TIDMSTX
RNS Number : 4931Q
Shield Therapeutics PLC
26 February 2021
THIS ANNOUNCEMENT (INCLUDING THE APPICES) AND THE INFORMATION
CONTAINED HEREIN IS RESTRICTED AND IS NOT FOR PUBLICATION, RELEASE
OR DISTRIBUTION, DIRECTLY OR INDIRECTLY, IN WHOLE OR IN PART, IN OR
INTO THE UNITED STATES, AUSTRALIA, CANADA, JAPAN OR THE REPUBLIC OF
SOUTH AFRICA OR IN OR INTO ANY OTHER JURISDICTION WHERE TO DO SO
WOULD BREACH ANY APPLICABLE LAW OR REGULATION.
THIS ANNOUNCEMENT (INCLUDING THE APPICES) IS FOR INFORMATION
PURPOSES ONLY AND DOES NOT ITSELF CONSTITUTE AN OFFER FOR SALE OR
SUBSCRIPTION OF ANY SECURITIES IN THE COMPANY OR CONTAIN ANY
INVITATION, SOLICITATION, RECOMMATION, OFFER OR ADVICE TO ANY
PERSON TO SUBSCRIBE FOR, OTHERWISE ACQUIRE OR DISPOSE OF ANY
SECURITIES OF SHIELD THERAPEUTICS PLC IN ANY JURISDICTION.
THIS ANNOUNCEMENT CONTAINS INSIDE INFORMATION AS DEFINED IN
ARTICLE 7 OF REGULATION (EU) NO 596/2014 OF THE EUROPEAN PARLIAMENT
AND OF THE COUNCIL OF 16 APRIL 2014 ON MARKET ABUSE (MARKET ABUSE
REGULATION) AS RETAINED AS PART OF UK LAW BY VIRTUE OF THE EUROPEAN
UNION (WITHDRAWAL) ACT 2018 AS AMED.
UPON THE PUBLICATION OF THIS ANNOUNCEMENT, THIS INSIDE
INFORMATION IS NOW CONSIDERED TO BE IN THE PUBLIC DOMAIN.
26 February 2021
Shield Therapeutics plc
("Shield", the "Group" or the "Company")
Placing and Subscription to raise up to GBP25 million,
Open Offer to raise up to GBP4.2 million
and
Notice of General Meeting
London, UK - 26 February 2021 : Shield Therapeutics plc (LSE:
STX), a commercial stage pharmaceutical company, announces a
proposed fundraising by way of a subscription (the "Subscription")
and placing (the "Placing") to raise up to GBP25.0 million in
aggregate at a price per share of 30.0 pence (the "Issue Price").
The Company also announces it will be making an Open Offer to
Qualifying Shareholders at the Issue Price to raise up to GBP4.2
million (the "Open Offer", and together with the Subscription and
the Placing, the "Fundraising").
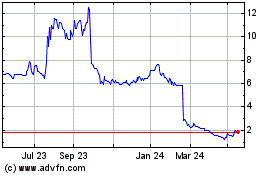
The Fundraising is being undertaken at an issue price of 30.0
pence per share which represents a discount of approximately 43.7
per cent. to the closing price on 25 February 2021 (being the
latest practicable date prior to this Announcement).
Reasons for the Fundraising
In recent months, and as outlined in the announcement of 10
December 2020, Shield has developed a comprehensive plan for the
self-commercialisation of Accrufer(R) in the US and the Placing and
Subscription will provide significant funding to execute this plan.
Sales estimates generated by management consultants and other third
parties support the potential for sales of Accrufer(R) in the US to
exceed $100 million from the third year following launch and to
reach $300 million-$400 million by years five to six.
The Group's cash flow forecasts, including the costs of the US
launch of Accrufer(R) and the paediatric study, indicate that the
Group should start to breakeven on a monthly basis within 15-18
months after launch of the drug in the US provided sales and costs
are within the range anticipated by the Directors . As previously
announced, Shield believes that around $30 million to $40 million
(c GBP21 million to GBP29 million) should provide the finance
necessary to reach the breakeven point.
The net proceeds of the Subscription and the Placing will
finance the launch costs of Accrufer(R). The bulk of the net
proceeds received will be converted into US dollars promptly in
order to avoid currency risk. US launch costs will include sales
representatives, ongoing market research and data analysis,
marketing spend and other US operational costs. The paediatric
study and other non-US expenditure is expected to be partly funded
out of gross margin generated by Shield in the US and Norgine BV
royalty revenues.
The Group has assessed a range of other options to fund the US
launch of Accrufer(R), including several debt and equity based
financing solutions. However the Board believes that the funding
requirements are best suited to an equity based financing solution
which the Fundraising will provide. Further, the Directors believe
that selling Accrufer(R) directly in the US has the potential to
generate significant returns for Shareholders.
The Subscription, the Placing and the Open Offer
The Subscription of 16,934,613 new Ordinary Shares (the
"Subscription Shares") will be subject to new shareholder approvals
to be sought at a general meeting of the Company (the "General
Meeting"). AOP and two of the Company's Directors, Tim Watts and
Dr. Christian Schweiger, have indicated that they will subscribe
for an aggregate of 16,634,613 Subscription Shares at the Issue
Price pursuant to the Subscription. Further, certain employees of
the Company who are existing shareholders have indicated that they
will subscribe for an aggregate of 300,000 Subscription Shares at
the Issue Price pursuant to the Subscription.
The Placing of up to 66,398,720 new Ordinary Shares (the
"Placing Shares") at the Issue Price will be subject to new
shareholder approvals to be sought at the General Meeting.
The gross proceeds of the Placing and the Subscription are up to
GBP25 million.
Details of the General Meeting will be set out in the Circular
to be sent to shareholders on or around 1 March 2021. The date of
the General Meeting will be confirmed at that time.
Peel Hunt LLP ("Peel Hunt") and finnCap Ltd ("finnCap") are
acting as joint bookrunners, and Peel Hunt is acting as nominated
adviser, in connection with the Placing. The Placing Shares are
being offered by way of an accelerated bookbuild, which will be
launched immediately following this Announcement (as defined
below), in accordance with the terms and conditions set out in
Appendix II to this Announcement.
The final number of Placing Shares will be agreed by Peel Hunt,
finnCap and the Company at the close of the Bookbuild and the
result will be announced as soon as practicable thereafter. It is
envisaged that the Bookbuild will be closed no later than 4.30 p.m.
on the date of this Announcement. Neither the Placing nor the
Subscription will be underwritten.
In addition, in order to provide Qualifying Shareholders with an
opportunity to participate in the equity financing at the Issue
Price, the Company will also be conducting an Open Offer, on the
basis of 2 Open Offer Shares for every 17 Ordinary Shares held on
the Record Date, to raise gross proceeds of up to GBP4.2 million
for the Company. The Open Offer will be made to Qualifying
Shareholders pursuant to the Circular. Shareholders subscribing for
their full entitlement under the Open Offer may request additional
Open Offer Shares through the Excess Application Facility. The Open
Offer is primarily aimed at those Qualifying Shareholders who are
not given the opportunity to participate in the Placing.
The Open Offer is not underwritten so if there is no take up by
Qualifying Shareholders, no additional proceeds will be received by
Shield pursuant to the Open Offer element of the Fundraising.
The Subscription, the Placing and the Open Offer are
conditional, inter alia, upon Shareholders approving the
Fundraising Resolutions at the General Meeting and upon the Placing
and Open Offer Agreement not having been terminated and becoming
unconditional. The Company intends to publish and send a circular
(the "Circular") on or around 1 March 2021 to Shareholders, which
will convene the General Meeting in order to propose the necessary
resolutions to authorise the Directors to allot the Subscription
Shares, the Placing Shares and the Open Offer Shares for cash free
of statutory pre-emption rights. It will also contain the terms and
conditions of the Open Offer. The Circular will be available on the
Company's website: https://www.shieldtherapeutics.com/ .
Neither the Subscription Shares nor the Placing Shares are being
made available to the public and are only available to Relevant
Persons. The Open Offer Shares will only be available to Qualifying
Shareholders.
Set out below in Appendix I is an extract from the draft
Circular that is proposed to be sent to Shareholders in due
course.
This Announcement should be read in its entirety. In particular,
your attention is drawn to the detailed terms and conditions of the
Placing in Appendix II to this Announcement. Further information
relating to the Fundraising and use of proceeds is set out in
Appendix I to this Announcement.
By choosing to participate in the Placing and by making an oral
and legally binding offer to acquire Placing Shares, investors will
be deemed to have read and understood this Announcement in its
entirety (including the Appendices), and to be making such offer on
the terms and subject to the conditions of the Placing contained
herein, and to be providing the representations, warranties and
acknowledgements contained in Appendix II.
Unless otherwise indicated, capitalised terms in this
Announcement have the meaning given to them in the definitions
section included in Appendix III.
The ticker for the Company's ordinary shares is STX. The
Company's LEI is 213800G74QWY15FC3W71.
Enquiries:
Shield Therapeutics plc +44 (0) 191 511 8500
Tim Watts (CEO)
Peel Hunt LLP - Nominated Adviser, Joint
Broker and Bookrunner +44 (0) 20 7148 8900
James Steel / Oliver Jackson (Investment
Banking)
Jock Maxwell Macdonald / Sohail Akbar
(ECM)
finnCap Ltd - Joint Broker and Bookrunner +44 (0) 20 7220 0563
Geoff Nash / Matthew Radley (Corporate
Finance)
Alice Lane / Charlotte Sutcliffe (ECM)
Walbrook PR - Financial PR & IR Adviser +44 (0) 20 7933 8780
Paul McManus / Lianne Cawthorne or shield@walbrookpr.com
About Shield
Shield is a commercial stage, pharmaceutical company with a
focus on addressing iron deficiency with its lead product Feraccru
(R) /Accrufer (R) (ferric maltol) (R), a novel, stable, non-salt
based oral therapy for adults with iron deficiency with or without
anaemia. Shield is quoted on AIM (STX). For more information about
Shield, please see www.shieldtherapeutics.com
IMPORTANT NOTICES AND DISCLAIMER
This announcement including its appendices (together, this
"Announcement") and the information contained in it is not for
publication, release, transmission distribution or forwarding, in
whole or in part, directly or indirectly, in or into the United
States, Australia, Canada, Japan or the Republic of South Africa or
any other jurisdiction in which publication, release or
distribution would be unlawful. This Announcement is for
information purposes only and does not constitute an offer to sell
or issue, or the solicitation of an offer to buy, acquire or
subscribe for shares in the capital of the Company in the United
States, Australia, Canada, Japan or the Republic of South Africa or
any other state or jurisdiction where to do so would be unlawful.
Any failure to comply with these restrictions may constitute a
violation of the securities laws of such jurisdictions. This
Announcement has not been approved by London Stock Exchange or by
any other securities exchange.
The new Ordinary Shares, have not been, and will not be,
registered under the U.S. Securities Act of 1933, as amended (the
"Securities Act") or with any securities regulatory authority of
any state or other jurisdiction of the United States and may not be
offered, sold, pledged, taken up, exercised, resold, renounced,
transferred or delivered, directly or indirectly, in or into the
United States absent registration under the Securities Act, except
pursuant to an exemption from the registration requirements of the
Securities Act and in compliance with any applicable securities
laws of any state or other jurisdiction of the United States. The
Placing Shares are being offered and sold by the company outside
the United States in offshore transactions as defined in, and
pursuant to, Regulation S under the Securities Act.
This announcement is being directed to persons in the United
Kingdom only in circumstances in which section 21(1) of the
Financial Services and Markets Act 2000, as amended ("FSMA") does
not apply.
This announcement is for information purposes only and is
directed only at persons who are: (1) in Member States of the
European Economic Area, qualified investors as defined in article
2(e) of the Prospectus Regulation (EU) 2017/1129 (the "EU
Prospectus Regulation"); (2) in the United Kingdom, qualified
investors as defined in article 2(e) of Prospectus Regulation (EU)
2017/1129 as it forms part of UK domestic law by virtue of the
European Union (Withdrawal) Act 2018, as amended (the "UK
Prospectus Regulation"), who (A) fall within article 19(5)
("investment professionals") of the Financial Services and Markets
Act 2000 (Financial Promotion) Order 2005, as amended (the
"Order"), or (B) fall within article 49(2)(a) to (d) ("high net
worth companies, unincorporated associations, etc.") of the Order;
or (3) are persons to whom it may otherwise be lawfully
communicated; (all such persons together being referred to as
"relevant persons"). This announcement and the terms and conditions
set out herein must not be acted on or relied on by persons who are
not relevant persons. Persons distributing this announcement must
satisfy themselves that it is lawful to do so. Any investment or
investment activity to which this announcement and the terms and
conditions set out herein relates is available only to relevant
persons and will be engaged in only with relevant persons.
The new Ordinary Shares have not been approved, disapproved or
recommended by the U.S. Securities and Exchange Commission, any
state securities commission in the United States or any other U.S.
regulatory authority, nor have any of the foregoing authorities
passed upon or endorsed the merits of the offering of new Ordinary
Shares. Subject to certain exceptions, the securities referred to
herein may not be offered or sold in the United States, Australia,
Canada, Japan or the Republic of South Africa or to, or for the
account or benefit of, any national, resident or citizen of the
United States, Australia, Canada, Japan or the Republic of South
Africa.
