during the
year ended 30 September 2023. The Company's share price and the
level of interest in the Company's shares, as measured by average
daily trading volumes, increased significantly following the launch
of the PSE test in September 2023. The Directors consider that
these developments tend to increase confidence that the Company
will be able to access further cash resources from investors in
future. However, as at the date of publication of this report, this
is not guaranteed.
The Directors do not believe that any of the factors above is
unusual or unexpected for the Group at this point in its
development. However, shareholders should be aware that there is
uncertainty around its ability to generate sufficient revenues and
the timing of receipts from customers, as well as the ability of
the Group to raise sufficient finance to meet its expected costs.
These conditions present a material uncertainty which may cast
significant doubt on the Group and Parent Company's ability to
continue as a going concern and, therefore, it may be unable to
realize its assets and discharge its liabilities in the normal
course of business.
Critical judgements in applying the Group's accounting
policies
The following are the critical judgements that the Directors
have made in the process of applying the Group's accounting
policies and that have the most significant effect on the amounts
recognized in the consolidated financial statements.
Treatment of revenue arising from test sales reimbursed by US
insurance payors
The Group recognizes revenue when or as the relevant performance
obligations in its contracts with customers are completed. Sales of
the Group's proprietary tests can be paid for by patients, payors
with whom the Group has direct agreements in place, or by US
insurers through the reimbursement process. In this final case, the
Group may also obtain an acknowledgement of financial
responsibility from a patient before processing a test.
EpiSwitch(R) CiRT tests were regularly reimbursed by several US
insurers throughout the year for a range of amounts and this has
continued post-year end. The amount received is influenced by
several factors, including the terms of individual patients'
policies such as requirements for co-payment, the price listed for
the test, if any, in the Centers for Medicare and Medicaid Services
(CMS) Clinical Laboratory Fee Schedule (CLFS), insurers' own
coverage policies in respect of the test, and claim denials. Where
reimbursement for a test is initially denied, or reimbursed at a
lower-than-expected amount, the Group avails itself of the appeals
process that exists in the reimbursement system. At the year end, a
number of appeals were in process but not yet complete.
Reimbursement claims for a further group of processed tests were
held by the Group pending confirmation of coverage decisions by
insurers or the relevant Medicare Administrative Contractor (MAC),
in order to ensure the most positive likely outcome in terms of
eventual reimbursement.
The above factors are relevant to Management's decision on
whether a contract with a customer exists and therefore whether the
five-step process of revenue recognition included in IFRS 15
Revenue from Contracts with Customers should be followed or whether
instead revenue should be recognized on final receipt of funds from
a payor.
Management exercised judgement in determining that, for most of
the Group's test orders in the period, the appropriate accounting
treatment is to recognize revenue on receipt of funds and not to
follow the five-step process.
Management anticipate that in future periods, as the Group's
historical collections experience increases in volume and
specificity in relation to particular payors and policies and the
proportion of test sales for which an acknowledgement of financial
responsibility is obtained from patients also increases, it is
likely that the five-step process will apply to an increased
proportion of test sales and that judgement will be required in
determining the extent to which variable consideration relating to
those tests is unconstrained and should therefore be
recognized.
Identification of the Group's cash-generating unit
In carrying out the impairment review of patent assets set out
in more detail below, Management exercised judgement in determining
that the Group currently has one cash-generating unit (CGU).
Guidance states that CGUs are "the smallest identifiable group of
assets that generates cash inflows that are largely independent of
the cash inflows for other assets or groups of assets".
The Group's strategy was expanded in December 2020, to include
the development and commercialization of proprietary tests. As at
30 September 2023, three lab developed test products had been
launched, with two of these (EpiSwitch(R) CiRT and EpiSwitch(R)
PSE) being actively marketed as well as the Group's EpiSwitch(R)
Explorer Array Kit, which is marketed to the life science research
community. Revenue from products and customer contracts is reported
separately to Directors in the Group's internal management
accounts. However, it is not currently possible to assign separate
groups of OBD assets to particular cashflows. With very limited
exceptions, people, premises, equipment and patents are generally
applied to both product and customer contract revenue streams. This
position may change as i) dedicated product sales and marketing
teams are more fully developed, ii) the Group's LDTs are
consistently processed through the Group's US and UK clinical
laboratories and iii) test-specific revenue streams become more
predictable.
