Ocular Therapeutix™ to Present at the Piper Jaffray 31st Annual Healthcare Conference
27 November 2019 - 12:00AM
Business Wire
Ocular Therapeutix, Inc. (NASDAQ:OCUL), a biopharmaceutical
company focused on the formulation, development, and
commercialization of innovative therapies for diseases and
conditions of the eye, today announced that Antony Mattessich,
President and Chief Executive Officer of Ocular Therapeutix, will
present and host investor meetings at the Piper Jaffray 31st Annual
Healthcare Conference being held at the Lotte New York Palace in
New York, NY. The Company is scheduled to present on Thursday,
December 5, 2019, at 12:30 p.m. ET.
About Ocular Therapeutix, Inc. Ocular Therapeutix, Inc.
is a biopharmaceutical company focused on the formulation,
development, and commercialization of innovative therapies for
diseases and conditions of the eye using its proprietary
bioresorbable hydrogel-based formulation technology. Ocular
Therapeutix’s first commercial drug product, DEXTENZA®, is
FDA-approved for the treatment of ocular inflammation and pain
following ophthalmic surgery. Ocular Therapeutix has also begun a
Phase 3 clinical trial evaluating DEXTENZA for the treatment of
ocular itching associated with allergic conjunctivitis. OTX-TP
(intracanalicular travoprost insert) is an intracanalicular insert
in Phase 3 clinical development for the reduction of intraocular
pressure in patients with primary open-angle glaucoma and ocular
hypertension. The Company’s earlier stage assets include OTX-TIC,
an extended-delivery intracameral travoprost implant for the
reduction of intraocular pressure in patients with glaucoma and
ocular hypertension, as well as sustained release intravitreal
implants for the treatment of retinal diseases. These intravitreal
implants include OTX-TKI, containing a tyrosine kinase inhibitor
(TKI), and, in collaboration with Regeneron, OTX-IVT, an
extended-delivery protein-based anti-vascular endothelial growth
factor (VEGF) trap. Ocular Therapeutix's first product, ReSure®
Sealant, is FDA-approved to seal corneal incisions following
cataract surgery.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20191126005254/en/
Investors Ocular Therapeutix Donald Notman Chief
Financial Officer dnotman@ocutx.com or Westwicke, an ICR Company
Chris Brinzey, 339-970-2843 chris.brinzey@westwicke.com
Media Ocular Therapeutix Scott Corning Senior Vice
President, Commercial scorning@ocutx.com
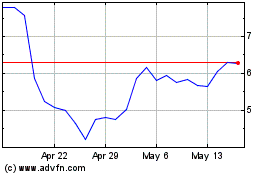
Ocular Therapeutix (NASDAQ:OCUL)
Historical Stock Chart
From Apr 2024 to May 2024
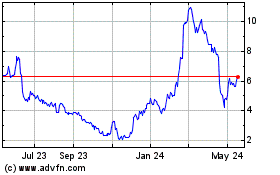
Ocular Therapeutix (NASDAQ:OCUL)
Historical Stock Chart
From May 2023 to May 2024