Merck: FDA to Review Gefapixant for Refractory, Unexplained Chronic Cough
01 March 2021 - 11:48PM
Dow Jones News
By Colin Kellaher
Merck & Co. on Monday said the U.S. Food and Drug
Administration accepted for review its new-drug application for
gefapixant for the treatment of refractory chronic cough or
unexplained chronic cough in adults.
The Kenilworth, N.J., drugmaker said the FDA set a target action
date of Dec. 21 for the review, adding that the agency plans to
discuss the application at an advisory committee meeting, which has
yet to be scheduled.
Merck said there are currently no approved therapies for the
treatment of refractory or unexplained chronic cough.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
March 01, 2021 07:33 ET (12:33 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
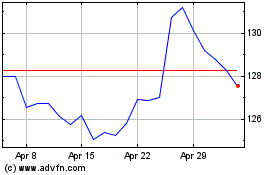
Merck (NYSE:MRK)
Historical Stock Chart
From Mar 2024 to Apr 2024
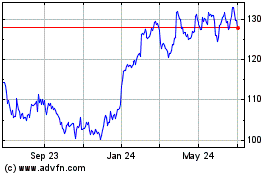
Merck (NYSE:MRK)
Historical Stock Chart
From Apr 2023 to Apr 2024