NovaBay Pharmaceuticals Launches First of Several New Products to Expand Avenova Line
17 May 2021 - 8:50PM
Business Wire
Self-diagnostic device designed to assist
Avenova customers with management of common lid and lash
problems
NovaBay® Pharmaceuticals, Inc. (NYSE American: NBY), a
pharmaceutical company focusing on expanding its presence in the
eye and skin care market, announces the availability of i-Chek
Illuminated Eye Examination Mirror on Amazon. i-Chek is a unique
portable, lightweight, handheld lighted mirror featuring 10x
magnification that is specifically designed for at-home precise
visual resolution and high magnification of the eyes, eyelashes and
eyelids.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20210517005310/en/
i-Chek, First of Several New Products to
Expand Avenova Line (Photo: Business Wire)
“i-Chek is the ideal companion product to Avenova, our premier
antimicrobial lid and lash spray, providing a personalized
before-and-after look at the effectiveness of Avenova,” said Justin
Hall, President and CEO of NovaBay Pharmaceuticals. “Those
afflicted with bacterial dry eye can now examine their own eyes and
view for themselves the buildup of scruff and debris on their lids
and lashes before using Avenova. Then after applying our popular,
soothing and highly effective spray, they can visualize what they
are feeling and the successful elimination of those conditions.
Visually examining the lid and lash area with i-Chek before and
after using Avenova twice-daily can improve the management of
painful conditions such as burning, inflammation, itching and
unsightly redness that can accompany bacterial dry eye.
“Expanding the Avenova product line in the direct-to-consumer
sales channel is a key element in our strategy to accelerate the
broadening of our customer base while also providing new and
complementary products for our many loyal Avenova lid and lash
spray users. Complementary products such as the Warm Eye Compress
and i-Chek will not only solidify the Avenova brand in the eyecare
market, but will also diversify our sources of revenue. Gross
margins for these product line extensions are in line with those of
Avenova. In addition, we are developing a lubricating dry eye drop
that will launch later this year. Once it becomes available,
customers will be able to use our eye drop for immediate relief
from dry eye symptoms, and then use the antimicrobial spray to
address the underlying cause. We are developing our marketing
strategy to promote NovaBay as a one stop shop for premier
over-the-counter dry eye treatments that can be easily accessed by
the general public who increasingly want to be more hands-on in
their own health care.”
i-Chek can also be useful in examining for lipase
saponification, which results when the lipid layer of tear film
breaks down into soap due to bacterial action associated with
blepharitis,” he added. “Professional eye care practitioners can
typically see saponification in their slit lamps as tiny bubbles
that sit on top of the eyelid, but most patients are unable to see
these bubbles with the unaided eye. i-Chek provides an at-home
solution for demonstrating Avenova’s effectiveness on this
condition, as well.”
Additional uses for i-Chek include help with eliminating scleral
contact lens bubbles and contact insertion misalignments, as well
as helping to prevent eye infections when removing makeup. i-Chek
features four LED ECO bulbs for more than 15,000 hours of use and a
durable carrying case. The product is available on Amazon.com and
Avenova.com for $29.99. The addition of i-Chek to the Avenova
product line follows the launch of the Avenova Warm Eye Compress on
Amazon.com and on Avenova.com in March 2021.
About NovaBay Pharmaceuticals, Inc.: Going Beyond
Antibiotics®
NovaBay Pharmaceuticals, Inc. is a biopharmaceutical company
focusing on high-quality, differentiated, anti-infective consumer
products: Avenova®, the premier antimicrobial lid and lash spray,
CelleRx® Clinical Reset™, a breakthrough product in the beauty
category, and NeutroPhase® Skin and Wound Cleanser for wound
healing. NovaBay’s products are formulated with its patented, pure,
stable, pharmaceutical-grade hypochlorous acid that replicates the
antimicrobial chemicals used by white blood cells to fight
infection. NovaBay’s hypochlorous acid products do not cause
stinging or irritation, are non-toxic and non-sensitizing, making
them completely safe for regular use. Avenova is the only
commercial hypochlorous acid lid and lash spray product clinically
proven to reduce bacterial load on ocular skin surfaces, thus
effectively addressing the underlying cause of bacterial dry
eye.
Forward-Looking Statements
Except for historical information herein, matters set forth in
this press release are forward-looking within the meaning of the
“safe harbor” provisions of the Private Securities Litigation
Reform Act of 1995, including statements about the commercial
progress and future financial performance of NovaBay
Pharmaceuticals, Inc. This release contains forward-looking
statements that are based upon management’s current expectations,
assumptions, estimates, projections and beliefs. These statements
include, but are not limited to, statements regarding our current
product offerings, potential future product offerings, and any
future revenue that may result from selling these products, as well
as generally the Company’s expected future financial results. These
statements involve known and unknown risks, uncertainties and other
factors that may cause actual results or achievements to be
materially different and adverse from those expressed in or implied
by the forward-looking statements. Factors that might cause or
contribute to such differences include, but are not limited to,
risks and uncertainties relating to the size of the potential
market for our products, the possibility that the available market
for the Company’s products will not be as large as expected, the
Company’s products will not be able to penetrate one or more
targeted markets, revenues will not be sufficient to meet the
Company’s cash needs, the effect on sales and potential
reputational damage resulting from decisions or actions taken by
regulators, including Warning Letters issued by the FDA, and any
other potential regulatory problems that may arise. Other risks
relating to NovaBay’s business, including risks that could cause
results to differ materially from those projected in the
forward-looking statements in this press release, are detailed in
NovaBay’s latest Form 10-Q/K filings with the Securities and
Exchange Commission, especially under the heading “Risk Factors.”
The forward-looking statements in this release speak only as of
this date, and NovaBay disclaims any intent or obligation to revise
or update publicly any forward-looking statement except as required
by law.
Socialize and Stay informed on
NovaBay’s progress
Like us on Facebook
Follow us on Twitter
Connect with NovaBay on LinkedIn
Visit NovaBay’s Website
Avenova Purchasing
Information
For NovaBay Avenova purchasing information:
Please call 800-890-0329 or email sales@avenova.com.
Avenova.com
CelleRx Clinical Reset Purchasing
Information
For NovaBay CelleRx Clinical Reset purchasing information
Please call 877-CELLERX
www.CelleRx.com
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210517005310/en/
NovaBay Contact Justin Hall Chief Executive Officer and
General Counsel 510-899-8800 jhall@novabay.com
Investor Contact LHA Investor Relations Jody Cain
310-691-7100 jcain@lhai.com
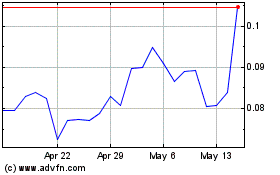
NovaBay Pharmaceuticals (AMEX:NBY)
Historical Stock Chart
From Apr 2024 to May 2024
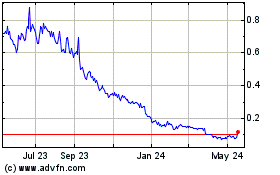
NovaBay Pharmaceuticals (AMEX:NBY)
Historical Stock Chart
From May 2023 to May 2024