At present, Management continues to conclude that the Group has
one CGU, relating to all commercial exploitation of its
EpiSwitch(R) technology. If this judgement were to be incorrect and
the Group determined to contain more than one separately
identifiable CGU, as part of the impairment review of the Group's
patent assets conducted at the year end, it would have been
necessary to estimate the recoverable value of each CGU separately
and to allocate patents to those CGUs.
Impairment review
Intangible assets are reviewed for indicators of impairment at
the end of each reporting period. An impairment review of patent
assets was conducted as at the year end, principally because the
Group's financial performance for the year resulted in a larger
than budgeted loss and this was considered to be an indicator of
potential impairment. In addition, an impairment review is required
for any assets not yet being amortized and certain patent assets
fall into this category.
As noted above, Management identified that at the current stage
in the Group's development, it includes a single CGU, to which all
patent assets are allocated. Management consider that the
recoverable amount of the Group's single CGU is based on its fair
value less cost of disposal (FVLCOD), and that this value is
attributable to its intellectual property, including patents and
know-how, and its other assets, including property plant and
equipment. The most reliable available estimate for the fair value
of the Group's CGU as a whole is the enterprise value of the Group,
which is in turn given by the market value of the Company on a
cash- and debt-free basis.
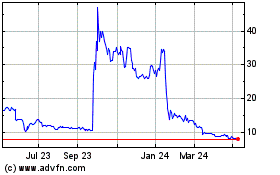
The Group had a year-end market capitalisation of GBP74.9m (37p
x 202,303,415 shares then in issue). For the 30 September 2023 year
end, Management also considered the significant increase in the
share price and market capitalization of the Company following the
announcement of the launch on 26 September 2023 of the EpiSwitch(R)
PSE test and for the purposes of the impairment review, the
increase in enterprise value that followed the launch announcement
was assumed to relate only to the PSE test. On 25 September 2023,
the latest date prior to the announcement of the launch, the Group
had a market capitalisation of GBP21.1m (10.45p x 202,303,415
shares in issue).
Cash/cash equivalents and term deposits at 30 September 2023 of
GBP5.3m are deducted from market value in arriving at the
enterprise value. Following review of available guidance,
Management determined that neither the warrant not the lease
liabilities associated with the Group's rented property should be
added back to the market value in determining the enterprise value.
This results in an estimate of the year-end enterprise value of the
Group as a whole of approximately GBP69m and an enterprise value
shortly pre-year-end of the underlying business "excluding" the PSE
test, of approximately GBP16m.
In estimating the cost of disposal (COD), Management used a
round sum estimate of GBP2.5m, representing a COD of approximately
12% of the pre-uplift market value, which is within the range of
estimates of disposal costs reviewed by Management. The FVLCOD of
the Company as at 30 September 2023 was therefore estimated to be
GBP13.5m prior to the launch of PSE and GBP66.5m at the year end,
after the launch of PSE. Management then compared the FVLCOD of the
Company to the gross value of the Group's assets excluding patents
(GBP9m as at 30 September 2023). The excess of the Company's FVLCOD
over its gross assets excluding patents, prior to the launch of
PSE, was therefore approximately GBP7m, compared to a carrying
value of patent assets (including patents linked to the PSE test)
of GBP1.82m. Management further reviewed each of the Company's
patent families for other indicators of impairment, principally
obsolescence, and determined that no such indicators existed at the
year end. Management therefore concluded that no impairment of the
Company's capitalized patents existed at the year end.
Management considers that a reduction in the Company's estimated
FVLCOD to an amount comparable to the carrying value of its
non-patent assets would lead to a reduction in the recoverable
amount of its patent assets, potentially to nil. Management will
continue to assess, at the end of each reporting period and more
frequently if necessary, whether there are indicators that any of
the Group's assets may be impaired.
3. Revenue
All revenue is derived from the Group's principal activities,
namely sales of proprietary products and biomarker research and
development. Analysis of the Group's revenue by principal
activities, geography and pattern of revenue recognition is as
follows:
2023 2022
GBP000 GBP000
Continuing operations:
Sales of proprietary products
USA 160 -
Rest of World 34 -
------- -------
194 -
Biomarker research and development
USA 228 107
Rest of World 88 47
------- -------
316 154
------- -------
Consolidated revenue 510 154
======= =======
2023 2022
GBP000 GBP000
Continuing operations:
Revenue recognized at a point in time 194 -
Revenue recognized over time 316 154
------- -------
510 154
======= =======
2023 2022
GBP000 GBP000
Revenue from individual customers each representing
more than 10%
of revenue for the period: 280 152
======= =======
Number Number
Number of individual customers each representing
more than 10%
of revenue for the period: 2 2
======= =======
4. Other operating income
2023 2022
GBP000 GBP000
Continuing operations:
Other operating income (awards and grants)
- recognized at a point in time - -
- recognized over time 827 351
------- -------
827 351
======= =======
During the year, the Company was granted a second FNIH
Partnership for Accelerating Cancer Therapies (PACT) Award. Income
was recognized in respect of each of the Company's PACT awards and
OBD's involvement in the EU-funded HIPPOCRATES consortium (income
in the prior year related only to the Company's first PACT
award).
