ACADIA Pharmaceuticals Presents Caregiver Burden Data at the International Congress of Non-Motor Dysfunctions in Parkinson’...
09 December 2014 - 1:00AM
Business Wire
Data Demonstrate a Decreased Burden Among
Caregivers of Patients with Parkinson’s Disease Psychosis Treated
with NUPLAZID™ (Pimavanserin)
ACADIA Pharmaceuticals Inc. (NASDAQ: ACAD) today announced the
presentation of caregiver burden data from its Phase III program
with NUPLAZID™ (pimavanserin) for Parkinson’s disease psychosis
(PDP) at the 10th Annual International Congress of Non-Motor
Dysfunctions in Parkinson’s Disease and Related Disorders held from
December 5-7 in Nice, France. Data from an analysis of Phase III
studies in patients with PDP showed that treatment with NUPLAZID
reduced caregiver burden compared to PDP patients on placebo.
“The symptoms of Parkinson’s disease psychosis are a significant
cause of distress to patients and their caregivers, and are
associated with greater caregiver burden, nursing home placement,
functional impairment and increased mortality,” said Dag Aarsland,
M.D., Professor of Clinical Dementia Research at the Department of
Neurobiology, Care Sciences and Society, Karolinska Institute. “The
benefit to caregivers observed with NUPLAZID may translate into a
reduced or delayed nursing home admission for patients with
Parkinson’s disease psychosis.”
In a poster presentation titled “Decreased Burden Among
Caregivers of People with Parkinson’s Disease Psychosis Treated
with Pimavanserin, a Selective 5-HT2A Inverse Agonist,” an
integrated analysis of Phase III clinical trials with NUPLAZID in
PDP was performed on caregiver burden among 268 caregivers of PDP
patients from North America. Caregiver burden was assessed using
the Zarit 22-item Caregiver Burden Scale. The scale was completed
by the caregiver to provide a quantitative assessment of burden
associated with the patient’s functional behavioral impairments,
the circumstances of at-home care, as well as the caregiver’s
health, social life and interpersonal relations. The objective of
the analysis was to explore the effects of NUPLAZID, which has
shown antipsychotic efficacy and non-sedative sleep benefits, on
perceived burden in caregivers of people with PDP.
In 6-week randomized placebo-controlled Phase III trials,
NUPLAZID demonstrated a significant improvement in caregiver burden
compared to placebo (p=0.001), as assessed by the Caregiver Burden
Scale. NUPLAZID showed the strongest improvement on the Life-Upset
subscale, which evaluates strains on personal/life relationships
and effects on caregiver health and anxiety over care. Although no
single item drove the Caregiver Burden Scale, the strongest benefit
of NUPLAZID was observed on Item 16 of the Responsibility subscale
(p<0.001), which asked the caregiver about the inability to take
care of the relative for much longer. In subgroup analyses,
caregiver benefit with NUPLAZID was observed to be greater for
spouses than non-spouses and for caregivers with more severe burden
at baseline.
Additionally, an interim analysis performed on the open-label
safety extension study in patients with PDP showed that caregivers
of NUPLAZID-treated patients demonstrated reduced caregiver burden
that persisted through 9 months, as assessed by the Caregiver
Burden Scale.
About NUPLAZID™ (pimavanserin)
NUPLAZID is ACADIA’s proprietary small molecule that is a
selective serotonin inverse agonist preferentially targeting 5-HT2A
receptors that play an important role in psychosis. ACADIA has
reported positive Phase III trial results with NUPLAZID, which has
the potential to be the first drug approved in the United States
for psychosis associated with Parkinson’s disease. NUPLAZID is
administered orally once-a-day. ACADIA discovered NUPLAZID and
holds worldwide rights to this new chemical entity. The trade name
NUPLAZID has been provisionally accepted by the FDA.
