Alterity Therapeutics Receives a $4.74 million Research & Development Tax Incentive Refund
15 August 2023 - 9:25PM

Alterity Therapeutics Limited (ASX: ATH, NASDAQ: ATHE) (“Alterity”
or “the Company”), a biotechnology company dedicated to developing
disease modifying treatments for neurodegenerative diseases, today
announced that it has received a A$4.74 million cash refund from
the Australian Taxation Office under the Australian Government’s
Research and Development Tax Incentive (R&DTI) Scheme.
The cash refund relates to the cost of eligible
research and development activities conducted during the financial
year ended 30 June 2022, and represents the amount disclosed in the
company’s audited financial statements. These funds will be used to
further Alterity’s clinical development and research activities,
including the ongoing Phase 2 clinical trials for the Company’s
lead drug candidate ATH434 in Multiple System Atrophy (MSA), a
Parkinsonian disorder with no approved therapy.
About ATH434
Alterity’s lead candidate, ATH434, is an oral
agent designed to inhibit the aggregation of pathological proteins
implicated in neurodegeneration. ATH434 has been shown
preclinically to reduce α-synuclein pathology and preserve nerve
cells by restoring normal iron balance in the brain. As an iron
chaperone, it has excellent potential to treat Parkinson’s disease
as well as various Parkinsonian disorders such as Multiple System
Atrophy (MSA). ATH434 successfully completed Phase 1 studies
demonstrating the agent is well tolerated and achieved brain levels
comparable to efficacious levels in animal models of MSA. ATH434 is
currently being studied in two clinical trials: Study ATH434-201 is
a randomized, double-blind, placebo-controlled Phase 2 clinical
trial in patients with early-stage MSA and Study ATH434-202 is an
open-label Phase 2 Biomarker trial in patients with MSA. ATH434 has
been granted Orphan drug designation for the treatment of MSA by
the U.S. FDA and the European Commission.
About Alterity Therapeutics
Limited
Alterity Therapeutics is a clinical stage
biotechnology company dedicated to creating an alternate future for
people living with neurodegenerative diseases. The Company’s
lead asset, ATH434, has the potential to treat various Parkinsonian
disorders and is currently being evaluated in two Phase 2 clinical
trials in Multiple System Atrophy. Alterity also has a broad drug
discovery platform generating patentable chemical compounds to
treat the underlying pathology of neurological diseases. The
Company is based in Melbourne, Australia, and San Francisco,
California, USA. For further information please visit the Company’s
web site at www.alteritytherapeutics.com.
Authorisation & Additional information
This announcement was authorized by David Stamler, CEO of
Alterity Therapeutics Limited.
Investor and Media Contacts:
Australia
Hannah Howlett
we-aualteritytherapeutics@we-worldwide.com
+61 450 648 064
U.S.
Remy Bernarda
remy.bernarda@iradvisory.com
+1 (415) 203-6386
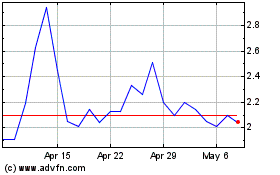
Alterity Therapeutics (NASDAQ:ATHE)
Historical Stock Chart
From Mar 2024 to Apr 2024
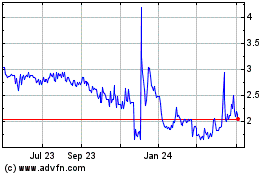
Alterity Therapeutics (NASDAQ:ATHE)
Historical Stock Chart
From Apr 2023 to Apr 2024