GLUCOTRACK EXPANDS LEADERSHIP TEAM WITH NEW VICE PRESIDENT OF QUALITY
13 March 2024 - 12:30AM

Glucotrack, Inc. (Nasdaq: GCTK) (“Glucotrack” or the “Company”), a
medical device company focused on the design, development, and
commercialization of novel technologies for people with diabetes,
announced today that Vincent Wong has joined the Company as Vice
President of Quality.
Mr. Wong has extensive experience in life
sciences and medical device manufacturing, with expertise in
establishing Quality Management functions, managing FDA
inspections, and driving product & process risk management
activities to support sustainable product launch of Class III
active implantable devices. He most recently served as Chief
Quality Officer at TOMZ Corporation, where he led the strategic
development and deployment of the Quality and Business Management
System at one of the world’s leading high-volume medical device
manufacturers and acquired entities. Prior to that, Mr. Wong served
on the Senior Management team as Director of Quality at Cirtec
Medical where he was responsible for overseeing all corporate
Quality functions including the organization’s Product Development
Process, FDA inspections, ISO 13485:2016 transition and
establishment of the Corporate Quality Management System across all
sites, facilitating the integration of multiple acquisitions and
new site launches. Mr. Wong earned both a Master and Bachelor of
Science in Biomedical Engineering from the University of
Connecticut.
Mr. Wong will be responsible for establishing
and implementing the company’s Quality System and all related
processes, including Risk Management, Product and Process
Validation, Design Transfer, and Sustainability into Commercial
Manufacturing. He will lead ISO-13485 certification and all related
quality activities for the Company’s implantable Continuous Blood
Glucose Monitoring (CBGM) technology.
“We are thrilled that Vincent has joined our
growing team in this important leadership role.” said Paul Goode,
PhD, CEO of Glucotrack. “Vincent’s experience with implantable
medical devices, from early feasibility through sustained
manufacturing and commercial launch will be a tremendous asset as
we transition to human clinical trials and move towards
commercialization of our CBGM which we believe will simplify and
improve diabetes management for people living with diabetes.”
“Glucotrack is an innovative organization that
is committed to incorporating quality and compliance best practices
from the very beginning. Implementing a sustainable Quality
Management System that will grow with us is essential as we now
embark on product development activities for our commercial
product. I look forward to joining this leadership team to bring
this differentiated technology to people with diabetes,” said Mr.
Wong.
# # #
About GlucoTrack, Inc.
GlucoTrack, Inc. (NASDAQ: GCTK) is focused on
the design, development, and commercialization of novel
technologies for people with diabetes. The Company is currently
developing a long-term implantable continuous glucose monitoring
system for people living with diabetes.
Glucotrack’s CBGM is a long-term, implantable
system that continually measures blood glucose levels with a sensor
longevity of 2+ years, no on-body wearable component and with a
once-only calibration event. For more information, please
visit http://www.glucotrack.com.
Investor Contact: investors@glucotrack.com
Forward-Looking Statements
This news release contains forward-looking
statements within the meaning of the Private Securities Litigation
Reform Act of 1995. Statements contained in this news release that
are not statements of historical fact may be deemed to be
forward-looking statements. Without limiting the generality of the
foregoing, words such as “believe”, “expect”, “plan” and “will” are
intended to identify forward-looking statements. Such
forward-looking statements are based on the beliefs of management,
as well as assumptions made by, and information currently available
to, management. Readers are cautioned that certain important
factors may affect Glucotrack’s actual results and could cause such
results to differ materially from any forward-looking statements
that may be made in this news release. Factors that may affect
Glucotrack’s results include, but are not limited to, the ability
of GlucoTrack to raise additional capital to finance its operations
(whether through public or private equity offerings, debt
financings, strategic collaborations or otherwise); risks relating
to the receipt (and timing) of regulatory approvals (including FDA
approval); risks relating to enrollment of patients in, and the
conduct of, clinical trials; risks relating to its current and
future distribution agreements; risks relating to its ability to
hire and retain qualified personnel, including sales and
distribution personnel; and the additional risk factors described
in Glucotrack’s filings with the U.S. Securities and Exchange
Commission (the “SEC”), including its Annual Report on Form 10-K
for the year ended December 31, 2022 as filed with the SEC on March
31, 2023.
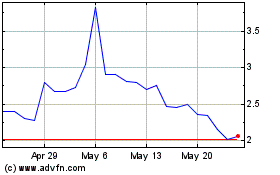
GlucoTrack (NASDAQ:GCTK)
Historical Stock Chart
From Apr 2024 to May 2024
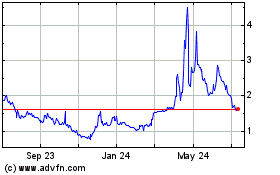
GlucoTrack (NASDAQ:GCTK)
Historical Stock Chart
From May 2023 to May 2024