No public offering of securities is being made in the United
Kingdom, the United States or any other jurisdiction. Offers of the
new Ordinary Shares will either be made pursuant to an exemption
under the EU Prospectus Regulation and the UK Prospectus Regulation
(as such terms are defined below) from the requirement to produce a
prospectus or otherwise in circumstances not resulting in an offer
of transferable securities to the public under section 102B of
FSMA.
This Announcement has been issued by, and is the sole
responsibility of, the Company. No representation or warranty,
express or implied, is or will be made as to, or in relation to,
and no responsibility or liability is or will be accepted by the
directors or the Company, or by any of its or their respective
partners, employees, advisers, affiliates or agents as to or in
relation to, the accuracy or completeness of this Announcement or
any other written or oral information made available to or publicly
available to any interested party or its advisers, and any
liability therefore is expressly disclaimed.
This Announcement contains inside information for the purposes
of Article 7 of the Market Abuse Regulation (EU) 596/2014 as it
forms part of UK domestic law by virtue of the European Union
(Withdrawal) Act 2018, as amended ("UK MAR"), encompassing
information relating to the Placing described above, and is
disclosed in accordance with the Company's obligations under
Article 17 of UK MAR. In addition, market soundings (as defined in
UK MAR) were taken in respect of the Placing with the result that
certain persons became aware of inside information, as permitted by
UK MAR. This inside information is set out in this Announcement.
Therefore, upon publication of this announcement, those persons
that received such inside information in a market sounding are no
longer in possession of such inside information relating to the
Company and its securities.
Peel Hunt LLP, which is authorised and regulated in the United
Kingdom by the Financial Conduct Authority is acting as nominated
adviser and joint bookrunner to the Company and no one else in
connection with the Placing and is not acting for and will not be
responsible to any person other than the Company for providing the
protections afforded to its clients or for providing advice in
relation to the Placing. Peel Hunt's responsibilities as the
Company's nominated adviser under the AIM Rules for Nominated
Advisers are owed solely to London Stock Exchange and are not owed
to the Company or to any Director or to any other person in respect
of his decision to acquire shares in the Company in reliance on any
part of this Announcement.
finnCap Ltd, which is authorised and regulated by the FCA for
the conduct of regulated activities in the United Kingdom, is
acting as joint bookrunner to the Company and no one else in
connection with the Placing and is not acting for and will not be
responsible to any person other than the Company for providing the
protections afforded to its clients or for providing advice in
relation to the Placing.
Except as required under applicable law, neither Peel Hunt,
finnCap nor any of their directors, officers, partners, members,
employees, advisers, affiliates or agents assume or accept any
responsibility whatsoever for the contents of the information
contained in this Announcement or for any other statement made or
purported to be made by or on behalf of Peel Hunt, finnCap or any
of their affiliates in connection with the Company, the new
Ordinary Shares or the Placing. Peel Hunt, finnCap and each of
their directors, officers, partners, members, employees, advisers,
affiliates and agents accordingly disclaim all and any
responsibility and liability whatsoever, whether arising in tort,
contract or otherwise (save as referred to above) in respect of any
statements or other information contained in this Announcement and
no representation or warranty, express or implied, is made by Peel
Hunt, FinnCap or any of their directors, officers, partners,
employees, advisers, affiliates or agents as to the accuracy,
completeness or sufficiency of the information contained in this
Announcement.
The distribution of this Announcement and/or the Placing in
certain jurisdictions may be restricted by law. No action has been
taken by the Company, Peel Hunt, finnCap or any of their respective
affiliates that would, or which is intended to, permit an offering
of the new Ordinary Shares in any jurisdiction or result in the
possession or distribution of this Announcement or any other
offering or publicity material relating to new Ordinary Shares in
any jurisdiction where action for that purpose is required.
This Announcement does not constitute a recommendation
concerning any investor's option with respect to the Placing. Each
investor or prospective investor should conduct his, her or its own
investigation, analysis and evaluation of the business and data
described in this Announcement and publicly available information.
The price and value of securities can go down as well as up. Past
performance is not a guide to future performance. The contents of
this Announcement are not to be construed as legal, business,
financial or tax advice. Each investor or prospective investor
should consult with his or her or its own legal adviser, business
adviser, financial adviser or tax adviser for legal, financial,
business or tax advice.
Neither the content of the Company's website nor any website
accessible by hyperlinks on the Company's website is incorporated
in, or forms part of, this Announcement.
Product Governance Requirements
Solely for the purposes of the product governance requirements
contained within: (a) EU Directive 2014/65/EU on markets in
financial instruments, as amended, as it forms part of UK domestic
law by virtue of the European Union (Withdrawal) Act 2018, as
amended ("MiFID II"); (b) Articles 9 and 10 of Commission Delegated
Directive (EU) 2017/593 supplementing MiFID II, as amended, as it
forms part of UK domestic law by virtue of the European Union
(Withdrawal) Act 2018; and (c) local implementing measures
(together, the "MiFID II Product Governance Requirements"), and
disclaiming all and any liability, whether arising in tort,
contract or otherwise, which any "manufacturer" (for the purposes
of the MiFID II Product Governance Requirements) may otherwise have
with respect thereto, the Placing Shares have been subject to a
product approval process, which has determined that the Placing
Shares are: (i) compatible with an end target market of: (a) retail
investors; (b) investors who meet the criteria of professional
clients; and (c) eligible counterparties, each as defined in MiFID
II; and (ii) eligible for distribution through all distribution
channels as are permitted by MiFID II (the "Target Market
Assessment"). Notwithstanding the Target Market Assessment,
distributors should note that: the price of the Placing Shares may
decline and investors could lose all or part of their investment;
the Placing Shares offer no guaranteed income and no capital
protection; and an investment in the Placing Shares is compatible
only with investors who do not need a guaranteed income or capital
protection, who (either alone or in conjunction with an appropriate
financial or other adviser) are capable of evaluating the merits
and risks of such an investment and who have sufficient resources
to be able to bear any losses that may result therefrom. The Target
Market Assessment is without prejudice to the requirements of any
contractual, legal or regulatory selling restrictions in relation
to the Placing. Furthermore, it is noted that, notwithstanding the
Target Market Assessment, Peel Hunt and finnCap will only procure
investors who meet the criteria of professional clients and
eligible counterparties.
For the avoidance of doubt, the Target Market Assessment does
not constitute: (a) an assessment of suitability or appropriateness
for the purposes of MiFID II; or (b) a recommendation to any
investor or group of investors to invest in, or purchase, or take
any other action whatsoever with respect to the Placing Shares.
Forward Looking Statements
This Announcement contains "forward-looking statements" which
include all statements (other than statements of historical facts)
including, without limitation, those regarding the Group's
financial position, business strategy, plans and objectives of
management for future operations, and any statements preceded by,
followed by or that include the words "targets", "believes",
"expects", "aims", "intends", "will", "may", "anticipates",
"would", "could" or "similar" expressions or negatives thereof.
Such forward-looking statements involve known and unknown risks,
uncertainties and other important factors beyond the Company's
control that could cause the actual results, performance or
achievements of the Group to be materially different from future
results, performance or achievements expressed or implied by such
forward-looking statements. Such forward-looking statements are
based on numerous assumptions regarding the Group's present and
future business strategies and the environment in which the Group
will operate in the future. These forward-looking statements speak
only as at the date of this Announcement. The Company expressly
disclaims any obligation or undertaking to disseminate any updates
or revisions to any forward looking statements contained herein to
reflect any change in the Company's expectations with regard
thereto or any change in events, conditions or circumstances on
which any such statements are based unless required to do so by
applicable law or the AIM Rules for Companies.
The following text has been extracted from the draft
Circular:
Appendix I - EXTRACTS FROM THE CIRCULAR
LETTER FROM THE CHAIRMAN
1) BACKGROUND TO AND REASONS FOR THE FUNDRAISING AND USE OF
PROCEEDS
OVERVIEW
Shield's lead product is Feraccru(R)/Accrufer(R), a novel oral
therapy for the treatment of iron deficiency. The active ingredient
of Feraccru(R)/Accrufer(R) is a molecule called ferric maltol and
the product is formulated as a capsule with an iron content of
30mg. Dosing is two capsules per day taken before food. Feraccru(R)
is the brand name in Europe where the product was approved for the
treatment of iron deficiency in adults in 2018. In the US the
product will be known as Accrufer(R) and it was approved by the FDA
in 2019 and has NCE status until 2024, with the same broad
indication of the treatment of iron deficiency in adults. The
product is protected by a range of patents extending to 2035.
Feraccru(R) is being marketed and sold in Europe by Norgine BV,
who also have the commercialisation rights in Australia and New
Zealand, and it has been licensed to Aosaikang Pharm ("ASK Pharm")
for development and commercialisation in China, Taiwan, Hong Kong
and Macau. Shield is now planning to launch Accrufer(R) in the US
and the purpose of the Fundraising is to provide the finance
necessary for the launch and to fund Shield to the point where the
Group expects to generate positive cash flows. Based on the Group's
cash flow forecasts, including the costs of the US launch of
Accrufer(R) and the paediatric study, the Group could start to
breakeven on a monthly basis within 15-18 months after launch
provided sales and costs are within the range anticipated by the
Directors.
Iron Deficiency
Iron is a key component of haemoglobin (Hb), a protein found in
red blood cells. Iron deficiency, which is caused by failure to
absorb enough iron through diet or the loss of iron through
bleeding, and consequential low Hb levels, leads to iron deficiency
anaemia (IDA). Mild levels of iron deficiency can be treated by
non-prescription iron supplements. More significant iron deficiency
and anaemia have historically been treated initially with
prescription oral iron salt products and subsequently, if
necessary, with intravenous (IV) infusions of iron. Oral iron salts
are mostly generic products and therefore inexpensive, but their
chemical composition means that they dissociate rapidly when
ingested. The iron freed up by this dissociation can cause a range
of adverse effects in patients such as diarrhoea, constipation,
nausea, abdominal pain and potentially more serious damage to the
intestinal lining which makes them unsuitable for patients with
inflammatory bowel diseases such as Crohn's disease or colitis.
These adverse effects result in oral iron salts typically being
poorly tolerated and ultimately in non-compliance by patients. IV
infusions typically deliver from 500mg to 1,000mg of iron in one or
two infusion sittings. IV infusions are required to be administered
in a hospital or clinic setting where resuscitation facilities are
available due to the small risk of allergic reactions. IV infusions
are therefore less convenient for patients than oral administration
and may require repeat infusions after the benefits of earlier
infusions have dissipated.
Shield believes that Accrufer(R) can address both oral and IV
segments of the markets. Up to 70% of patients treated with oral
iron salts experience gastrointestinal side effects and might be
considered oral iron intolerant. Accrufer(R) has been shown to be
well tolerated and effective which therefore makes it a credible
alternative to oral iron salt products. Compared with IV infusions,
Accrufer(R) offers the advantage of convenient administration and
has demonstrated that, taken twice daily, it can maintain Hb at
normal levels.
FERACCRU(R) /ACCRUFER(R) DIFFERENTIATION AND POSITIONING
Feraccru(R) /Accrufer(R) (ferric maltol) is an oral product but
is not an iron salt and has a different chemical composition from
oral iron salt products. The ferric maltol molecule does not
dissociate on ingestion but remains bound until it reaches the
duodenum. If the body is iron deficient, the body's natural
processes overcome the ferric maltol binding and iron is absorbed
into the blood stream leaving maltol, which is a naturally
occurring compound often used in food products as a flavour
enhancer, to be harmlessly excreted naturally. If the body is no
longer iron deficient, the ferric maltol molecule remains intact
and is excreted. Ferric maltol is well tolerated and has been shown
in clinical studies to have a side effect profile similar to
placebo.
Three Phase III clinical studies have been conducted on
Feraccru(R) /Accrufer(R) . The first two studies, in patients
suffering from inflammatory bowel disease and chronic kidney
disease, were used for the approval of the product in Europe and
the US. These studies, conducted against placebo, demonstrated that
Feraccru(R) /Accrufer(R) increases Hb levels by clinically relevant
amounts over 12/16 weeks, maintains Hb levels over the long term
and is well tolerated. The third study was a comparison against IV
iron. This study showed similar results to the first two studies in
terms of restoration of Hb levels, maintenance of Hb over the long
term and tolerability. In the comparison against IV therapy, the
restoration of Hb levels in the first 12 weeks was not as fast as
that shown by the IV treatment and the study therefore did not meet
this primary endpoint. However the Directors were encouraged that
over the longer term, Feraccru(R) /Accrufer(R) showed that it was
able to maintain Hb levels steadily and avoided anaemia in those
patients on the Feraccru(R) /Accrufer(R) arm, whereas 58% of
patients on the IV arm who were monitored from the week 12 visit
required at least one further IV infusion.
US market opportunity
The US presents a substantial market opportunity for Accrufer(R)
and Shield believes there is significant and well-documented unmet
market need for an effective, well-tolerated oral iron therapeutic
option. An estimated 10 million patients suffer from iron
deficiency anaemia at any time in the United States with some 5
million of these treated annually. Patients typically come from a
range of therapeutic areas including chronic kidney disease,
gastrointestinal disorders, women's health, cardiology and
oncology, and in the US each year there are approximately 10-11
million prescriptions for first line oral iron salts and around 2.3
million IV doses infused. Annual US IV iron sales are estimated to
be around $1.2 billion annually and can be less convenient for
patients with expensive administration costs.
