Voyager Therapeutics Announces Positive Preclinical Data from New Vectorized anti-HER2 Antibody Program at the 26th Society for Neuro-Oncology Annual Meeting
19 November 2021 - 11:30PM

Voyager Therapeutics, Inc. (Nasdaq: VYGR), a gene therapy company
developing life-changing treatments and next-generation
adeno-associated virus (AAV) platform technologies, today will
present preclinical data demonstrating that IV dosing of a
proprietary AAV vector identified by the TRACER™️ screening
technology, encoding an anti-HER2 antibody payload, prevented tumor
growth in models of HER2+ brain metastases. Dan R. Laks, Ph.D.,
Voyager Scientist II, will present key findings at 4:15 p.m. ET
during the 26th Society for Neuro-Oncology (SNO) Annual Meeting.
“Today’s findings demonstrate that AAV gene transfer of
HER2-directed antibodies to the central nervous system shows
promise to treat brain metastases in patients with HER2+ breast
cancer,” said Glenn Pierce, M.D., Ph.D., interim chief scientific
officer of Voyager. “While approved anti-HER2 therapies are
effective for peripheral disease, they have limited ability to
reach the CNS in sufficient concentrations to treat brain
metastases. By using a proprietary, TRACER AAV capsid to deliver
Voyager’s vectorized payload, and by utilizing brain cells to
produce therapeutic antibodies, we have demonstrated a durable
effect against CNS tumors in several mouse models. We look forward
to continued optimization of this innovative gene therapy approach
as we progress toward clinical development.”
Voyager’s vectorized anti-HER2 antibody was shown to inhibit
proliferation and promote antibody-dependent cell cytotoxicity
(ADCC), a process that recruits natural killer cells, macrophages
and/or brain-resident innate immune cells called microglia to
eliminate tumor cells. IV administration of a proprietary AAV
vector incorporating a TRACER blood-brain barrier penetrant capsid
and a payload encoding an ADCC enhanced anti-HER2 antibody
conferred a survival benefit in a mouse model of HER2+ brain
metastasis.
SNO Oral Presentation DetailsTitle: AAV
mediated brain delivery of an ADCC-enhanced antibody obviates
xenograft growth in mouse models of HER2+ breast cancer brain
metastasisAbstract Number: EXTH-02Abstract Session: CNS
MetastasesPresenter: Dan R. Laks, Ph.D., Scientist II,
VoyagerPresentation Time: 4:15 p.m. ET
Key Findings
- Peripheral administration of a proprietary TRACER AAV vector
achieved widespread transduction of brain tissue such that
efficacious levels of HER2-directed antibodies were produced for
extended periods.
- In mouse models of HER2+ breast cancer brain metastasis, IV
administration of a HER2 antibody-encoding AAV vector resulted in
>1 ug/mL of the antibody in cerebrospinal fluid.
- AAV-mediated expression of the ADCC-enhanced HER2-directed
antibody significantly blocked tumor growth in multiple orthotopic
xenograft models.
Voyager intends to further optimize this gene therapy approach
in ongoing studies progressing towards application of a human
therapeutic.
About HER2+ Brain MetastasesHER2+ breast
cancer, or tumors that overexpress the HER2 growth receptor,
account for approximately 20% of all breast cancers, and brain
metastases are a major cause of mortality in up to 50% of HER2+
metastatic breast cancer patients. While approved anti-HER2
therapies are effective for peripheral disease, they have limited
ability to reach the central nervous system in sufficient
concentrations to treat brain metastases.
About the TRACER™ AAV Capsid Discovery
PlatformVoyager’s TRACER™ (Tropism Redirection of AAV by
Cell-type-specific Expression of RNA) system is a broadly
applicable, RNA-based functional screening platform that allows for
rapid in vivo evolution of AAV capsids with enhanced tropisms and
cell- and tissue-specific transduction properties in multiple
species, including non-human primates (NHPs). Initial data from the
first of many libraries screened in NHPs demonstrated the
proprietary capsid variants effectively penetrated the blood-brain
barrier and achieved widespread biodistribution and transduction of
multiple regions of the brain. Separate results have demonstrated
the ability of certain capsids to transduce cardiac muscle and to
de-target the dorsal root ganglia. Voyager is proceeding with
additional capsid campaigns derived from unique capsid serotypes to
identify novel AAV vectors optimized for specific therapeutic
applications.
