Medtronic Launches New Endoscopic Ablation Catheter for Barrett's Esophagus
21 April 2016 - 10:35PM


BarrxTM 360 Express
RFA Balloon Catheter's Adjustable Custom Fit Provides Targeted
Precision Therapy - Allowing Physician Ease and Efficiency
DUBLIN - April 21, 2016 -
Medtronic (NYSE: MDT) today announced the launch of the new
BarrxTM 360 Express
radiofrequency ablation (RFA) balloon catheter, which can help in
the treatment of Barrett's esophagus.
The Barrx 360 Express catheter, with its
self-adjusting circumferential RFA catheter, lets
gastroenterologists and surgeons provide RFA treatment more easily
and efficiently. RFA therapy removes diseased tissue while
minimizing injury1 to healthy
esophageal tissue. This treatment has been shown to reduce the risk
of Barrett's esophagus with low grade dysplasia -- a precancerous
condition that causes abnormal cell growth in the esophagus,
progressing to high grade dysplasia or esophageal adenocarcinoma, a
type of cancer -- by approximately 90%.2
Esophageal cancer is the fastest-growing cancer in
the U.S., having increased sixfold since the 1970s, outpacing
breast cancer, prostate cancer and melanoma.3
Barrett's esophagus, which affects more than 12 million American
adults, develops as a result of chronic injury from
gastroesophageal reflux disease (GERD).1,4
The new design helps physicians lower procedural
time by up to 20 percent 2,5 by reducing
procedural steps and enhanced ease-of-use features, including the
ability to custom fit the device to various esophagus diameter
sizes. With this tool, physicians are able to ablate Barrett's
tissue more efficiently. Using RFA to treat dysplastic Barrett's
tissue has been shown to reduce the risk for progression to
high-grade dysplasia and esophageal adenocarcinoma,2,6 the most
common type of esophageal cancer in the U.S.
"The biggest advantage of the Barrx 360 Express
catheter is that it's easier to use because of the larger, longer
and adjustable balloon size," said Anthony Infantolino, MD, AGAF,
FACG, FACP, of Thomas Jefferson University Hospital in
Philadelphia. "These new features provide for a reduction in
surgery time2,5 and the
number of catheters required during RFA procedures, allowing me to
concentrate on increasing the quality of the procedure."
"In reducing the progression of dysplastic
Barrett's esophagus to esophageal cancer, the launch of the Barrx
360 Express RFA balloon catheter underscores our commitment to
patients," said Vafa Jamali, president, Early Technologies business
in the Medtronic Minimally Invasive Therapies Group. "We believe
patients and physicians will benefit from the easier-to-use
technology of the Barrx 360 Express catheter, which provides a more
precise delivery of RF energy to the targeted area."
The company is currently launching the Barrx 360
Express RFA balloon catheter in the United States. The catheter is
also expected to be available in the EU, Australia and New Zealand
in 2016.

BarrxTM 360 Express
radiofrequency ablation (RFA) balloon catheter
Click the image for a larger image.
About Medtronic
Medtronic plc (www.medtronic.com), headquartered in Dublin,
Ireland, is among the world's largest medical technology, services
and solutions companies - alleviating pain, restoring health and
extending life for millions of people around the world. Medtronic
employs more than 85,000 people worldwide, serving physicians,
hospitals and patients in approximately 160 countries. The company
is focused on collaborating with stakeholders around the world to
take healthcare Further, Together.
Any forward-looking statements
are subject to risks and uncertainties such as those described in
Medtronic's periodic reports on file with the Securities and
Exchange Commission. Actual results may differ materially from
anticipated results.
-end-
1 Gilbert
EW, Luna RA, Harrison VL, Hunter JG. Barrett's Esophagus: A Review
of the Literature. J Gastrointest Surg. 2011 May;15(5):708-18.
2 Phoa KN,
van Vilsteren FG, Weusten BL, et al. Radiofrequency Ablation vs
Endoscopic Surveillance for Patients With Barrett Esophagus and
Low-Grade Dysplasia: A Randomized Clinical Trial. JAMA.
2014;311:1209-17.
3 Pohl H,
Welch HG. The Role of Overdiagnosis and Reclassification in the
Marked Increase of Esophageal Adenocarcinoma Incidence. J Natl
Cancer Inst. 2005 Jan 19;97(2):142-6.
4 Hayeck
TJ et al. The Prevalence of Barrett's Esophagus in the US:
Estimates from a Simulation Model
Confirmed by SEER Data. Dis Esophagus 2010; 23(6):451-7.
5 Bergman
JJ, Weusten BL, Schoon EJ, Koch AD. Safety and Efficacy of the Self
Sizing RFA balloon: an uncontrolled multicenter feasibility study
(B-550).
6 Shaheen
NJ, Sharma P, Overholt BF, et al. Radiofrequency ablation in
Barrett's esophagus with dysplasia. N Engl J
Med. 2009.360:2277-88.
Contacts:
John Jordan
Public Relations
+1-508-452-4891
Ryan Weispfenning
Investor Relations
+1-763-505-4626
This
announcement is distributed by NASDAQ OMX Corporate Solutions on
behalf of NASDAQ OMX Corporate Solutions clients.
The issuer of this announcement warrants that they are solely
responsible for the content, accuracy and originality of the
information contained therein.
Source: Medtronic plc via Globenewswire
HUG#2005345
Medtronic (NYSE:MDT)
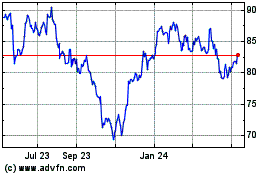
Historical Stock Chart
From Apr 2024 to May 2024
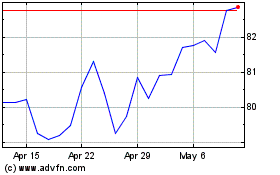
Medtronic (NYSE:MDT)
Historical Stock Chart
From May 2023 to May 2024