Market research shows that physicians believe there is an unmet
need in the market with key needs being effectiveness and GI
tolerability. Accrufer(R)'s product profile, with its good
tolerability profile and efficacy data, was viewed favourably
compared with existing oral and IV therapies giving potential for
first line usage if allowed by insurance plans. Further, payer
research has indicated that Accrufer(R) should have few
restrictions and non-preferred formulary status at tested price
points ensuring good patient access.
The COVID-19 pandemic has been changing healthcare delivery and
recommendations for the care of at-risk patients. Many ID/IDA
patients have an underlying condition making them susceptible to
COVID-19, including conditions such as CKD, cancer, heart failure,
those taking immunosuppressant drugs and the elderly. This has led
to a strong preference for home treatments and switching patients
from IV to oral therapies to minimize exposure risk of vulnerable
patients to the virus. Shield believes this dynamic will be helpful
for the take-up of Accrufer(R) as long as COVID-19 presents a
threat to these at-risk patients.
Furthermore, Shield believes that the first US launch of an oral
hypoxia-inducible factor-prolyl hydroxylase (HIF-PH) inhibitor(1)
(Roxadustat) for CKD patients, anticipated in 2021 (subject to FDA
approval), is likely to increase the need for effective and well
tolerated oral iron replacement therapy. HIF-PH inhibitors are
orally administered products that promote erythropoiesis, the
production of red blood cells, by increasing endogenous production
of erythropoietin. Since supplemental iron is still likely to be
required for many patients that in time may be prescribed
Roxadustat, an effective and well-tolerated oral iron product such
as Accrufer(R) may be preferred to IV iron.
Qualitative market research with ten US payers representing
around 200 million covered lives indicated that Accrufer(R) is
expected to have few restrictions at tested price points and that
an orally administered therapy with better gastrointestinal
tolerability is the key unmet need perceived by payers being
addressed by Accrufer(R).
Net sales estimates generated by management consultants and
other third parties support the potential for Accrufer(R) US sales
to exceed $100 million in the third year following launch and to
reach $300 million to $400 million by years five to six.
Shield anticipates that patients taking Accrufer(R) will use, on
average, four monthly packs in a year which is likely to be a
combination of using Accrufer(R) to restore Hb levels and also for
maintenance of Hb at normal, non-anaemic, levels. Assuming a net US
pack price of $250, which was supported by the payer market
research, the average usage of four packs per year implies net
sales of $1,000 per patient per year in the US. Net sales of $100
million in a given year would therefore require approximately
100,000 patients to be treated annually or 400,000 prescriptions
which equates to 2% of the 5 million US IDA patients treated
annually.
The gross margin of Accrufer(R) is expected to be around 90% of
net sales after deducting both the cost of goods and a 5% royalty
on net sales payable to Vitra Pharmaceuticals Limited, the original
owner of the ferric maltol intellectual property. Shield's launch
plan for the US expects annual SG&A costs in the US to be
around $40 million to $45 million by year three. Based on the
Group's cash flow forecasts, including the costs of the US launch
of Accrufer(R) and the paediatric study, the Group could start to
breakeven on a monthly basis within 15-18 months after launch
provided sales and costs are within the range anticipated by the
Directors. The Directors believe that the US opportunity has the
potential to generate significant cash for Shield .
Planning for a successful US launch
Shield has engaged four US pharmaceutical commercial executives
with deep experience of the key areas necessary such as pricing and
market access, marketing, medical affairs, and supply chain.
Detailed planning for Accrufer(R) 's launch is being undertaken
across all of these areas. In particular market analysis has been
conducted into the current prescribers of iron therapy. The
Company's US commercial operations will be run by Brian Groch who
has some 30 years' commercial experience gained from working with a
range of pharmaceutical and biotechnology companies.
Whilst there are around 460,000 healthcare professionals
prescribing oral and IV iron, around 11,000 high prescribers have
been identified who account for 30% of oral iron prescriptions.
These high prescribers each average 252 iron prescriptions per
annum. Further, 100 "super prescribers" have been identified, each
of whom writes over 1,000 prescriptions per annum in the US. Shield
plans to target these prescribers, initially with 30 sales
representatives rising to 60 as sales momentum grows - this is
expected to cover 80% of the target list of prescribers. The Group
is also planning to conduct a significant proportion of sales and
marketing interactions by using remote/e-detailing technology
rather than in-person meetings. This is a trend which has been
developing in recent years and has been accelerated by the
coronavirus pandemic and which enables more efficient communication
with prescribers and key opinion leaders than was previously
possible. Launch stocks of Accrufer(R) have been manufactured and
are ready to be shipped to the US and first sales are expected to
be made in Q2 2021.
Feraccru(R) in Europe and China
Feraccru(R) was initially approved in Europe in 2016 for the
treatment of iron deficiency in adults suffering from inflammatory
bowel disease but the label was broadened in 2018 to allow the
treatment of all iron deficiency in adults. Feraccru(R) was
licensed to Norgine BV in September 2018 for commercialisation in
most of Europe, Australia and New Zealand. Shield received an GBP11
million upfront payment on signing and is entitled to royalties
ranging from 25% to 40% based on tiers of Norgine BV's net sales.
Shield is responsible for paying for the cost of manufacturing. In
addition, Shield could receive sales milestones up to EUR50 million
based on Norgine BV achieving defined annual targets. Feraccru(R)
is currently being marketed by Norgine BV in Germany, the UK,
Scandinavia and Belgium. Sales volumes in Germany and the UK in
2020 grew by 70% compared with 2019, and royalty income to Shield
amounted to GBP0.7 million in 2020. Norgine BV are using the
updated AEGIS H2H detailed study results to reconfirm pricing and
reimbursement strategy for Feraccru(R) in the major European
markets of France, Italy and Spain.
Feraccru(R) is not yet approved in China. It has been licensed
to ASK Pharm who will complete the development, seek marketing
approval and then commercialise the product. Shield received $11.4
million on signing the license agreement in 2020 and will receive a
further $11.4 million upon approval. When launched, Shield will be
entitled to royalties of 10% and 15% based on tiers of ASK Pharm's
net sales, and up to $40 million in sales milestones. ASK Pharm
will pay the cost of manufacturing the product. ASK Pharm have
recently submitted the Investigational New Drug (IND) application
to the Chinese regulatory authorities and anticipate that one
12-week Phase III study will be required in 120 inflammatory bowel
disease patients before the product can be approved, potentially in
2023.
Paediatric study
When Feraccru(R) /Accrufer(R) was approved by the EMA and the
FDA, both agencies imposed a post-approval commitment on Shield to
evaluate the safety and tolerability of the product in infants,
children and adolescents. Secondary endpoints include the change in
Hb concentration and achieving Hb concentration within normal range
by week 12. A liquid formulation has been developed and tested in
healthy adult volunteers for equivalence with the capsule version.
The Clinical Study Report from this first stage is expected by the
end of February 2021. The main study is expected to start
recruiting 110 subjects in summer 2021 and is expected to cost
around GBP4.5 million and take up to 30 months. A successful
outcome could lead to the product's label being expanded to include
children.
Development pipeline - PT20
PT20 is a Phase III-ready phosphate binder in development for
the treatment of hyperphosphatemia. Hyperphosphatemia is a
metabolic disorder characterized by elevated serum phosphorus
levels. Older generation phosphate binders have been based on
metals (lanthanum, aluminium), calcium salts, and polymers and have
side effects, poor tolerance and lack of effectiveness. Latest
generation products like Velphoro and Auryxia are iron-based. The
overall market size of the US market is around $1 billion per
annum. PT20 is a novel formulation iron-based phosphate binder
which enhances phosphate binding with similar side effects compared
to latest generation iron-based products. PT20 has completed one
Phase II pivotal study and now requires one further Phase III study
to allow an NDA to be filed. Development of PT20 has been
constrained in recent years due to lack of finance. Prior to
beginning a Phase III study it will be necessary to develop a
formulation suitable for commercial use. The Phase III would be
expected to cost around GBP20 million and would take 2-3 years.
2) CASH REQUIREMENT, SOURCES AND USE OF PROCEEDS
The Group's cash flow forecasts, including the costs of the US
launch of Accrufer(R) and the paediatric study, indicate that the
Group should start to breakeven on a monthly basis within 15-18
months after launch of the drug in the US provided sales and costs
are within the range anticipated by the Directors . As previously
announced Shield believes that around $30 million to $40 million (c
GBP21 million to GBP29 million) should provide the finance
necessary to reach the breakeven point.
The net proceeds of the Subscription and the Placing will
finance the launch costs of Accrufer(R). The bulk of the net
proceeds received will be converted into US dollars in order to
avoid currency risk. US launch costs will include sales
representatives, ongoing market research and data analysis,
marketing spend and other US operational costs. The paediatric
study and other non-US expenditure is expected to be partly funded
out of gross margin generated by Shield in the US and Norgine BV
royalty revenues.
The Shareholder Loan Facilities announced on 10 December 2020
and confirmed on 29 January 2021 which amounted to approximately
GBP4.4 million will be cancelled on completion of the
Fundraising.
3) CURRENT TRADING AND OUTLOOK
On 15 January 2021 the Group issued a trading update for the
year ended 31 December 2020 which highlighted that Feraccru(R) 2020
sales volumes in Europe had increased by c.70% compared with 2019,
the IND application in China had been submitted, and the first
stage of the paediatric study plan had been completed other than
the finalisation of the study report. Unaudited revenues in 2020
were GBP9.4 million, which included GBP8.7 million from the licence
upfront payment received from ASK Pharm. Unaudited cash balances at
the end of December 2020 and January 2021 were GBP2.9 million and
GBP2.3 million respectively, the outflow in January 2021 impacted
by the payment of a significant portion of the costs of the first
stage of the paediatric study. Trading in January and February 2021
has been in line with management expectations. Although the US has
been the recent commercialisation priority, during 2020 Shield
continued to have discussions with potential partners in several
other countries and the Company is aiming to complete a new licence
transaction in 2021.
4) DETAILS OF THE PLACING, THE SUBSCRIPTION AND THE OPEN
OFFER
The Directors gave careful consideration to the structure of the
Fundraising and concluded that the Placing, together with the
Subscription and the Open Offer was the most suitable option
available to the Company and its Shareholders at this time.
The Placing will be made available to certain eligible existing
institutional shareholders and certain new institutional investors
to raise gross proceeds of up to approximately GBP19.9 million.
AOP, a major shareholder of the Company, have indicated that
they will subscribe for 14,333,333 Subscription Shares. Dr.
Christian Schweiger, a non-executive Director of the Company and
major shareholder and Tim Watts, Chief Executive Officer of the
Company, have also indicated that they will subscribe for 1,301,280
Subscription Shares and 1,000,000 Subscription Shares respectively.
Further, certain employees of the Company who are existing
Shareholders have indicated that they will subscribe for an
aggregate of 300,000 Subscription Shares at the Issue Price
pursuant to the Subscription. The aggregate gross proceeds of the
Subscription will amount to approximately GBP5.1 million.
The gross proceeds of the Placing and the Subscription are up to
GBP25 million.
The Open Offer is being made for up to 13,946,397 Open Offer
Shares at the Issue Price on the basis of 2 Open Offer Shares for
every 17 Ordinary Shares held by Qualifying Shareholders at the
Record Date, to raise up to approximately GBP4.2 million before
expenses.
Shareholders subscribing for their full entitlement under the
Open Offer may also request additional Open Offer Shares as an
Excess Entitlement, up to the total number of Open Offer Shares
available to Qualifying Shareholders under the Open Offer.
The Placing, the Subscription and the Open Offer are conditional
upon the Placing and Open Offer Agreement becoming unconditional
and not having been terminated in accordance with its terms. The
Placing and Open Offer Agreement is conditional, amongst other
things, upon the following:
-- the passing without amendment of the Fundraising Resolutions at the General Meeting;
-- the Company having allotted, subject only to Admission, the
Placing Shares, the Subscription Shares and such number of Open
Offer Shares subscribed for in accordance with the Placing and Open
Offer Agreement;
-- the Company having complied with its obligations under the
Placing and Open Offer Agreement to be performed on or prior to
Admission and not being in breach of the Placing and Open Offer
Agreement; and
-- Admission occurring by 8.00 a.m. on 19 March 2021 or such
later time and date (being not later than 8.00 a.m. on 31 March
2021) as the Joint Bookrunners and the Company may agree.
If the conditions set out above are not satisfied or waived
(where capable of waiver), the Placing, the Subscription and the
Open Offer will lapse and the New Shares will not be allotted and
issued and no monies will be received by the Company from investors
in respect of the New Shares.
5) THE PLACING AND OPEN OFFER AGREEMENT
Pursuant to the terms and subject to the conditions of the
Placing and Open Offer Agreement, the Joint Bookrunners, as agents
for the Company, have severally (and not jointly or jointly and
severally) agreed to use their reasonable endeavours to procure
Placees to take up the Placing Shares, at the Issue Price. The
Placing and Open Offer Agreement is conditional upon, amongst other
things, the conditions set out in paragraph 4 above.