About Voyager TherapeuticsVoyager Therapeutics
(Nasdaq: VYGR) is leading the next generation of AAV gene therapy
to unlock the potential of the technology to treat devastating
diseases. Proprietary capsids born from the Company’s TRACER
screening platform are powering a rich early-stage pipeline of new
and second-generation programs and may elevate the field to
overcome the limitations of conventional gene therapy vectors
across neurologic disorders and other therapeutic areas.
voyagertherapeutics.com
LinkedIn Twitter
Voyager Therapeutics® is a registered trademark, and
TRACER™ is a trademark, of Voyager Therapeutics,
Inc.
Forward-Looking StatementsThis press release
contains forward-looking statements for the purposes of the safe
harbor provisions under The Private Securities Litigation Reform
Act of 1995 and other federal securities laws. The use of words
such as “may,” “might,” “will,” “would,” “should,” “expect,”
“plan,” “anticipate,” “believe,” “estimate,” “undoubtedly,”
“project,” “intend,” “future,” “potential,” or “continue,” and
other similar expressions are intended to identify forward-looking
statements.
For example, all statements Voyager makes regarding Voyager’s
ability to continue to identify and develop proprietary capsids
from its TRACER AAV screening platform; Voyager’s ability to
identify and develop proprietary capsids from its TRACER AAV
screening platform with increased transgene expression, increased
blood-brain barrier penetration and increased biodistribution
compared to conventional AAV9 capsids; the initiation, timing,
progress, activities, goals and reporting of results of its
research and development programs; Voyager’s ability to continue to
develop preclinical data on its early pipeline programs relying
upon its novel capsid discovery efforts; and Voyager’s ability to
utilize its novel proprietary capsids in its own product
development programs are forward looking.
All forward-looking statements are based on estimates and
assumptions by Voyager’s management that, although Voyager believes
such forward-looking statements to be reasonable, are inherently
uncertain. All forward-looking statements are subject to risks and
uncertainties that may cause actual results to differ materially
from those that Voyager expected. Such risks and uncertainties
include, among others, the severity and length of the COVID-19
health crisis; the continued development of Voyager’s technology
platforms, including Voyager’s TRACER platform; the ability to
initiate and conduct of preclinical studies in more advanced
pre-clinical animal models; the ability to attract and retain
talented contractors and employees; the ability to create and
protect intellectual property; and the sufficiency of cash
resources.
These statements are also subject to a number of material risks
and uncertainties that are described in Voyager’s most recent
Annual Report on Form 10-K filed with the Securities and
Exchange Commission, as updated by its subsequent filings with
the Securities and Exchange Commission. All information in the
press release is as of the date of this press release, and any
forward-looking statement speaks only as of the date on which it
was made. Voyager undertakes no obligation to publicly update or
revise this information or any forward-looking statement, whether
as a result of new information, future events or otherwise, except
as required by law.
Voyager Contacts
Investors
Investors@voyagertherapeutics.com
Media
Scott Santiamo
ssantiamo@vygr.com
Voyager Therapeutics (NASDAQ:VYGR)
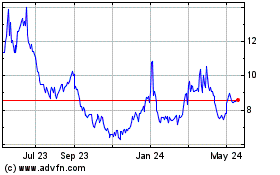
Historical Stock Chart
From Apr 2024 to May 2024
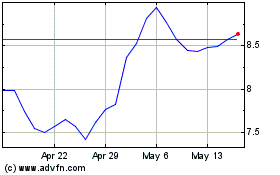
Voyager Therapeutics (NASDAQ:VYGR)
Historical Stock Chart
From May 2023 to May 2024