5. Business segments
Products and services from which reportable segments derive
their revenues
Information reported to the Group's Chief Executive Officer (who
has been determined to be the Group's Chief Operating Decision
Maker) for the purposes of resource allocation and assessment of
segment performance is focused on costs incurred to support the
Group's main activities. The Group is currently determined to have
one reportable segment under IFRS 8, that of sales of proprietary
products and biomarker research and development. This assessment
will be kept under review as the Group's activity expands.
The Group's operating expenses and non-current assets, analysed
by geographical location were as follows:
2023 2022
GBP000 GBP000
Staff costs
UK 2,614 2,572
USA 2,692 1,815
Rest of World 97 96
------- -------
Total staff costs 5,403 4,483
======= =======
Research & development costs
UK 680 523
USA 77 -
Rest of World 1 4
------- -------
Total research & development costs 758 527
======= =======
General & other admin costs
UK 2,399 1,898
USA 969 479
Rest of World 43 75
------- -------
Total general & other admin costs 3,411 2,452
======= =======
Non-current assets
UK 7,446 7,954
USA 1,478 564
Malaysia 36 61
------- -------
Total non-current assets 8,960 8,579
======= =======
6. Loss for the year
Loss for the year has been arrived at after
charging/(crediting):
2023 2022
GBP000 GBP000
Net foreign exchange losses (31) (123)
Research and development costs (excluding
staff costs) 758 526
Amortization of intangible assets 146 100
Depreciation of property, plant and equipment 548 539
Depreciation of right-of-use assets 663 574
Staff costs 5,403 4,483
Share-based payments charged to profit
and loss 332 394
Fair value loss / (gain) on financial liabilities
designated as FVTPL 1,246 (1,095)
Gain reclassified to profit or loss on (113) -
disposal of foreign operation
======= ========
7. Earnings per share
From continuing operations
The calculation of the basic and diluted earnings per share is
based on the following data:
2023 2022
GBP000 GBP000
Earnings for the purposes of basic earnings per
share being net loss attributable to owners of
the Company (10,826) (6,710)
----------- -----------
Earnings for the purposes of diluted earnings
per share (10,826) (6,710)
=========== ===========
2023 2022
No No
Number of shares
Weighted average number of ordinary shares for
the purposes of
basic and diluted earnings per share* 147,481,566 99,702,257
============ ===========
Pence Pence
Earnings per share
Basic and diluted earnings per share (7.3) (6.7)
============ ===========
*Ordinary shares that may be issued on the exercise of options
or warrants are not treated as dilutive as the entity is
loss-making.
8. Intangible fixed assets
Website Software
development development
Group costs costs Patents Total
GBP000 GBP000 GBP000 GBP000
Cost
At 1 October 2022 62 144 1,674 1,880
Additions - 39 427 466
Exchange differences - (10) - (10)
------------- -------- -------
At 30 September
2023 62 173 2,101 2,336
------------- ------------- -------- -------
Accumulated amortization
At 1 October 2022 62 65 152 279
Charge for the year - 36 110 146
Exchange differences - (2) - (2)
-------- -------
At 30 September
2023 62 99 262 423
------------- ------------- -------- -------
Carrying amount
At 30 September
2023 - 74 1,839 1,913
============= ============= ======== =======
Website Software
development development
Group costs costs Patents Total
GBP000 GBP000 GBP000 GBP000
Cost
At 1 October 2021 62 57 1,208 1,327
Additions - 72 466 538
Exchange differences - 15 - 15
------------- -------- -------
At 30 September
2022 62 144 1,674 1,880
------------- ------------- -------- -------
Accumulated amortization
At 1 October 2021 54 36 85 175
Charge for the year 8 25 67 100
Exchange differences - 4 - 4
-------- -------
At 30 September
2022 62 65 152 279
------------- ------------- -------- -------
Carrying amount
At 30 September
2022 - 79 1,522 1,601
============= ============= ======== =======
As at 30 September 2023, in the Group and Company, a total of
GBP304,000 (2021: GBP263,000) of patent assets were not yet being
amortized because their useful life was determined not to have
begun.