The Placing and Open Offer Agreement contains customary
warranties given by the Company in favour of the Joint Bookrunners
in relation to, amongst other things, the accuracy of the
information in this document and other matters relating to the
Group and its business. In addition, the Company has agreed to
indemnify the Joint Bookrunners (and certain of their affiliates)
in relation to certain liabilities which they may incur in respect
of the Placing, the Open Offer and the Subscription.
Each of the Joint Bookrunners has the right to terminate its
obligations under the Placing and Open Offer Agreement in certain
circumstances prior to Admission. In particular, in the event of
breach of the warranties or if the Placing and Open Offer Agreement
does not become unconditional, the Joint Bookrunners may terminate
their obligations under the Placing, in which case the Fundraising
will terminate and the New Shares will not be issued.
6) THE GENERAL MEETING
The Directors do not currently have authority to allot all of
the New Shares and, accordingly, the Board is seeking the approval
of Shareholders, at the General Meeting, to allot the New
Shares.
AOP, W. Health and Dr. Christian Schweiger, who hold in
aggregate 61.3% of the Company's existing issued share capital have
irrevocably agreed to vote in favour of the Resolutions at the
General Meeting pursuant to the Voting Undertakings.
The General Meeting is being held inter alia for the purpose of
considering and, if thought fit, passing the Fundraising
Resolutions in order to approve the authorities required to allot
and issue the New Shares.
7) ACTION TO BE TAKEN IN RESPECT OF THE GENERAL MEETING
Shareholders should complete and submit a Form of Proxy (whether
online or by requesting a hard copy form from Link Group) in
accordance with the instructions printed on it. Shareholders will
not receive a hard copy form of proxy for the General Meeting in
the post. Instead, Shareholders will be able to vote electronically
using the link www.signalshares.com. Shareholders will need to log
into their Signal Shares account, or register if they have not
previously done so. To register they will need their Investor Code,
detailed on their share certificate or available from the
Registrar, Link Group. The Form of Proxy (if completed in hard
copy) must be received by the Company's registrars, Link Group,
PXS, 10(th) Floor, Central Square, 29 Wellington Street, Leeds LS1
4DL, by no later than the date set out in the Circular. In light of
the social distancing measures imposed by the UK Government as a
result of the current COVID-19 pandemic, any proxy you appoint
other than the chairman of the General Meeting will be refused
entry to the General Meeting. You should therefore appoint the
chairman of the General Meeting as your proxy to ensure your vote
is counted.
Shareholders are reminded that the Fundraising is conditional,
inter alia, on the passing of the Fundraising Resolutions to be
proposed at the General Meeting. Should the Fundraising Resolutions
not be passed, the Fundraising will not proceed and any associated
subscription monies in respect of the New Shares will be returned
to investors.
8) RECOMMATION
If the Fundraising Resolutions are not passed at the General
Meeting, the Fundraising will not take place and the proceeds of
the Fundraising will not be received by the Company. This would
materially and adversely affect the Company's business plans and
severely impact its ability to launch Accrufer(R) in the US as
currently intended. The Company also would not be able to commence
the proposed stage two paediatric study which would breach
commitments made to the EMA and FDA and prevent
Feraccru(R)/Accrufer(R) from receiving EMA and FDA approval for use
by infants, children and adolescents.
If this were to happen, in order to have sufficient working
capital to continue to trade, the Company would draw down under the
Shareholder Loan Facilities. Whilst these loans can provide the
Company with a cash runway through to approximately the end of 2021
they will not provide sufficient funding for the Company to launch
Accrufer(R) in the US directly or to start the paediatric study.
The Directors would need to explore alternative forms of funding
for its medium term operations which may not be available at all or
if available, may be on commercially unacceptable terms. The
Directors would also have to explore alternative options for
maximising value from Accrufer(R) in the United States and the
Company as a whole, having recently concluded that a Shield-led
launch of Accrufer(R) in the US was likely to provide greater value
to Shareholders compared to other options that the Company has been
assessing.
The Directors consider that the Fundraising and the passing of
the Resolutions are in the best interests of the Company and its
Shareholders as a whole. Accordingly, the Directors unanimously
recommend that Shareholders vote in favour of all of the
Resolutions, as they intend to do in respect of their beneficial
holdings of an aggregate of 4,973,000 Existing Ordinary Shares,
representing approximately 4.2 per cent, of the Existing Ordinary
Shares.
The Fundraising is conditional, inter alia, upon the passing of
the Fundraising Resolutions at the General Meeting.
APPIX II - TERMS AND CONDITIONS OF THE PLACING
IMPORTANT INFORMATION ON THE PLACING FOR INVITED PLACEES
ONLY.
THIS ANNOUNCEMENT, INCLUDING THE APPICES AND THE TERMS AND
CONDITIONS SET OUT HEREIN (TOGETHER, THIS "ANNOUNCEMENT") IS FOR
INFORMATION PURPOSES ONLY AND IS DIRECTED ONLY AT PERSONS WHO ARE
(1) IF IN A MEMBER STATE OF THE EUROPEAN ECONOMIC AREA, QUALIFIED
INVESTORS AS DEFINED IN ARTICLE 2(E) OF THE PROSPECTUS REGULATION
(EU) 2017/1129 (THE "EU PROSPECTUS REGULATION"); (2) IF IN THE
UNITED KINGDOM, QUALIFIED INVESTORS AS DEFINED IN ARTICLE 2(E) OF
PROSPECTUS REGULATION (EU) 2017/1129 AS IT FORMS PART OF UK
DOMESTIC LAW BY VIRTUE OF THE EUROPEAN UNION (WITHDRAWAL) ACT 2018,
AS AMED (THE "UK PROSPECTUS REGULATION), WHO (A) FALL WITHIN
ARTICLE 19(5) ("INVESTMENT PROFESSIONALS") OF THE FINANCIAL
SERVICES AND MARKETS ACT 2000 (FINANCIAL PROMOTION) ORDER 2005, AS
AMED (THE "ORDER"), OR (B) FALL WITHIN ARTICLE 49(2)(A) TO (D)
("HIGH NET WORTH COMPANIES, UNINCORPORATED ASSOCIATIONS, ETC.") OF
THE ORDER; OR (3) ARE PERSONS TO WHOM IT MAY OTHERWISE BE LAWFULLY
COMMUNICATED; (ALL SUCH PERSONS TOGETHER BEING REFERRED TO AS
"RELEVANT PERSONS").
THIS ANNOUNCEMENT MUST NOT BE ACTED ON OR RELIED ON BY PERSONS
WHO ARE NOT RELEVANT PERSONS. PERSONS DISTRIBUTING THIS
ANNOUNCEMENT MUST SATISFY THEMSELVES THAT IT IS LAWFUL TO DO SO.
ANY INVESTMENT OR INVESTMENT ACTIVITY TO WHICH THIS ANNOUNCEMENT
AND THE TERMS AND CONDITIONS SET OUT HEREIN RELATES IS AVAILABLE
ONLY TO RELEVANT PERSONS AND WILL BE ENGAGED IN ONLY WITH RELEVANT
PERSONS. THIS ANNOUNCEMENT DOES NOT ITSELF CONSTITUTE AN OFFER FOR
THE SALE OR SUBSCRIPTION OF ANY SECURITIES IN THE COMPANY.
The New Shares have not been and will not be registered under
the US Securities Act of 1933, as amended (the "US Securities Act")
or under any securities laws of any state or other jurisdiction of
the United States and may not be offered, sold, pledged, taken up,
exercised, resold, renounced, transferred or delivered, directly or
indirectly, in or into the United States except pursuant to an
applicable exemption from the registration requirements of the US
Securities Act and in compliance with the securities laws of any
state or other jurisdiction of the United States. There will be no
public offer of the securities mentioned herein in the United
States.
This Announcement and the information contained herein is
restricted and is not for release, publication or distribution, in
whole or in part, directly or indirectly, in or into or from the
United States, Australia, Canada, Japan, the Republic of South
Africa or any other jurisdiction in which such release publication
or distribution would be unlawful.
The distribution of this Announcement and/or the Placing and/or
the issue of the New Shares in certain jurisdictions may be
restricted by law. No action has been taken by the Company, the
Joint Bookrunners or any of their respective affiliates, agents
directors, officers or employees that would permit an offer of the
New Shares or possession or distribution of this Announcement or
any other offering or publicity material relating to such New
Shares in any jurisdiction where action for that purpose is
required. Persons into whose possession this Announcement comes are
required by the Company and the Joint Bookrunners to inform
themselves about and to observe any such restrictions.
This Announcement or any part of it does not constitute or form
part of any offer to issue or sell, or the solicitation of an offer
to acquire, purchase or subscribe for any securities in the United
States, Australia, Canada, Japan or the Republic of South Africa or
any other jurisdiction in which the same would be unlawful. No
public offering of the New Shares is being made in any such
jurisdiction.
The relevant clearances have not been, nor will they be,
obtained from the securities commission of any province or
territory of Canada, no prospectus has been lodged with, or
registered by, the Australian Securities and Investments
Commission, the Japanese Ministry of Finance; the relevant
clearances have not been, and will not be, obtained for the South
Africa Reserve Bank or any other applicable body in the Republic of
South Africa in relation to the New Shares and the New Shares have
not been, nor will they be registered under or offered in
compliance with the securities laws of any state, province or
territory of Australia, Canada, Japan or the Republic of South
Africa. Accordingly, the New Shares may not (unless an exemption
under the relevant securities laws is applicable) be offered, sold,
pledged, taken up, exercised, resold, renounced, transferred or
delivered, directly or indirectly, in or into Australia, Canada,
Japan or the Republic of South Africa or any other jurisdiction
outside the United Kingdom and the EEA.
This Announcement should be read in its entirety. In particular,
you should read and understand the information provided in the
'Important Notices' section of this Announcement.
By participating in the Bookbuild and/or the Placing, each
Placee will be deemed to have read and understood this Announcement
in its entirety, to be participating, making an offer and acquiring
Placing Shares on the terms and conditions contained herein and to
be providing the representations, warranties, indemnities,
acknowledgements and undertakings contained in this Appendix.
In particular, each such Placee represents, warrants,
undertakes, agrees and acknowledges, inter alia, that:
1. it is a Relevant Person and undertakes that it will acquire,
hold, manage or dispose of any Placing Shares that are allocated to
it for the purposes of its business;
2. in the case of a Relevant Person in the United Kingdom who
acquires any Placing Shares pursuant to the Placing:
(a) it is a Qualified Investor within the meaning of Article
2(e) of the UK Prospectus Regulation; and
(b) in the case of any Placing Shares acquired by it as a
financial intermediary, as that term is used in Article 5(1) of the
UK Prospectus Regulation:
(i) the Placing Shares acquired by it in the Placing have not
been acquired on behalf of, nor have they been acquired with a view
to their offer or resale to, persons in the United Kingdom other
than Qualified Investors or in circumstances in which the prior
consent of the Joint Bookrunners have been given to the offer or
resale; or
(ii) where Placing Shares have been acquired by it on behalf of
persons in the United Kingdom other than Qualified Investors, the
offer of those Placing Shares to it is not treated under the UK
Prospectus Regulation as having been made to such persons;
3. in the case of a Relevant Person in a member state of the EEA
(each, a "Relevant Member State") who acquires any Placing Shares
pursuant to the Placing:
(a) it is a Qualified Investor within the meaning of Article
2(e) of the EU Prospectus Regulation; and
(b) in the case of any Placing Shares acquired by it as a
financial intermediary, as that term is used in Article 5(1) of the
EU Prospectus Regulation:
(i) the Placing Shares acquired by it in the Placing have not
been acquired on behalf of, nor have they been acquired with a view
to their offer or resale to, persons in any Relevant Member State
other than Qualified Investors or in circumstances in which the
prior consent of the Joint Bookrunners have been given to the offer
or resale; or
(ii) where Placing Shares have been acquired by it on behalf of
persons in any Relevant Member State other than Qualified
Investors, the offer of those Placing Shares to it is not treated
under the EU Prospectus Regulation as having been made to such
persons;
4. it is acquiring the Placing Shares for its own account or is
acquiring the Placing Shares for an account with respect to which
it has authority to exercise, and is exercising, investment
discretion and has the authority to make and does make the
representations, warranties, indemnities, acknowledgements,
undertakings and agreements contained in this Announcement;
5. it understands (or if acting for the account of another
person, such person has confirmed that such person understands) the
resale and transfer restrictions set out in this Appendix;
6. except as otherwise permitted by the Company and subject to
any available exemptions from applicable securities laws, it (and
any person on whose account it is acting, as referred to in
paragraph 5 above) is located outside the United States and is
acquiring the Placing Shares in an "offshore transaction" as
defined in, and in accordance with, Regulation S under the US
Securities Act ("Regulation S"); and
7. it has not offered, sold or delivered and will not offer to
sell or deliver any of the Placing Shares to persons within the
United States, directly or indirectly; neither it, its affiliates,
nor any persons acting on its behalf, have engaged or will engage
in any directed selling efforts (as defined in Regulation S) with
respect to the Placing Shares; and it is not taking up the Placing
Shares for resale in or into the United States.
