Catalent Completes $25 Million Expansion of its Clinical Supply Facility in Schorndorf, Germany
16 July 2024 - 4:00PM
Business Wire
Expansion adds more temperature-controlled
storage and dedicated space for FastChain® demand-led supply
service
Catalent, Inc. (NYSE: CTLT), the leader in enabling the
development and supply of better treatments for patients worldwide,
today announced it has completed expansion of its clinical supply
facility in Schorndorf, Germany. The Schorndorf site, Catalent’s
flagship European facility, provides comprehensive clinical supply
services, including packaging, storage and distribution.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20240716141571/en/
Catalent Schorndorf Expansion (Photo:
Business Wire)
The expansion increases the site’s footprint by 32,000 square
feet (3,000 square meters), adding storage and handling of clinical
trial supply materials at controlled room temperatures between 15
and 25 degrees Celsius. It also creates space in the original
building to accommodate the installation of a new, fully automatic
bottle filling line, as well as a dedicated area for Catalent’s
FastChain® demand-led supply service. These added capabilities will
support a continued high level of service to Catalent’s pharma and
biotech customers and the patients they serve.
“The Schorndorf site plays an important role in the strategic
focus of the company and our ability to meet customer demand for
clinical packaging solutions, including temperature-controlled
services,” said Lorenzo Carletti, President, Clinical Development
& Supply, Catalent, Inc. “We are pleased to bring additional
capacity to the site for temperature-controlled storage and
Catalent’s FastChain service so that we can meet the current and
future needs of our customers.”
Opened in 1996, Catalent’s Schorndorf facility provides
comprehensive clinical supply services including multi-lingual
project services – including English, French and German –
comparator sourcing, primary and secondary packaging, labeling,
clinical storage and distribution, and returns and destruction.
This co-located site also offers late-stage pharma product
development and commercial services, including oral solid dose
manufacturing and specialty packaging.
The company’s FastChain service is a continuous Good
Manufacturing Practices (GMP) approach that decouples primary from
secondary packaging to enable timely and cost-effective adaptation
and regional supply of clinical materials on demand.
ABOUT CATALENT
Catalent, Inc. is a global leader in enabling pharma, biotech,
and consumer health partners to optimize product development,
launch, and full life-cycle supply for patients around the world.
With broad and deep scale and expertise in development sciences,
delivery technologies, and multi-modality manufacturing, Catalent
is a preferred industry partner for personalized medicines,
consumer health brand extensions, and blockbuster drugs.
Catalent helps accelerate over 1,500 partner programs and launch
over 150 new products every year. Its flexible manufacturing
platforms at over 50 global sites supply approximately 70 billion
doses of nearly 8,000 products annually. Catalent’s expert
workforce of nearly 18,000 includes more than 3,000 scientists and
technicians.
Headquartered in Somerset, New Jersey, the company generated
nearly $4.3 billion in revenue in its 2023 fiscal year.
MORE PRODUCTS. BETTER TREATMENTS. RELIABLY SUPPLIED.™
View source
version on businesswire.com: https://www.businesswire.com/news/home/20240716141571/en/
Investor Contact: Paul Surdez +1 (732) 537-6325
investors@catalent.com
Media Contact: Laura Hortas +1 (609) 240-7025
media@catalent.com
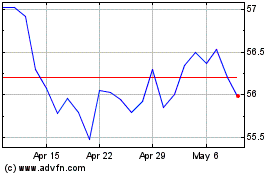
Catalent (NYSE:CTLT)
Historical Stock Chart
From Jun 2024 to Jul 2024
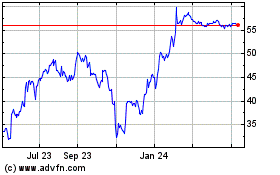
Catalent (NYSE:CTLT)
Historical Stock Chart
From Jul 2023 to Jul 2024