The Group and Company hold no intangible assets that are
determined to have indefinite useful life.
9. Property, plant and equipment
Leasehold Office Fixtures Laboratory
Group improvements equipment and fittings equipment Total
GBP000 GBP000 GBP000 GBP000 GBP000
Cost
At 1 October 2022 2,041 182 172 2,318 4,713
Additions 45 58 15 125 243
Disposals - (47) - (88) (135)
Exchange differences (2) (2) (2) (55) (61)
-------------- ----------- -------------- ----------- -------
At 30 September
2023 2,084 191 185 2,300 4,760
-------------- ----------- -------------- ----------- -------
Accumulated depreciation
At 1 October 2022 231 139 44 1,717 2,131
Charge for the year 208 37 34 269 548
Eliminated on disposals - (47) - (84) (131)
Exchange differences (2) (2) (1) (21) (26)
-------------- ----------- -------------- ----------- -------
At 30 September
2023 437 127 77 1,881 2,522
-------------- ----------- -------------- ----------- -------
Carrying amount
At 30 September
2023 1,647 64 108 419 2,238
============== =========== ============== =========== =======
Leasehold Office Fixtures Laboratory
Group improvements equipment and fittings equipment Total
GBP000 GBP000 GBP000 GBP000 GBP000
Cost
At 1 October 2021 2,001 160 106 2,140 4,407
Additions 38 24 65 102 229
Disposals - (7) - (9) (16)
Exchange differences 2 5 1 85 93
-------------- ----------- -------------- ----------- -------
At 30 September
2022 2,041 182 172 2,318 4,713
-------------- ----------- -------------- ----------- -------
Accumulated depreciation
At 1 October 2021 26 102 12 1,439 1,579
Charge for the year 204 42 31 262 539
Eliminated on disposals - (7) - (8) (15)
Exchange differences 1 2 1 24 28
-------------- ----------- -------------- ----------- -------
At 30 September
2022 231 139 44 1,717 2,131
-------------- ----------- -------------- ----------- -------
Carrying amount
At 30 September
2022 1,810 43 128 601 2,582
============== =========== ============== =========== =======
10. Right-of-use assets
Group Buildings Other Total
GBP000 GBP000 GBP000
Cost
At 1 October 2022 5,224 18 5,242
Additions 1,029 - 1,029
Exchange differences (12) - (12)
---------- ------- -------
At 30 September
2023 6,241 18 6,259
---------- ------- -------
Accumulated depreciation
At 1 October 2022 835 11 846
Charge for the year 657 6 663
Exchange Differences (9) - (9)
---------- ------- -------
At 30 September
2023 1,483 17 1,500
---------- ------- -------
Carrying amount
At 30 September
2023 4,758 1 4,759
========== ======= =======
Group Buildings Other Total
GBP000 GBP000 GBP000
Cost
At 1 October 2021 4,968 18 4,986
Additions 226 - 226
Derecognition (9) - (9)
Exchange differences 39 - 39
---------- ------- -------
At 30 September
2022 5,224 18 5,242
---------- ------- -------
Accumulated depreciation
At 1 October 2021 263 5 268
Charge for the year 568 6 574
Eliminated on derecognition (9) - (9)
Exchange Differences 13 - 13
---------- ------- -------
At 30 September
2022 835 11 846
---------- ------- -------
Carrying amount
At 30 September
2022 4,389 7 4,396
========== ======= =======
11. Share capital of the company
2023 2023 2022 2022
Number GBP Number GBP
Authorized shares
Ordinary shares of GBP0.01 each
- allotted and fully paid 202,303,415 2,023,034 100,351,574 1,003,516
Total 202,303,415 2,023,034 100,351,574 1,003,516
============ ========== ============ ==========
The Company has one class of ordinary shares which carry no
right to fixed income.
On 28 October 2022 and 31 October 2022, the Company issued a
total of 46,360,806 new ordinary shares .
On 21 August 2023 and 22 August 2023, the Company issued a total
of 55,591,035 new ordinary shares.
No shares were issued on the exercise of share options or
warrants during the year (2022: nil).
12. Warrants
As at 30 September 2023 there were 7,791,803 shares reserved for
issue under warrants (30 September 2022: 7,791,803).
The Warrants were issued during the prior year, on 11 November
2021. The Warrants have an exercise price of 58.125p and may be
exercised for a period beginning one year and ending five years
after the issue date.