No prospectus
The New Shares are being offered to a limited number of
specifically invited persons only and will not be offered in such a
way as to require any prospectus or other offering document to be
published. No prospectus or other offering document has been or
will be submitted to be approved by the FCA in relation to the
Placing or the New Shares and Placees' commitments will be made
solely on the basis of their own assessment of the Company, the New
Shares and the Placing based on the information contained in this
Announcement, the announcement of the closing of the Placing (the
"Results Announcement") (together, the "Placing Documents") and any
other information publicly announced through a regulatory
information service ("RIS") by or on behalf of the Company on or
prior to the date of this Announcement (the "Publicly Available
Information") and subject to any further terms set forth in the
contract note sent to individual Placees.
Each Placee, by participating in the Placing, agrees that the
content of the Placing Documents is exclusively the responsibility
of the Company and confirms that it has neither received nor relied
on any information (other than the Publicly Available Information),
representation, warranty or statement made by or on behalf of the
Joint Bookrunners or the Company or any other person and none of
the Joint Bookrunners, the Company nor any other person acting on
such person's behalf nor any of their respective affiliates has or
shall have any responsibility or liability for any Placee's
decision to participate in the Placing based on any other
information, representation, warranty or statement. Each Placee
acknowledges and agrees that it has relied on its own investigation
of the business, financial or other position of the Company in
accepting a participation in the Placing. No Placee should consider
any information in this Announcement to be legal, tax or business
advice. Each Placee should consult its own attorney, tax advisor,
and business advisor for legal, tax and business advice regarding
an investment in the New Shares. Nothing in this paragraph shall
exclude the liability of any person for fraudulent
misrepresentation.
Details of the Placing and Open Offer Agreement and the Placing
Shares
The Joint Bookrunners are acting as joint bookrunners and
brokers in connection with the Placing and have today entered into
the Placing and Open Offer Agreement with the Company under which,
on the terms and subject to the conditions set out in the Placing
and Open Offer Agreement, the Joint Bookrunners, as agents for and
on behalf of the Company, have severally (and not jointly or
jointly and severally) agreed to use their respective reasonable
endeavours to procure placees for the Placing Shares at the Issue
Price.
Further details of the placing procedure and terms on which the
Placing Shares are being offered are set out below.
No element of the Placing is being underwritten.
The Placing Shares will, when issued, be credited as fully paid
up and will be issued subject to the Company's articles of
association and rank pari passu in all respects with the Existing
Ordinary Shares, including the right to receive all dividends and
other distributions declared, made or paid on or in respect of the
Ordinary Shares after the date of issue of the Placing Shares, and
will on issue be free of all claims, liens, charges, encumbrances
and equities.
Application for Admission
Application will be made to London Stock Exchange plc (the
"London Stock Exchange") for Admission.
It is expected that Admission of the Placing Shares will occur
at or before 8.00 a.m. on 19 March 2021 (or such later time and/or
date as the Joint Bookrunners may agree with the Company) and that
dealings in the Placing Shares on AIM will commence at that
time.
Bookbuild
The Joint Bookrunners will today commence the Bookbuild to
determine demand for Placing Shares by Placees. This Announcement
gives details of the terms and conditions of, and the mechanics of
participation in, the Placing. No commissions will be paid to
Placees or by Placees in respect of any Placing Shares.
The Joint Bookrunners shall be entitled to effect the Placing by
such alternative method to the Bookbuild as they may, in their
absolute discretion following consultation with the Company,
determine.
Participation in, and principal terms of, the Placing
1. The Joint Bookrunners are arranging the Placing severally,
and not jointly, or jointly and severally, as joint bookrunners,
brokers and placing agents of the Company. Participation in the
Placing will only be available to persons who may lawfully be, and
are, invited to participate by either of the Joint Bookrunners.
Each of the Joint Bookrunners may itself agree to be a Placee in
respect of all or some of the Placing Shares or may nominate any
member of its group to do so.
2. The number of Placing Shares will be agreed by the Joint
Bookrunners (in consultation with the Company) following completion
of the Bookbuild. The number of Placing Shares to be issued will be
announced on an RIS following the completion of the Bookbuild via
the Results Announcement.
3. To participate in the Bookbuild, prospective Placees should
communicate their bid orally by telephone or in writing to their
usual sales contact at the relevant Joint Bookrunner. Each bid
should state the number of Placing Shares which the prospective
Placee wishes to subscribe for at the Issue Price. The Joint
Bookrunners reserve the right not to accept bids or to accept bids
in part rather than in whole. The acceptance of the bids will be at
the Joint Bookrunners' absolute discretion, subject to agreement
with the Company.
4. The Bookbuild is expected to close no later than 4.30 p.m. on
the date of this Announcement but may be closed earlier or later at
the sole discretion of the Joint Bookrunners. The Joint Bookrunners
may, in agreement with the Company, accept bids that are received
after the Bookbuild has closed. The Company reserves the right
(upon the agreement of the Joint Bookrunners) to reduce the number
of Placing Shares to be issued pursuant to the Placing, in its
absolute discretion.
5. Allocations of the Placing Shares will be determined by the
Joint Bookrunners after consultation with the Company (the proposed
allocations having been supplied by the Joint Bookrunners to the
Company in advance of such consultation). Allocations in respect of
the Placing Shares will be confirmed orally by the Joint
Bookrunners and a contract note will be despatched as soon as
possible thereafter. A Joint Bookrunner's oral confirmation to such
Placee constitutes an irrevocable legally binding commitment upon
such person (who will at that point become a Placee), in favour of
the Joint Bookrunners and the Company, on the terms and conditions
set out in this Appendix and in accordance with the Company's
articles of association to subscribe for such number of Placing
Shares as are confirmed by the Joint Bookrunners and to pay in
cleared funds an amount equal to the product of the Issue Price and
the number of Placing Shares for which such Placee has agreed to
subscribe. Except with the relevant Joint Bookrunner's consent,
such commitment will not be capable of variation or revocation
after the time at which it is submitted.
6. As set out above, each Placee's allocation and commitment
will be evidenced by a contract note issued to such Placee by the
relevant Joint Bookrunner. The terms of this Appendix will be
deemed incorporated in that contract note.
7. Irrespective of the time at which a Placee's allocation
pursuant to the Placing is confirmed, settlement for all Placing
Shares to be subscribed for pursuant to the Placing will be
required to be made at the same time, on the basis explained below
under 'Registration and Settlement'.
8. All obligations under the Bookbuild and/or the Placing will
be subject to fulfilment or (where applicable) waiver of the
conditions referred to below under 'Conditions of the Placing' and
to the Placing not being terminated on the basis referred to below
under 'Right to terminate under the Placing and Open Offer
Agreement'.
9. By participating in the Placing, each Placee agrees that its
rights and obligations in respect of the Placing will terminate
only in the circumstances described below and will not be capable
of rescission or termination by the Placee.
10. To the fullest extent permissible by law and the applicable
rules of the FCA, neither the Joint Bookrunners, nor the Company,
nor any of their respective affiliates, agents, directors, officers
or employees shall have any responsibility or liability to Placees
(or to any other person whether acting on behalf of a Placee or
otherwise). In particular, none of the Joint Bookrunners, nor the
Company, nor any of their respective affiliates, agents, directors,
officers or employees shall have any responsibility or liability
(including to the extent permissible by law, any fiduciary duties)
in respect of the Joint Bookrunners' conduct of the Placing or of
such alternative method of effecting the Placing as the Joint
Bookrunners and the Company may determine.
11. The Placing Shares will be issued subject to the terms and
conditions of this Announcement and each Placee's commitment to
subscribe for Placing Shares on the terms set out herein will
continue notwithstanding any amendment that may in future be made
to the terms and conditions of the Placing and Placees will have no
right to be consulted or require that their consent be obtained
with respect to the Company's or the Joint Bookrunners' conduct of
the Placing.
12. All times and dates in this Announcement may be subject to
amendment. The Joint Bookrunners shall notify the Placees and any
person acting on behalf of the Placees of any changes.
Conditions of the Placing
The Placing is conditional upon the Placing and Open Offer
Agreement becoming unconditional and not having been terminated in
accordance with its terms. The Joint Bookrunners' obligations under
the Placing and Open Offer Agreement are conditional on customary
conditions including, inter alia, (the "Conditions"):
1. the passing (without amendment) of the resolutions required
to complete the Placing, Subscription and Open Offer to be set out
in the Notice of General Meeting;
2. the Company having complied with all its obligations and
having satisfied all conditions under the Placing and Open Offer
Agreement or under the terms or conditions of the Placing and Open
Offer, which fall to be performed or satisfied on or prior to
Admission;
3. the warranties contained in the Placing and Open Offer
Agreement, being true, accurate and not misleading at the date of
the Placing and Open Offer Agreement (and remaining true, accurate
and not misleading at any time between such date and Admission) by
reference to the facts then subsisting; and
4. Admission occurring no later than 8.00 a.m. on 19 March 2021
(or such later time and/or date, not being later than 8.00 a.m. on
31 March 2021, as the Joint Bookrunners may otherwise agree with
the Company).
The Joint Bookrunners (if they both agree) may, at their
absolute discretion and upon such terms as they think fit, waive
compliance by the Company with the whole or any part of certain of
the Company's obligations in relation to the Conditions or extend
the time or date provided for fulfilment of certain such Conditions
in respect of all or any part of the performance thereof. Certain
Conditions including, inter alia, Admission taking place, may not
be waived. Any such extension or waiver will not affect Placees'
commitments as set out in this Announcement.
If: (i) any of the Conditions are not fulfilled or (where
permitted) waived by the Joint Bookrunners by the relevant time or
date specified (or such later time or date as the Company and the
Joint Bookrunners may agree); or (ii) the Placing and Open Offer
Agreement is terminated in the circumstances specified below under
'Right to terminate under the Placing and Open Offer Agreement',
the Placing will not proceed and the Placees' rights and
obligations hereunder in relation to the Placing Shares shall cease
and terminate at such time and each Placee agrees that no claim can
be made by it or on its behalf (or any person on whose behalf the
Placee is acting) in respect thereof.
Neither of the Joint Bookrunners, nor the Company, nor any of
their respective affiliates, agents, directors, officers or
employees shall have any liability to any Placee (or to any other
person whether acting on behalf of a Placee or otherwise) in
respect of any decision they may make as to whether or not to waive
or to extend the time and/or date for the satisfaction of any
Condition to the Placing, nor for any decision they may make as to
the satisfaction of any Condition or in respect of the Placing
generally, and by participating in the Placing each Placee agrees
that any such decision is within the absolute discretion of the
Joint Bookrunners.
Right to terminate under the Placing and Open Offer
Agreement
Each of the Joint Bookrunners is entitled, at any time on or
before Admission, to terminate its obligations under the Placing
and Open Offer Agreement in accordance with its terms in certain
circumstances, including, inter alia, if at any time on or before
Admission:
1. in the opinion of the Joint Bookrunners (acting in good
faith), the Company has breached any of its obligations under the
Placing and Open Offer Agreement; or
2. in the opinion of the Joint Bookrunners (acting in good
faith), any of the warranties contained in the Placing and Open
Offer Agreement has become untrue, inaccurate or misleading (or
would be untrue, inaccurate or misleading if repeated at any time
up to Admission) by reference to the facts and circumstances then
existing; or
3. there has occurred, in the opinion of the Joint Bookrunners
(acting in good faith), a material adverse effect (whether or not
foreseeable at the date of the Placing and Open Offer Agreement);
or
4. the occurrence, in the opinion of the Joint Bookrunners
(acting in good faith), of certain force majeure events (including
material deterioration in, or material escalation in the response
to, the Covid-19 pandemic), which would, inter alia, in the opinion
of the Joint Bookrunners (acting in good faith), be likely to
prejudice the success of the Fundraising and/or Admission, or make
it impractical to proceed with the Placing and/or Open Offer and/or
Admission.
Upon termination, such terminating Joint Bookrunner shall be
released and discharged (except for any liability arising before or
in relation to such termination) from their respective obligations
under or pursuant to the Placing and Open Offer Agreement, subject
to certain exceptions. If both Joint Bookrunners terminate their
obligations under the Placing and Open Offer Agreement, then the
Placing and Open Offer Agreement shall cease and terminate and the
Placing will not proceed.
By participating in the Placing, each Placee agrees that (i) the
exercise by either of the Joint Bookrunners of any right of
termination or of any other discretion under the Placing and Open
Offer Agreement shall be within the absolute discretion of such
Joint Bookrunner and that it need not make any reference to, or
consult with, Placees and that it shall have no liability to
Placees whatsoever in connection with any such exercise or failure
to so exercise and (ii) its rights and obligations terminate only
in the circumstances described above under 'Right to terminate
under the Placing and Open Offer Agreement' and 'Conditions of the
Placing', and its participation will not be capable of rescission
or termination by it after oral confirmation by the Joint
Bookrunners of the allocation and commitments following the close
of the Bookbuild.
Lock-up Arrangements
The Company has undertaken to the Joint Bookrunners that,
between the date of the Placing Agreement and 90 days after
Admission, it will not, without the prior written consent from the
Joint Bookrunners, directly or indirectly, offer, issue, lend, sell
or contract to sell, issue options in respect of or otherwise
dispose of, or announce an offer or issue of any shares of the
Company (or any interest therein or in respect thereof) or any
other securities exchangeable or convertible into, or substantially
similar to, shares of the Company, or enter into any transaction
with the same economic effect as, or agree to do, any of the
foregoing. However, this undertaking shall not prevent or restrict
the grant or exercise of options or other rights related to shares
of the Company and/or the issue of shares of the Company pursuant
to the exercise of options, in each case under employee share
incentive schemes where such schemes are in existence on the date
of Admission.
