Adagene Announces Poster Presentation on Optimal Dose Selection for Masked Anti-CTLA-4 SAFEbody® ADG126 at Upcoming Society for Immunotherapy of Cancer (SITC) 38th Annual Meeting
12 October 2023 - 11:00PM

Adagene Inc. (“Adagene”) (Nasdaq: ADAG), a company transforming the
discovery and development of novel antibody-based therapies, today
announced its poster presentation at the SITC 38th Annual Meeting
taking place November 1-5, 2023 in San Diego.
Details for the poster include:
Title: Optimal Dose Selection of ADG126 (a Masked anti-CTLA-4
SAFEbody®) with Significantly Widened Therapeutic Index compared to
Ipilimumab in Combination with anti-PD-1 Antibodies Informed by QSP
Modeling
- Date: Friday, November 3
- Poster Hall Hours: 9:00 a.m. – 7:00 p.m. (Reception: 5:10 p.m.
– 6:40 p.m.)
- Onsite Location: Exhibit Halls A and B1 – San Diego Convention
Center
- Abstract Number: 847
About Adagene
Adagene Inc. (Nasdaq: ADAG) is a platform-driven,
clinical-stage biotechnology company committed to transforming the
discovery and development of novel antibody-based cancer
immunotherapies. Adagene combines computational biology
and artificial intelligence to design novel antibodies that address
unmet patient needs. Powered by its proprietary Dynamic
Precision Library (DPL) platform, composed of NEObody™,
SAFEbody®, and POWERbody™ technologies, Adagene’s highly
differentiated pipeline features novel immunotherapy
programs. Adagene has forged strategic collaborations
with reputable global partners that leverage its technology in
multiple approaches at the vanguard of science.
For more information, please
visit: https://investor.adagene.com.
Follow Adagene on WeChat, LinkedIn and Twitter.
SAFEbody® is a registered trademark in the United
States, China, Australia, Japan, Singapore, and
the European Union.
Safe Harbor Statement
This press release contains forward-looking statements,
including statements regarding certain clinical results of ADG126,
the potential implications of clinical data for patients, and
Adagene’s advancement of, and anticipated preclinical activities,
clinical development, regulatory milestones, and commercialization
of its product candidates. Actual results may differ materially
from those indicated in the forward-looking statements as a result
of various important factors, including but not limited to
Adagene’s ability to demonstrate the safety and efficacy of its
drug candidates; the clinical results for its drug candidates,
which may not support further development or regulatory approval;
the content and timing of decisions made by the relevant regulatory
authorities regarding regulatory approval of Adagene’s drug
candidates; Adagene’s ability to achieve commercial success for its
drug candidates, if approved; Adagene’s ability to obtain and
maintain protection of intellectual property for its technology and
drugs; Adagene’s reliance on third parties to conduct drug
development, manufacturing and other services; Adagene’s limited
operating history and Adagene’s ability to obtain additional
funding for operations and to complete the development and
commercialization of its drug candidates; Adagene’s ability to
enter into additional collaboration agreements beyond its existing
strategic partnerships or collaborations, and the impact of the
COVID-19 pandemic on Adagene’s clinical development, commercial and
other operations, as well as those risks more fully discussed in
the “Risk Factors” section in Adagene’s filings with the U.S.
Securities and Exchange Commission. All forward-looking statements
are based on information currently available to Adagene, and
Adagene undertakes no obligation to publicly update or revise any
forward-looking statements, whether as a result of new information,
future events or otherwise, except as may be required by law.
Investor & Media Contact:Ami
KnoeflerAdagene650-739-9952ir@adagene.com
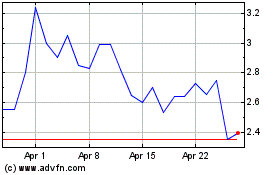
Adagene (NASDAQ:ADAG)
Historical Stock Chart
From Mar 2024 to Apr 2024
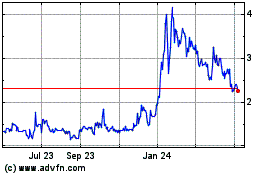
Adagene (NASDAQ:ADAG)
Historical Stock Chart
From Apr 2023 to Apr 2024