In certain circumstances, the Warrants may be exercised by way
of a 'cashless exercise' whereby holders are entitled to receive a
number of warrant shares equal to [(A-B) x 7,791,803]/(A), where A
is the value of the Company's ordinary shares at the time, and B is
the warrant exercise price of 58.125p. Anti-dilution provisions are
also in place such that if there is an adjustment for any dividends
paid or changes to ordinary share capital at any time whilst the
warrant is outstanding, the number of shares issued on exercise of
the warrant is adjusted to take into account the proportionate
change (with a limitation on fractional shares).
On award and at each subsequent reporting date, the fair value
of the Warrants has been estimated using the Black-Scholes option
pricing model. Volatility has been estimated by reference to
historical share price data over a period commensurate with the
expected term of the options awarded (effectively the remaining
term at each reporting date).
The fair value of the Warrants and the assumptions used in
estimating it are shown below:
30 September 30 September
2023 2022
Share price at date of award
/ value date (p) 37 11.5
Exercise price (p) 58.125 58.125
Expected volatility 84.39% 59.86%
Dividend yield 0% 0%
Expected life of option 3.11 years 4.11 years
Risk free interest rate 4.55% 4.40%
Fair value per Warrant 17p 1p
Warrant liability GBP1,360,000 GBP114,000
Warrant liability - Group and Company Total
GBP000
At 1 October 2022 114
Issue of warrants -
Fair value loss on financial liability designated
as FVTPL 1,246
------------------------
At 30 September 2023 1,360
========================
At 1 October 2021 -
Issue of warrants 1,209
Fair value gain on financial liability designated
as FVTPL (1,095)
------------------------
At 30 September 2022 114
========================
13. Lease liabilities
Group 2023 2022
Maturity analysis: GBP000 GBP000
Year 1 1,045 910
Year 2 1,052 908
Year 3 1,051 820
Year 4 1,058 813
Year 5+ 3,101 3,470
7,307 6,921
Less: future interest charges (868) (785)
------- -------
6,439 6,136
======= =======
Analysed as:
Current 818 736
Non-current 5,621 5,400
6,439 6,136
======= =======
14. Share-based payments
Equity-settled share option scheme
In November 2016, the Company established an Enterprise
Management Incentive ("EMI") share option scheme, under which
options have been granted to certain employees, and a non-employee
option scheme with similar terms, except that options granted under
it do not have EMI status. EMI and non-EMI share options were also
previously granted under a share option scheme established in
October 2008 ("the 2008 Scheme"). The Company does not intend to
grant any further options under the 2008 Scheme. All of the schemes
are equity-settled share-based payment arrangements, whereby the
individuals are granted share options of the Company's equity
instruments, namely ordinary shares of 1 pence each.
The schemes include non-market-based vesting conditions only,
whereby the share options may be exercised from the date of vesting
until the 10(th) anniversary of the date of the grant. In most
cases options vest under the following pattern: one-third of
options granted vest on the first anniversary of the grant date;
one-third on the second anniversary and one-third on the third
anniversary. The only exception to this pattern is 84,000 options
which were granted in the year ended 30 September 2016 which vested
immediately upon grant.
The options outstanding as at 30 September 2023 have exercise
prices in the range of GBP0.156 to GBP2.10.
2023 2022
Weighted Weighted
Number average Number average
of exercise of exercise
options price Options price
GBP GBP
Outstanding at start of period 9,447,658 0.67 8,526,484 0.76
Granted during the period 2,721,061 0.18 1,556,757 0.28
Forfeited during the period (2,185,576) (0.48) (635,583) (0.93)
Exercised during the period - - - -
------------ ---------- ---------- ----------
Outstanding at end of period 9,983,143 0.57 9,447,658 0.67
============ ========== ========== ==========
Exercisable at end of period 5,983,853 0.76 6,622,162 0.68
============ ========== ========== ==========
Weighted average remaining contractual
life (in years) of options outstanding
at the period end 6.60 5.36
========== ==========
2023 2022
GBP000 GBP000
Expense arising from share-based payment
transactions 332 394
========== ==========
The fair value of share options has been estimated using the
Black-Scholes option pricing model. Volatility has been estimated
by reference to historical share price data over a period
commensurate with the expected term of the options awarded. The
assumptions for the options granted during the current and prior
periods were as follows:
2023 2022
GBP000 GBP000
Share price at date of grant GBP0.156 to GBP0.189 GBP0.17 to GBP0.40
Exercise price GBP0.156 to GBP0.189 GBP0.17 to GBP0.40
Expected volatility 55% to 56% 52% to 54%
Dividend yield 0% 0%
Expected life of option 8.7 to 9.0 years 8.6 to 8.7 years
Risk free interest rate 3.45% to 3.70% 0.73% to 1.87%
===================== ===================
15. Events after the balance sheet date
There were no events after the balance sheet requiring
disclosure in these financial statements.