By participating in the Placing, Placees agree that the exercise
by any Joint Bookrunner of any power to grant consent to the
undertaking by the Company of a transaction which would otherwise
be subject to the lock-up provisions under the Placing Agreement
shall be within the absolute discretion of that Joint Bookrunner
and that it need not make any reference to, or consult with,
Placees and that it shall have no liability to Placees whatsoever
in connection with any such exercise of the power to grant
consent.
Placing Procedure
Placees shall acquire the Placing Shares to be issued pursuant
to the Placing and any allocation of Placing Shares will be
notified to them on or around the date of this Announcement (or
such other time and/or date as the Company and the Joint
Bookrunners may agree).
Payment in full for any Placing Shares so allocated in respect
of the Placing at the Issue Price must be made by no later than
11.00 am on 19 March 2021 (or such other date as shall be notified
to each Placee by the Joint Bookrunners). The Joint Bookrunners
will notify Placees if any of the dates in these terms and
conditions should change, including as a result of delay in
Admission or otherwise.
Registration and Settlement
Settlement of transactions in the Placing Shares (ISIN:
GB00BYV81293) following Admission will take place within CREST,
subject to certain exceptions. The Joint Bookrunners reserve the
right to require settlement for, and delivery of, the Placing
Shares (or any part thereof) to Placees by such other means that
they may deem necessary if delivery or settlement is not possible
or practicable within the CREST system or would not be consistent
with the regulatory requirements in the Placee's jurisdiction.
Following the close of the Bookbuild, each Placee to be
allocated Placing Shares in the Placing will be sent a contract
note in accordance with the standing arrangements in place with the
relevant Joint Bookrunner stating the number of Placing Shares
allocated to them at the Issue Price, the aggregate amount owed by
such Placee to the Joint Bookrunner and settlement instructions.
Each Placee agrees that it will do all things necessary to ensure
that delivery and payment is completed in accordance with the
standing CREST or certificated settlement instructions in respect
of the Placing Shares that it has in place with the relevant Joint
Bookrunner.
The Company will deliver the Placing Shares to a CREST account
operated by the relevant Joint Bookrunner as agent for the Company
and the relevant Joint Bookrunner will enter its delivery
instruction into the CREST system. The input to CREST by a Placee
of a matching or acceptance instruction will then allow delivery of
the relevant Placing Shares to that Placee against payment.
It is anticipated that settlement in respect of the Placing
Shares will take place on 19 March 2021 on a delivery versus
payment basis.
Interest is chargeable daily on payments not received from
Placees on the due date in accordance with the arrangements set out
above at the rate of two percentage points above LIBOR as
determined by the Joint Bookrunners.
Each Placee is deemed to agree that, if it does not comply with
these obligations, the relevant Joint Bookrunner may sell any or
all of the Placing Shares allocated to that Placee on such Placee's
behalf and retain from the proceeds, for the Joint Bookrunners'
account and benefit, an amount equal to the aggregate amount owed
by the Placee plus any interest due. The relevant Placee will,
however, remain liable for any shortfall below the aggregate amount
owed by it and will be required to bear any stamp duty or stamp
duty reserve tax or other taxes or duties (together with any
interest or penalties) imposed in any jurisdiction which may arise
upon the sale of such Placing Shares on such Placee's behalf.
If Placing Shares are to be delivered to a custodian or
settlement agent, Placees should ensure that the contract note is
copied and delivered immediately to the relevant person within that
organisation. Insofar as Placing Shares are issued in a Placee's
name or that of its nominee or in the name of any person for whom a
Placee is contracting as agent or that of a nominee for such
person, such Placing Shares should, subject as provided below, be
so registered free from any liability to stamp duty or stamp duty
reserve tax. If there are any circumstances in which any stamp duty
or stamp duty reserve tax or other similar taxes or duties
(including any interest and penalties relating thereto) is payable
in respect of the allocation, allotment, issue, sale, transfer or
delivery of the Placing Shares (or, for the avoidance of doubt, if
any stamp duty or stamp duty reserve tax is payable in connection
with any subsequent transfer of or agreement to transfer Placing
Shares), none of the Joint Bookrunners nor the Company shall be
responsible for payment thereof.
Representations, warranties, undertakings and
acknowledgements
By participating in the Placing each Placee (and any person
acting on such Placee's behalf) irrevocably acknowledges, confirms,
undertakes, represents, warrants and agrees (as the case may be)
with the Joint Bookrunners (in their capacity as Joint Bookrunners
and placing agents of the Company in respect of the Placing) and
the Company, in each case as a fundamental term of their
application for Placing Shares, the following:
General
1. it has read and understood this Announcement in its entirety
and its subscription for Placing Shares is subject to and based
upon all the terms, conditions, representations, warranties,
acknowledgements, agreements and undertakings and other information
contained herein and it has not relied on, and will not rely on,
any information given or any representations, warranties or
statements made at any time by any person in connection with the
Placing, the Company, the Placing Shares, Admission or otherwise
other than the information contained in the Placing Documents and
the Publicly Available Information;
2. the person whom it specifies for registration as holder of
the Placing Shares will be (a) itself or (b) its nominee, as the
case may be. None of the Joint Bookrunners nor the Company will be
responsible for any liability to stamp duty or stamp duty reserve
tax or other similar taxes or duties imposed in any jurisdiction
(including interest and penalties relating thereto) ("Indemnified
Taxes"). Each Placee and any person acting on behalf of such Placee
agrees to indemnify the Company and the Joint Bookrunners on an
after-tax basis in respect of any Indemnified Taxes;
3. neither the Joint Bookrunners nor any of their respective
affiliates, agents, directors, officers and employees accepts any
responsibility for any acts or omissions of the Company or any of
the directors of the Company or any other person (other than the
relevant Joint Bookrunner) in connection with the Placing;
4. time is of the essence as regards its obligations under this Announcement;
5. any document that is to be sent to it in connection with the
Placing will be sent at its risk and may be sent to it at any
address provided by it to the Joint Bookrunners;
No distribution of Announcement
6. it will not redistribute, forward, transfer, duplicate or
otherwise transmit this Announcement or any part of it, or any
other presentational or other material concerning the Placing
(including electronic copies thereof) to any person and represents
that it has not redistributed, forwarded, transferred, duplicated,
or otherwise transmitted any such materials to any person;
No prospectus
7. no prospectus or other offering document is required under
the UK Prospectus Regulation or the EU Prospectus Regulation, nor
will one be prepared in connection with the Bookbuild, the Placing
or the Placing Shares and it has not received and will not receive
a prospectus or other offering document in connection with the
Bookbuild, the Placing or the Placing Shares;
Purchases by Joint Bookrunners for their own account
8. in connection with the Placing, the Joint Bookrunners and any
of their affiliates acting as an investor for its own account may
subscribe for Placing Shares in the Company and in that capacity
may retain, purchase or sell for its own account such Placing
Shares in the Company and any securities of the Company or related
investments and may offer or sell such securities or other
investments otherwise than in connection with the Placing.
Accordingly, references in this Announcement to the Placing Shares
being issued, offered or placed should be read as including any
issue, offering or placement of such shares in the Company to each
of the Joint Bookrunners or any of their affiliates acting in such
capacity;
9. each of the Joint Bookrunners and their affiliates may enter
into financing arrangements and swaps with investors in connection
with which each of the Joint Bookrunners and any of their
affiliates may from time to time acquire, hold or dispose of such
securities of the Company, including the Placing Shares;
10. the Joint Bookrunners do not intend to disclose the extent
of any investment or transactions referred to in paragraphs 8 and 9
above otherwise than in accordance with any legal or regulatory
obligation to do so;
No fiduciary duty or client of the Joint Bookrunners
11. the Joint Bookrunners do not owe any fiduciary or other
duties to any Placee in respect of any representations, warranties,
undertakings or indemnities in the Placing and Open Offer
Agreement;
12. its participation in the Placing is on the basis that it is
not and will not be a client of any of the Joint Bookrunners in
connection with its participation in the Placing and that the Joint
Bookrunners have no duties or responsibilities to it for providing
the protections afforded to their respective clients or customers
or for providing advice in relation to the Placing nor in respect
of any representations, warranties, undertakings or indemnities
contained in the Placing and Open Offer Agreement nor for the
exercise or performance of any of their respective rights and
obligations thereunder including any rights to waive or vary any
conditions or exercise any termination right;
No responsibility of the Joint Bookrunners for information
13. the content of this Announcement and the Publicly Available
Information has been prepared by and is exclusively the
responsibility of the Company and neither Joint Bookrunner nor
their respective affiliates, agents, directors, officers or
employees nor any person acting on behalf of any of them is
responsible for or has or shall have any responsibility or
liability for any information, representation or statement
contained in, or omission from, this Announcement, the Publicly
Available Information or otherwise nor will they be liable for any
Placee's decision to participate in the Placing based on any
information, representation, warranty or statement contained in
this Announcement, the Publicly Available Information or otherwise,
provided that nothing in this paragraph excludes the liability of
any person for fraudulent misrepresentation made by such
person;
Reliance on information regarding the Placing
14.