About Parkinson’s Disease Psychosis
According to the National Parkinson Foundation, about one
million people in the United States and from four to six million
people worldwide suffer from Parkinson’s disease. Parkinson’s
disease psychosis (PDP) is a debilitating disorder that occurs in
an estimated 40 percent of Parkinson’s patients. Currently, there
is no FDA-approved therapy to treat PDP in the United States. PDP,
which commonly consists of visual hallucinations and delusions,
substantially contributes to the burden of Parkinson’s disease and
deeply affects the quality of life of patients. PDP also is
associated with increased caregiver stress and burden, nursing home
placement, and increased morbidity and mortality. There is a large
unmet medical need for new therapies that will effectively treat
PDP without compromising motor control in patients with Parkinson’s
disease.
About ACADIA Pharmaceuticals
ACADIA is a biopharmaceutical company focused on the development
and commercialization of innovative medicines to address unmet
medical needs in neurological and related central nervous system
disorders. ACADIA has a pipeline of product candidates led by
NUPLAZID™ (pimavanserin), for which we have reported positive Phase
III trial results in Parkinson’s disease psychosis and which has
the potential to be the first drug approved in the United States
for this disorder. Pimavanserin is also in Phase II development for
Alzheimer’s disease psychosis and has successfully completed a
Phase II trial in schizophrenia. ACADIA also has clinical-stage
programs for chronic pain and glaucoma in collaboration with
Allergan, Inc. and two preclinical programs directed at Parkinson’s
disease and other neurological disorders. All product candidates
are small molecules that emanate from internal discoveries. ACADIA
maintains a website at www.acadia-pharm.com to which we regularly
post copies of our press releases as well as additional information
and through which interested parties can subscribe to receive
e-mail alerts.
Forward-Looking Statements
Statements in this press release that are not strictly
historical in nature are forward-looking statements. These
statements include but are not limited to statements related to the
progress and timing of ACADIA’s drug discovery and development
programs, either alone or with a partner, including clinical
trials, the benefits to be derived from ACADIA’s product
candidates, in each case including NUPLAZID (pimavanserin), and
whether a reduction in caregiver burden would translate into a
reduced or delayed nursing home admission for patients with
Parkinson’s disease. These statements are only predictions based on
current information and expectations and involve a number of risks
and uncertainties. Actual events or results may differ materially
from those projected in any of such statements due to various
factors, including the risks and uncertainties inherent in drug
discovery, development and commercialization, and collaborations
with others, and the fact that past results of clinical trials may
not be indicative of future trial results. For a discussion of
these and other factors, please refer to ACADIA’s annual report on
Form 10-K for the year ended December 31, 2013 as well as ACADIA’s
subsequent filings with the Securities and Exchange Commission. You
are cautioned not to place undue reliance on these forward-looking
statements, which speak only as of the date hereof. This caution is
made under the safe harbor provisions of the Private Securities
Litigation Reform Act of 1995. All forward-looking statements are
qualified in their entirety by this cautionary statement and ACADIA
undertakes no obligation to revise or update this press release to
reflect events or circumstances after the date hereof, except as
required by law.
Investor Contacts:ACADIA Pharmaceuticals Inc.Steve Davis,
Executive Vice President,Chief Financial Officer and Chief Business
OfficerLisa Barthelemy, Director of Investor Relations(858)
558-2871orMedia Contact:Chandler Chicco CompaniesDavid Polk(310)
309-1029 or (805) 428-5775
Acadia Pharmaceuticals (NASDAQ:ACAD)
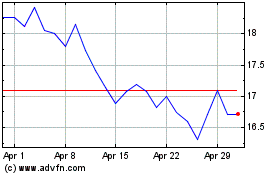
Historical Stock Chart
From Mar 2024 to Apr 2024
Acadia Pharmaceuticals (NASDAQ:ACAD)
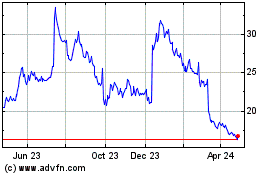
Historical Stock Chart
From Apr 2023 to Apr 2024