Notes for Editors
About Oxford BioDynamics Plc
Oxford BioDynamics Plc (AIM: OBD) is a global biotechnology
company, advancing personalized healthcare by developing and
commercializing precision medicine tests for life-changing
diseases.
Its flagship products are the EpiSwitch(R) CiRT (Checkpoint
Inhibitor Response Test) and EpiSwitch(R) PSE (EpiSwitch Prostate
Screening test) blood tests. CiRT is a predictive immune response
profile for immuno-oncology (IO) checkpoint inhibitor treatments,
launched in February 2022. PSE is a blood test that boosts the
predictive accuracy of a PSA test from 55% to 94% when testing the
presence or absence of prostate cancer, which has been launched in
the US and UK in September 2023.
In March 2021, the Company launched its first commercial
prognostic test, EpiSwitch(R) CST (Covid Severity Test) and the
first commercially available microarray kit for high-resolution 3D
genome profiling and biomarker discovery, EpiSwitch(R) Explorer
Array Kit , which is available for purchase by the life science
research community.
The Company's product portfolio is based on a proprietary 3D
genomic biomarker platform, EpiSwitch(R), which can build molecular
diagnostic classifiers for the prediction of response to therapy,
patient prognosis, disease diagnosis and subtyping, and residual
disease monitoring in a wide range of indications.
Oxford BioDynamics has participated in more than 40 partnerships
with big pharma and leading institutions including Pfizer, EMD
Serono, Genentech, Roche, Biogen, Mayo Clinic, Massachusetts
General Hospital and Mitsubishi Tanabe Pharma.
The Company has created a valuable technology portfolio,
including biomarker arrays, molecular diagnostic tests,
bioinformatic tools for 3D genomics and an expertly curated 3D
genome knowledgebase comprising hundreds of millions of data points
from over 15,000 samples in more than 30 human diseases.
OBD is headquartered in Oxford, UK and is listed on AIM of the
London Stock Exchange. It also has a commercial office in
Gaithersburg and a clinical laboratory in Frederick, MD, USA, and a
reference laboratory in Penang, Malaysia.
For more information, please visit the Company's website,
www.oxfordbiodynamics.com , or follow OBD on Twitter
(@OxBioDynamics) and LinkedIn .
About EpiSwitch(R)
The 3D configuration of the genome plays a crucial role in gene
regulation. By mapping this architecture and identifying abnormal
configurations, EpiSwitch(R) can be used to diagnose patients or
determine how individuals might respond to a disease or
treatment.
Built on over 10 years of research, EpiSwitch(R) is Oxford
Biodynamics' award-winning, proprietary platform that enables
screening, evaluation, validation and monitoring of 3D genomic
biomarkers. The technology is fully developed, based on testing of
over 15,000 samples in 30 disease areas, and reduced to
practice.
In addition to stratifying patients with respect to anticipated
clinical outcomes, EpiSwitch(R) data offer insights into systems
biology and the physiological manifestation of disease that are
beyond the scope of other molecular modalities. The technology has
performed well in academic medical research settings and has been
validated through its integration in biomarker discovery and
clinical development with big pharma.
This information is provided by RNS, the news service of the
London Stock Exchange. RNS is approved by the Financial Conduct
Authority to act as a Primary Information Provider in the United
Kingdom. Terms and conditions relating to the use and distribution
of this information may apply. For further information, please
contact rns@lseg.com or visit www.rns.com.
RNS may use your IP address to confirm compliance with the terms
and conditions, to analyse how you engage with the information
contained in this communication, and to share such analysis on an
anonymised basis with others as part of our commercial services.
For further information about how RNS and the London Stock Exchange
use the personal data you provide us, please see our Privacy
Policy.
END
FR DXGDBLDBDGSL
(END) Dow Jones Newswires
January 17, 2024 02:00 ET (07:00 GMT)
Oxford Biodynamics (LSE:OBD)
Historical Stock Chart
From Mar 2024 to Apr 2024
Oxford Biodynamics (LSE:OBD)
Historical Stock Chart
From Apr 2023 to Apr 2024