(a) the only information on which it is entitled to rely and on
which such Placee has relied in committing itself to subscribe for
Placing Shares is contained in this Announcement, or any Publicly
Available Information (save that in the case of Publicly Available
Information, a Placee's right to rely on that information is
limited to the right that such Placee would have as a matter of law
in the absence of this paragraph 16(a)), such information being all
that such Placee deems necessary or appropriate and sufficient to
make an investment decision in respect of the Placing Shares;
(b) it has neither received nor relied on any other information
given, or representations, warranties or statements, express or
implied, made, by any of the Joint Bookrunners or the Company nor
any of their respective affiliates, agents, directors, officers or
employees acting on behalf of any of them (including in any
management presentation delivered in respect of the Bookbuild) with
respect to the Company, the Placing or the Placing Shares or the
accuracy, completeness or adequacy of any information contained in
this Announcement, or the Publicly Available Information or
otherwise;
(c) none of the Joint Bookrunners, nor the Company, nor any of
their respective affiliates, agents, directors, officers or
employees or any person acting on behalf of any of them has
provided, nor will provide, it with any material or information
regarding the Placing Shares or the Company or any other person
other than the information in the Placing Documents or the Publicly
Available Information; nor has it requested any of the Joint
Bookrunners, the Company, any of their respective affiliates or any
person acting on behalf of any of them to provide it with any such
material or information; and
(d) none of the Joint Bookrunners or the Company will be liable
for any Placee's decision to participate in the Placing based on
any other information, representation, warranty or statement,
provided that nothing in this paragraph 16 excludes the
liability of any person for fraudulent misrepresentation made by
that person;
Conducted own investigation and due diligence
15. it may not rely, and has not relied, on any investigation
that the Joint Bookrunners, any of their affiliates or any person
acting on their behalf, may have conducted with respect to the
Placing Shares, the terms of the Placing or the Company, and none
of such persons has made any representation, express or implied,
with respect to the Company, the Placing, the Placing Shares or the
accuracy, completeness or adequacy of the information in this
Announcement, the Publicly Available Information or any other
information;
16. in making any decision to subscribe for Placing Shares it:
(a) has such knowledge and experience in financial and business
matters to be capable of evaluating the merits and risks of
subscribing for the Placing Shares;
(b) will not look to the Joint Bookrunners for all or part of
any such loss it may suffer;
(c) is experienced in investing in securities of this nature in
this sector and is aware that it may be required to bear, and is
able to bear, the economic risk of an investment in the Placing
Shares;
(d) is able to sustain a complete loss of an investment in the Placing Shares;
(e) has no need for liquidity with respect to its investment in the Placing Shares;
(f) has made its own assessment and has satisfied itself
concerning the relevant tax, legal, currency and other economic
considerations relevant to its investment in the Placing Shares;
and
(g) has conducted its own due diligence, examination,
investigation and assessment of the Company, the Placing Shares and
the terms of the Placing and has satisfied itself that the
information resulting from such investigation is still current and
relied on that investigation for the purposes of its decision to
participate in the Placing;
17. the Ordinary Shares are admitted to trading on AIM, and that
the Company is therefore required to publish certain business and
financial information in accordance with the AIM Rules, which
includes a description of the nature of the Company's business and
the Company's most recent balance sheet and profit and loss account
and that it is able to obtain or access such information without
undue difficulty, and is able to obtain access to such information
or comparable information concerning any other publicly traded
company, without undue difficulty;
Capacity and authority
18. it is subscribing for the Placing Shares for its own account
or for an account with respect to which it exercises sole
investment discretion and has the authority to make and does make
the acknowledgements, representations and agreements contained in
this Announcement;
19. it is acting as principal only in respect of the Placing or,
if it is acting for any other person, it:
(a) is duly authorised to do so and has full power to make the
acknowledgments, representations and agreements herein on behalf of
each such person; and
(b) will remain liable to the Company and/or the Joint
Bookrunners for the performance of all its obligations as a Placee
in respect of the Placing (regardless of the fact that it is acting
for another person);
20. it and any person acting on its behalf is entitled to
subscribe for the Placing Shares under the laws and regulations of
all relevant jurisdictions that apply to it and that it has fully
observed such laws and regulations, has capacity and authority and
is entitled to enter into and perform its obligations as a
subscriber of Placing Shares and will honour such obligations, and
has obtained all such governmental and other guarantees, permits,
authorisations, approvals and consents which may be required
thereunder and complied with all necessary formalities to enable it
to commit to this participation in the Placing and to perform its
obligations in relation thereto (including, without limitation, in
the case of any person on whose behalf it is acting, all necessary
consents and authorities to agree to the terms set out or referred
to in this Announcement) and will honour such obligations and that
it has not taken any action or omitted to take any action which
will or may result in the Joint Bookrunners, the Company or any of
their respective directors, officers, agents, employees or advisers
acting in breach of the legal or regulatory requirements of any
jurisdiction in connection with the Placing;
21. where it is subscribing for Placing Shares for one or more
managed accounts, it is authorised in writing by each managed
account to subscribe for the Placing Shares for each managed
account;
22. it irrevocably appoints any duly authorised officer of each
Joint Bookrunner as its agent for the purpose of executing and
delivering to the Company and/or its registrars any documents on
its behalf necessary to enable it to be registered as the holder of
any of the Placing Shares for which it agrees to subscribe upon the
terms of this Announcement;
Excluded territories
23. the Placing Shares have not been and will not be registered
or otherwise qualified and that a prospectus will not be cleared in
respect of any of the Placing Shares under the securities laws or
legislation of the United States, Australia, Canada, Japan or the
Republic of South Africa, or any state, province, territory or
jurisdiction thereof;
24. the Placing Shares may not be offered, sold, or delivered or
transferred, directly or indirectly, in or into the above
jurisdictions or any jurisdiction in which it would be unlawful to
do so and no action has been or will be taken by any of the
Company, the Joint Bookrunners or any person acting on behalf of
the Company or the Joint Bookrunners that would, or is intended to,
permit a public offer of the Placing Shares in the United States,
Australia, Canada, Japan, or the Republic of South Africa or any
country or jurisdiction, or any state, province, territory or
jurisdiction thereof, where any such action for that purpose is
required;
25. unless otherwise specifically agreed with the Joint
Bookrunners, it is not and at the time the Placing Shares are
subscribed for, neither it nor the beneficial owner of the Placing
Shares will be, a resident of, nor have an address in, Australia,
Japan, the Republic of South Africa or any province or territory of
Canada;
26. it may be asked to disclose in writing or orally to the Joint Bookrunners:
(a) if he or she is an individual, his or her nationality; or
(b) if he or she is a discretionary fund manager, the
jurisdiction in which the funds are managed or owned;
Compliance with US securities laws
27. it, and any prospective beneficial owner for whose account
or benefit it is purchasing the Placing Shares, is (i) located
outside the United States and is acquiring the Placing Shares in an
"offshore transaction" as defined in, and in accordance with,
Regulation S; (ii) has not been offered to purchase or subscribe
for Placing Shares by means of any "directed selling efforts" as
defined in Regulation S;
28. it understands that the Placing Shares have not been, and
will not be, registered under the US Securities Act and may not be
offered, sold or resold in or into or from the United States except
pursuant to an effective registration under the US Securities Act,
or pursuant to an exemption from the registration requirements of
the US Securities Act and in accordance with applicable state
securities laws;
29. it will not distribute, forward, transfer or otherwise
transmit this Announcement or any part of it, or any other
presentational or other materials concerning the Placing in or into
or from the United States (including electronic copies thereof) to
any person, and it has not distributed, forwarded, transferred or
otherwise transmitted any such materials to any person;
Compliance with selling restrictions and the EU Prospectus
Regulation and UK Prospectus Regulation
30. if in the United Kingdom, it is a Relevant Person and it is
a Qualified Investor (as such term is defined in Article 2(e) of
the UK Prospectus Regulation);
31. if in Relevant Member State, it is a Relevant Person and it
is a Qualified Investor (as such term is defined in the EU
Prospectus Regulation);
32. it has not offered or sold and will not offer or sell any
Placing Shares to persons in the United Kingdom except to Qualified
Investors (as such term is defined in Article 2(e) of the UK
Prospectus Regulation) or otherwise in circumstances which have not
resulted in and which will not result in an offer to the public in
the United Kingdom within the meaning of the UK Prospectus
Regulation;
33. it has not offered or sold and will not offer or sell any
Placing Shares to persons in a Relevant Member State except to
Qualified Investors (as such term is defined in Article 2(e) of the
EU Prospectus Regulation) or otherwise in circumstances which have
not resulted in and which will not result in an offer to the public
in a Relevant Member State within the meaning of the EU Prospectus
Regulation;
34. if a financial intermediary, as that term is used in Article
5(1) of the UK Prospectus Regulation, the Placing Shares subscribed
for by it in the Placing will not be acquired on a
non-discretionary basis on behalf of, nor will they be acquired
with a view to their offer or resale to, persons in the United
Kingdom other than Qualified Investors;
35. if a financial intermediary, as that term is used in Article
5(1) of the EU Prospectus Regulation, the Placing Shares subscribed
for by it in the Placing will not be acquired on a
non-discretionary basis on behalf of, nor will they be acquired
with a view to their offer or resale to, persons in a Relevant
Member State other than Qualified Investors;
Compliance with FSMA, the UK financial promotion regime and UK
MAR
36. if in the United Kingdom, that it is a person (i) having
professional experience in matters relating to investments who
falls within the definition of "investment professionals" in
Article 19(5) of the Order or (ii) who falls within Article 49(2)
(a) to (d) ("High Net Worth Companies, Unincorporated Associations,
etc") of the Order, or (iii) to whom it may otherwise lawfully be
communicated;
37. it has not offered or sold and will not offer or sell any
Placing Shares to persons in the United Kingdom, except to persons
whose ordinary activities involve them in acquiring, holding,
managing or disposing of investments (as principal or agent) for
the purposes of their business or otherwise in circumstances which
have not resulted and which will not result in an offer to the
public in the United Kingdom within the meaning of section 85(1) of
FSMA;
38. it has only communicated or caused to be communicated and
will only communicate or cause to be communicated any invitation or
inducement to engage in investment activity (within the meaning of
section 21 of FSMA) relating to the Placing Shares in circumstances
in which section 21(1) of FSMA does not require approval of the
communication by an authorised person and it acknowledges and
agrees that the Placing Documents have not and will not have been
approved by either Joint Bookrunner in its capacity as an
authorised person under section 21 of the FSMA and it may not
therefore be subject to the controls which would apply if it was
made or approved as a financial promotion by an authorised
person;
39. it has complied and will comply with all applicable laws
with respect to anything done by it or on its behalf in relation to
the Placing Shares (including all applicable provisions in FSMA and
Regulation (EU) No. 596/2014 of the European Parliament and of the
Council of 16 April 2014 on market abuse, as it forms part of UK
domestic law by virtue of the European Union (Withdrawal) Act 2018,
("UK MAR")) in respect of anything done in, from or otherwise
involving, the United Kingdom);
Compliance with laws
40. if it is a pension fund or investment company, its
subscription for Placing Shares is in full compliance with
applicable laws and regulations;
41. it is not a (i) a person named on the Consolidated List of
Financial Sanctions Targets maintained by HM Treasury of the United
Kingdom; or (ii) a person subject to financial sanctions imposed
pursuant to a regulation of the European Union or a regulation
adopted by the United Nations;
42. it has complied with its obligations under the Criminal
Justice Act 1993 and Articles 8, 10 and 12 of UK MAR and in
connection with money laundering and terrorist financing under the
Proceeds of Crime Act 2002 (as amended), the Terrorism Act 2000 (as
amended), the Terrorism Act 2006 and the Money Laundering,
Terrorist Financing and Transfer of Funds (Information on the
Payer) Regulations 2017 (as amended) and any related or similar
rules, regulations or guidelines, issued, administered or enforced
by any government agency having jurisdiction in respect thereof
(the "Regulations") and, if making payment on behalf of a third
party, that satisfactory evidence has been obtained and recorded by
it to verify the identity of the third party as required by the
Regulations;
43. in order to ensure compliance with the Regulations, each
Joint Bookrunner (for itself and as agent on behalf of the Company)
or the Company's registrars may, in their absolute discretion,
require verification of its identity. Pending the provision to the
relevant Joint Bookrunner or the Company's registrars, as
applicable, of evidence of identity, definitive certificates in
respect of the Placing Shares may be retained at the relevant Joint
Bookrunner's absolute discretion or, where appropriate, delivery of
the Placing Shares to it in uncertificated form may be delayed at
the relevant Joint Bookrunner's or the Company's registrars', as
the case may be, absolute discretion. If within a reasonable time
after a request for verification of identify the relevant Joint
Bookrunner (for itself and as agent on behalf of the Company) or
the Company's registrars have not received evidence satisfactory to
them, either the relevant Joint Bookrunner and/or the Company may,
at its absolute discretion, terminate its commitment in respect of
the Placing, in which event the monies payable on acceptance of
allotment will, if already paid, be returned without interest to
the account of the drawee's bank from which they were originally
debited;
Depositary receipts and clearance services
44. the allocation, allotment, issue and delivery to it, or the
person specified by it for registration as holder, of Placing
Shares will not give rise to a stamp duty or stamp duty reserve tax
liability under (or at a rate determined under) any of sections 67,
70, 93 or 96 of the Finance Act 1986 (depositary receipts and
clearance services) and that the Placing Shares are not being
acquired in connection with arrangements to issue depositary
receipts or to issue or transfer Placing Shares into a clearance
service;
Undertaking to make payment
45. it (and any person acting on its behalf) has the funds
available to pay for the Placing Shares for which it has agreed to
subscribe and acknowledges and agrees that it will make payment in
respect of the Placing Shares allocated to it in accordance with
this Announcement on the due time and date set out herein, failing
which the relevant Placing Shares may be placed with other
subscribers or sold as the Joint Bookrunners may in their sole
discretion determine and without liability to such Placee, who will
remain liable for any amount by which the net proceeds of such sale
falls short of the product of the relevant Issue Price and the
number of Placing Shares allocated to it and will be required to
bear any stamp duty, stamp duty reserve tax or other taxes or
duties (together with any interest, fines or penalties) imposed in
any jurisdiction which may arise upon the sale of such Placee's
Placing Shares;
Money held on account
46. any money held in an account with the relevant Joint
Bookrunners on behalf of the Placee and/or any person acting on
behalf of the Placee and/or any person acting on behalf of the
Placee will not be treated as client money within the meaning of
the relevant rules and regulations of the FCA made under the FSMA.
Each Placee acknowledges that the money will not be subject to the
protections conferred by the client money rules: as a consequence
this money will not be segregated from the relevant Joint
Bookrunner's money in accordance with the client money rules and
will be held by it under a banking relationship and not as
trustee;
Allocation
47. its allocation (if any) of Placing Shares will represent a
maximum number of Placing Shares to which it will be entitled, and
required, to subscribe for, and that the Joint Bookrunners or the
Company may call upon it to subscribe for a lower number of Placing
Shares but in no event in aggregate more than the aforementioned
maximum;
No recommendation
48. none of the Joint Bookrunners, nor any of their respective
affiliates, nor any person acting on behalf of them, is making any
recommendations to it or advising it regarding the suitability of
any transactions it may enter into in connection with the
Placing;
Inside information
49. if it has received any 'inside information' (for the
purposes of UK MAR and section 56 of the Criminal Justice Act 1993)
in relation to the Company and its securities in advance of the
Placing, it confirms that it has received such information within
the market soundings regime provided for in article 11 of UK MAR
and associated delegated regulations and it has not:
(a) used that inside information to acquire or dispose of
securities of the Company or financial instruments related thereto
or cancel or amend an order concerning the Company's securities or
any such financial instruments;
(b) used that inside information to encourage, require,
recommend or induce another person to deal in the securities of the
Company or financial instruments related thereto or to cancel or
amend an order concerning the Company's securities or such
financial instruments; or
(c) disclosed such information to any person, prior to the
information being made publicly available;
Rights and remedies
50. the rights and remedies of the Company and the Joint
Bookrunners under the terms and conditions in this Announcement are
in addition to any rights and remedies which would otherwise be
available to each of them and the exercise or partial exercise of
one will not prevent the exercise of others; and
Governing law and jurisdiction
51. these terms and conditions of the Placing and any agreements
entered into by it pursuant to the terms and conditions of the
Placing, and all non-contractual or other obligations arising out
of or in connection with them, shall be governed by and construed
in accordance with the laws of England and Wales, and it submits
(on behalf of itself and on behalf of any person on whose behalf it
is acting) to the exclusive jurisdiction of the English courts as
regards any claim, dispute or matter arising out of any such
contract (including any dispute regarding the existence, validity
or termination of such contract or relating to any non-contractual
or other obligation arising out of or in connection with such
contract), except that enforcement proceedings in respect of the
obligation to make payment for the Placing Shares (together with
any interest chargeable thereon) may be taken by either the Company
or the Joint Bookrunners in any jurisdiction in which the relevant
Placee is incorporated or in which any of its securities have a
quotation on a recognised stock exchange.
The foregoing representations, warranties, confirmations,
acknowledgements, agreements and undertakings are given for the
benefit of the Company as well as each of the Joint Bookrunners and
are irrevocable. The Joint Bookrunners, the Company and their
respective affiliates and others will rely upon the truth and
accuracy of the foregoing representations, warranties,
confirmations, acknowledgements, agreements and undertakings. Each
prospective Placee, and any person acting on behalf of such Placee,
irrevocably authorises the Company and the Joint Bookrunners to
produce this Announcement, pursuant to, in connection with, or as
may be required by any applicable law or regulation, administrative
or legal proceeding or official inquiry with respect to the matters
set forth herein.
Indemnity
By participating in the Placing, each Placee (and any person
acting on such Placee's behalf) agrees to indemnify on an after tax
basis and hold the Company, the Joint Bookrunners and their
respective affiliates, agents, directors, officers and employees
harmless from any and all costs, claims, liabilities and expenses
(including legal fees and expenses) arising out of or in connection
with any breach of the representations, warranties,
acknowledgements, agreements and undertakings given by the Placee
(and any person acting on such Placee's behalf) in this
Announcement or incurred by the Joint Bookrunners, the Company or
each of their respective affiliates, agents, directors, officers or
employees arising from the performance of the Placees' obligations
as set out in this Announcement, and further agrees that the
provisions of this Announcement shall survive after completion of
the Placing.
Taxation
The agreement to allot and issue Placing Shares to Placees
(and/or to persons for whom such Placee is contracting as agent)
free of stamp duty and stamp duty reserve tax relates only to their
allotment and issue to Placees, or such persons as they nominate as
their agents, direct from the Company for the Placing Shares in
question. Such agreement also assumes that the Placing Shares are
not being acquired in connection with arrangements to issue
depositary receipts or to issue or transfer the Placing Shares into
a clearance service. If there are any such arrangements, or the
settlement relates to any other dealing in the Placing Shares,
stamp duty or stamp duty reserve tax or other similar taxes or
duties may be payable, for which neither the Company nor the Joint
Bookrunners will be responsible and the Placees shall indemnify the
Company and the Joint Bookrunners on an after-tax basis for any
stamp duty or stamp duty reserve tax or other similar taxes or
duties (together with interest, fines and penalties) in any
jurisdiction paid by the Company or the Joint Bookrunners in
respect of any such arrangements or dealings. If this is the case,
each Placee should seek its own advice and notify the Joint
Bookrunners accordingly. Placees are advised to consult with their
own advisers regarding the tax aspects of the subscription for
Placing Shares.
The Company and the Joint Bookrunners are not liable to bear any
taxes that arise on a sale of Placing Shares subsequent to their
acquisition by Placees, including any taxes arising otherwise than
under the laws of the United Kingdom. Each prospective Placee
should, therefore, take its own advice as to whether any such tax
liability arises and notify the Joint Bookrunners and the Company
accordingly. Furthermore, each prospective Placee agrees to
indemnify on an after-tax basis and hold each of the Joint
Bookrunners and/or the Company and their respective affiliates
harmless from any and all interest, fines or penalties in relation
to stamp duty, stamp duty reserve tax and all other similar duties
or taxes in any jurisdiction to the extent that such interest,
fines or penalties arise from the unreasonable default or delay of
that Placee or its agent.
In addition, Placees should note that they will be liable for
any stamp duty and all other stamp, issue, securities, transfer,
registration, documentary or other duties or taxes (including any
interest, fines or penalties relating thereto) payable, whether
inside or outside the United Kingdom, by them or any other person
on the subscription, acquisition, transfer or sale by them of any
Placing Shares or the agreement by them to subscribe for, acquire,
transfer or sell any Placing Shares.
No statement in this Announcement is intended to be a profit
forecast or estimate, and no statement in this Announcement should
be interpreted to mean that earnings per share of the Company for
the current or future financial years would necessarily match or
exceed the historical published earnings per share of the Company.
Past performance is no guide to future performance and persons
needing advice should consult an independent financial adviser.
The price of shares and any income expected from them may go
down as well as up and investors may not get back the full amount
invested upon disposal of the shares. Past performance is no guide
to future performance, and persons needing advice should consult an
independent financial adviser.
The Placing Shares to be issued pursuant to the Placing will not
be admitted to trading on any stock exchange other than AIM.
Neither the content of the Company's website nor any website
accessible by hyperlinks on the Company's website is incorporated
in, or forms part of, this Announcement.
APPIX III - DEFINITIONS
The following definitions apply throughout this Announcement
unless the context otherwise requires:
"Admission" the admission of (i) the Placing Shares to be issued pursuant to
the Placing, (ii) the Open
Offer Shares to be issued pursuant to the Open Offer and (iii) the
Subscription Shares, to
trading on AIM becoming effective by means of the issue by London
Stock Exchange of a dealing
notice under Rule 6 of the AIM Rules;
"AIM" AIM, the market of that name operated by London Stock Exchange
"AIM Rules" the 'AIM Rules for Companies' and/or the AIM Rules for Nominated
Advisers (as the context
may require)
"AIM Rules for Companies" the rules of AIM as set out in the publication entitled "AIM Rules
for Companies" published
by the London Stock Exchange from time to time
"AIM Rules for Nominated Advisers" the rules of AIM as set out in the publication entitled "AIM Rules
for Nominated Advisers"
published by the London Stock Exchange from time to time
"AOP" AOP Orphan International AG
"Basic Entitlements" or "Open Offer Entitlements" the pro rata entitlement for Qualifying Shareholders to subscribe
for Open Offer Shares, pursuant
to the Open Offer as described in Part IV of the Circular
"Bookbuild" the accelerated bookbuilding process which will be launched
immediately following this Announcement
"certificated" or "in certificated form" an Ordinary Share or other security recorded on a company's share
register as being held in
certificated form (that is not in CREST)
"Circular" the circular to be posted to Shareholders shortly in relation to
the Fundraising and incorporating
the Notice of General Meeting
"Company" or "Shield" Shield Therapeutics plc, a public limited company incorporated in
England and Wales under
registered number 09761509
"CREST" the relevant system (as defined in the Regulations) which enables
title to units of relevant
securities (as defined in the Regulations) to be evidenced and
transferred without a written
instrument and in respect of which Euroclear is the Operator (as
defined in the CREST Regulations)
"CREST Regulations" the Uncertificated Securities Regulations 2001 (SI 2001/3755) as
amended
"EMA" European Medicines Agency
"Euroclear" Euroclear UK & Ireland Limited, the operator (as defined in the
CREST Regulations) of CREST
"Excess Application Facility" the arrangement pursuant to which Qualifying Shareholders may apply
for additional Open Offer
Shares in excess of the Basic Entitlement in accordance with the
terms and conditions of the
Open Offer
"Excess Entitlement(s)" Open Offer Shares in excess of the Basic Entitlement allocated to a
Qualifying Shareholder
pursuant to the Open Offer as described in Part IV of the Circular
"Existing Ordinary Shares" the Ordinary Shares in issue immediately prior to the Fundraising,
all of which are admitted
to trading on AIM
"FCA" the Financial Conduct Authority of the United Kingdom
"FDA" the US Food and Drug Administration
"finnCap" finnCap Ltd, the Company's joint bookrunner and broker in
connection with the Placing and
Open Offer
"Form of Proxy" the form of proxy for use by Shareholders in relation to the
General Meeting, either online
or a hard copy requested from Link Group
"FSMA" the Financial Services and Markets Act 2000, as amended
"Fundraising" together, the Placing, the Subscription and the Open Offer
"Fundraising Resolutions" the resolutions numbered 1 and 2 to be proposed at the General
Meeting as set out in the Notice
of General Meeting in the Circular
"General Meeting" the general meeting of the Shareholders to be convened by the
Notice of General Meeting
"Group" the Company, its subsidiaries and subsidiary undertakings
"IND application" investigational new drug application, part of the pharmaceutical
marketing regulatory approval
process
"Issue Price" 30 pence per New Share
"Joint Bookrunners" finnCap and Peel Hunt
"London Stock Exchange" London Stock Exchange plc
"New Shares" up to 97,279,730 new Ordinary Shares to be issued pursuant to the
Placing, the Subscription
and Open Offer
"NCE" New Chemical Entity status granted by the FDA
"Notice of General Meeting" the notice of the General Meeting which will be set out in the
Circular
"Open Offer" the conditional invitation to Qualifying Shareholders to apply to
subscribe for Open Offer
Shares at the Issue Price on the terms and subject to the
conditions to be set out or referred
to in the Circular
"Open Offer Shares" up to 13,946,397 New Shares to be offered to Qualifying
Shareholders pursuant to the Open
Offer whose allotment and issue is conditional (amongst other
things) on the passing of the
Fundraising Resolutions
"Ordinary Shares" ordinary shares of 1.5 pence each in the capital of the Company
"Overseas Shareholders" Shareholders with registered addresses in a Restricted Jurisdiction
or any other jurisdiction
where the extension or availability of the Open Offer would breach
any applicable law
"Peel Hunt" Peel Hunt LLP, the Company's nominated adviser and joint bookrunner
and broker in connection
with the Placing and Open Offer
"Placee" eligible institutional investors procured by the Joint Bookrunners
and subscribing for Placing
Shares in the Placing
"Placing" the conditional placing by Peel Hunt and finnCap (on behalf of the
Company) of 66,398,720
Placing Shares pursuant to the Placing and Open Offer Agreement to
raise approximately GBP19.9
million before expenses
"Placing and Open Offer Agreement" the placing and open offer agreement dated 26 February 2021 made
between the Company and the
Joint Bookrunners in relation to the Placing and Open Offer
"Placing Shares" up to 66,398,720 new Ordinary Shares to be issued to Placees by the
Company pursuant to the
Placing whose allotment and issue is conditional (amongst other
things) on the passing of
the Fundraising Resolutions
"Qualifying CREST Shareholders" Qualifying Shareholders holding Ordinary Shares in uncertificated
form at the Record Date
"Qualifying non-CREST Shareholders" Qualifying Shareholders holding Ordinary Shares in certificated
form at the Record Date
"Qualifying Shareholders" Shareholders at the Record Date other than Overseas Shareholders
"Record Date" 6.00 p.m. on 25 February 2021
"Regulatory Information Service" or "RIS" has the meaning given in the AIM Rules
"Relevant Persons" has the meaning set out in Appendix II of this Announcement
"Resolutions" the resolutions to be proposed at the General Meeting as set out in
the Notice of General
Meeting
"Restricted Jurisdictions" each and any of the United States, Australia, Canada, Japan and the
Republic of South Africa
"Shareholder Loan Facilities" the loan facilities from each of AOP and Dr. Christian Schweiger to
the Company announced
on 10 December 2020 and confirmed on 29 January 2021
"Shareholders" the holders of Ordinary Shares for the time being (each
individually a "Shareholder")
"Subscribers" each of Tim Watts, Dr Christian Schweiger, AOP and certain
employees of the Company who are
participating in the Subscription
"Subscription" the conditional subscription by the Subscribers for Subscription
Shares at the Issue Price
in accordance with the Subscription Letters to raise approximately
GBP5.1 million before expenses
"Subscription Letters" the subscription letters entered into between the Subscribers and
the Company on 26 February
2021 in relation to the Subscription
"Subscription Shares" the 16,934,613 new Ordinary Shares to be issued pursuant to the
Subscription whose allotment
and issue is conditional (amongst other things) on the passing of
the Fundraising Resolutions
"United Kingdom" or "UK" the United Kingdom of Great Britain and Northern Ireland
"UK MAR" Regulation (EU) No. 596/2014 of the European Parliament and of the
Council of 16 April 2014
on market abuse, as it forms part of UK domestic law by virtue of
the European Union (Withdrawal)
Act 2018, as amended
"uncertificated" or "in uncertificated form" recorded on the register of members of the Company as being held in
uncertificated form in
CREST and title to which, by virtue of the CREST Regulations, may
be transferred by means
of CREST
"United States" or "US" the United States of America, its territories and possessions, any
state of the United States
and the District of Columbia
"Voting Undertakings" the voting undertakings entered into dated 25 and 26 February 2021
by each of AOP, W. Health
and Christian Schweiger undertaking to vote in favour of the
Resolutions at the General Meeting
"W. Health" W. Health L.P. of Winterbotham Place Marlborough & Queen Streets
NASSAU C5 N-3026, an investment
vehicle of Inventages Wealth Management Inc
This information is provided by RNS, the news service of the
London Stock Exchange. RNS is approved by the Financial Conduct
Authority to act as a Primary Information Provider in the United
Kingdom. Terms and conditions relating to the use and distribution
of this information may apply. For further information, please
contact rns@lseg.com or visit www.rns.com.
RNS may use your IP address to confirm compliance with the terms
and conditions, to analyse how you engage with the information
contained in this communication, and to share such analysis on an
anonymised basis with others as part of our commercial services.
For further information about how RNS and the London Stock Exchange
use the personal data you provide us, please see our Privacy
Policy.
END
IOEFIFSTFIIRFIL
(END) Dow Jones Newswires
February 26, 2021 04:30 ET (09:30 GMT)
Shield Therapeutics (LSE:STX)
Historical Stock Chart
From Apr 2024 to May 2024
Shield Therapeutics (LSE:STX)
Historical Stock Chart
From May 2023 to May 2024